Irisin, an Effective Treatment for Cardiovascular Diseases?
Abstract
:1. Introduction
2. FNDC5 and Irisin
3. Physiological Function of Irisin
4. Regulation of Irisin by Exercise
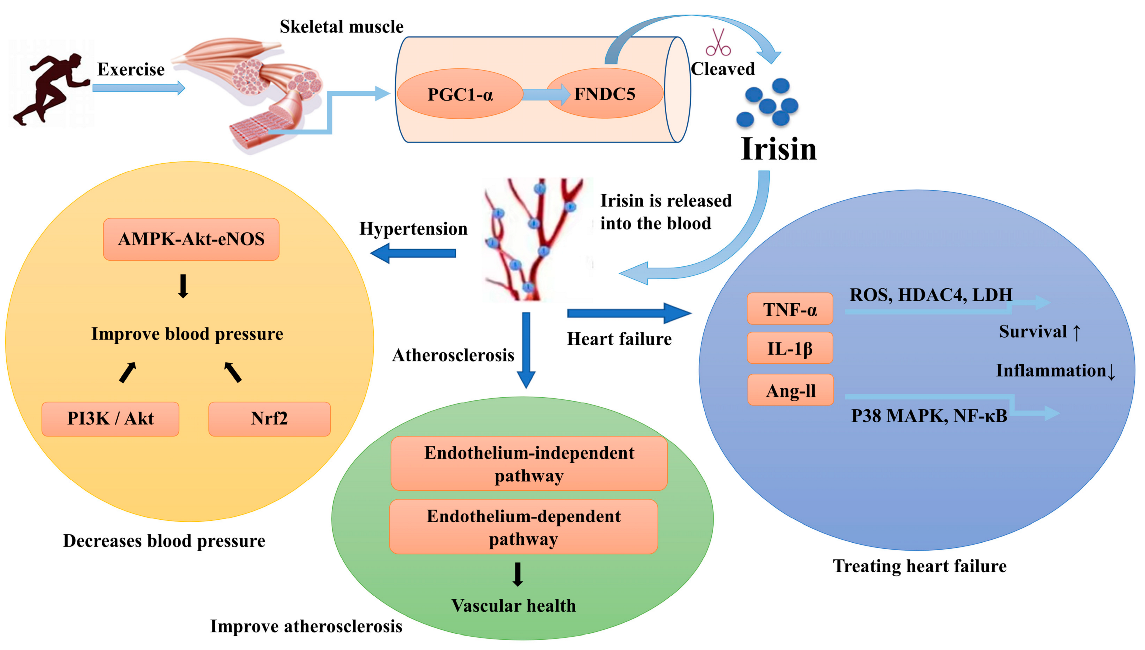
5. Irisin and CVDs
5.1. Irisin, Biomarker for CVD Diagnosis
5.2. The Regulation of Irisin on CVDs
5.2.1. Hypertension
5.2.2. Atherosclerosis
5.2.3. Heart Failure
5.2.4. Other Cardiovascular Diseases
6. New Treatments and Strategies Based on Irisin
7. Conclusions and Prospects
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Wang, Z.; Chen, Z.; Zhang, L.; Wang, X.; Hao, G.; Zhang, Z.; Shao, L.; Tian, Y.; Dong, Y.; Zheng, C.; et al. Status of Hypertension in China: Results From the China Hypertension Survey, 2012–2015. Circulation 2018, 137, 2344–2356. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wu, C.; Lu, J.; Chen, B.; Li, Y.; Yang, Y.; Hu, S.; Li, J. Cardiovascular risk factors in China: A nationwide population-based cohort study. Lancet Public Health 2020, 5, e672–e681. [Google Scholar] [CrossRef]
- Egan, B.M. Combination therapy with an angiotensin-converting enzyme inhibitor and a calcium channel blocker. J. Clin. Hypertens. 2007, 9, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Chen, C.; Lv, S.; Zhu, Y.; Fang, T. Comparing the Efficacy of Angiotensin Converting Enzyme Inhibitors with Calcium Channel Blockers on the Treatment of Diabetic Nephropathy: A Meta-Analysis. Iran. J. Public Health 2019, 48, 189–197. [Google Scholar] [CrossRef]
- Tian, D.; Meng, J. Exercise for Prevention and Relief of Cardiovascular Disease: Prognoses, Mechanisms, and Approaches. Oxidative Med. Cell. Longev. 2019, 2019, 3756750. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Saltin, B. Exercise as medicine–Evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand. J. Med. Sci. Sports 2015, 25 (Suppl. S3), 1–72. [Google Scholar] [CrossRef]
- Cassidy, S.; Thoma, C.; Hallsworth, K.; Parikh, J.; Hollingsworth, K.G.; Taylor, R.M.R.; Jakovljevic, D.; Trenell, M.I. High intensity intermittent exercise improves cardiac structure and function and reduces liver fat in patients with type 2 diabetes: A randomized controlled trial. Diabetologia 2016, 59, 56–66. [Google Scholar] [CrossRef]
- Gubert, C.; Hannan, A.J. Exercise mimetics: Harnessing the therapeutic effects of physical activity. Nat. Rev. Drug Discov. 2021, 20, 862–879. [Google Scholar] [CrossRef]
- Ma, C.; Ding, H.; Deng, Y.; Liu, H.; Xiong, X.; Yang, Y. Irisin: A New Code Uncover the Relationship of Skeletal Muscle and Cardiovascular Health During Exercise. Front. Physiol. 2021, 12, 620608. [Google Scholar] [CrossRef]
- Hsieh, I.-C.; Ho, M.-Y.; Wen, M.-S.; Chen, C.-C.; Hsieh, M.-J.; Lin, C.-P.; Yeh, J.-K.; Tsai, M.-L.; Yang, C.-H.; Wu, V.C.-C.; et al. Serum irisin levels are associated with adverse cardiovascular outcomes in patients with acute myocardial infarction. Int. J. Cardiol. 2018, 261, 12–17. [Google Scholar] [CrossRef]
