Relative Frequency of Paradoxical Growth and Trailing Effect with Caspofungin, Micafungin, Anidulafungin, and the Novel Echinocandin Rezafungin against Candida Species
Abstract
1. Introduction
2. Materials and Methods
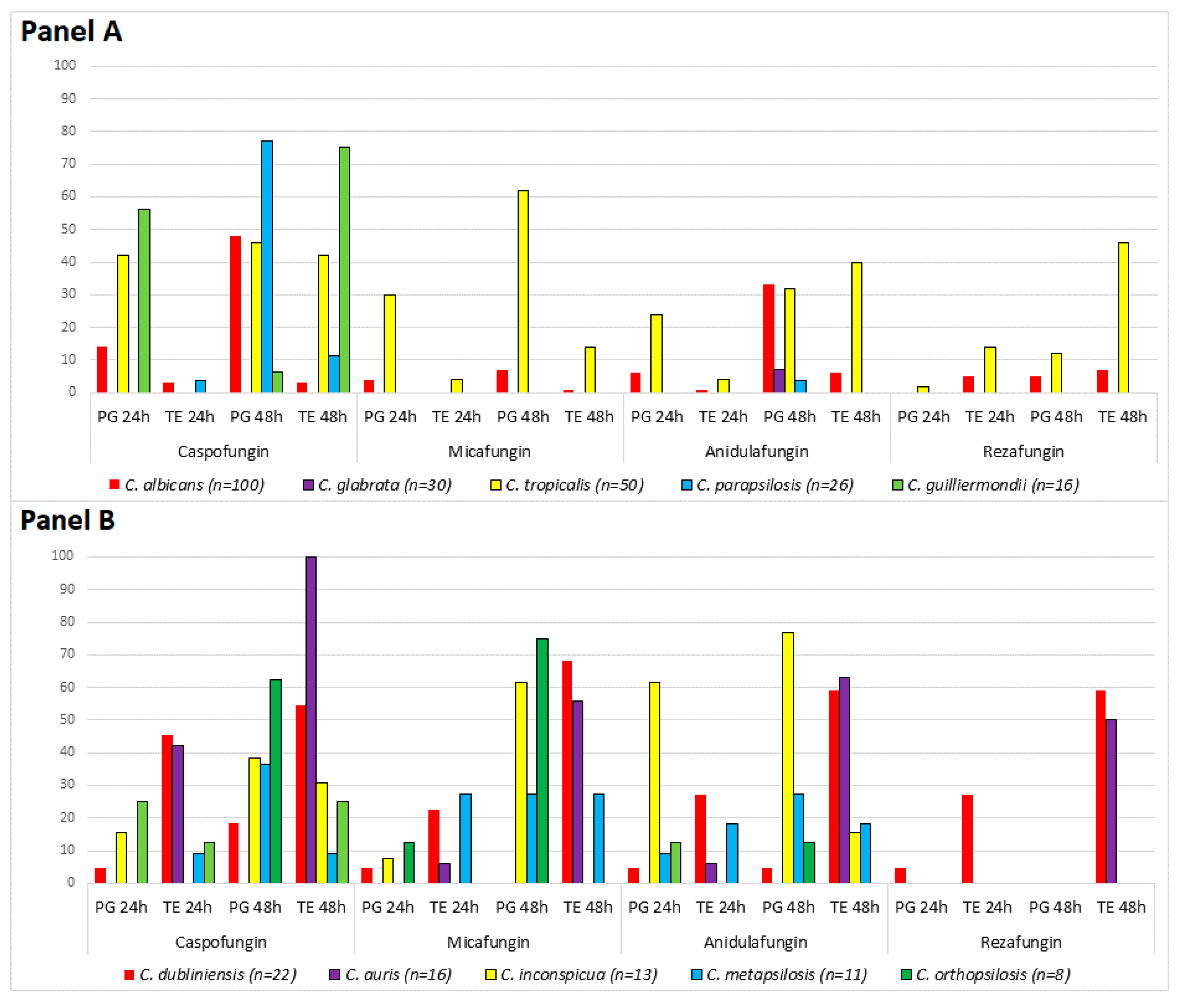
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Executive Summary: Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2016, 62. [Google Scholar] [CrossRef] [PubMed]
- Prépost, E.; Tóth, Z.; Perlin, D.S.; Gesztelyi, R.; Kardos, G.; Kovács, R.; Nagy, F.; Forgács, L.; Majoros, L. Efficacy of humanized single large doses of caspofungin on the lethality and fungal tissue burden in a deeply neutropenic murine model against Candida albicans and Candida dubliniensis. Infect. Drug Resist. 2019, 12, 1805–1814. [Google Scholar] [CrossRef] [PubMed]
- Domán, M.; Kovács, R.; Kardos, G.; Gesztelyi, R.; Juhasz, B.; Bozó, A.; Kardos, T.; Saleh, Q.; Majoros, L. Killing Rates of Caspofungin in 50 Percent Serum Correlate with Caspofungin Efficacy Against Candida albicans in a Neutropenic Murine Model. Curr. Drug Deliv. 2016, 13, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Prideaux, B.; Nagasaki, Y. Unraveling drug penetration of echinocandin antifungals at the site of infection in an intra-abdominal abscess model. Antimicrob. Agents Chemother. 2017, 61, e01009. [Google Scholar] [CrossRef] [PubMed]
- Bader, J.C.; Bhavnani, S.M.; Andes, D.R.; Ambrose, P.G. We can do better: A fresh look at echinocandin dosing. J. Antimicrob. Chemother. 2018, 73, i44–i50. [Google Scholar] [CrossRef]
- Wagener, J.; Loiko, V. Recent Insights into the Paradoxical Effect of Echinocandins. J. Fungi 2017, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Shields, R.K.; Nguyen, M.H.; Du, C.; Press, E.; Cheng, S.; Clancy, C.J. Paradoxical Effect of Caspofungin against Candida Bloodstream Isolates Is Mediated by Multiple Pathways but Eliminated in Human Serum. Antimicrob. Agents Chemother. 2011, 55, 2641–2647. [Google Scholar] [CrossRef]
- Rueda, C.; Cuenca-Estrella, M.; Zaragoza, O. Paradoxical Growth of Candida albicans in the Presence of Caspofungin Is Associated with Multiple Cell Wall Rearrangements and Decreased Virulence. Antimicrob. Agents Chemother. 2013, 58, 1071–1083. [Google Scholar] [CrossRef] [PubMed]
