Improvement of Monacolin K and Pigment Production in Monascus by 5-Azacytidine
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Materials
2.2. Addition of 5-Azacytidine
2.3. Determination of Pigment Value
2.4. Determination of Monacolin K Yield
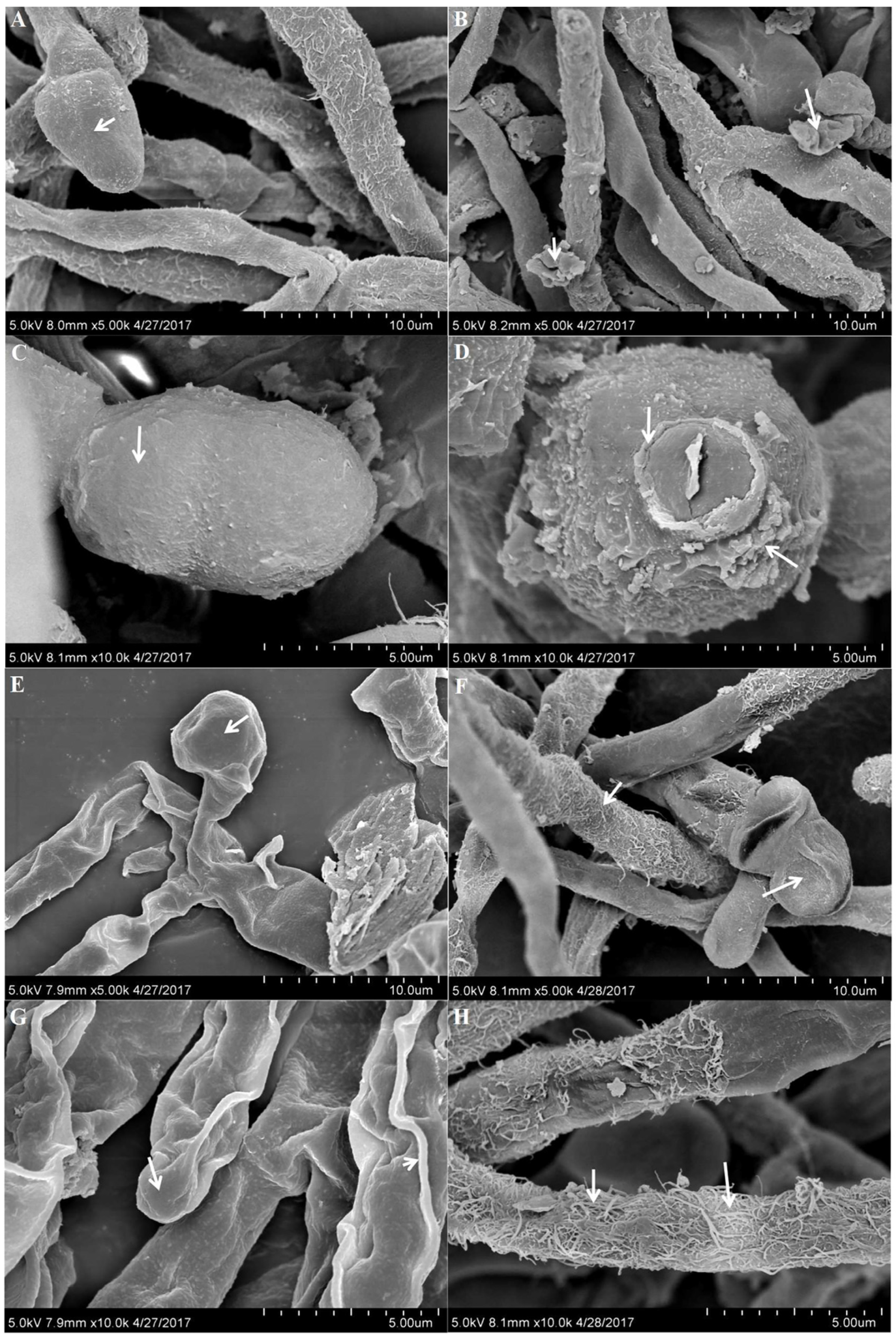
2.5. Scanning Electron Microscopy (SEM)
2.6. Statistical Analysis
3. Results
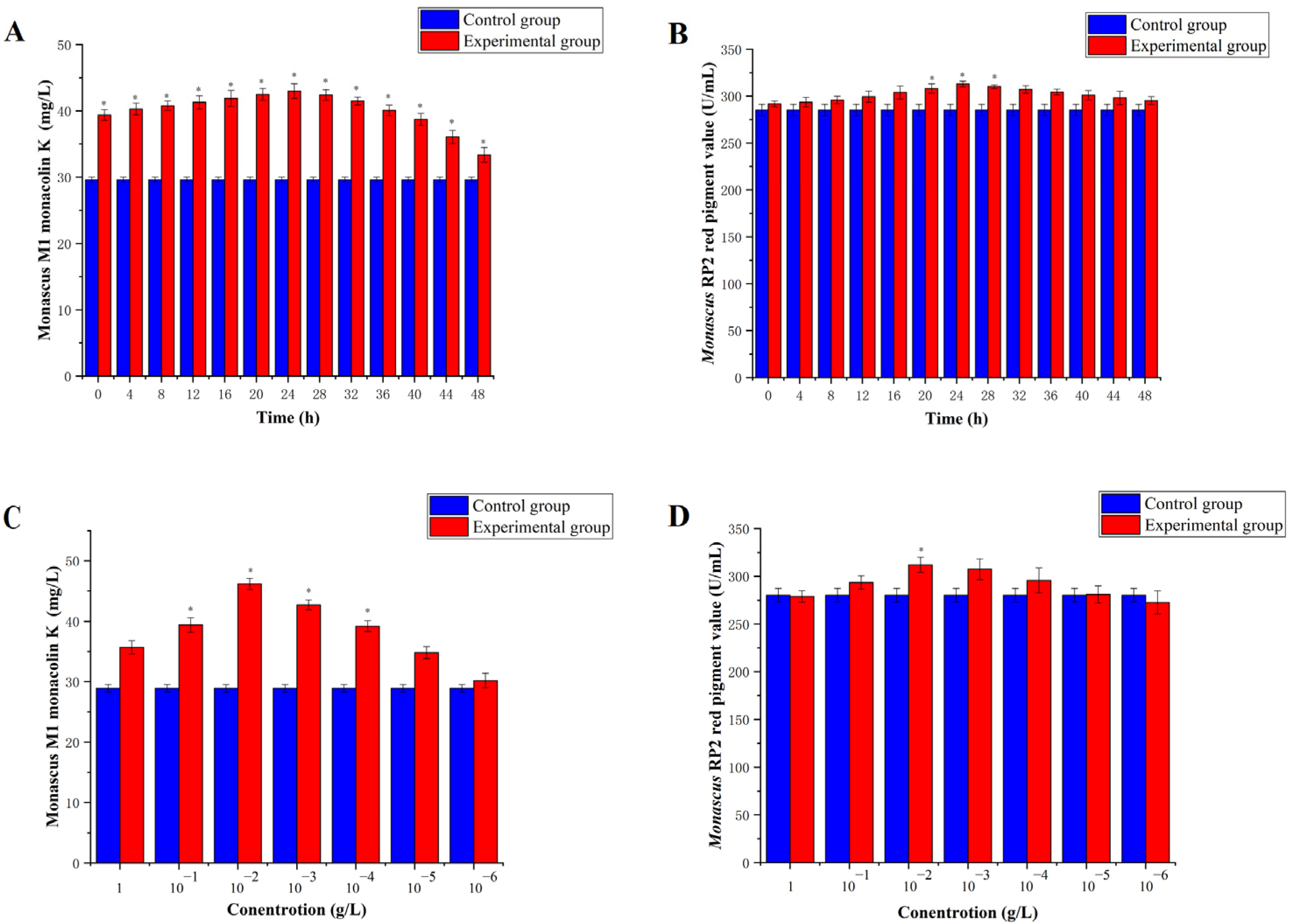
3.1. Addition of 5-Azacytidine to Monascus M1 and RP2 at Different Time Points and Concentrations
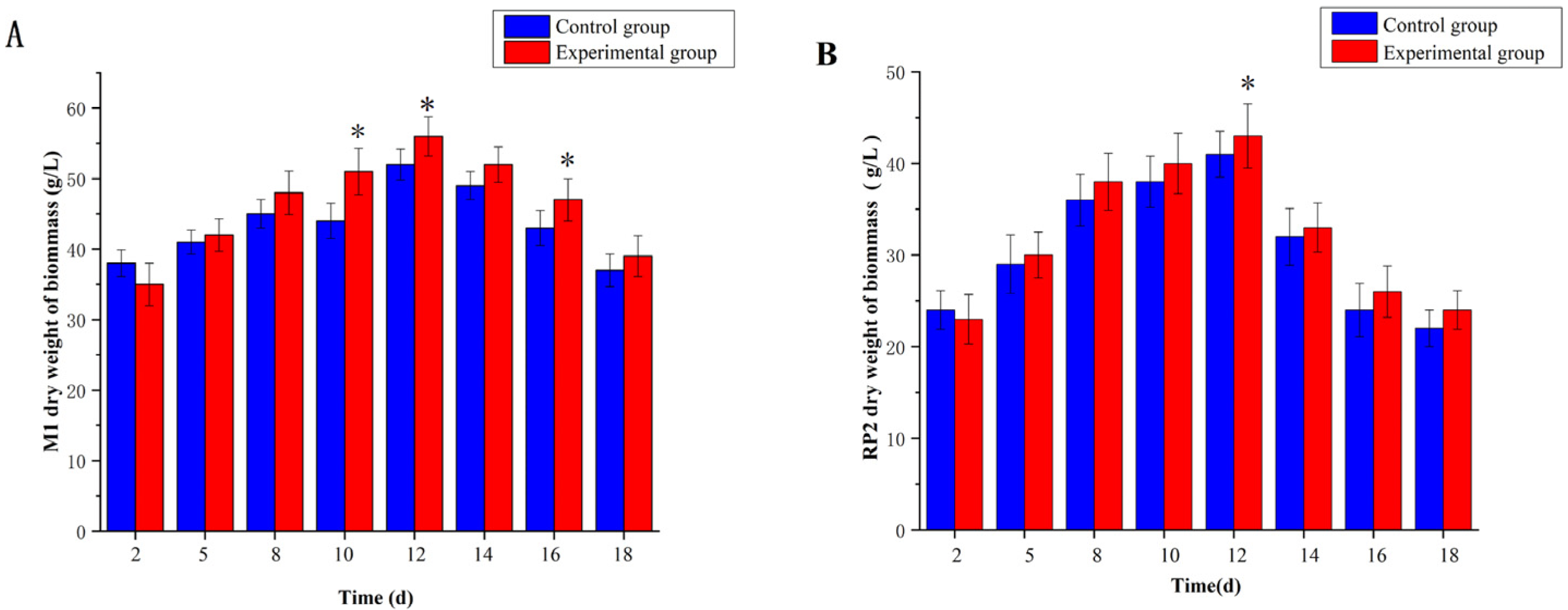
3.2. Effect of 5-Azacytidine on the Dry Weights of Monascus M1 and RP2
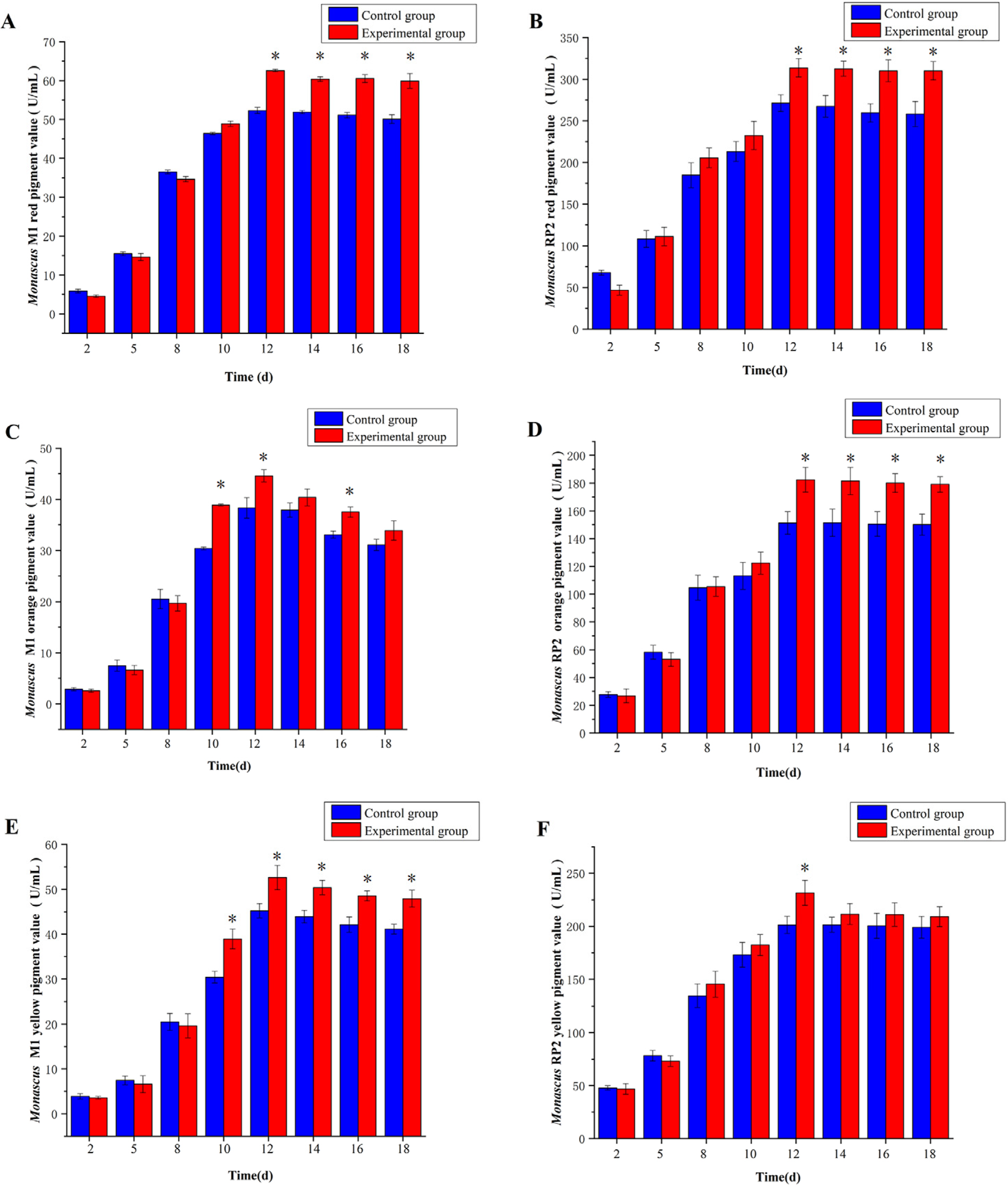
3.3. Effect of 5-Azacytidine on Red, Orange, and Yellow Pigment Values of Monascus M1 and RP2
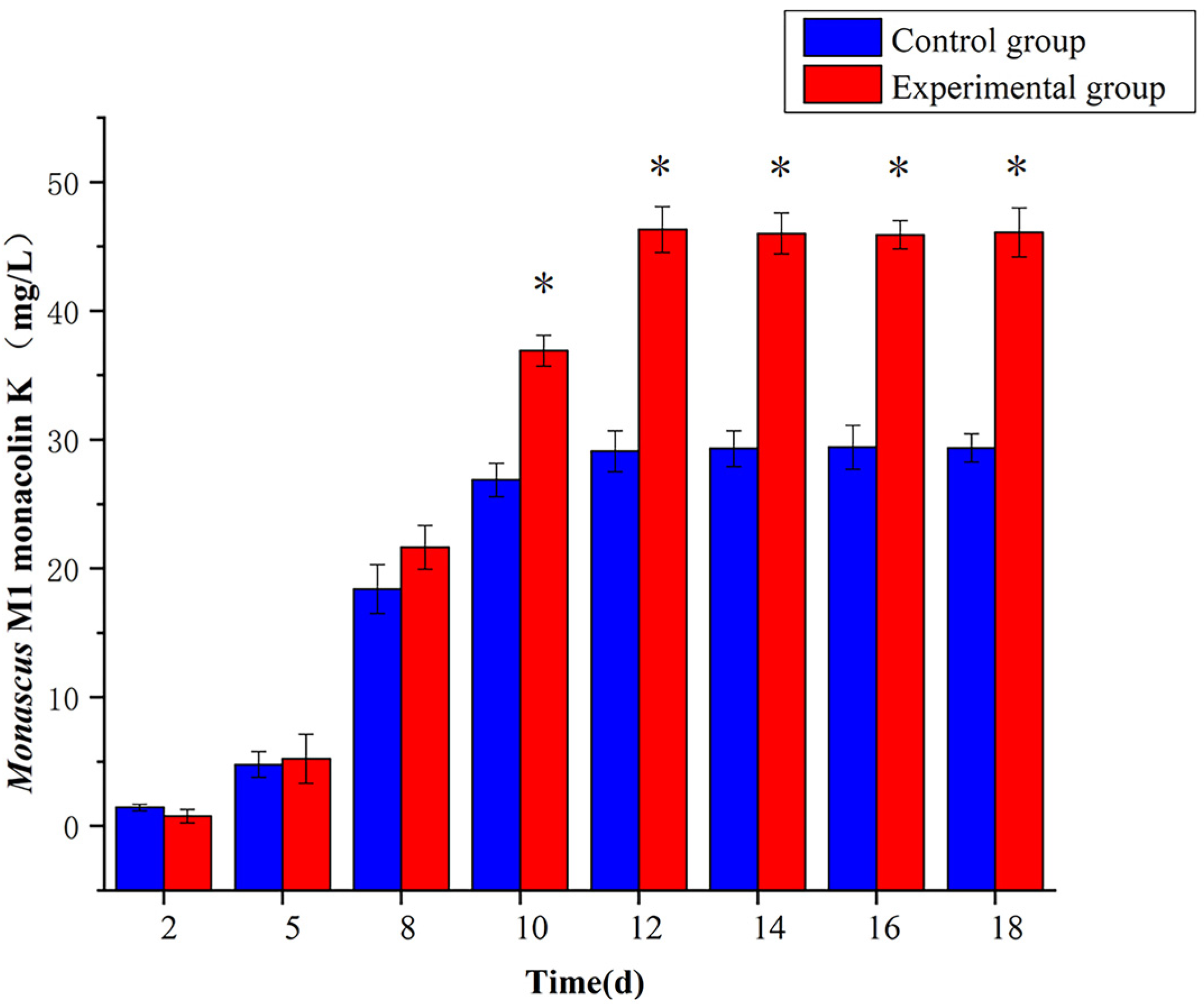
3.4. Effect of 5-Azacytidine on the Monacolin K Yield of Monascus M1
3.5. Effect of 5-Azacytidine on Mycelial Morphology of Monascus M1 and RP2
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, J.J.; Shang, X.Y.; Li, L.L.; Liu, M.T.; Zheng, J.Q.; Jin, Z.L. New Cytotoxic Azaphilones from Monascus Purpureus-Fermented Rice (Red Yeast Rice). Molecules 2010, 15, 1958–1966. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Shao, Y.; Chen, F. Monascus Pigments. Appl. Microbiol. Biotechnol. 