- Yu, Q.; Kou, W.; Xu, X.; Zhou, S.; Luan, P.; Xu, X.; Li, H.; Zhuang, J.; Wang, J.; Zhao, Y.; et al. FNDC5/Irisin inhibits pathological cardiac hypertrophy. Clin. Sci. 2019, 133, 611–627. [Google Scholar] [CrossRef] [PubMed]
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Bubak, M.P.; Heesch, M.W.; Shute, R.J.; Dinan, N.E.; Laursen, T.L.; La Salle, D.T.; Slivka, D.R. Irisin and Fibronectin Type III Domain-Containing 5 Responses to Exercise in Different Environmental Conditions. Int. J. Exerc. Sci. 2017, 10, 666–680. [Google Scholar]
- Tang, S.; Zhang, R.; Jiang, F.; Wang, J.; Chen, M.; Peng, D.; Yan, J.; Wang, S.; Bao, Y.; Hu, C.; et al. Circulating irisin levels are associated with lipid and uric acid metabolism in a Chinese population. Clin. Exp. Pharmacol. Physiol. 2015, 42, 896–901. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, X.; Liang, J.; Kirberger, M.; Chen, N. Irisin, an exercise-induced bioactive peptide beneficial for health promotion during aging process. Ageing Res. Rev. 2022, 80, 101680. [Google Scholar] [CrossRef]
- Raschke, S.; Elsen, M.; Gassenhuber, H.; Sommerfeld, M.; Schwahn, U.; Brockmann, B.; Jung, R.; Wisloff, U.; Tjonna, A.E.; Raastad, T.; et al. Evidence against a beneficial effect of irisin in humans. PLoS ONE 2013, 8, e73680. [Google Scholar] [CrossRef]
- Kozak, M. Context effects and inefficient initiation at non-AUG codons in eucaryotic cell-free translation systems. Mol. Cell. Biol. 1989, 9, 5073–5080. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Wang, P.; Chen, Q.; Li, C. Exercise enhances mitochondrial fission and mitophagy to improve myopathy following critical limb ischemia in elderly mice via the PGC1a/FNDC5/irisin pathway. Skelet. Muscle 2020, 10, 25. [Google Scholar] [CrossRef]
- Tiano, J.P.; Springer, D.A.; Rane, S.G. SMAD3 Negatively Regulates Serum Irisin and Skeletal Muscle FNDC5 and Peroxisome Proliferator-Activated Receptor γ Coactivator 1-α (PGC-1α) during Exercise. J. Biol. Chem. 2015, 290, 7671–7684. [Google Scholar] [CrossRef]
- Kim, H.; Wrann, C.D.; Jedrychowski, M.; Vidoni, S.; Kitase, Y.; Nagano, K.; Zhou, C.; Chou, J.; Parkman, V.A.; Novick, S.J.; et al. Irisin mediates effects on bone and fat via αV integrin receptors. Cell 2018, 175, 1756–1768.e17. [Google Scholar] [CrossRef]
- Schumacher, M.A.; Chinnam, N.; Ohashi, T.; Shah, R.S.; Erickson, H.P. The Structure of Irisin Reveals a Novel Intersubunit β-Sheet Fibronectin Type III (FNIII) Dimer: Implications for receptor activation. J. Biol. Chem. 2013, 288, 33738–33744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinkowska, A.; Podhorska-Okołów, M.; Dzięgiel, P.; Nowińska, K. The Role of Irisin in Cancer Disease. Cells 2021, 10, 1479. [Google Scholar] [CrossRef] [PubMed]
- Arhire, L.I.; Mihalache, L.; Covasa, M. Irisin: A Hope in Understanding and Managing Obesity and Metabolic Syndrome. Front. Endocrinol. 2019, 10, 524. [Google Scholar] [CrossRef]
