Analysis of the Effects of Beauveria bassiana Appressorium Formation on Insect Cuticle Metabolism Based on LC-MS
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Strains and Insects
2.2. Regulation of the Proportion of Appressorium Formation in Beauveria bassiana
2.3. Determination of the Pathogenicity of Different Appressorium Formation Ratios of Beauveria bassiana to Insects
2.4. Effects of Beauveria bassiana Appressorium Formation on Insect Cuticle Metabolism
2.4.1. Appressorium Formation Rate of Beauveria bassiana on Larval Cuticles
2.4.2. Sample Processing
2.4.3. LC-MS Analysis
2.5. Data Processing
3. Results
3.1. Germination and Appressorium Formation Rates of B. bassiana at Different Sulforaphane Concentrations
3.2. Correlation Analysis Between Beauveria bassiana Appressorium Formation Rate and Pathogenicity
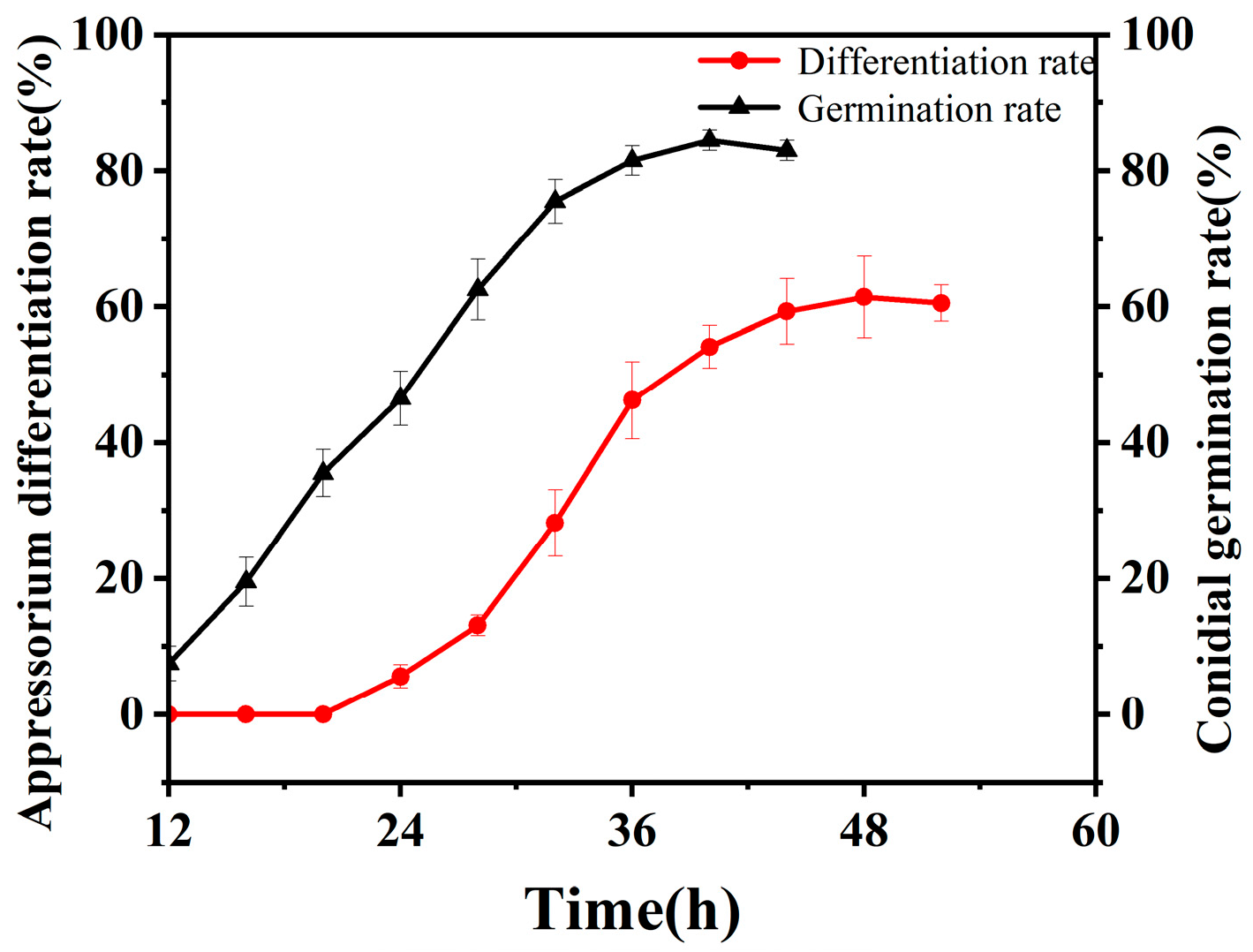
3.3. Dynamics of Appressorium Formation by B. bassiana on the Cuticle of O. arenosella
3.4. Effects of B. bassiana Infection on Differences in the Cuticle Compounds of the Larvae of the O. arenosella
3.4.1. Orthogonal Partial Least Squares Discriminant Analysis (OPLS-DA)
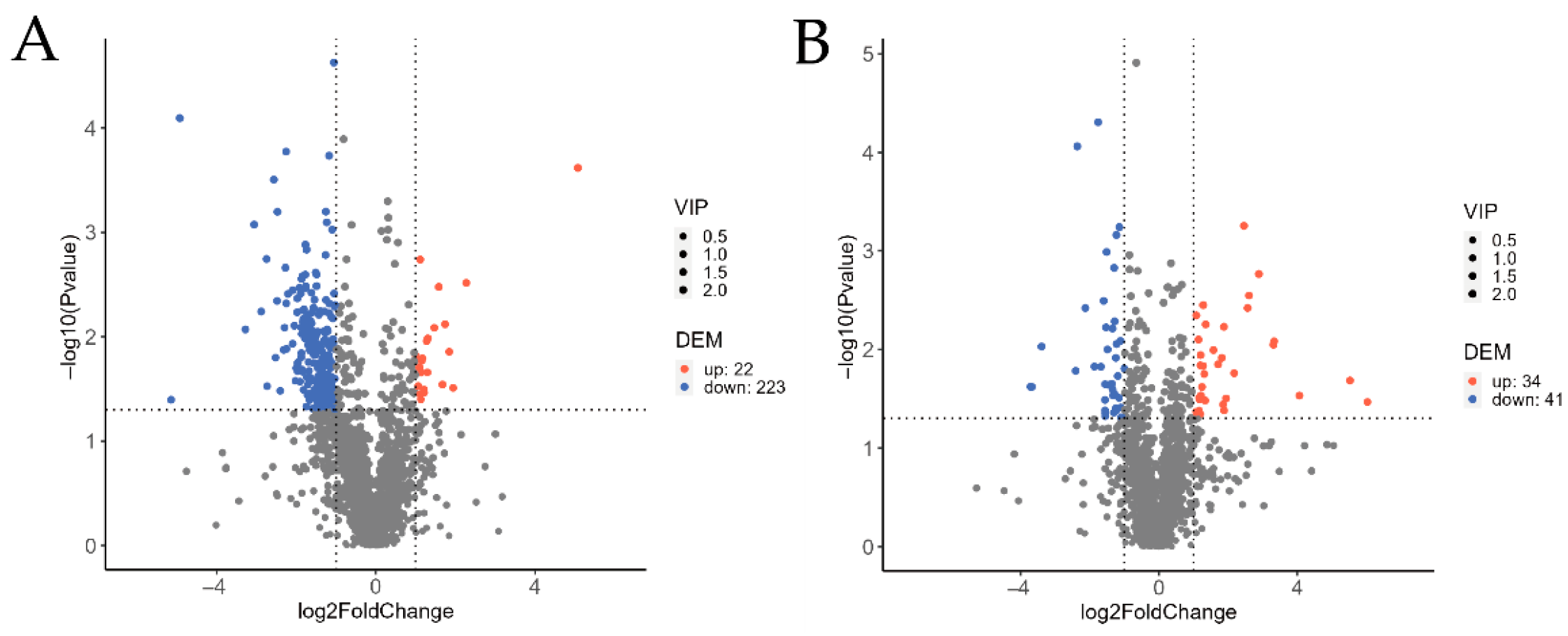
3.4.2. Volcano Plot Analysis
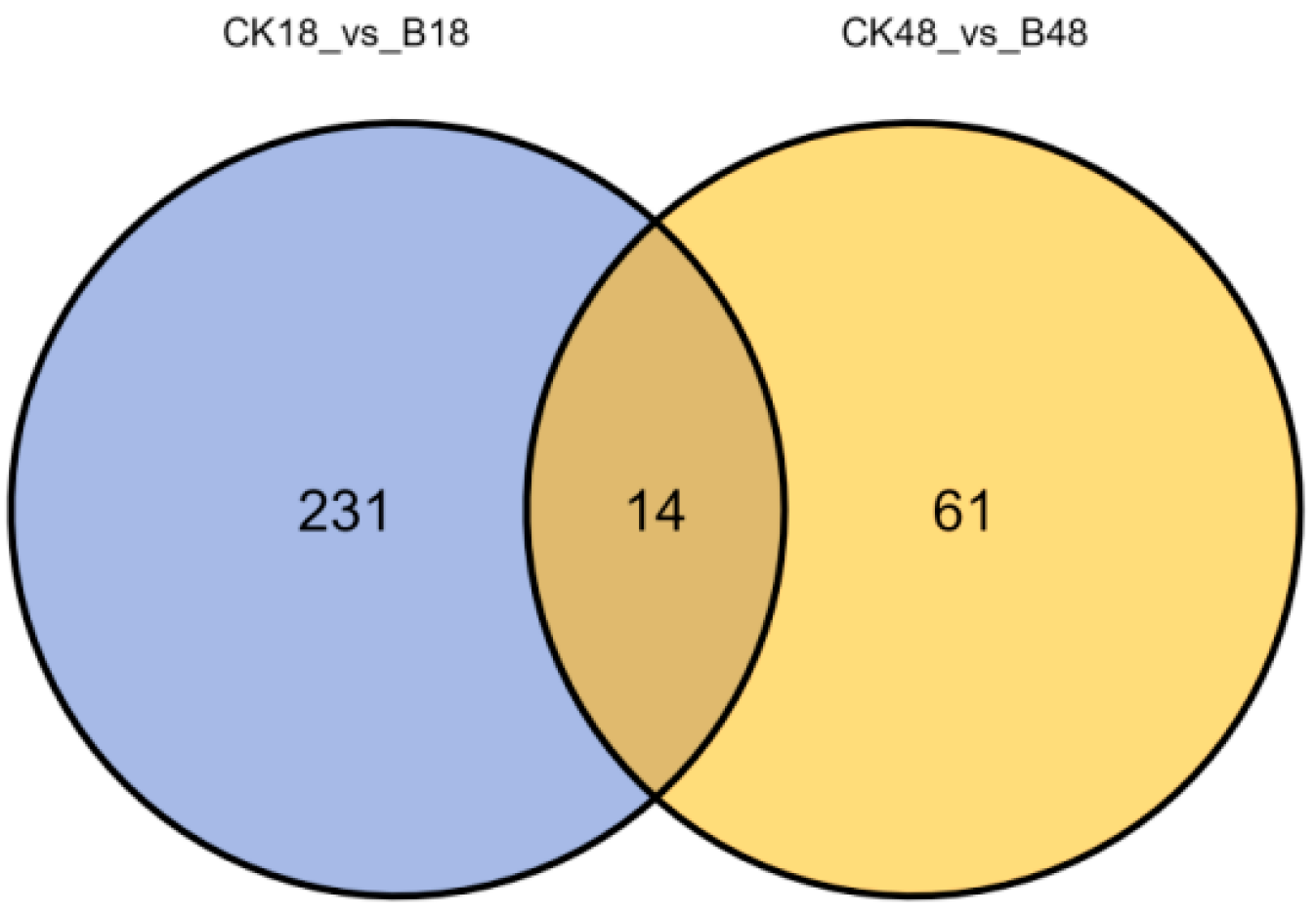
3.4.3. Differential Compounds Unique to the Larval Epidermis After the Formation of B. bassiana Appressorium
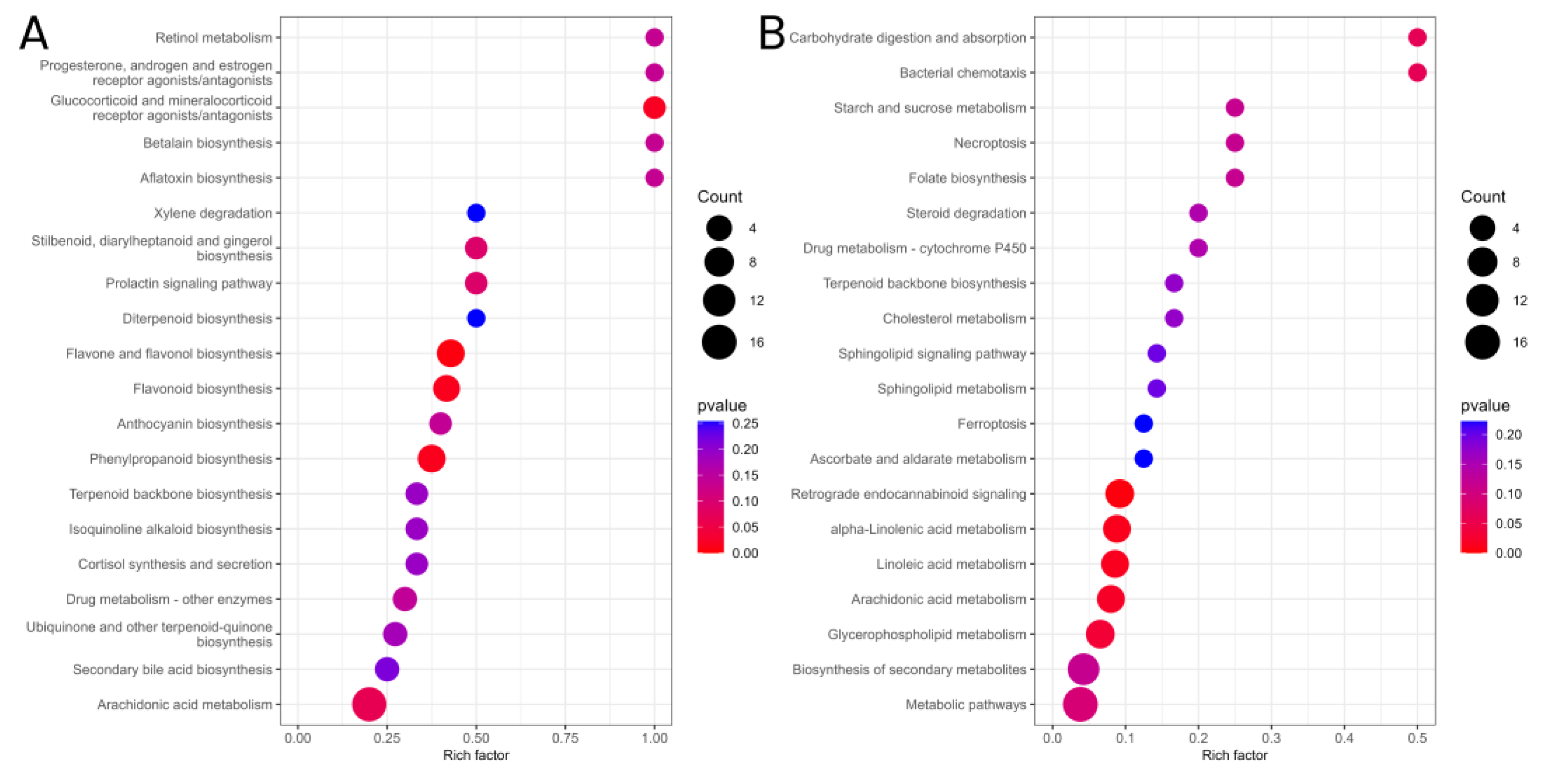
3.4.4. KEGG Enrichment Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zimmermann, G. Review on safety of the entomopathogenic fungi Beauveria bassiana and Beauveria brongniartii. Biocontrol Sci. Technol. 2007, 17, 553–596. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Chen, J.; Huang, J.; Zheng, L. Screening for highly virulent Beauveria bassiana Walker strains against Dendrolimus punctatus. J. South. Agric. 2016, 47, 662–666. [Google Scholar]