2012, 96, 1421–1440. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.L.; Wang, T.H.; Lee, M.H.; Su, N.W. Biologically Active Components and Nutraceuticals in the Monascus-Fermented Rice: A Review. Appl. Microbiol. Biotechnol. 2008, 77, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Jo, D.; Choe, D.; Nam, K.; Shin, C.S. Biological Evaluation of Novel Derivatives of the Orange Pigments from Monascus Sp. as Inhibitors of Melanogenesis. Biotechnol. Lett. 2014, 36, 1605–1613. [Google Scholar] [CrossRef]
- Liu, M.T.; Wang, A.L.; Sun, Z.; Li, J.J.; Wu, X.L.; Liu, Y.X.; Shang, X.Y. Cytotoxic Monacolin Analogs from Monascus Purpureus-Fermented Rice. J. Asian Nat. Prod. Res. 2013, 15, 600–609. [Google Scholar] [CrossRef]
- Radu, N.; Salageanu, A.; Ferdes, M.; Rau, I. Cytotoxicity Study Regarding Some Products Derived from Monascus sp. Mol. Cryst. Liq. Cryst. 2012, 555, 189–194. [Google Scholar] [CrossRef]
- Su, Y.C.; Wang, J.J.; Lin, T.T.; Pan, T.M. Production of the Secondary Metabolites γ-Aminobutyric Acid and Monacolin K by Monascus. J. Ind. Microbiol. Biotechnol. 2003, 30, 41–46. [Google Scholar] [CrossRef]