- Wang, R.; Liu, H. Association Between Serum Irisin and Diabetic Nephropathy in Patients with Type 2 Diabetes Mellitus: A Meta-Analysis. Horm. Metab. Res. 2021, 53, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Cui, Y.; Zhai, Y.; Xin, W.; Yu, Y.; Liang, J.; Li, S.; Sun, H. Neuroprotective Effects of Exogenous Irisin in Kainic Acid-Induced Status Epilepticus. Front. Cell. Neurosci. 2021, 15, 738533. [Google Scholar] [CrossRef] [PubMed]
- Briganti, S.I.; Gianluigi, G.; Gaia, T.; Naciu, A.M.; Roberto, C.; Silvia, M.; Andrea, P. Irisin as a regulator of bone and glucose metabolism. Minerva Endocrinol. 2018, 43, 489–500. [Google Scholar] [CrossRef]
- Li, Q.; Tan, Y.; Chen, S.; Xiao, X.; Zhang, M.; Wu, Q.; Dong, M. Irisin alleviates LPS-induced liver injury and inflammation through inhibition of NLRP3 inflammasome and NF-κB signaling. J. Recept. Signal Transduct. Res. 2021, 41, 294–303. [Google Scholar] [CrossRef]
- Cheng, Z.-B.; Huang, L.; Xiao, X.; Sun, J.-X.; Zou, Z.-K.; Jiang, J.-F.; Lu, C.; Zhang, H.-Y.; Zhang, C. Irisin in atherosclerosis. Clin. Chim. Acta 2021, 522, 158–166. [Google Scholar] [CrossRef]
- Pan, J.-A.; Zhang, H.; Lin, H.; Gao, L.; Zhang, J.-F.; Wang, C.-Q.; Gu, J. Irisin ameliorates doxorubicin-induced cardiac perivascular fibrosis through inhibiting endothelial-to-mesenchymal transition by regulating ROS accumulation and autophagy disorder in endothelial cells. Redox Biol. 2021, 46, 102120. [Google Scholar] [CrossRef]
- Korta, P.; Pocheć, E.; Mazur-Biały, A. Irisin as a Multifunctional Protein: Implications for Health and Certain Diseases. Medicina 2019, 55, 485. [Google Scholar] [CrossRef]
- Löffler, D.; Müller, U.; Scheuermann, K.; Friebe, D.; Gesing, J.; Bielitz, J.; Erbs, S.; Landgraf, K.; Wagner, I.V.; Kiess, W.; et al. Serum Irisin Levels Are Regulated by Acute Strenuous Exercise. J. Clin. Endocrinol. Metab. 2015, 100, 1289–1299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daskalopoulou, S.S.; Cooke, A.B.; Gomez, Y.-H.; Mutter, A.F.; Filippaios, A.; Mesfum, E.T.; Mantzoros, C.S. Plasma irisin levels progressively increase in response to increasing exercise workloads in young, healthy, active subjects. Eur. J. Endocrinol. 2014, 171, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Norheim, F.; Langleite, T.M.; Hjorth, M.; Holen, T.; Kielland, A.; Stadheim, H.K.; Gulseth, H.L.; Birkeland, K.I.; Jensen, J.; Drevon, C.A. The effects of acute and chronic exercise on PGC-1α, irisin and browning of subcutaneous adipose tissue in humans. FEBS J. 2014, 281, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Ando, D.; Takamatsu, K.; Goto, K. Resistance exercise induces a greater irisin response than endurance exercise. Metabolism 2015, 64, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Pekkala, S.; Wiklund, P.K.; Hulmi, J.J.; Ahtiainen, J.P.; Horttanainen, M.; Pöllänen, E.; Mäkelä, K.A.; Kainulainen, H.; Häkkinen, K.; Nyman, K.; et al. Are skeletal muscle FNDC5 gene expression and irisin release regulated by exercise and related to health? J. Physiol. 2013, 591, 5393–5400. [Google Scholar] [CrossRef]