- Chamilos, G.; Lewis, R.E.; Albert, N.; Kontoyiannis, D.P. Paradoxical Effect of Echinocandins across Candida Species In Vitro: Evidence for Echinocandin-Specific and Candida Species-Related Differences. Antimicrob. Agents Chemother. 2007, 51, 2257–2259. [Google Scholar] [CrossRef]
- Marcos-Zambrano, L.J.; Escribano, P.; Sánchez-Carrillo, C.; Bouza, E.; Guinea, J. Frequency of the Paradoxical Effect Measured Using the EUCAST Procedure with Micafungin, Anidulafungin, and Caspofungin against Candida Species Isolates Causing Candidemia. Antimicrob. Agents Chemother. 2016, 61. [Google Scholar] [CrossRef]
- Fleischhacker, M.; Radecke, C.; Schulz, B.; Ruhnke, M. Paradoxical growth effects of the echinocandins caspofungin and micafungin, but not of anidulafungin, on clinical isolates of Candida albicans and C. dubliniensis. Eur. J. Clin. Microbiol. Infect. Dis. 2007, 27, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, M.D.; Whyte, J.A.; Odds, F.C. Candida albicans and Candida dubliniensis Respond Differently to Echinocandin Antifungal Agents In Vitro. Antimicrob. Agents Chemother. 2007, 51, 1882–1884. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rueda, C.; Puig-Asensio, M.; Padilla, B.; Muñoz, P.; Guinea, J.; Pardo, J.P.; García-Rodríguez, J.; Cerrada, C.G.; Fortún, J.; Martín, P.; et al. Evaluation of the possible influence of trailing and paradoxical effects on the clinical outcome of patients with candidemia. Clin. Microbiol. Infect. 2017, 49. [Google Scholar] [CrossRef] [PubMed]
- Varga, I.; Sóczó, G.; Kardos, G.; Majoros, L. Time-Kill studies investigating the killing activity of caspofungin against Candida dubliniensis: Comparing RPMI-1640 and antibiotic medium 3. J. Antimicrob. Chemother. 2008, 62, 149–152. [Google Scholar] [CrossRef]
- Betts, R.F.; Nucci, M.; Talwar, D.; Gareca, M.; Queiroz-Telles, F.; Bedimo, R.J.; Herbrecht, R.; Ruiz-Palacios, G.; Young, J.-A.H.; Baddley, J.W.; et al. A Multicenter, Double-Blind Trial of a High-Dose Caspofungin Treatment Regimen versus a Standard Caspofungin Treatment Regimen for Adult Patients with Invasive Candidiasis. Clin. Infect. Dis. 2009, 48, 1676–1684. [Google Scholar] [CrossRef]
- Pappas, P.G.; Rotstein, C.M.F.; Betts, R.F.; Nucci, M.; Talwar, D.; De Waele, J.J.; Vazquez, J.A.; Dupont, B.F.; Horn, D.L.; Ostrosky-Zeichner, L.; et al. Micafungin versus Caspofungin for Treatment of Candidemia and Other Forms of Invasive Candidiasis. Clin. Infect. Dis. 2007, 45, 883–893. [Google Scholar] [CrossRef]