- Zerikly, M.; Challis, G.L. Strategies for the Discovery of New Natural Products by Genome Mining. ChemBioChem 2009, 10, 625–633. [Google Scholar] [CrossRef]
- Sakai, K.; Kinoshita, H.; Shimizu, T.; Nihira, T. Construction of a Citrinin Gene Cluster Expression System in Heterologous Aspergillus Oryzae. J. Biosci. Bioeng. 2008, 106, 466–472. [Google Scholar] [CrossRef]
- Bode, H.B.; Bethe, B.; Höfs, R.; Zeeck, A. Big Effects from Small Changes: Possible Ways to Explore Nature’s Chemical Diversity. ChemBioChem 2002, 3, 619–627. [Google Scholar] [CrossRef]
- Chiang, Y.M.; Szewczyk, E.; Davidson, A.D.; Keller, N.; Oakley, B.R.; Wang, C.C.C. A Gene Cluster Containing Two Fungal Polyketide Synthases Encodes the Biosynthetic Pathway for a Polyketide, Asperfuranone, in Aspergillus Nidulans. J. Am. Chem. Soc. 2009, 131, 2965–2970. [Google Scholar] [CrossRef] [PubMed]
- Gacek, A.; Strauss, J. The Chromatin Code of Fungal Secondary Metabolite Gene Clusters. Appl. Microbiol. Biotechnol. 2012, 95, 1389–1404. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.C.; Matsen, C.B.; Gonzales, F.A.; Ye, W.; Greer, S.; Marquez, V.E.; Jones, P.A.; Selker, E.U. Inhibition of DNA Methylation and Reactivation of Silenced Genes by Zebularine. JNCI J. Nalt. Cancer Inst. 2003, 95, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Birch, P.R.J.; Sims, P.F.G.; Broda, P. A Reporter System for Analysis of Regulatable Promoter Functions in the Basidiomycete Fungus Phanerochaete Chrysosporium. J. Appl. Microbiol. 1998, 85, 417–424. [Google Scholar] [CrossRef]
- Bok, J.W.; Hoffmeister, D.; Maggio-Hall, L.A.; Murillo, R.; Glasner, J.D.; Keller, N.P. Genomic Mining for Aspergillus Natural Products. Chem. Biol. 2006, 13, 31–37. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, H.; Zhu, Q.; Hao, S.; Chai, S.; Li, Y.; Jiao, Z.; Shi, J.; Sun, B.; Wang, C. Overexpression of Global Regulator LaeA Increases Secondary Metabolite Production in Monascus Purpureus. Appl. Microbiol. Biotechnol. 2020, 104, 3049–3060. [Google Scholar] [CrossRef]
- Dannenberg, L.O.; Edenberg, H.J. Epigenetics of Gene Expression in Human Hepatoma Cells: Expression Profiling the Response to Inhibition of DNA Methylation and Histone Deacetylation. BMC Genom. 2006, 7, 181. [Google Scholar] [CrossRef]
- Ma, Y.J.; Lu, C.S.; Wang, J.W. Effects of 5-Azacytidine on Growth and Hypocrellin Production of Shiraia Bambusicola. Front. Microbiol. 2018, 9, 2508. [Google Scholar] [CrossRef]