- Bizjak, D.A.; Zügel, M.; Schumann, U.; Tully, M.A.; Dallmeier, D.; Denkinger, M.; Steinacker, J.M. Do skeletal muscle composition and gene expression as well as acute exercise-induced serum adaptations in older adults depend on fitness status? BMC Geriatr. 2021, 21, 697. [Google Scholar] [CrossRef]
- Tibana, R.A.; Nascimento, D.D.C.; de Souza, N.M.F.; de Souza, V.C.; Neto, I.V.D.S.; Voltarelli, F.A.; Pereira, G.B.; Navalta, J.W.; Prestes, J. Irisin levels are not associated to resistance training-induced alterations in body mass composition in older untrained women with and without obesity. J. Nutr. Health Aging 2017, 21, 241–246. [Google Scholar] [CrossRef]
- Huh, J.Y.; Mougios, V.; Skraparlis, A.; Kabasakalis, A.; Mantzoros, C.S. Irisin in response to acute and chronic whole-body vibration exercise in humans. Metabolism 2014, 63, 918–921. [Google Scholar] [CrossRef]
- Inoue, K.; Fujie, S.; Hasegawa, N.; Horii, N.; Uchida, M.; Iemitsu, K.; Sanada, K.; Hamaoka, T.; Iemitsu, M. Aerobic exercise training-induced irisin secretion is associated with the reduction of arterial stiffness via nitric oxide production in adults with obesity. Appl. Physiol. Nutr. Metab. 2020, 45, 715–722. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, D.-Y. Aquarobic exercises improve the serum blood irisin and brain-derived neurotrophic factor levels in elderly women. Exp. Gerontol. 2018, 104, 60–65. [Google Scholar] [CrossRef]
- Kim, H.-J.; So, B.; Choi, M.; Kang, D.; Song, W. Resistance exercise training increases the expression of irisin concomitant with improvement of muscle function in aging mice and humans. Exp. Gerontol. 2015, 70, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Su, Z.; Qu, C.; Dong, Y. Effects of 12 Weeks Resistance Training on Serum Irisin in Older Male Adults. Front. Physiol. 2017, 8, 171. [Google Scholar] [CrossRef] [PubMed]
- Coletta, A.M.; Agha, N.H.; Baker, F.L.; Niemiro, G.M.; Mylabathula, P.L.; Brewster, A.M.; Bevers, T.B.; Fuentes-Mattei, E.; Basen-Engquist, K.; Gilchrist, S.C.; et al. The impact of high-intensity interval exercise training on NK-cell function and circulating myokines for breast cancer prevention among women at high risk for breast cancer. Breast Cancer Res. Treat. 2021, 187, 407–416. [Google Scholar] [CrossRef]
- Ellefsen, S.; Vikmoen, O.; Slettaløkken, G.; Whist, J.E.; Nygaard, H.; Hollan, I.; Rauk, I.; Vegge, G.; Strand, T.; Raastad, T.; et al. Irisin and FNDC5: Effects of 12-week strength training, and relations to muscle phenotype and body mass composition in untrained women. Eur. J. Appl. Physiol. 2014, 114, 1875–1888. [Google Scholar] [CrossRef] [PubMed]
- Nygaard, H.; Slettaløkken, G.; Vegge, G.; Hollan, I.; Whist, J.E.; Strand, T.; Rønnestad, B.R.; Ellefsen, S. Irisin in Blood Increases Transiently after Single Sessions of Intense Endurance Exercise and Heavy Strength Training. PLoS ONE 2015, 10, e0121367. [Google Scholar] [CrossRef]