- Zhang, L.-W.; Kang, K.; Liu, Y.-J.; Zhang, T.; Sun, L.; Zhan, C.; Huang, C.-C.; Jiang, L.-Y.; Ye, K.-Y.; Ding, D.-G. Evaluation of Beauveria bassiana isolates as potential agents for control of Hyphantria cunea (Lepidoptera: Arctiidae). Acta Entomol. Sin. 2016, 59, 111–118. [Google Scholar]
- He, R.; Cui, X.; Ying, Y.; Qu, L.-J.; Wang, R.-Z.; Zhang, Y. Screening and identification of Beauveria bassiana strains for biocontrol of Monochamus alternatus adults (Coleoptera: Cerambycidae). Sci. Silvae Sin. 2020, 56, 129–134. [Google Scholar]
- He, X.-Y.; Cai, S.-P.; Tong, Y.-H.; Xiong, Y.; Huang, Y.; Xie, J.-D.; Chen, S.-L. Pathogenicity evaluation of the entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae against adults of Basilepta melanopus (Coleoptera: Eumolpidae). Acta Entomol. Sin. 2011, 54, 1281–1287. [Google Scholar]
- Xun, W.; Sui, L.; Gao, P.; Zhang, R.; Wang, Z.; Zhang, Z.; Li, Q. Study and Application of Wettable Powder of Beauveria bassiana to Control Corn Borer. Chin. J. Biol. Control. 2020, 36, 862–865. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, J.; Jiang, X.; Wang, G.; Luo, Z.; Fan, Y.; Wu, Z.; Pei, Y. Requirement of a mitogen-activated protein kinase for appressorium formation and penetration of insect cuticle by the entomopathogenic fungus Beauveria bassiana. Appl. Environ. Microbiol. 2010, 76, 2262–2270. [Google Scholar] [CrossRef]
- Chethana, K.T.; Jayawardena, R.S.; Chen, Y.-J.; Konta, S.; Tibpromma, S.; Phukhamsakda, C.; Abeywickrama, P.D.; Samarakoon, M.C.; Senwanna, C.; Mapook, A. Appressorial interactions with host and their evolution. Fungal Divers. 2021, 110, 75–107. [Google Scholar] [CrossRef]