- Cui, Y.; Lu, C.; Liu, L.; Sun, D.; Yao, N.; Tan, S.; Bai, S.; Ma, X. Reactivation of Methylation-Silenced Tumor Suppressor Gene P16INK4a by Nordihydroguaiaretic Acid and Its Implication in G1 Cell Cycle Arrest. Life Sci. 2008, 82, 247–255. [Google Scholar] [CrossRef]
- Zhang, C.; Liang, J.; Yang, L.; Chai, S.; Zhang, C.; Sun, B.; Wang, C. Glutamic Acid Promotes Monacolin K Production and Monacolin K Biosynthetic Gene Cluster Expression in Monascus. AMB Express 2017, 7, 22. [Google Scholar] [CrossRef]
- Zhang, Z.; Ali, Z.; Khan, S.I.; Khan, I.A. Cytotoxic Monacolins from Red Yeast Rice, a Chinese Medicine and Food. Food Chem. 2016, 202, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.; Kim, C.; Kim, K.; Shin, C.S. Color Characteristics of Monascus Pigments Derived by Fermentation with Various Amino Acids. J. Agric. Food. Chem. 2003, 51, 1302–1306. [Google Scholar] [CrossRef]
- Hsu, Y.W.; Hsu, L.C.; Liang, Y.H.; Kuo, Y.H.; Pan, T.M. Monaphilones A-C, Three New Antiproliferative Azaphilone Derivatives from Monascus Purpureus NTU 568. J. Agric. Food. Chem. 2010, 58, 8211–8216. [Google Scholar] [CrossRef]