- Tóth, Z.; Forgács, L.; Locke, J.B.; Kardos, G.; Nagy, F.; Kovács, R.; Szekely, A.; Borman, A.M.; Majoros, L. In Vitro activity of rezafungin against common and rare Candida species and Saccharomyces cerevisiae. J. Antimicrob. Chemother. 2019, 74, 3505–3510. [Google Scholar] [CrossRef]
- Zhao, Y.; Perez, W.B.; Jiménez-Ortigosa, C.; Hough, G.; Locke, J.B.; Ong, V.; Bartizal, K.; Perlin, D.S. CD101: A novel long-acting echinocandin. Cell. Microbiol. 2016, 18, 1308–1316. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts: Fourth Edition M27-Ed4; CLSI: Wayne, PA, USA, 2017. [Google Scholar]
- Kordalewska, M.; Lee, A.; Park, S.; Berrio, I.; Chowdhary, A.; Zhao, Y.; Perlin, D.S. Understanding Echinocandin Resistance in the Emerging Pathogen Candida auris. Antimicrob. Agents Chemother. 2018, 62. [Google Scholar] [CrossRef]
- Sofjan, A.K.; Mitchell, A.; Shah, D.N.; Nguyen, T.; Sim, M.; Trojcak, A.; Beyda, N.D.; Garey, K.W. Rezafungin (CD101), a next-generation echinocandin: A systematic literature review and assessment of possible place in therapy. J. Glob. Antimicrob. Resist. 2018, 14, 58–64. [Google Scholar] [CrossRef]
- Dudiuk, C.; Berrio, I.; Leonardelli, F.; Morales-Lopez, S.; Theill, L.; Macedo, D.; Rodriguez, J.Y.; Salcedo, S.; Marin, A.; Gamarra, S.; et al. Antifungal activity and killing kinetics of anidulafungin, caspofungin and amphotericin B against Candida auris. J. Antimicrob. Chemother. 2019, 74, 2295–2302. [Google Scholar] [CrossRef] [PubMed]
- Van Hal, S.J.; Stark, D.; Harkness, J.; Marriott, D. Candida dubliniensis Meningitis as Delayed Sequela of TreatedC. dubliniensis Fungemia. Emerg. Infect. Dis. 2008, 14, 327–329. [Google Scholar] [CrossRef]
- Alatoom, A.; Sartawi, M.; Lawlor, K.; AbdelWareth, L.; Thomsen, J.; Nusair, A.; Mirza, I. Persistent candidemia despite appropriate fungal therapy: First case of Candida auris from the United Arab Emirates. Int. J. Infect. Dis. 2018, 70, 36–37. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Bradley, N.; Brooks, S.; Burney, S.; Wassner, C. Management of Patients with Candida auris Fungemia at Community Hospital, Brooklyn, New York, USA, 2016–2018. Emerg. Infect. Dis. 2019, 25, 601–602. [Google Scholar] [CrossRef]