- Luo, F.; Li, S.; Chen, L.; Zhang, W.; Wang, B.; Huang, B.; Hu, F. Identification of spore germination and virulence related biomarkers from Beauveria bassiana using an LC-MS-based metabolomic technique. Acta Microbiol. Sin. 2014, 54, 33–41. [Google Scholar]
- Fan, Y.; Cao, Z.; Gu, S.; Dong, J. Effect of different induction factors on appressorium of Setosphaeria turcica. Sci. Agric. Sin. 2004, 37, 769–772. [Google Scholar]
- Wang, H.; Lin, F.; Wang, Z. Mechanical penetrating force of appressoria of plant pathogenic fungi. Mycosystema 2004, 23, 151–157. [Google Scholar]
- Kleemann, J.; Takahara, H.; Stüber, K.; O’Connell, R. Identification of soluble secreted proteins from appressoria of Colletotrichum higginsianum by analysis of expressed sequence tags. Microbiology 2008, 154, 1204–1217. [Google Scholar] [CrossRef][Green Version]
- Kim, Y.-K.; Kawano, T.; Li, D.; Kolattukudy, P.E. A mitogen-activated protein kinase kinase required for induction of cytokinesis and appressorium formation by host signals in the conidia of Colletotrichum gloeosporioides. Plant Cell 2000, 12, 1331–1343. [Google Scholar] [CrossRef] [PubMed]
- Ni, D. Preliminary Study on Chemical Interaction Between Secondary Metabolites and Insect Body Wall Compounds During Infection of Beauveria bassiana; Fujian Agriculture and Forestry University: Fuzhou, China, 2024. [Google Scholar]
- Li, Y.; Dang, Z.; Gao, Z.; Li, Y.; Xu, G.; Ma, Y.; Pan, W. Laboratory screening of highly-toxic insecticides for controlling Athetis lepigone Möschler. Agrochemicals 2012, 51, 213–214. [Google Scholar]
- Zhao, X.; Wang, J.; Shao, H. An Insect Epidermal Cell Line and Its Construction Method and Application. CN101182487A, 14 November 2007. [Google Scholar]
