The Caryopsis of Red-Grained Rice Has Enhanced Resistance to Fungal Attack
Abstract
1. Introduction
2. Materials and Methods
2.1. Rice Genotypes and Kernel Preparation
2.2. Effect of Red Rice Pigment Extract on Fungal Growth
2.3. Fungi Used for Infection Tests
2.4. Effect of E. nigrum on Rice Germination and Seedling Growth
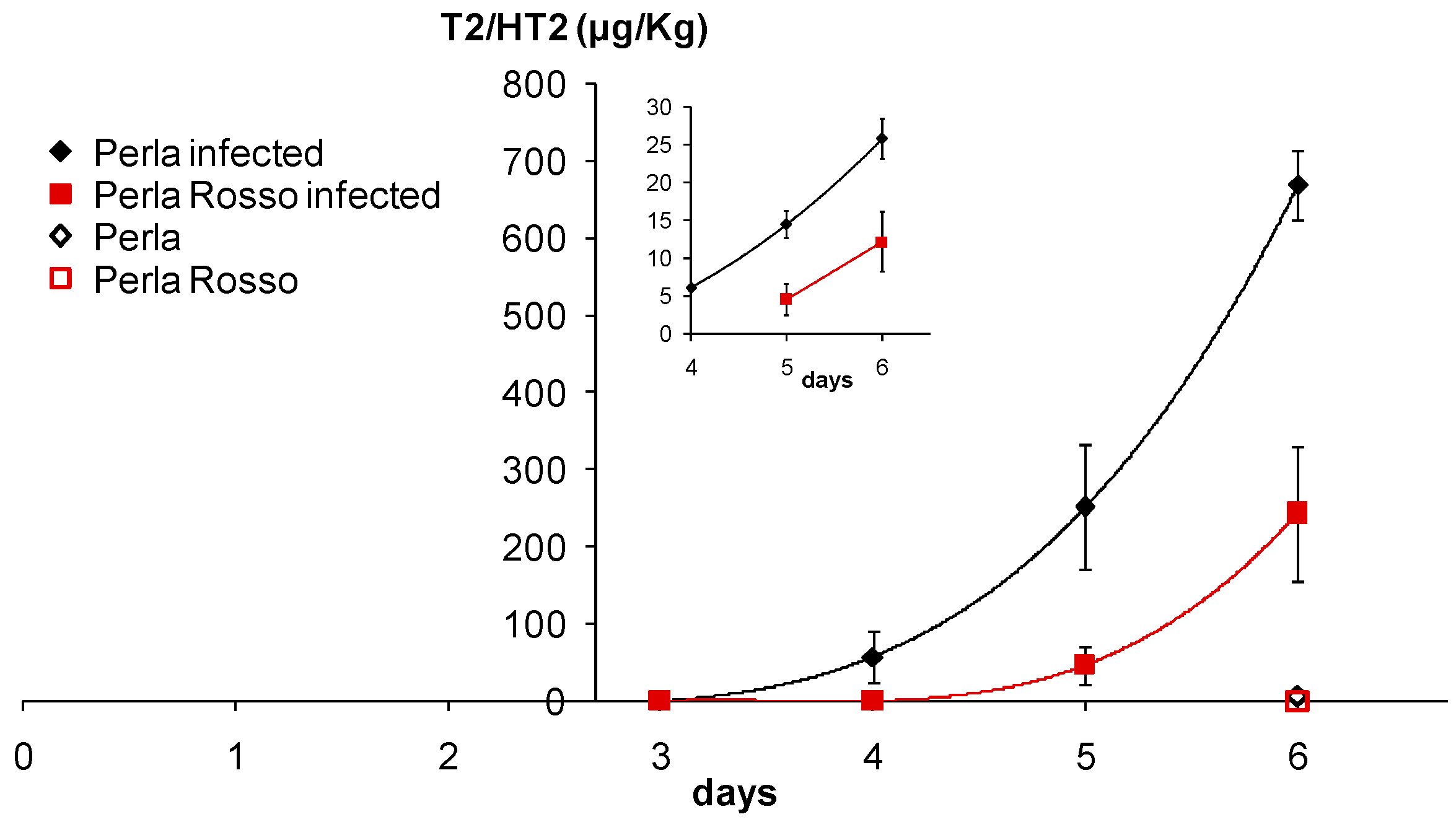
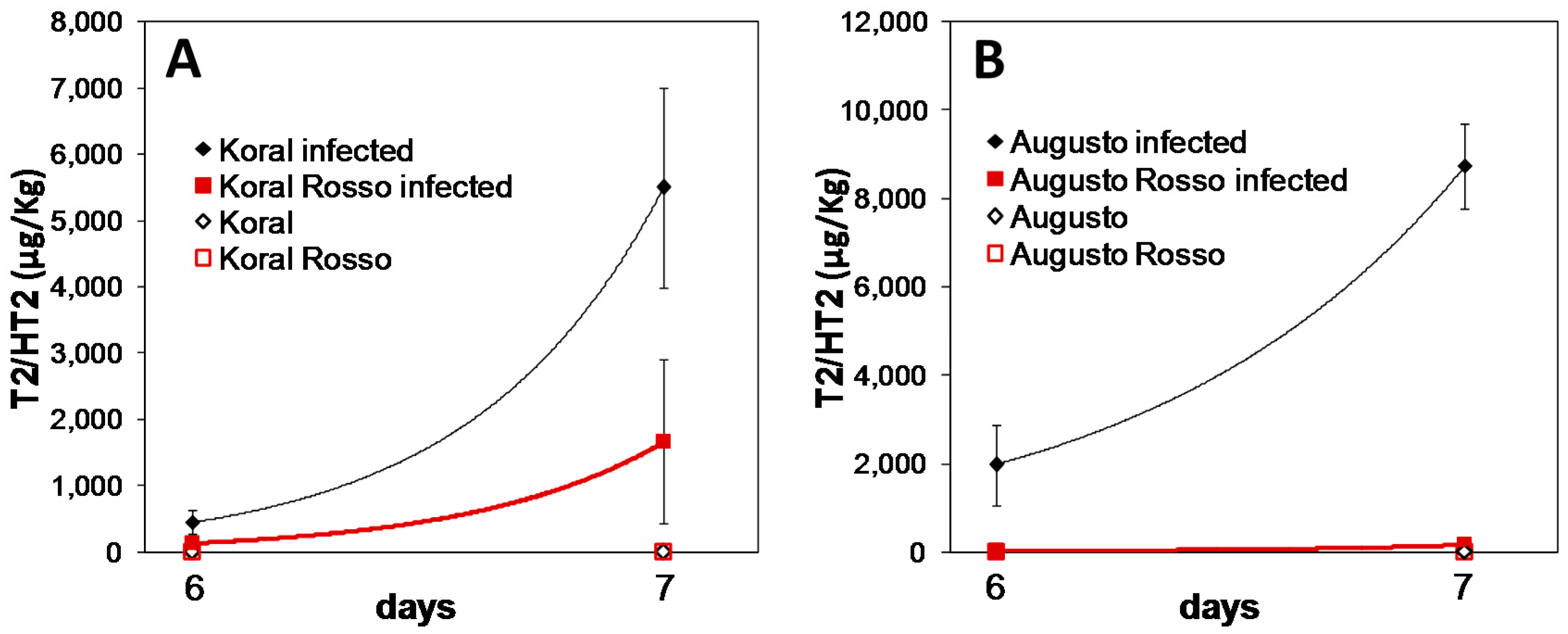
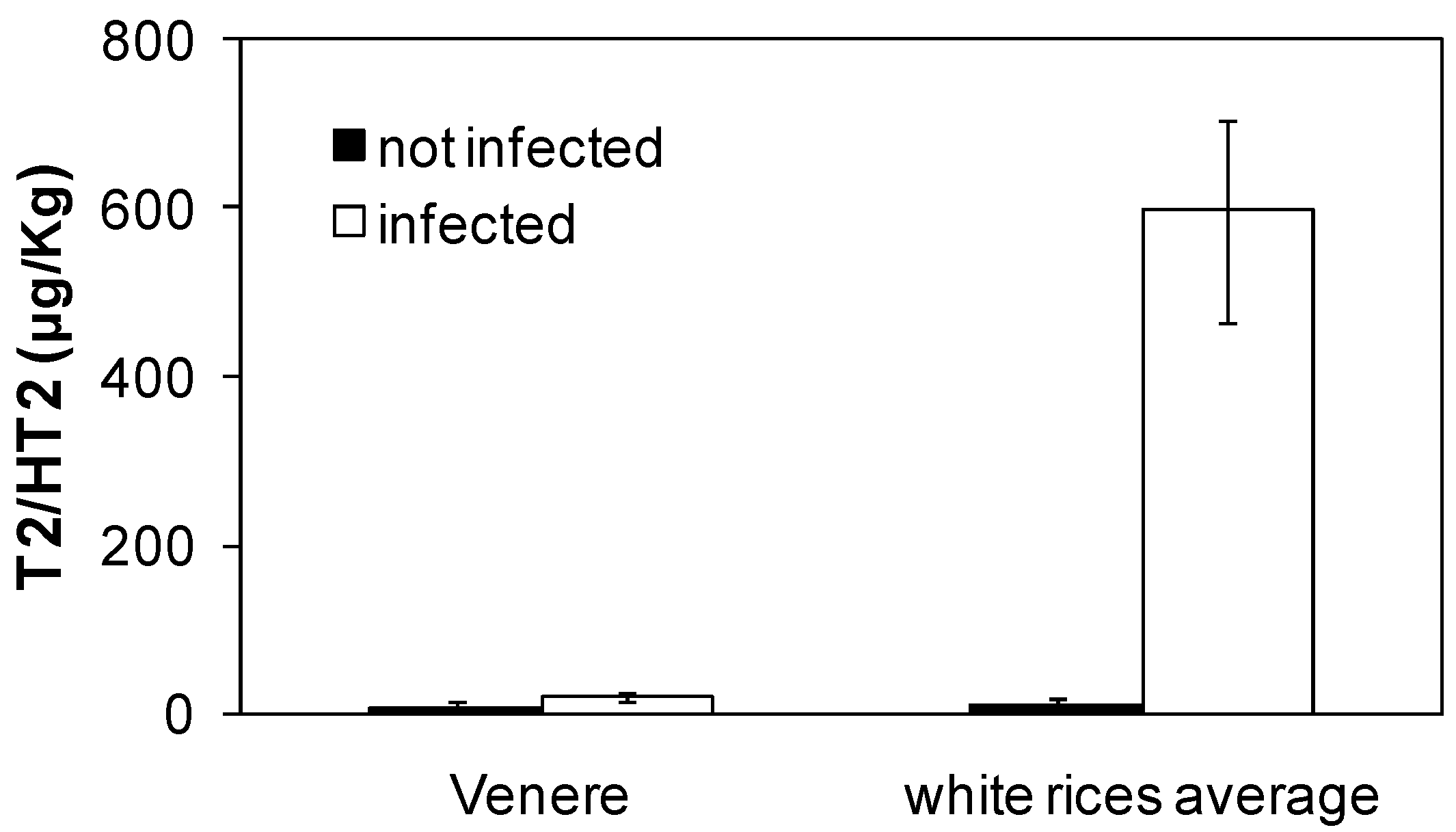
2.5. Accumulation of T-2/HT-2 from F. sporotrichioides in Rice Kernels
2.6. Mycotoxin Quantification
2.7. Statistical Analysis
3. Results and Discussion
3.1. Germination and Growth of Caryopses Infected with E. nigrum
3.2. Accumulation of T-2/HT-2 in Caryopses Infected with F. sporotrichioides
4. Conclusions and Perspectives
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Krishnan, S.; Dayanandan, P. Structural and histochemical studies on grain-filling in the caryopsis of rice (Oryza sativa L.). J. Biosci. 2003, 28, 455–469. [Google Scholar] [CrossRef] [PubMed]
- Finocchiaro, F.; Ferrari, B.; Gianinetti, A. A study of biodiversity of flavonoid content in the rice caryopsis evidencing simultaneous accumulation of anthocyanins and proanthocyanidins in a black-grained genotype. J. Cereal Sci. 2010, 51, 28–34. [Google Scholar] [CrossRef]
- Finocchiaro, F.; Ferrari, B.; Gianinetti, A.; Dall’Asta, C.; Galaverna, G.; Scazzina, F.; Pellegrini, N. Characterization of antioxidant compounds of red and white rice and changes in total antioxidant capacity during processing. Mol. Nutr. Food Res. 2007, 51, 1006–1019. [Google Scholar] [CrossRef] [PubMed]
