First Report of Agroathelia rolfsii Causing White Fruit Rot in Oil Palm Hybrid OxG in Colombia
Abstract
1. Introduction
2. Materials and Methods
2.1. Symptom Description and Isolate Collection
2.2. Pathogenicity Test
2.3. Morphological Characterization
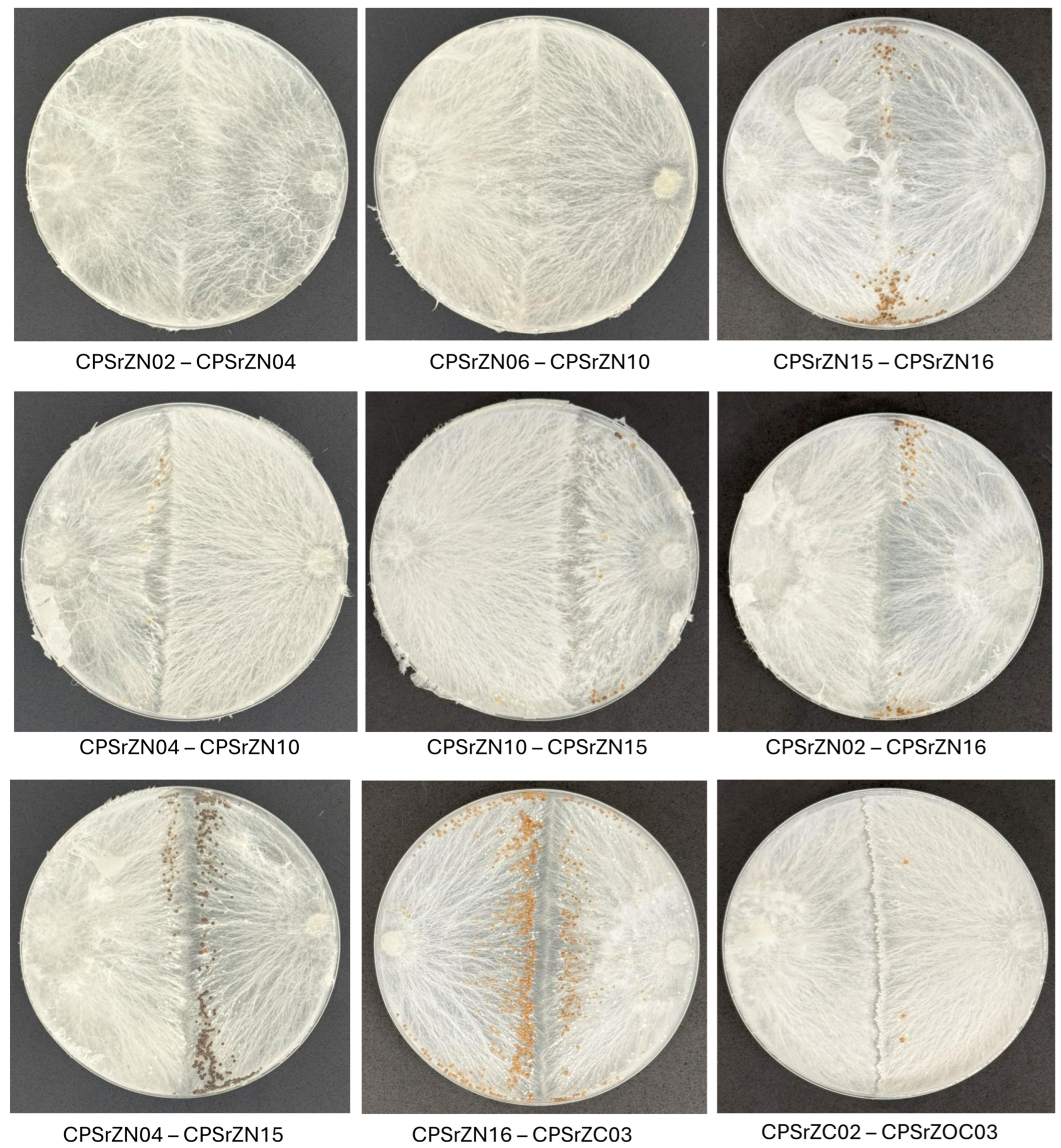
2.4. Evaluation of Mycelial Compatibility
2.5. Molecular Characterization, DNA Extraction, and Amplification
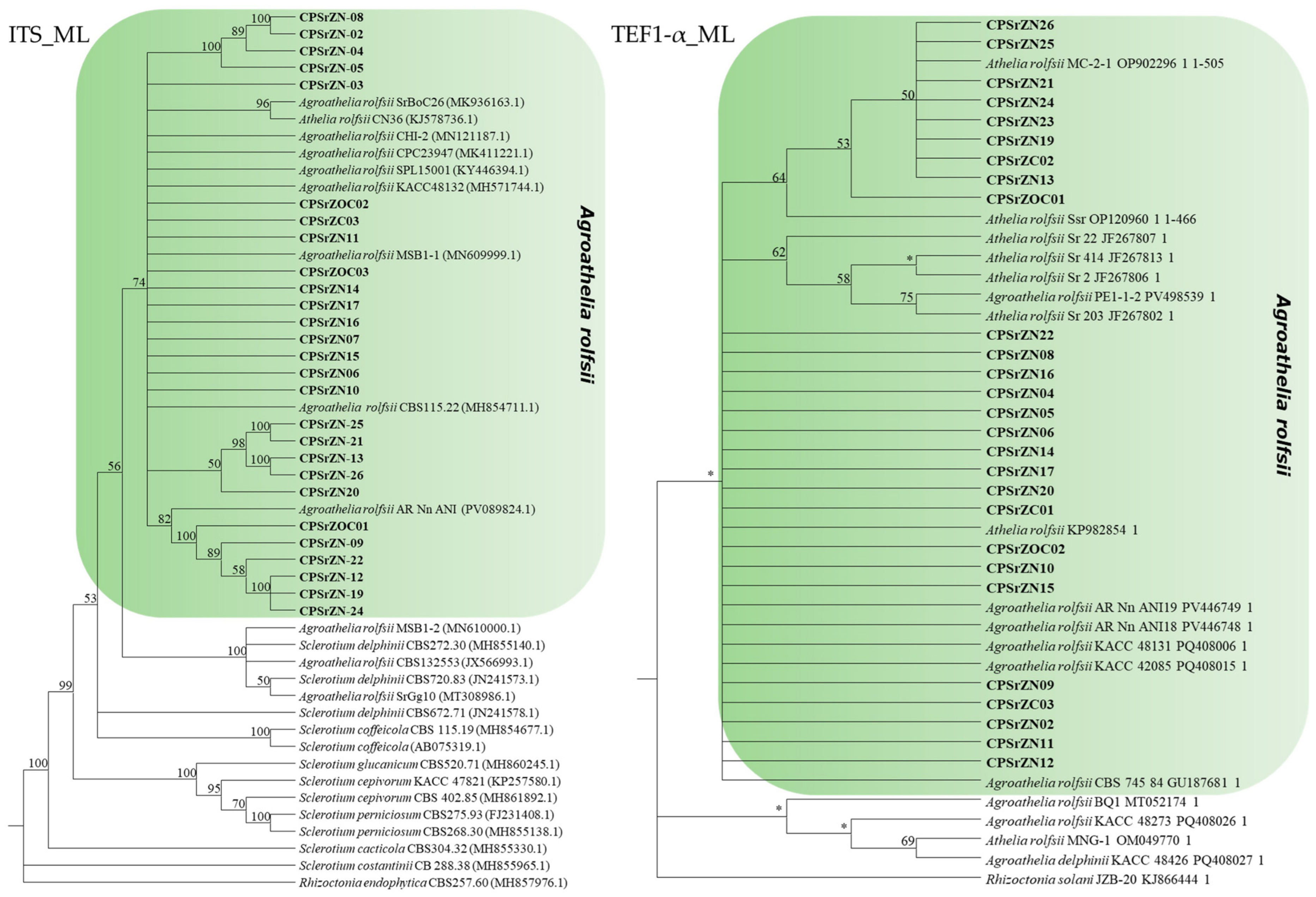
2.6. Phylogenetic Analysis
3. Results
3.1. Symptom Description
3.2. Isolated Microorganisms
3.3. Pathogenicity Tests
3.4. Mycelial Compatibility Assessment
3.5. Molecular Identification
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- SISPA Producción y Rendimiento, Historico. Available online: https://sispaplus.fedepalma.org/Reportes_Publicos/Produccion_Rendimiento (accessed on 28 November 2025).
- Bojacá, C.R. Parte 1 Variabilidad Climática En Las Zonas Palmeras de Colombia ¿cómo Se Ha Comportado La Temperatura Durante Los Últimos 30 Años. Available online: https://elpalmicultor.com/variabilidad-climatica-zonas-palmeras-colombia/ (accessed on 16 December 2025).