- Kim, H.-J.; Lee, H.-J.; So, B.; Son, J.S.; Yoon, D.; Song, W. Effect of Aerobic Training and Resistance Training on Circulating Irisin Level and Their Association with Change of Body Composition in Overweight/Obese Adults: A Pilot Study. Physiol. Res. 2016, 65, 271–279. [Google Scholar] [CrossRef]
- Kurdiova, T.; Balaz, M.; Vician, M.; Maderova, D.; Vlcek, M.; Valkovic, L.; Srbecky, M.; Imrich, R.; Kyselovicova, O.; Belan, V.; et al. Effects of obesity, diabetes and exercise on Fndc5 gene expression and irisin release in human skeletal muscle and adipose tissue: In vivo and in vitro studies. J. Physiol. 2014, 592, 1091–1107. [Google Scholar] [CrossRef]
- Liu, J.-J.; Wong, M.D.; Toy, W.C.; Tan, C.S.; Liu, S.; Ng, X.W.; Tavintharan, S.; Sum, C.F.; Lim, S.C. Lower circulating irisin is associated with type 2 diabetes mellitus. J. Diabetes Complicat. 2013, 27, 365–369. [Google Scholar] [CrossRef]
- Huh, J.Y.; Panagiotou, G.; Mougios, V.; Brinkoetter, M.; Vamvini, M.T.; Schneider, B.E.; Mantzoros, C.S. FNDC5 and irisin in humans: I. Predictors of circulating concentrations in serum and plasma and II. mRNA expression and circulating concentrations in response to weight loss and exercise. Metabolism 2012, 61, 1725–1738. [Google Scholar] [CrossRef]
- Hisamatsu, T.; Miura, K.; Arima, H.; Fujiyoshi, A.; Kadota, A.; Kadowaki, S.; Zaid, M.; Miyagawa, N.; Satoh, A.; Kunimura, A.; et al. Relationship of serum irisin levels to prevalence and progression of coronary artery calcification: A prospective, population-based study. Int. J. Cardiol. 2018, 267, 177–182. [Google Scholar] [CrossRef]
- Deng, W. Association of Serum Irisin Concentrations with Presence and Severity of Coronary Artery Disease. Med. Sci. Monit. 2016, 22, 4193–4197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Efe, T.H.; Açar, B.; Ertem, A.G.; Yayla, K.G.; Algül, E.; Yayla, Ç.; Ünal, S.; Bilgin, M.; Çimen, T.; Kirbaş, O.; et al. Serum Irisin Level Can Predict the Severity of Coronary Artery Disease in Patients with Stable Angina. Korean Circ. J. 2017, 47, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Wenwen, G.; Baihui, Z.; Xia, W. Lower irisin levels in coronary artery disease: A meta-analysis. Minerva Endocrinol. 2020, 45, 61–69. [Google Scholar] [CrossRef]
- Saadeldin, M.K.; Elshaer, S.S.; Emara, I.A.; Maged, M.; Abdel-Aziz, A.K. Serum sclerostin and irisin as predictive markers for atherosclerosis in Egyptian type II diabetic female patients: A case control study. PLoS ONE 2018, 13, e0206761. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.-J.; Xie, Q.; Tang, C.-S.; Zhang, A.-H. Expressions of irisin and urotensin II and their relationships with blood pressure in patients with preeclampsia. Clin. Exp. Hypertens. 2017, 39, 460–467. [Google Scholar] [CrossRef]