- Imolehin, E.D. Rice seedborne fungi and their effect on seed germination. Plant Dis. 1983, 67, 1334–1336. [Google Scholar] [CrossRef]
- Ou, S.H. Rice Diseases, 2nd ed.; Commonwealth Mycological Institute: Kew, UK, 1985; ISBN 978-0-85198-545-9. [Google Scholar]
- Webster, R.K.; Gunnell, P.S. (Eds.) Compendium of Rice Diseases; The Disease Compendium Series of the American Phytopathological Society; APS Press: St. Paul, MN, USA, 1992; ISBN 978-0-89054-126-5. [Google Scholar]
- Lanning, F.C.; Eleuterius, L.N. Silica and ash in seeds of cultivated grains and native plants. Ann. Bot. 1992, 69, 151–160. [Google Scholar] [CrossRef]
- Dixon, R.A.; Xie, D.-Y.; Sharma, S.B. Proanthocyanidins—A final frontier in flavonoid research? New Phytol. 2005, 165, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Pourcel, L.; Routaboul, J.; Cheynier, V.; Lepiniec, L.; Debeaujon, I. Flavonoid oxidation in plants: From biochemical properties to physiological functions. Trends Plant. Sci. 2007, 12, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Skadhauge, B.; Thomsen, K.K.; von Wettstein, D. The role of the barley testa layer and its flavonoid content in resistance to Fusarium infections. Hereditas 1997, 126, 147–160. [Google Scholar] [CrossRef]
- Boutigny, A.-L.; Richard-Forget, F.; Barreau, C. Natural mechanisms for cereal resistance to the accumulation of Fusarium trichothecenes. Eur. J. Plant Pathol. 2008, 121, 411–423. [Google Scholar] [CrossRef]