- IDEAM Sistema de Información Para La Gestión de Datos Hidrológicos y Meteorológicos. Available online: https://www.ideam.gov.co/dhime (accessed on 16 December 2025).
- Maizatul-Suriza, M.; Suhanah, J.; Madihah, A.Z.; Idris, A.S.; Mohidin, H. Phylogenetic and Pathogenicity Evaluation of the Marasmioid Fungus Marasmius palmivorus Causing Fruit Bunch Rot Disease of Oil Palm. For. Pathol. 2021, 51, e12660. [Google Scholar] [CrossRef]
- Pong, V.M.; Zainal Abidin, M.A.; Almaliky, B.S.A.; Kadir, J.; Wong, M.Y. Isolation, Fruiting and Pathogenicity of Marasmiellus palmivorus (Sharples) Desjardin (comb. prov.) in Oil Palm Plantations in West Malaysia. Pertanika J. Trop. Agric. Sci. 2012, 35, 37–48. Available online: http://psasir.upm.edu.my/id/eprint/19208/1/80.%20Isolation%2C%20Fruiting%20and%20Pathogenicity%20of%20Marasmiellus%20palmivorus.pdf (accessed on 16 December 2025).
- Almaliky, B.S.A.; MiorAhmad, Z.A.; Kadir, J.; Mui, W. Pathogenicity of Marasmiellus palmivorus (Sharples) Desjardin Comb. Prov. on oil palm Elaeis guineensis. Wulfenia 2012, 19, 144–160. Available online: https://www.researchgate.net/publication/263582258_Pathogenicity_of_Marasmiellus_palmivorus_Sharples_Desjardin_comb_Prov_on_Oil_Palm_Elaeis_guineensis (accessed on 16 December 2025).