- Fan, M.; Bo, H.; Wang, J.; Bin, W. Fluorescence and scanning electron microscopy study on appresorium morphology of some entomogenous fungi. Mycosystema 1999, 18, 249–253. [Google Scholar]
- Cao, Y.; Zhu, X. Test and Determination of Entomopathogenic Fungal Spore and Appressorium on Its Host with Calcofluor White M2R. J. Microbiol. 2010, 30, 91–92. [Google Scholar]
- Want, E.J.; Masson, P.; Michopoulos, F.; Wilson, I.D.; Theodoridis, G.; Plumb, R.S.; Shockcor, J.; Loftus, N.; Holmes, E.; Nicholson, J.K. Global metabolic profiling of animal and human tissues via UPLC-MS. Nat. Protoc. 2013, 8, 17–32. [Google Scholar] [CrossRef] [PubMed]
- Boulesteix, A.-L.; Strimmer, K. Partial least squares: A versatile tool for the analysis of high-dimensional genomic data. Brief. Bioinform. 2007, 8, 32–44. [Google Scholar] [CrossRef]
- Fang, X.; Zeng, X.; Wang, M.; Qin, L.; Tan, C.; Wu, J. Enrichment analysis of differentially expressed genes in chronic heart failure. Ann. Palliat. Med. 2021, 10, 9049–9056. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Rao, J.; Cheng, F.; Hu, C.; Quan, S.; Lin, H.; Wang, J.; Chen, G.; Zhao, X.; Alexander, D.; Guo, L. Metabolic map of mature maize kernels. Metabolomics 2014, 10, 775–787. [Google Scholar] [CrossRef]
- Jia, S.; Wang, Y.; Hu, J.; Ding, Z.; Liang, Q.; Zhang, Y.; Wang, H. Mineral and metabolic profiles in tea leaves and flowers during flower development. Plant Physiol. Biochem. 2016, 106, 316–326. [Google Scholar] [CrossRef]
- Lin, H.; Rao, J.; Shi, J.; Hu, C.; Cheng, F.; Wilson, Z.A.; Zhang, D.; Quan, S. Seed metabolomic study reveals significant metabolite variations and correlations among different soybean cultivars. J. Integr. Plant Biol. 2014, 56, 826–836. [Google Scholar] [CrossRef] [PubMed]