- Hsu, Y.W.; Hsu, L.C.; Liang, Y.H.; Kuo, Y.H.; Pan, T.M. New Bioactive Orange Pigments with Yellow Fluorescence from Monascus-Fermented Dioscorea. J. Agric. Food. Chem. 2011, 59, 4512–4518. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, G.; Singh, S.K. Purification and Characterization of a New Red Pigment from Monascus Purpureus in Submerged Fermentation. Process. Biochem. 2011, 46, 188–192. [Google Scholar] [CrossRef]
- Wild, D.; Tóth, G.; Humpf, H.U. New Monascus Metabolite Isolated from Red Yeast Rice (Angkak, Red Koji). J. Agric. Food Chem. 2002, 50, 3999–4002. [Google Scholar] [CrossRef] [PubMed]
- Shin, C.S.; Kim, H.J.; Kim, M.J.; Ju, J.Y. Morphological Change and Enhanced Pigment Production of Monascus When Cocultured with Saccharomyces Cerevisiae or Aspergillus Oryzae. Biotechnol. Bioeng. 1998, 59, 576–581. [Google Scholar] [CrossRef]
- Chang, Y.N.; Huang, J.C.; Lee, C.C.; Shih, I.-L.; Tzeng, Y.-M. Use of Response Surface Methodology to Optimize Culture Medium for Production of Lovastatin by Monascus Ruber. Enzyme Microb. Technol. 2002, 30, 889–894. [Google Scholar] [CrossRef]
- Hajjaj, H.; Blanc, P.J.; Groussac, E.; Goma, G.; Uribelarrea, J.L.; Loubiere, P. Improvement of Red Pigment/Citrinin Production Ratio as a Function of Environmental Conditions by Monascus Ruber. Biotechnol Bioeng. 1999, 64, 497–501. [Google Scholar] [CrossRef]