- Balanagouda, P.; Sridhara, S.; Shil, S.; Hegde, V.; Naik, M.K.; Narayanaswamy, H.; Balasundram, S.K. Assessment of the Spatial Distribution and Risk Associated with Fruit Rot Disease in Areca catechu L. J. Fungi 2021, 7, 797. [Google Scholar] [CrossRef]
- dos Santos, P.H.D.; Mussi-Dias, V.; Freire, M.D.; Carvalho, B.M.; da Silveira, S.F. Diagrammatic Scale of Severity for Postharvest Black Rot (Ceratocystis paradoxa) in Coconut Palm Fruits. Summa Phytopathol. 2017, 43, 269–275. [Google Scholar] [CrossRef][Green Version]
- Mahadevakumar, S.; Yadav, V.; Tejaswini, G.S.; Janardhana, G.R. Morphological and Molecular Characterization of Sclerotium rolfsii. Associated with Fruit Rot of Cucurbita maxima. Eur. J. Plant Pathol. 2015, 145, 215–219. [Google Scholar] [CrossRef]
- Okabe, I.; Matsumoto, N. Population Structure of Sclerotium rolfsii in Peanut Fields. Mycoscience 2000, 41, 145–148. [Google Scholar] [CrossRef]
- Kator, L.; Yula Hosea, Z.; Daniel Oche, O. Sclerotium rolfsii; Causative Organism of Southern Blight, Stem Rot, White Mold and Sclerotia Rot Disease. Sch. Res. Libr. Ann. Biol. Res. 2015, 6, 78–89. Available online: https://www.researchgate.net/publication/343268195_Sclerotium_rolfsii_Causative_organism_of_southern_blight_stem_rot_white_mold_and_sclerotia_rot_disease (accessed on 16 December 2025).
- Remesal, E.; Jordán-Ramírez, R.; Jiménez-Díaz, R.M.; Navas-Cortés, J.A. Mycelial Compatibility Groups and Pathogenic Diversity in Sclerotium rolfsii Populations from Sugar Beet Crops in Mediterranean-Type Climate Regions. Plant Pathol. 2012, 61, 739–753. [Google Scholar] [CrossRef]
- Elahi, F.E.; Islam, M.; Alam, M.; Islam, N.; Tipu, M.M.H.; Khatun, F.; Chowdhury, S.R.; Sarker, S. First Report of Athelia rolfsii on Jackfruit (Artocarpus heterophyllus). Australas. Plant Dis. Notes 2021, 16, 16–18. [Google Scholar] [CrossRef]
- Chowdary, G.B.S.M.; Jameema, G.; Charishma, K.V. Collar and Stem Pathogen Sclerotium rolfsii: A Review. Plant Arch. 2024, 24, 67–72. [Google Scholar] [CrossRef]
- Okon, O.G.; Uwaidem, Y.I.; Rhouma, A.; Antia, U.E.; Okon, J.E.; Archibong, B.F. Unraveling the Biology, Effects and Management Methods of Sclerotium rolfsii Infection in Plants for Sustainable Agriculture. Eur. J. Biol. Res. 2025, 15, 1–10. [Google Scholar] [CrossRef]
- Forero, D.C.; Hormaza, P.A.; Moreno, L.P.; Ruíz, R. Generalidades Sobre La Morfología y Fenología de La Palma de Aceite; Centro de Investigación en Palma de Aceite: Bogotá, Colombia, 2012; ISBN 978-958-8360-40-9. [Google Scholar]
- Marshall, D.; Tunali, B.; Nelson, L.R. Occurrence of Fungal Endophytes in Species of Wild Triticum. Crop Sci. 1999, 39, 1507–1512. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 Years of Image Analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Punja, Z.K. Hyphal Interactions and Antagonism Among Field Isolates and Single-Basidiospore Strains of Athelia (Sclerotium) rolfsii. Phytopathology 1983, 73, 1279. [Google Scholar] [CrossRef]
- Adaskaveg, J.E.; Gilbertson, R.L. Vegetative Incompatibility between Intraspecific Dikaryotic Pairings of Ganoderma lucidum and G. Tsugae. Mycologia 1987, 79, 603–613. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Academic Press: Cambridge, MA, USA, 1990; pp. 315–322. ISBN 0-12-372180-6. [Google Scholar]
- Carbone, I.; Kohn, L.M. A Method for Designing Primer Sets for Speciation Studies in Filamentous Ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Edler, D.; Klein, J.; Antonelli, A.; Silvestro, D. RaxmlGUI 2.0: A Graphical Interface and Toolkit for Phylogenetic Analyses Using RAxML. Methods Ecol. Evol. 2021, 12, 373–377. [Google Scholar] [CrossRef]