- Terzi, V.; Tumino, G.; Stanca, A.M.; Morcia, C. Reducing the incidence of cereal head infection and mycotoxins in small grain cereal species. J. Cereal Sci. 2014, 59, 284–293. [Google Scholar] [CrossRef]
- Sempere Ferre, F. Worldwide occurrence of mycotoxins in rice. Food Control 2016, 62, 291–298. [Google Scholar] [CrossRef]
- Cho, D.-H.; Lim, S.-T. Germinated brown rice and its bio-functional compounds. Food Chem. 2016, 196, 259–271. [Google Scholar] [CrossRef] [PubMed]
- De Lima Favaro, L.C..; de Melo, F.L.; Aguilar-Vildoso, C.I.; Araújo, W.L. Polyphasic analysis of intraspecific diversity in Epicoccum nigrum warrants reclassification into separate species. PLoS ONE 2011, 6, e14828. [Google Scholar] [CrossRef]
- Lee, F.N. Red Blotch of Grains. In Compendium of Rice Diseases; Webster, R.K., Gunnell, P.S., Eds.; The Disease Compendium Series of the American Phytopathological Society; APS Press: St. Paul, MN, USA, 1992; p. 31. ISBN 978-0-89054-126-5. [Google Scholar]
- Mateo, J.J.; Mateo, R.; Jiménez, M. Accumulation of type A trichothecenes in maize, wheat and rice by Fusarium sporotrichioides isolates under diverse culture conditions. Int. J. Food Microbiol. 2002, 72, 115–123. [Google Scholar] [CrossRef]
- Sweeney, M.T.; Thomson, M.J.; Pfeil, B.E.; McCouch, S. Caught red-handed: Rc encodes a basic helix-loop-helix protein conditioning red pericarp in rice. Plant Cell 2006, 18, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Lupotto, E.; Powell, W. G-string slippage turns white rice red. Genome 2009, 52, 490–493. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhou, X.-Y.; Qiang, Q.-Q.; Zhang, Z.-Q. Ultrasound-assisted extraction and preliminary purification of proanthocyanidins and chlorogenic acid from almond (Prunus dulcis) skin. J. Sep. Sci. 2014, 37, 1834–1841. [Google Scholar] [CrossRef] [PubMed]
- Morcia, C.; Tumino, G.; Ghizzoni, R.; Bara, A.; Salhi, N.; Terzi, V. In vitro evaluation of sub-lethal concentrations of plant-derived antifungal compounds on FUSARIA growth and mycotoxin production. Molecules 2017, 22, 1271. [Google Scholar] [CrossRef] [PubMed]
- Sweet, H.C.; Bolton, W.E. The surface decontamination of seeds to produce axenic seedlings. Am. J. Bot. 1979, 66, 692–698. [Google Scholar] [CrossRef]
- Chun, S.-C.; Schneider, R.W.; Cohn, M.A. Sodium hypochlorite: Effect of solution pH on rice seed disinfestation and its direct effect on seedling growth. Plant Dis. 1997, 81, 821–824. [Google Scholar] [CrossRef]
- Abdul-Baki, A.A.; Anderson, J.D. Vigor determination in soybean seed by multiple criteria. Crop Sci. 1973, 13, 630–633. [Google Scholar] [CrossRef]
- Morcia, C.; Tumino, G.; Ghizzoni, R.; Badeck, F.W.; Lattanzio, V.M.T.; Pascale, M.; Terzi, V. Occurrence of Fusarium langsethiae and T-2 and HT-2 toxins in Italian malting barley. Toxins 2016, 8, 247. [Google Scholar] [CrossRef] [PubMed]
- Duchateau, L.; Janssen, P.; Rowlands, G.J. Linear Mixed Models. An Introduction with Applications in Veterinary Research; International Livestock Research Institute: Nairobi, Kenya, 1998; ISBN 978-92-9146-038-0. [Google Scholar]