- Li, W. Physical and Chemiscal Factors Effecting Appressorium Formination of Metarhizium anisopliae and Beauveria bassiana. Master’s Thesis, Southwest Agriculture University, Chongqing, China, 2003. [Google Scholar]
- Li, W.; Wang, Z.; Yin, Y.; Xia, Y.; Peng, G. Effection of physical and chemical factors on appressorium formation of Beauveria bassiana. J. Chongqing Univ. (Nat. Sci. Ed.) 2004, 27, 102–106. [Google Scholar]
- He, Y.; Li, X.; Meng, Q.; Tao, B.; Wang, S.; Jia, H.; Cao, Z. Mechanism of Tricyclazole Effect on Setosphaeria turcica Pathogenicity. J. Maize Sci. 2015, 23, 149–153. [Google Scholar]
- Duan, Z.B.; Xia, Y.L.; Shang, Y.F.; Huang, W.; Wang, C.S. Appressorial differentiation, virulence and their association with phosphorylation site mutation of MPL1 in the insect pathogenic fungus Metarhizium anisopliae. In Proceedings of the Abstracts of the Annual Meeting 2009 Mycological Society of China, Hohhot, China, 15–18 August 2009. [Google Scholar]
- Meng, S. Mechanisms of MoWhi2 Involved in Appressorium Formation and Mitophagy in Regulaion of the Pathogenicity of Magnaporthe oryzae; Huazhong Agricultureal University: Wuhan, China, 2022. [Google Scholar]
- Hu, J.; Yang, B.; Zhao, J.; Zhang, H.; Chen, W. Effect of Lactobacillus gasseri with Different Bacteriocins Production on the Intestinal Micro-Ecology in Mice. J. Food Sci. Biotechnol. 2024, 43, 1–10. [Google Scholar]
- Nappi, J.; Goncalves, P.; Khan, T.; Majzoub, M.E.; Grobler, A.S.; Marzinelli, E.M.; Thomas, T.; Egan, S. Differential priority effects impact taxonomy and functionality of host-associated microbiomes. Mol. Ecol. 2023, 32, 6278–6293. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, X.; Fan, Y.; Huo, P.; Huang, S.; Wang, H.; Lu, Z.; Luo, Z.; Zhang, Y. Transcriptome analysis of Beauveria bassiana interaction with Nicotiana benthamiana reveals signatures of N. benthamiana growth promotion and enhanced defense responses. J. Invertebr. Pathol. 2025, 211, 108334, Erratum in J. Invertebr. Pathol. 2025, 211, 108347. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.-J.; Mou, Y.-N.; Tong, S.-M.; Ying, S.-H.; Feng, M.-G. Subtilisin-like Pr1 proteases marking the evolution of pathogenicity in a wide-spectrum insect-pathogenic fungus. Virulence 2020, 11, 365–380. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Typas, M.A.; Butt, T.M. Detection and characterisation of pr1 virulent gene deficiencies in the insect pathogenic fungus Metarhizium anisopliae. FEMS Microbiol. Lett. 2002, 213, 251–255. [Google Scholar] [CrossRef]
- Zhang, Y.-Y.; Cheng, H.; Wang, J.-E.; Wu, Z.-Y.; Pei, R.-J. Engineering of thiamine pyrophosphate fluorescent biosensors based on ribozyme switches in mammalian cells. Chin. J. Anal. Chem. 2017, 45, 157–162. [Google Scholar] [CrossRef]
- Gutiérrez Acosta, O.B.; Hardt, N.; Hacker, S.M.; Strittmatter, T.; Schink, B.; Marx, A. Thiamine pyrophosphate stimulates acetone activation by Desulfococcus biacutus as monitored by a fluorogenic ATP analogue. ACS Chem. Biol. 2014, 9, 1263–1266. [Google Scholar] [CrossRef]
- Wang, C.; Leger, R.J.S. The Metarhizium anisopliae perilipin homolog MPL1 regulates lipid metabolism, appressorial turgor pressure, and virulence. J. Biol. Chem. 2007, 282, 21110–21115. [Google Scholar] [CrossRef] [PubMed]
- Ryder, L.S.; Dagdas, Y.F.; Mentlak, T.A.; Kershaw, M.J.; Thornton, C.R.; Schuster, M.; Chen, J.; Wang, Z.; Talbot, N.J. NADPH oxidases regulate septin-mediated cytoskeletal remodeling during plant infection by the rice blast fungus. Proc. Natl. Acad. Sci. USA 2013, 110, 3179–3184. [Google Scholar] [CrossRef] [PubMed]