- Çelik, H.T.; Akkaya, N.; Erdamar, H.; Gok, S.; Kazanci, F.; Demircelik, B.; Cakmak, M.; Yigitoglu, R. The Effects of Valsartan and Amlodipine on the Levels of Irisin, Adropin, and Perilipin. Clin. Lab. 2015, 61, 1889–1895. [Google Scholar] [CrossRef]
- Lee, M.J.; A Lee, S.; Nam, B.Y.; Park, S.; Lee, S.-H.; Ryu, H.J.; Kwon, Y.E.; Kim, Y.L.; Park, K.S.; Oh, H.J.; et al. Irisin, a novel myokine is an independent predictor for sarcopenia and carotid atherosclerosis in dialysis patients. Atherosclerosis 2015, 242, 476–482. [Google Scholar] [CrossRef]
- Shen, S.; Gao, R.; Bei, Y.; Li, J.; Zhang, H.; Zhou, Y.; Yao, W.; Xu, D.; Zhou, F.; Jin, M.; et al. Serum Irisin Predicts Mortality Risk in Acute Heart Failure Patients. Cell. Physiol. Biochem. 2017, 42, 615–622. [Google Scholar] [CrossRef]
- Kalkan, A.K.; Cakmak, H.A.; Erturk, M.; Kalkan, K.E.; Uzun, F.; Tasbulak, O.; Diker, V.O.; Aydin, S.; Celik, A. Adropin and Irisin in Patients with Cardiac Cachexia. Arq. Bras. Cardiol. 2018, 111, 39–47. [Google Scholar] [CrossRef]
- Sobieszek, G.; Powrózek, T.; Mazurek, M.; Skwarek-Dziekanowska, A.; Małecka-Massalska, T. Electrical and Hormonal Biomarkers in Cachectic Elderly Women with Chronic Heart Failure. J. Clin. Med. 2020, 9, 1021. [Google Scholar] [CrossRef]
- Lin, Y.-Y.; Lee, S.-D. Cardiovascular Benefits of Exercise Training in Postmenopausal Hypertension. Int. J. Mol. Sci. 2018, 19, 2523. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Zhou, M.; Wang, X.; Li, S.; Yang, D. The Role of Myokines and Adipokines in Hypertension and Hypertension-related Complications. Hypertens. Res. 2019, 42, 1544–1551. [Google Scholar] [CrossRef] [PubMed]
- Huo, C.-J.; Yu, X.-J.; Sun, Y.-J.; Li, H.-B.; Su, Q.; Bai, J.; Li, Y.; Liu, K.-L.; Qi, J.; Zhou, S.-W.; et al. Irisin lowers blood pressure by activating the Nrf2 signaling pathway in the hypothalamic paraventricular nucleus of spontaneously hypertensive rats. Toxicol. Appl. Pharmacol. 2020, 394, 114953. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Wang, J.; Song, M.; Zhou, F.; Fu, D.; Ruan, G.; Zhu, X.; Bai, Y.; Huang, L.; Pang, R.; et al. Irisin Increased the Number and Improved the Function of Endothelial Progenitor Cells in Diabetes Mellitus Mice. J. Cardiovasc. Pharmacol. 2016, 68, 67–73. [Google Scholar] [CrossRef]
- Ling, L.; Chen, D.; Tong, Y.; Zang, Y.-H.; Ren, X.-S.; Zhou, H.; Qi, X.-H.; Chen, Q.; Li, Y.-H.; Kang, Y.-M.; et al. Fibronectin type III domain containing 5 attenuates NLRP3 inflammasome activation and phenotypic transformation of adventitial fibroblasts in spontaneously hypertensive rats. J. Hypertens. 2018, 36, 1104–1114. [Google Scholar] [CrossRef]
- Zhang, W.; Chang, L.; Zhang, C.; Zhang, R.; Li, Z.; Chai, B.; Li, J.; Chen, E.; Mulholland, M. Central and Peripheral Irisin Differentially Regulate Blood Pressure. Cardiovasc. Drugs Ther. 2015, 29, 121–127. [Google Scholar] [CrossRef]