- Zar, J.H. Biostatistical Analysis, 4th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1999; ISBN 978-0-13-081542-2. [Google Scholar]
- Butsat, S.; Siriamornpun, S. Antioxidant capacities and phenolic compounds of the husk, bran and endosperm of Thai rice. Food Chem. 2010, 119, 606–613. [Google Scholar] [CrossRef]
- Hashem, M.; Ali, E. Epicoccum nigrum as biocontrol agent of Pythium damping-off and root-rot of cotton seedlings. Arch. Phytopathol. Plant Prot. 2004, 37, 283–297. [Google Scholar] [CrossRef]
- Nelson, E.B. The seed microbiome: Origins, interactions, and impacts. Plant Soil 2017, 422, 7–34. [Google Scholar] [CrossRef]
- Bamford, P.C.; Norris, G.L.F.; Ward, G. Flavipin production by Epicoccum spp. Trans. Br. Mycol. Soc. 1961, 44, 354–356. [Google Scholar] [CrossRef]
- Brown, A.E.; Finlay, R.; Ward, J.S. Antifungal compounds produced by Epicoccum purpurascens against soil-borne plant pathogenic fungi. Soil Biol. Biochem. 1987, 19, 657–664. [Google Scholar] [CrossRef]
- Madrigal, C.; Tadeo, J.L.; Melgarejo, P. Relationship between flavipin production by Epicoccum nigrum and antagonism against Monilinia laxa. Mycol. Res. 1991, 95, 1375–1381. [Google Scholar] [CrossRef]
- De Lima Favaro, L.C.; de Souza Sebastianes, F.L.; Araújo, W.L. Epicoccum nigrum P16, a sugarcane endophyte, produces antifungal compounds and induces root growth. PLoS ONE 2012, 7, e36826. [Google Scholar] [CrossRef]
- Li, Y.; Xia, L.Q.; Wang, Y.N.; Liu, X.Y.; Zhang, C.H.; Hu, T.L.; Cao, K.Q. The inhibitory effect of Epicoccum nigrum strain XF1 against Phytophthora infestans. Biol. Control 2013, 67, 462–468. [Google Scholar] [CrossRef]