- Caicedo, A.F.; Millan, E.S.; Ruiz, R.; Romero, H.M. Criterios de Cosecha En Cultivares Híbrido: Características Que Evalúan El Punto de Cosecha En Palma de Aceite, 2nd ed.; Federación Nacional de Cultivadores de Palma de Aceite de Colombia: Bogotá, Colombia, 2020; ISBN 978-958-8360-75-1. [Google Scholar]
- Romero, H.M.; Ruiz-Romero, R.; Caicedo-Zambrano, A.F.; Ayala-Diaz, I.; Rodríguez, J.L. Determining the Optimum Harvest Point in Oil Palm Interspecific Hybrids (O × G) to Maximize Oil Content. Agronomy 2025, 15, 887. [Google Scholar] [CrossRef]
- Adhikari, P.; Shrestha, S.M.; Manandhar, H.K.; Marahatta, S. Morphology and Cross Infectivity of Sclerotium rolfsii Sacc. Isolated from Different Host Plants in Nepal. J. Agric. Environ. 2022, 23, 177–187. [Google Scholar] [CrossRef]
- Ablormeti, F.K.; Souza, P.G.C.; Awuah, R.T.; Kwoseh, C.K.; Agbetiameh, D.; Kena, A.W.; Aidoo, K.A.S.; Lutuf, H.; Sossah, F.L.; da Silva, R.S.; et al. Modeling Global Habitat Suitability of Agroathelia rolfsii Causing Sclerotium Wilt Disease of Tomato with Emphasis on Ghana. Sci. Rep. 2025, 15, 34034. [Google Scholar] [CrossRef]
- Ke, Z.; Cai, G.; Zhang, F.; Yang, S.; Cheng, M.; Lu, W.; Chen, Y. First Report of Agroathelia (syn. Sclerotium) rolfsii Associated with Southern Blight Disease of Daphne tangutica (Gansu Ruixiang) in China. Plant Dis. 2025, 109, 2216. [Google Scholar] [CrossRef]
- Acabal, B.D.; Dalisay, T.U.; Groenewald, J.Z.; Crous, P.W.; Cumagun, C.J.R. Athelia rolfsii (=Sclerotium rolfsii) Infects Banana in the Philippines. Australas. Plant Dis. Notes 2019, 14, 10. [Google Scholar] [CrossRef]
- Meena, P.N.; Meena, A.K.; Ram, C. Morphological and Molecular Diversity in Sclerotium rolfsii Sacc., Infecting Groundnut (Arachis hypogaea L.). Discov. Agric. 2023, 1, 3. [Google Scholar] [CrossRef]
- Paparu, P.; Acur, A.; Kato, F.; Acam, C.; Nakibuule, J.; Nkuboye, A.; Musoke, S.; Mukankusi, C. Morphological and Pathogenic Characterization of Sclerotium rolfsii, the Causal Agent of Southern Blight Disease on Common Bean in Uganda. Plant Dis. 2020, 104, 2130–2137. [Google Scholar] [CrossRef] [PubMed]
- Farhaoui, A.; Legrifi, I.; Taoussi, M.; Mokrini, F.; Tahiri, A.; Lahlali, R. Sclerotium rolfsii-Induced Damping off and Root Rot in Sugar Beet: Understanding the Biology, Pathogenesis, and Disease Management Strategies. Physiol. Mol. Plant Pathol. 2024, 134, 102456. [Google Scholar] [CrossRef]
- Huang, B.; Huang, J.; Guo, Z.; Zheng, J.; Liu, Q. First Report of Southern Blight Caused by Athelia rolfsii on Pepper in Yiyang, China. Plant Dis. 2023, 107, 3287. [Google Scholar] [CrossRef]
- Le, C.N.; Mendes, R.; Kruijt, M.; Raaijmakers, J.M. Genetic and Phenotypic Diversity of Sclerotium rolfsii in Groundnut Fields in Central Vietnam. Plant Dis. 2012, 96, 389–397. [Google Scholar] [CrossRef]
- Xie, C.; Huang, C.H.; Vallad, G.E. Mycelial Compatibility and Pathogenic Diversity among Sclerotium rolfsii Isolates in the Southern United States. Plant Dis. 2014, 98, 1685–1694. [Google Scholar] [CrossRef]
- Paul, S.K.; Mahmud, N.U.; Gupta, D.R.; Surovy, M.Z.; Rahman, M.; Islam, M.T. Characterization of Sclerotium rolfsii Causing Root Rot of Sugar Beet in Bangladesh. Sugar Tech 2021, 23, 1199–1205. [Google Scholar] [CrossRef]
- Okabe, I.; Chiharu, M.; Naoyuki, M.; Kazunari, Y. Variation in Sclerotium rolfsii Isolates in Japan. Mycoscience 1998, 39, 399–407. [Google Scholar] [CrossRef]