- Fu, J.; Han, Y.; Wang, J.; Liu, Y.; Zheng, S.; Zhou, L.; Jose, P.A.; Zeng, C. Irisin Lowers Blood Pressure by Improvement of Endothelial Dysfunction via AMPK-Akt-eNOS-NO Pathway in the Spontaneously Hypertensive Rat. J. Am. Heart Assoc. 2016, 5, e003433. [Google Scholar] [CrossRef]
- Han, F.; Zhang, S.; Hou, N.; Wang, D.; Sun, X. Irisin improves endothelial function in obese mice through the AMPK-eNOS pathway. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H1501–H1508. [Google Scholar] [CrossRef]
- Li, R.-L.; Wu, S.-S.; Wu, Y.; Wang, X.-X.; Chen, H.-Y.; Xin, J.-J.; Li, H.; Lan, J.; Xue, K.-Y.; Li, X.; et al. Irisin alleviates pressure overload-induced cardiac hypertrophy by inducing protective autophagy via mTOR-independent activation of the AMPK-ULK1 pathway. J. Mol. Cell. Cardiol. 2018, 121, 242–255. [Google Scholar] [CrossRef]
- Ho, M.-Y.; Wang, C.-Y. Role of Irisin in Myocardial Infarction, Heart Failure, and Cardiac Hypertrophy. Cells 2021, 10, 2103. [Google Scholar] [CrossRef]
- Zhu, Y.; Xian, X.; Wang, Z.; Bi, Y.; Chen, Q.; Han, X.; Tang, D.; Chen, R. Research Progress on the Relationship between Atherosclerosis and Inflammation. Biomolecules 2018, 8, 80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Icli, A.; Cure, E.; Cure, M.C.; Uslu, A.U.; Balta, S.; Arslan, S.; Sakiz, D.; Kucuk, A. Novel myokine: Irisin may be an independent predictor for subclinic atherosclerosis in Behçet’s disease. J. Investig. Med. 2016, 64, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Xiang, G.; Liu, M.; Mei, W.; Xiang, L.; Dong, J. Irisin protects against endothelial injury and ameliorates atherosclerosis in apolipoprotein E-Null diabetic mice. Atherosclerosis 2015, 243, 438–448. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Mu, Q.; Zhou, Z.; Song, H.; Zhang, Y.; Wu, F.; Jiang, M.; Wang, F.; Zhang, W.; Li, L.; et al. Protective Effect of Irisin on Atherosclerosis via Suppressing Oxidized Low Density Lipoprotein Induced Vascular Inflammation and Endothelial Dysfunction. PLoS ONE 2016, 11, e0158038. [Google Scholar] [CrossRef]
- Deng, X.; Huang, W.; Peng, J.; Zhu, T.-T.; Sun, X.-L.; Zhou, X.-Y.; Yang, H.; Xiong, J.-F.; He, H.-Q.; Xu, Y.-H.; et al. Irisin Alleviates Advanced Glycation End Products-Induced Inflammation and Endothelial Dysfunction via Inhibiting ROS-NLRP3 Inflammasome Signaling. Inflammation 2018, 41, 260–275. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, H.; Zhang, Y.; Wu, F.; Mu, Q.; Jiang, M.; Wang, F.; Zhang, W.; Li, L.; Shao, L.; et al. Irisin Inhibits Atherosclerosis by Promoting Endothelial Proliferation Through microRNA126-5p. J. Am. Heart Assoc. 2016, 5, e004031. [Google Scholar] [CrossRef]
- Zhu, D.; Wang, H.; Zhang, J.; Zhang, X.; Xin, C.; Zhang, F.; Lee, Y.; Zhang, L.; Lian, K.; Yan, W.; et al. Irisin improves endothelial function in type 2 diabetes through reducing oxidative/nitrative stresses. J. Mol. Cell. Cardiol. 2015, 87, 138–147. [Google Scholar] [CrossRef]
- Fu, J.; Li, F.; Tang, Y.; Cai, L.; Zeng, C.; Yang, Y.; Yang, J. The Emerging Role of Irisin in Cardiovascular Diseases. J. Am. Heart Assoc. 2021, 10, e022453. [Google Scholar] [CrossRef]