- Franke, J.; Geary, B.; Meyer, S.E. Identification of the infection route of a Fusarium seed pathogen into nondormant Bromus tectorum seeds. Phytopathology 2014, 104, 1306–1313. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Nan, Z.; Zhang, X.; Hou, F.; Christensen, M.; Baskin, C. Does dormancy protect seeds against attack by the pathogenic fungus Fusarium tricinctum in a semiarid grassland of Northwest China? Plant Soil 2017, 422, 155–168. [Google Scholar] [CrossRef]
- Dalling, J.W.; Davis, A.S.; Schutte, B.J.; Elizabeth Arnold, A. Seed survival in soil: Interacting effects of predation, dormancy and the soil microbial community. J. Ecol. 2011, 99, 89–95. [Google Scholar] [CrossRef]
- Nazari, L.; Pattori, E.; Terzi, V.; Morcia, C.; Rossi, V. Influence of temperature on infection, growth, and mycotoxin production by Fusarium langsethiae and F. sporotrichioides in durum wheat. Food Microbiol. 2014, 39, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Atanasova-Penichon, V.; Barreau, C.; Richard-Forget, F. Antioxidant secondary metabolites in cereals: Potential involvement in resistance to Fusarium and mycotoxin accumulation. Front. Microbiol. 2016, 7, 566. [Google Scholar] [CrossRef] [PubMed]
- Ghotbzadeh, S.; Gianinetti, A. A response of the imbibed dormant red rice caryopsis to biotic challenges involves extracellular pH increase to elicit superoxide production. Seed Sci. Res. 2018, in press. [Google Scholar]