- Cepero de García, M.C.; Restrepo, S.; Franco-Molano, A.E.; Vargas, N. Biología de Hongos; Ediciones Uniandes-Universidad de los Andes: Bogotá, Colombia, 2012; ISBN 978-958-695-701-4. [Google Scholar]
- Sun, S.; Sun, F.; Deng, D.; Zhu, X.; Duan, C.; Zhu, Z. First Report of Southern Blight of Mung Bean Caused by Sclerotium rolfsii in China. Crop Prot. 2020, 130, 105055. [Google Scholar] [CrossRef]
- Jamja, T.; Bora, S.; Tabing, R.; Basar, E.; Kotoky, U. Incidence, Symptomology, Morpho-Molecular Characterization of Agroathelia rolfsii (Sclerotium rolfsii Sacc.): The Causal Agent of Basal Rot in Chabaud Carnation. Indian J. Agric. Res. 2025, 1–7. [Google Scholar] [CrossRef]
- Mullen, J. Southern Blight, Southern Stem Blight, White Mold. Plant Health Instr. 2001, 1, 1–18. [Google Scholar] [CrossRef]
- Mordue, J.E.M. Corticium rolfsii. [Descriptions of Fungi and Bacteria]; CABI: Wallingford, UK, 1974; p. Sheet 410. [Google Scholar] [CrossRef]
- Mahadevakumar, S.; Savitha, A.S.; Danteswari, C.; Sarma, P.V.S.R.N.; Renuka, M.; Ajithkumar, K.; Mahesh, M.; Sampath Kumar, P.T.S. Morpho-Molecular Characterization of Agroathelia rolfsii Causing Leaf Blight of Sacred Lotus (Nelumbo nucifera) in the Andaman and Nicobar Islands. Physiol. Mol. Plant Pathol. 2025, 140, 102899. [Google Scholar] [CrossRef]
- Punja, Z.K.; Sun, L.J. Genetic Diversity among Mycelial Compatibility Groups of Sclerotium rolfsii (Teleomorph Athelia rolfsii) and S. delphinii. Mycol. Res. 2001, 105, 537–546. [Google Scholar] [CrossRef]
- Cilliers, A.J.; Herselman, L.; Pretorius, Z.A. Genetic Variability within and among Mycelial Compatibility Groups of Sclerotium rolfsii in South Africa. Phytopathology 2000, 90, 1026–1031. [Google Scholar] [CrossRef]
- Cilliers, A.J.; Pretorius, Z.A.; Van Wyk, P.S. Mycelial Compatibility Groups of Sclerotium rolfsii in South Africa. S. Afr. J. Bot. 2002, 68, 389–392. [Google Scholar] [CrossRef]
- Adandonon, A.; Aveling, T.A.S.; Van Der Merwe, N.A.; Sanders, G. Genetic Variation among Sclerotium Isolates from Benin and South Africa, Determined Using Mycelial Compatibility and ITS RDNA Sequence Data. Australas. Plant Pathol. 2005, 34, 19–25. [Google Scholar] [CrossRef]
- Nalim, F.A.; Starr, J.L.; Woodard, K.E.; Segner, S.; Keller, N.P. Mycelial Compatibility Groups in Texas Peanut Field Populations of Sclerotium rolfsii. Phytopathology 1995, 85, 1507–1512. [Google Scholar] [CrossRef]
- Harlton, C.E.; Levesque, C.A.; Punja, Z.K. Genetic Diversity in Sclerotium (Athelia) rolfsii and Related Species. Phytopathology 1995, 85, 1269–1281. [Google Scholar] [CrossRef]
- Tu, C.C.; Kimbrough, J.W. Systematics and Phylogeny of Fungi in the Rhizoctonia Complex. Bot. Gaz. 1978, 139, 454–466. [Google Scholar] [CrossRef]
- Mascarenhas, J.; Quesada-Ocampo, L.M. Diagnostic Guide for Sclerotial Blight and Circular Spot of Sweetpotato. Plant. Health. Prog. 2024, 25, 340–344. [Google Scholar] [CrossRef]
- Xu, Z.; Harrington, T.C.; Gleason, M.L.; Batzer, J.C. Phylogenetic Placement of Plant Pathogenic Sclerotium Species among Teleomorph Genera. Mycologia 2010, 102, 337–346. [Google Scholar] [CrossRef]
- Mahadevakumar, S.; Janardhana, G.R. Morphological and Molecular Characterization of Sclerotium rolfsii Associated with Leaf Blight Disease of Psychotria nervosa (Wild Coffee). J. Plant Pathol. 2016, 98, 351–354. Available online: https://www.researchgate.net/publication/301589890_Morphological_and_molecular_characterization_of_Sclerotium_rolfsii_associated_with_leaf_blight_disease_of_Psychotria_nervosa_Wild_coffee (accessed on 16 December 2025).