- Szilágyi, J.; Földi, R.; Bayegan, S.; Kardos, G.; Majoros, L. Effect of nikkomycin Z and 50% human serum on the killing activity of high-concentration caspofungin against Candida species using time-kill methodology. J. Chemother. 2012, 24, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Bayegan, S.; Majoros, L.; Kardos, G.; Kemény-Beke, Á.; Miszti, C.; Kovács, R.; Gesztelyi, R. In Vivo studies with a Candida tropicalis isolate exhibiting paradoxical growth in vitro in the presence of high concentration of caspofungin. J. Microbiol. 2010, 48, 170–173. [Google Scholar] [CrossRef] [PubMed]

| Species (n) | Drug | Number of Isolates Inhibited at Each MIC Value (µg/mL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| ≤0.06 | 0.12 | 0.25 | 0.5 | 1 | 2 | 4 | 8 | 16 | 32 | ||
| C. albicans(100) | RZF | 100 | |||||||||
| ANF | 100 | ||||||||||
| CSF | 9 | 5 | 52 | 34 | |||||||
| MCF | 99 | 1 | |||||||||
| C. glabrata(30) | RZF | 18 | 12 | ||||||||
| ANF | 30 | ||||||||||
| CSF | 1 | 11 | 18 | ||||||||
| MCF | 30 | ||||||||||
| C. parapsilosis sensu stricto(26) | RZF | 5 | 11 | 10 | |||||||
| ANF | 4 | 10 | 12 | ||||||||
| CSF | 4 | 16 | 6 | ||||||||
| MCF | 3 | 14 | 9 | ||||||||
| C. tropicalis(50) | RZF | 50 | |||||||||
| ANF | 50 | ||||||||||
| CSF | 2 | 9 | 24 | 15 | |||||||
| MCF | 48 | 2 | |||||||||
| C. krusei(30) | RZF | 16 | 14 | ||||||||
| ANF | 27 | 2 | 1 | ||||||||
| CSF | 2 | 6 | 22 | ||||||||
| MCF | 9 | 21 | |||||||||
| C. kefyr(16) | RZF | 10 | 6 | ||||||||
| ANF | 15 | 1 | |||||||||
| CSF | 12 | 4 | |||||||||
| MCF | 11 | 5 | |||||||||
| C. lusitaniae(27) | RZF | 3 | 17 | 7 | |||||||
| ANF | 26 | 1 | |||||||||
| CSF | 1 | 4 | 12 | 10 | |||||||
| MCF | 9 | 11 | 6 | 1 | |||||||
| C. guilliermondii(16) | RZF | 3 | 12 | 1 | |||||||
| ANF | 3 | 11 | 2 | ||||||||
| CSF | 1 | 9 | 6 | ||||||||
| MCF | 7 | 7 | 2 | ||||||||
| C. dubliniensis(22) | RZF | 22 | |||||||||
| ANF | 22 | ||||||||||
| CSF | 2 | 8 | 10 | 2 | |||||||
| MCF | 22 | ||||||||||
| C. auris(16) | RZF | 4 | 6 | 6 | |||||||
| ANF | 9 | 4 | 2 | 1 | |||||||
| CSF | 4 | 7 | 5 | ||||||||
| MCF | 6 | 10 | |||||||||
| C. orthopsilosis(8) | RZF | 3 | 2 | 3 | |||||||
| ANF | 1 | 1 | 3 | 3 | |||||||
| CSF | 3 | 5 | |||||||||
| MCF | 1 | 4 | 3 | ||||||||
| C. metapsilosis(11) | RZF | 2 | 9 | ||||||||
| ANF | 4 | 5 | 2 | ||||||||
| CSF | 1 | 5 | 5 | ||||||||
| MCF | 1 | 6 | 4 | ||||||||
| C. inconspicua(13) | RZF | 13 | |||||||||
| ANF | 13 | ||||||||||
| CSF | 2 | 4 | 7 | ||||||||
| MCF | 13 | ||||||||||
| Rezafungin | Anidulafungin | Caspofungin | Micafungin | |||||
|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 24 h | 48 h | 24 h | 48 h | 24 h | 48 h | |