- Li, L.; Shao, Y.; Li, Q.; Yang, S.; Chen, F. Identification of Mga1, a G-Protein α-Subunit Gene Involved in Regulating Citrinin and Pigment Production in Monascus Ruber M7. FEMS Microbiol. Lett. 2010, 308, 108–114. [Google Scholar] [CrossRef]
- Jia, L.; Yu, J.-H.; Chen, F.; Chen, W. Characterization of the Asexual Developmental Genes BrlA and WetA in Monascus Ruber M7. Fungal Genet. Biol. 2021, 151, 103564. [Google Scholar] [CrossRef]
- Liu, J.; Lei, M.; Zhou, Y.; Chen, F. A Comprehensive Analysis of the Small GTPases Ypt7 Involved in the Regulation of Fungal Development and Secondary Metabolism in Monascus Ruber M7. Front. Microbiol. 2019, 10, 452. [Google Scholar] [CrossRef] [PubMed]
- Xie, N.; Liu, Q.; Chen, F. Deletion of PigR Gene in Monascus Ruber Leads to Loss of Pigment Production. Biotechnol. Lett. 2013, 35, 1425–1432. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-Q.; Zhao, X.-X.; Zhi, Q.-Q.; Zhao, M.; He, Z.-M. Transcriptomic Profiling of Aspergillus Flavus in Response to 5-Azacytidine. Fungal Genet. Biol. 2013, 56, 78–86. [Google Scholar] [CrossRef]