- Okabe, I.; Arakawa, M.; Matsumoto, N. ITS Polymorphism within a Single Strain of Sclerotium rolfsii. Mycoscience 2001, 42, 107–113. [Google Scholar] [CrossRef]
- Song, J.; Qiu, H.; Meng, X.; Li, S.; Mao, Y.; Zhang, L.; Cai, Y.; Wang, J.; Zhou, M.; Duan, Y. Risk Assessment and Molecular Mechanism of Sclerotium rolfsii Resistance to Boscalid. Pestic. Biochem. Physiol. 2024, 200, 105806. [Google Scholar] [CrossRef] [PubMed]
- Tejaswini, G.S.; Mahadevakumar, S.; Sowmya, R.; Deepika, Y.S.; Meghavarshinigowda, B.R.; Nuthan, B.R.; Sharvani, K.A.; Amruthesh, K.N.; Sridhar, K.R. Molecular Detection and Pathological Investigations on Southern Blight Disease Caused by Sclerotium rolfsii on Cabbage (Brassica oleracea var. capitata): A New Record in India. J. Phytopathol. 2022, 170, 363–372. [Google Scholar] [CrossRef]
- Rahman, M.M.E.; Dey, T.K.; Hossain, D.M.; Nonaka, M.; Harada, N. First Report of White Mould Caused by Sclerotinia sclerotiorum on Jackfruit. Australas. Plant Dis. Notes 2015, 10, 10. [Google Scholar] [CrossRef][Green Version]
- Sennoi, R.; Jogloy, S.; Saksirirat, W.; Patanothai, A. Pathogenicity Test of Sclerotium rolfsii, a Causal Agent of Jerusalem Artichoke (Helianthus tuberosus L.) Stem Rot. Asian J. Plant Sci. 2010, 9, 281–284. [Google Scholar] [CrossRef]
- Shivalingappa, H.; Ashtaputre, S.A.; Jahagirdar, S.; Rao, M.S.L.; Motagi, B.N.; Jones, P.; Mahesha, H.S.; Raghunandhan, A.; Shivakumara, K.T. Deciphering the Morphological and Pathogenic Variability among Sclerotium rolfsii: A Causal Agent of Groundnut Stem Rot Disease. Phytoparasitica 2025, 53, 84. [Google Scholar] [CrossRef]
- Chaudhary, S.; Lal, M.; Sagar, S.; Sharma, S.; Meena, A.L.; Singh, R.K. Variability in Morphology, Oxalic Acid Production and Mycelial Compatibility Grouping Among Sclerotium rolfsii Isolates Causing Southern Blight Disease. Plant Pathol. 2025, 74, 2272–2288. [Google Scholar] [CrossRef]





| Location | Strain | Taxon | GenBank Accession Numbers | ||
|---|---|---|---|---|---|
| X Coordinate | Y Coordinate | ITS | TEF | ||
| 7.64100 | −76.7343 | CPSrZN02 | Agroathelia rolfsii | ON524853 | PX658743 |
| 7.64270 | −76.7343 | CPSrZN03 | A. rolfsii | ON524854 | |
| 7.63710 | −76.7383 | CPSrZN04 | A. rolfsii | ON524855 | PX658744 |
| 7.75690 | −76.6676 | CPSrZN05 | A. rolfsii | ON524856 | PX658745 |
| 7.56200 | −76.6401 | CPSrZN06 | A. rolfsii | ON524857 | PX658746 |
| 7.55304 | −76.6459 | CPSrZN07 | A. rolfsii | ON524858 | |
| 7.62000 | −76.6978 | CPSrZN08 | A. rolfsii | ON524859 | PX658747 |
| 7.56240 | −76.6409 | CPSrZN09 | A. rolfsii | PX658748 | |
| 10.73040 | −74.1297 | CPSrZN10 | A. rolfsii | PX635264 | PX658749 |
| 10.53947 | −74,18716 | CPSrZN11 | A. rolfsii | PX635265 | PX658750 |
| 10.53970 | −74.1871 | CPSrZN12 | A. rolfsii | PX658751 | |
| 10.53920 | −74.1875 | CPSrZN13 | A. rolfsii | PX635266 | PX658752 |
| 10.54150 | −74.1956 | CPSrZN14 | A. rolfsii | PX635267 | PX658753 |
| 10.53980 | −74.1869 | CPSrZN15 | A. rolfsii | PX635268 | PX658754 |
| 10.53970 | −74.1871 | CPSrZN16 | A. rolfsii | PX635269 | PX658755 |
| 10.54008 | −74.18745 | CPSrZN17 | A. rolfsii | PX635270 | PX658756 |
| 10.54000 | −74.1871 | CPSrZN19 | A. rolfsii | PX658757 | |
| 10.53743 | −74.18727 | CPSrZN20 | A. rolfsii | PX635271 | PX658758 |
| 10.53609 | −74.18736 | CPSrZN21 | A. rolfsii | PX635272 | PX658766 |
| 10.54000 | −74.1871 | CPSrZN22 | A. rolfsii | PX658759 | |
| 10.53440 | −74.1873 | CPSrZN23 | A. rolfsii | PX658760 | |
| 10.53514 | −74.18844 | CPSrZN24 | A. rolfsii | PX658761 | |
| 10.53477 | −74.18845 | CPSrZN25 | A. rolfsii | PX635273 | PX658762 |
| 10.73060 | −74.1315 | CPSrZN26 | A. rolfsii | PX635274 | PX658763 |
| 1.591944 | −78.663611 | CPSrZOC01 | A. rolfsii | PX658764 | |