| C. krusei ATCC 6258 | - | - | - | - | - | - | - | - |
| C. parapsilosis ATCC 22019 | - | - | - | PG (8–32) | - | - | - | - |
| C. albicans ATCC 10231 | - | - | - | - | - | PG (16–32) | - | - |
| C. glabrata ATCC 90030 | - | - | - | PG (16–32) | - | - | - | - |
| C. tropicalis ATCC 750 | - | - | - | - | PG (16–32) | PG (4–32) | - | - |
| C. orthopsilosis ATCC 96139 | - | - | - | - | PG (8) | PG (8) | PG (8) | PG (8) |
| C. metapsilosis ATCC 96144 | - | - | - | - | - | PG (8) | - | - |
| C. guilliermondii ATCC 6260 | - | - | - | - | - | PG (32) | - | - |
| C. dubliniensis CD36 | PG (16–32) | TE | TE | TE | TE | TE | - | - |
| C. auris NCPF 13029 = CBS 10913 (type strain) | - | TE | PG (32) | TE | PG (8–32) | TE | - | - |
| C. inconspicua ATCC 16783 | - | - | PG (8–32) | PG (1–32) | PG (4–8) | TE | - | PG (8–32) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tóth, Z.; Forgács, L.; Kardos, T.; Kovács, R.; Locke, J.B.; Kardos, G.; Nagy, F.; Borman, A.M.; Adnan, A.; Majoros, L. Relative Frequency of Paradoxical Growth and Trailing Effect with Caspofungin, Micafungin, Anidulafungin, and the Novel Echinocandin Rezafungin against Candida Species. J. Fungi 2020, 6, 136. https://doi.org/10.3390/jof6030136
Tóth Z, Forgács L, Kardos T, Kovács R, Locke JB, Kardos G, Nagy F, Borman AM, Adnan A, Majoros L. Relative Frequency of Paradoxical Growth and Trailing Effect with Caspofungin, Micafungin, Anidulafungin, and the Novel Echinocandin Rezafungin against Candida Species. Journal of Fungi. 2020; 6(3):136. https://doi.org/10.3390/jof6030136
Chicago/Turabian StyleTóth, Zoltán, Lajos Forgács, Tamás Kardos, Renátó Kovács, Jeffrey B. Locke, Gábor Kardos, Fruzsina Nagy, Andrew M. Borman, Awid Adnan, and László Majoros. 2020. "Relative Frequency of Paradoxical Growth and Trailing Effect with Caspofungin, Micafungin, Anidulafungin, and the Novel Echinocandin Rezafungin against Candida Species" Journal of Fungi 6, no. 3: 136. https://doi.org/10.3390/jof6030136
APA StyleTóth, Z., Forgács, L., Kardos, T., Kovács, R., Locke, J. B., Kardos, G., Nagy, F., Borman, A. M., Adnan, A., & Majoros, L. (2020). Relative Frequency of Paradoxical Growth and Trailing Effect with Caspofungin, Micafungin, Anidulafungin, and the Novel Echinocandin Rezafungin against Candida Species. Journal of Fungi, 6(3), 136. https://doi.org/10.3390/jof6030136