- Colzi, I.; Troyan, A.N.; Perito, B.; Casalone, E.; Romoli, R.; Pieraccini, G.; Škalko-Basnet, N.; Adessi, A.; Rossi, F.; Gonnelli, C. Antibiotic delivery by liposomes from prokaryotic microorganisms: Similia cum similis works better. Eur. J. Pharm. Biopharm. 2015, 94, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Perera, P.G.T.; Todorova, N.; Vilagosh, Z.; Bazaka, O.; Bazaka, K.; Crawford, R.J.; Croft, R.J.; Yarovsky, I.; Ivanova, E.P. Translocation of silica nanospheres through giant unilamellar vesicles (GUVs) induced by a high frequency electromagnetic field. RSC Adv. 2021, 11, 31408–31420. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.; Li, W.; Wu, C.; Wu, S.; Tong, Y. Analysis of the Effects of Beauveria bassiana Appressorium Formation on Insect Cuticle Metabolism Based on LC-MS. J. Fungi 2025, 11, 595. https://doi.org/10.3390/jof11080595
Chen J, Li W, Wu C, Wu S, Tong Y. Analysis of the Effects of Beauveria bassiana Appressorium Formation on Insect Cuticle Metabolism Based on LC-MS. Journal of Fungi. 2025; 11(8):595. https://doi.org/10.3390/jof11080595
Chicago/Turabian StyleChen, Jiarui, Wenzhe Li, Canxia Wu, Songqing Wu, and Yinghua Tong. 2025. "Analysis of the Effects of Beauveria bassiana Appressorium Formation on Insect Cuticle Metabolism Based on LC-MS" Journal of Fungi 11, no. 8: 595. https://doi.org/10.3390/jof11080595
APA StyleChen, J., Li, W., Wu, C., Wu, S., & Tong, Y. (2025). Analysis of the Effects of Beauveria bassiana Appressorium Formation on Insect Cuticle Metabolism Based on LC-MS. Journal of Fungi, 11(8), 595. https://doi.org/10.3390/jof11080595