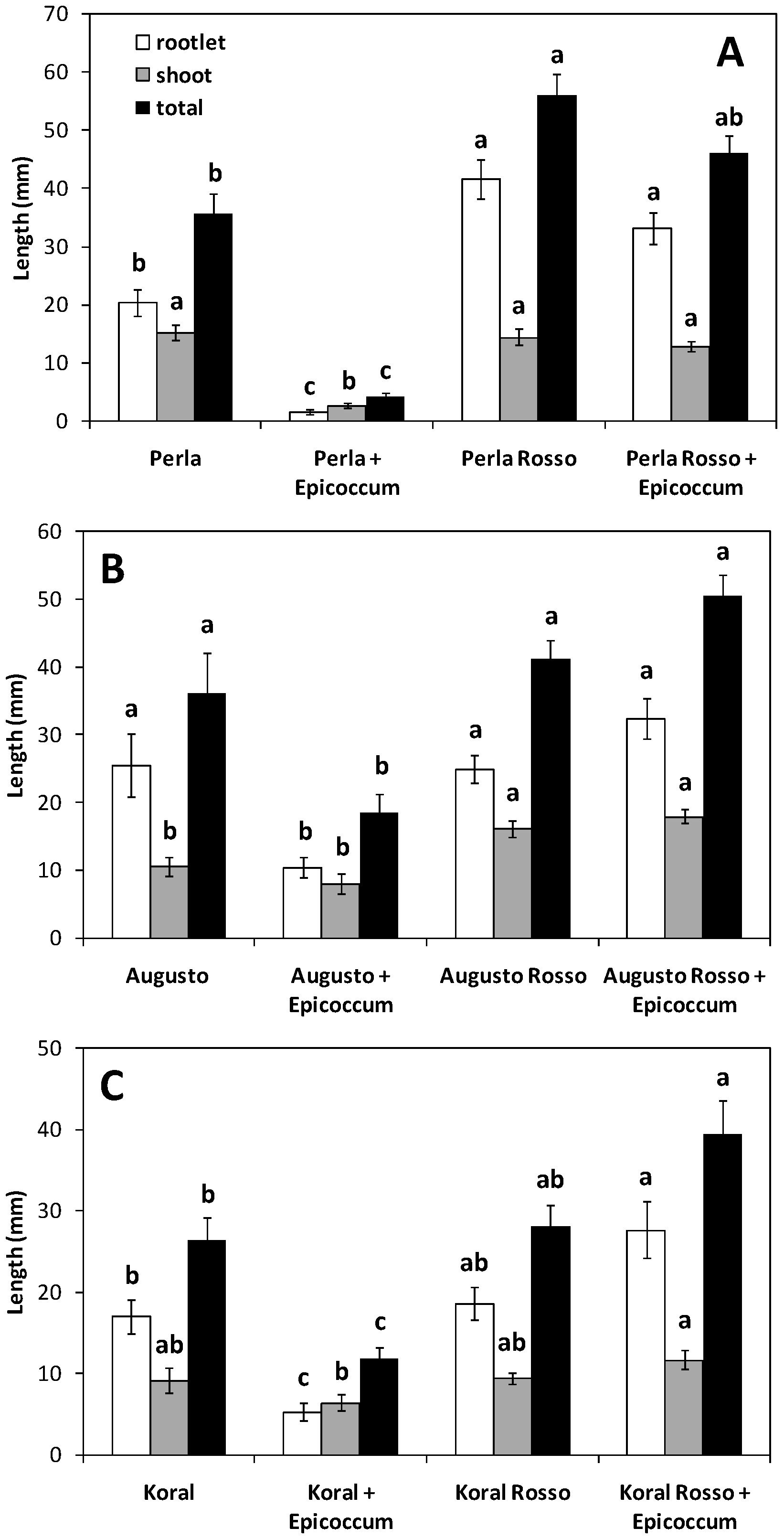
| Grain | Germination (%) | Mean Seedling Length (mm) | Vigour Index | |||
|---|---|---|---|---|---|---|
| Control | with E. nigrum | Control | with E. nigrum | Control | with E. nigrum | |
| White | 86.0 ± 7.9 a | 52.9 ± 22.6 b | 37.5 ± 4.9 a | 20.2 ± 6.5 b | 3262 ± 553 a | 1140 ± 716 b |
| Red | 96.0 ± 4.6 a | 91.5 ± 4.7 a | 43.0 ± 13.2 a | 49.0 ± 4.4 a | 4170 ± 1401 a | 4515 ± 552 a |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gianinetti, A.; Finocchiaro, F.; Maisenti, F.; Kouongni Satsap, D.; Morcia, C.; Ghizzoni, R.; Terzi, V. The Caryopsis of Red-Grained Rice Has Enhanced Resistance to Fungal Attack. J. Fungi 2018, 4, 71. https://doi.org/10.3390/jof4020071
Gianinetti A, Finocchiaro F, Maisenti F, Kouongni Satsap D, Morcia C, Ghizzoni R, Terzi V. The Caryopsis of Red-Grained Rice Has Enhanced Resistance to Fungal Attack. Journal of Fungi. 2018; 4(2):71. https://doi.org/10.3390/jof4020071
Chicago/Turabian StyleGianinetti, Alberto, Franca Finocchiaro, Fabio Maisenti, Dailly Kouongni Satsap, Caterina Morcia, Roberta Ghizzoni, and Valeria Terzi. 2018. "The Caryopsis of Red-Grained Rice Has Enhanced Resistance to Fungal Attack" Journal of Fungi 4, no. 2: 71. https://doi.org/10.3390/jof4020071
APA StyleGianinetti, A., Finocchiaro, F., Maisenti, F., Kouongni Satsap, D., Morcia, C., Ghizzoni, R., & Terzi, V. (2018). The Caryopsis of Red-Grained Rice Has Enhanced Resistance to Fungal Attack. Journal of Fungi, 4(2), 71. https://doi.org/10.3390/jof4020071