| 1.548478 | −78.716944 | CPSrZOC02 | A. rolfsii | PX635275 | PX658765 |
| 1.415833 | −78.741111 | CPSrZOC03 | A. rolfsii | PX635276 | |
| 6.972972 | −73.713222 | CPSrZC01 | A. rolfsii | PX658740 | |
| 7.297850 | −73.884240 | CPSrZC02 | A. rolfsii | PX658741 | |
| 7.382028 | −73.895194 | CPSrZC03 | A. rolfsii | PX635256 | PX658742 |
| 6.972972 | −73.713222 | CPRZC01 | Rhizoctonia sp. | ||
| 6.972972 | −73.713222 | CPFuZC01 | Fusarium sp. | ||
| 6.972972 | −73.713222 | CPPesZC01 | Pestalotiopsis sp. | ||
| Locality | Isolate | Number of Sclerotia | Average Sclerotia Diameter (±SD, Min–Max) | Growth Rate (mm/Day ± SD) |
|---|---|---|---|---|
| Barrancabermeja | CPSrZC01 | 53 | 1.01 ± 0.15 (0.76–1.26) | 19.3 ± 0.62 |
| Puerto Wilches | CPSrZC02 | 71 | 1.04 ± 0.13 (0.82–1.37) | 19.0 ± 0.26 |
| Puerto Wilches | CPSrZC03 | 192 | 1.11 ± 0.25 (0.72–1.57) | 26.2 ± 0.52 |
| Tumaco | CPSrZOC01 | 72 | 1.12 ± 0.19 (0.96–2.00) | 24.4 ± 0.40 |
| Tumaco | CPSrZOC02 | 4 | 1.25 ± 0.12 (1.13–1.38) | 22.1 ± 0.48 |
| Tumaco | CPSrZOC03 | 24 | 1.45 ± 0.37 (1.00–2.20) | 31.4 ± 0.84 |
| Chigorodó | CPSrZN02 | 104 | 0.81 ± 0.11 (0.57–0.99) | 23.7 ± 0.55 |
| Chigorodó | CPSrZN04 | 193 | 0.82 ± 0.10 (0.54–0.99) | 22.7 ± 0.41 |
| Chigorodó | CPSrZN06 | 49 | 1.11 ± 0.21 (0.62–1.53) | 20.7 ± 0.85 |
| Zona Bananera | CPSrZN10 | 122 | 1.00 ± 0.14 (0.73–1.27) | 20.6 ± 0.35 |
| Aracataca | CPSrZN15 | 425 | 0.88 ± 0.12 (0.61–1.12) | 22.5 ± 0.36 |
| Aracataca | CPSrZN16 | 449 | 0.74 ± 0.19 (0.46–1.28) | 24.7 ± 0.71 |
| Isolate | Strain | p_Ajust | Sig 1 | Isolate | Strain | p_Ajust | Sig |
|---|---|---|---|---|---|---|---|
| T1 | CPSrZN02 | 0.016 | * | T24 | CPSrZn26 | 0.0203 | * |
| T10 | CPSrZN11 | 0.0242 | * | T25 | CPSrZOC01 | 0.0103 | * |
| T11 | CPSrZN12 | 0.00528 | ** | T26 | CPSrZOC02 | 0.261 | Ns |
| T12 | CPSrZN13 | 0.00914 | ** | T27 | CPSrZOC03 | 0.144 | Ns |
| T13 | CPSrZN14 | 0.00914 | ** | T28 | CPSrZC01 | 0.00528 | ** |
| T14 | CPSrZN15 | 0.00528 | ** | T29 | CPSrZC02 | 0.00528 | ** |
| T15 | CPSrZN16 | 0.00528 | ** | T3 | CPSrZN04 | 0.00654 | ** |
| T16 | CPSrZN17 | 0.0877 | Ns | T30 | CPRZC01 | 0.767 | Ns |
| T17 | CPSrZN19 | 0.0701 | Ns | T31 | CPFuZC01 | 0.977 | Ns |
| T18 | CPSrZN20 | 0.11 | Ns | T32 | CPPesZC01 | 1 | Ns |
| T19 | CPSrZN21 | 0.00528 | ** | T4 | CPSrZN05 | 0.00628 | ** |
| T2 | CPSrZN03 | 0.338 | Ns | T5 | CPSrZN06 | 0.00103 | ** |
| T20 | CPSrZN22 | 0.00528 | ** | T6 | CPSrZN07 | 0.024 | * |
| T21 | CPSrZN23 | 0.00103 | ** | T7 | CPSrZN08 | 0.0933 | Ns |
| T22 | CPSrZN24 | 0.00654 | ** | T8 | CPSrZN09 | 0.0143 | * |
| T23 | CPSrZN25 | 0.00528 | ** | T9 | CPSrZN10 | 0.0298 | * |
| CPSr ZN02 | CPSr ZN04 | CPSr ZN06 | CPSr ZN10 | CPSr ZN15 | CPSr ZN16 | CPSr ZC01 | CPSr ZC02 | CPSr ZC03 | CPSr ZOC01 | CPSr ZOC02 | CPSr ZOC03 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CPSr ZN02 | 0 | |||||||||||
| CPSr ZN04 | 0 | 0 | ||||||||||
| CPSr ZN06 | 2 | 2 | 0 | |||||||||
| CPSr ZN10 | 2 | 2 | 0 | 0 | ||||||||
| CPSr ZN15 | 2 | 3 | 2 | 2 | 0 | |||||||
| CPSr ZN16 | 2 | 3 | 3 | 2 | 0 | 0 | ||||||
| CPSr ZC01 | 3 | 3 | 3 | 3 | 3 | 3 | 0 | |||||
| CPSr ZC02 | 3 | 3 | 3 | 2 | 3 | 3 | 3 | 0 | ||||
| CPSr ZC03 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 0 | |||
| CPSr ZOC01 | 3 | 3 | 3 | 3 | 3 | 2 | 3 | 3 | 3 | 0 | ||
| CPSr ZOC02 | 3 | 3 | 2 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 0 | |
| CPSr ZOC03 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 1 | 0 | 0 |
| Species | Isolates | Host | Country | Genbank Accession Number | |
|---|---|---|---|---|---|
| ITS | TEF1-α | ||||
| Agroathelia delphinii | CBS720.83 | Unknown | Unknown | JN241573.1 | |
| CBS672.71 | Unknown | Unknown | JN241578.1 | ||
| CBS272.30 | Unknown | Unknown | MH855140.1 | ||
| KACC:48426 | Malus domestica | South Korea | PQ408027.1 | ||