- Manfrão-Netto, J.H.C.; Mello-de-Sousa, T.M.; Mach-Aigner, A.R.; Mach, R.L.; Poças-Fonseca, M.J. The DNA-methyltransferase Inhibitor 5-aza-2-deoxycytidine Affects Humicola Grisea Enzyme Activities and the Glucose-mediated Gene Repression. J. Basic Microbiol. 2018, 58, 144–153. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, C.; Wang, H.; Sun, Q.; Ablimit, A.; Dong, H.; Wang, C.; Zhai, D.; Zhang, B.; Hu, W.; Liu, C.; et al. Improvement of Monacolin K and Pigment Production in Monascus by 5-Azacytidine. J. Fungi 2024, 10, 819. https://doi.org/10.3390/jof10120819
Zhang C, Wang H, Sun Q, Ablimit A, Dong H, Wang C, Zhai D, Zhang B, Hu W, Liu C, et al. Improvement of Monacolin K and Pigment Production in Monascus by 5-Azacytidine. Journal of Fungi. 2024; 10(12):819. https://doi.org/10.3390/jof10120819
Chicago/Turabian StyleZhang, Chan, Haijiao Wang, Qing Sun, Arzugul Ablimit, Huijun Dong, Congcong Wang, Duchen Zhai, Bobo Zhang, Wenlin Hu, Chengjian Liu, and et al. 2024. "Improvement of Monacolin K and Pigment Production in Monascus by 5-Azacytidine" Journal of Fungi 10, no. 12: 819. https://doi.org/10.3390/jof10120819
APA StyleZhang, C., Wang, H., Sun, Q., Ablimit, A., Dong, H., Wang, C., Zhai, D., Zhang, B., Hu, W., Liu, C., & Wang, C. (2024). Improvement of Monacolin K and Pigment Production in Monascus by 5-Azacytidine. Journal of Fungi, 10(12), 819. https://doi.org/10.3390/jof10120819