| Agroathelia rolfsii | SrBoC26 | Brassica oleracea | India | MK936163.1 | |
| CN36 | Hordeum vulgare | Brazil | KJ578736.1 | ||
| CHI-2 | Unknown | India | MN121187.1 | ||
| CPC:23947 | Musa sp. | Philippines | MK411221.1 | ||
| SPL15001 | Ipomoea batatas | South Korea | KY446394.1 | ||
| KACCC48132 | Oxalis purpurea | South Korea | MH571744.1 | ||
| MSB1-1 | Vigna radiata | China | MN609999.1 | ||
| AR Nn ANI | Unknown | India | PV089824.1 | ||
| MSB1-2 | Vigna radiata | China | MN610000.1 | ||
| CBS132553 | Vigna unguiculata | Laos | JX566993.1 | ||
| SrGg10 | Unknown | India | MT308986.1 | ||
| CBS115.22 | Unknown | USA | MH854711.1 | ||
| SPL15001 | Ipomoea batatas | South Korea | KY446394.1 | ||
| Sr_203 | Unknown | Spain | JF267802.1 | ||
| PE1-1-2 | Oryza sativa | China | PV498539.1 | ||
| CBS745.84 | Unknown | Unknown | GU187681.1 | ||
| Sr_2 | Unknown | Chile | JF267806.1 | ||
| BQ1 | Cynanchum stauntonii | China | MT052174.1 | ||
| KACC:48131 | Oxalis triangularis | South Korea | PQ408006.1 | ||
| ARNn ANI18 | Nelumbo nucifera | India | PV446748.1 | ||
| ARNn ANI19 | Nelumbo nucifera | India | PV446749.1 | ||
| KACC:42085 | Disporum smilacinum | South Korea | PQ408015.1 | ||
| KACC:48273 | Peucedanum japonicum | South Korea | PQ408026.1 | ||
| Sr_414 | Unknown | Portugal | JF267813.1 | ||
| Sr_22 | Unknown | Chile | JF267807.1 | ||
| SR7 | Stevia rebaudiana | Italy | KP982854.1 | ||
| MC-2-1 | OP902296.1 | ||||
| Ssr | OP120960.1 | ||||
| MNG-1 | Baccaurea ramiflora | China | OM049770.1 | ||
| S. cacticola | CBS304.32 | Unknown | Netherlands | MH855330.1 | |
| S. cepivorum | KACC:47821 | Allium hookeri | South Korea | KP257580 | |
| S. coffeicola | Coffea sp. | Suriname | AB075319.1 | ||
| S. costantinii | CBS288.38 | Unknown | France | MH855965.1 | |
| S. glucanicum | CBS520.71 | Unknown | USA | MH860245 | |
| S. perniciosum | CBS275.93 | Unknown | Unknown | FJ231408.1 | |
| CBS286.30 | Unknown | Netherlands | MH855138.1 | ||
| Rhizoctonia endophytica | CBS257.60 | Unknown | Unknown | MH857976.1 | |
| Rhizoctonia solani | JZB-20 | Solanum tuberosum | China | KJ866444.1 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Angel-Salazar, L.d.M.; Zuñiga-Perez, L.F.; Mestizo-Garzón, Y.A.; Ortega-Soto, C.S.; Garcia-Ruiz, D.A.; Medina-Cárdenas, H.C.; Padilla, J.L.; Vargas-Medina, L.E.; Morales-Rodríguez, A.; Sarria, G.A. First Report of Agroathelia rolfsii Causing White Fruit Rot in Oil Palm Hybrid OxG in Colombia. J. Fungi 2026, 12, 31. https://doi.org/10.3390/jof12010031
Angel-Salazar LdM, Zuñiga-Perez LF, Mestizo-Garzón YA, Ortega-Soto CS, Garcia-Ruiz DA, Medina-Cárdenas HC, Padilla JL, Vargas-Medina LE, Morales-Rodríguez A, Sarria GA. First Report of Agroathelia rolfsii Causing White Fruit Rot in Oil Palm Hybrid OxG in Colombia. Journal of Fungi. 2026; 12(1):31. https://doi.org/10.3390/jof12010031
Chicago/Turabian StyleAngel-Salazar, Lina del Mar, Leon Franky Zuñiga-Perez, Yuri Adriana Mestizo-Garzón, Cristian Steven Ortega-Soto, Daniela Alejandra Garcia-Ruiz, Hector Camilo Medina-Cárdenas, Jose Luis Padilla, Liseth Estefanía Vargas-Medina, Anuar Morales-Rodríguez, and Greicy Andrea Sarria. 2026. "First Report of Agroathelia rolfsii Causing White Fruit Rot in Oil Palm Hybrid OxG in Colombia" Journal of Fungi 12, no. 1: 31. https://doi.org/10.3390/jof12010031
APA StyleAngel-Salazar, L. d. M., Zuñiga-Perez, L. F., Mestizo-Garzón, Y. A., Ortega-Soto, C. S., Garcia-Ruiz, D. A., Medina-Cárdenas, H. C., Padilla, J. L., Vargas-Medina, L. E., Morales-Rodríguez, A., & Sarria, G. A. (2026). First Report of Agroathelia rolfsii Causing White Fruit Rot in Oil Palm Hybrid OxG in Colombia. Journal of Fungi, 12(1), 31. https://doi.org/10.3390/jof12010031