- Li, H.; Zhang, C.; Liu, J.; Xie, W.; Xu, W.; Liang, F.; Huang, K.; He, X. Intraperitoneal administration of follistatin promotes adipocyte browning in high-fat diet-induced obese mice. PLoS ONE 2019, 14, e0220310. [Google Scholar] [CrossRef]
- Tschöpe, C.; Kherad, B.; Klein, O.; Lipp, A.; Blaschke, F.; Gutterman, D.; Burkhoff, D.; Hamdani, N.; Spillmann, F.; Van Linthout, S. Cardiac contractility modulation: Mechanisms of action in heart failure with reduced ejection fraction and beyond. Eur. J. Heart Fail. 2019, 21, 14–22. [Google Scholar] [CrossRef]
- Lecker, S.H.; Zavin, A.; Cao, P.; Arena, R.; Allsup, K.; Daniels, K.M.; Joseph, J.; Schulze, P.C.; Forman, D.E. Expression of the Irisin Precursor FNDC5 in Skeletal Muscle Correlates With Aerobic Exercise Performance in Patients With Heart Failure. Circ. Heart Fail. 2012, 5, 812–818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuo, Y.; Gleitsmann, K.; Mangner, N.; Werner, S.; Fischer, T.; Bowen, T.S.; Kricke, A.; Matsumoto, Y.; Kurabayashi, M.; Schuler, G.; et al. Fibronectin type III domain containing 5 expression in skeletal muscle in chronic heart failure-relevance of inflammatory cytokines. J. Cachexia Sarcopenia Muscle 2015, 6, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Chen, K.; Han, Y.; Zhu, H.; Zhou, X.; Tan, T.; Zeng, J.; Zhang, J.; Liu, Y.; Li, Y.; et al. Irisin Protects Heart Against Ischemia-Reperfusion Injury Through a SOD2-Dependent Mitochondria Mechanism. J. Cardiovasc. Pharmacol. 2018, 72, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Liao, Q.; Qu, S.; Tang, L.-X.; Li, L.-P.; He, D.-F.; Zeng, C.Y.; Wang, W.E. Irisin exerts a therapeutic effect against myocardial infarction via promoting angiogenesis. Acta Pharmacol. Sin. 2019, 40, 1314–1321. [Google Scholar] [CrossRef]
- Li, R.; Wang, X.; Wu, S.; Wu, Y.; Chen, H.; Xin, J.; Li, H.; Lan, J.; Xue, K.; Li, X.; et al. Irisin ameliorates angiotensin II-induced cardiomyocyte apoptosis through autophagy. J. Cell. Physiol. 2019, 234, 17578–17588. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, C.; Wu, H.-M.; Ma, Z.-G.; Tang, Q.-Z. Fibronectin type III domain-containing 5 in cardiovascular and metabolic diseases: A promising biomarker and therapeutic target. Acta Pharmacol. Sin. 2021, 42, 1390–1400. [Google Scholar] [CrossRef]
- Brailoiu, E.; Deliu, E.; Sporici, R.A.; Brailoiu, G.C. Irisin evokes bradycardia by activating cardiac-projecting neurons of nucleus ambiguus. Physiol. Rep. 2015, 3, e12419. [Google Scholar] [CrossRef]
- Deng, J.; Zhang, N.; Wang, Y.; Yang, C.; Wang, Y.; Xin, C.; Zhao, J.; Jin, Z.; Cao, F.; Zhang, Z. FNDC5/irisin improves the therapeutic efficacy of bone marrow-derived mesenchymal stem cells for myocardial infarction. Stem Cell Res. Ther. 2020, 11, 228. [Google Scholar] [CrossRef]
- Chen, R.-R.; Fan, X.-H.; Chen, G.; Zeng, G.-W.; Xue, Y.-G.; Liu, X.-T.; Wang, C.-Y. Irisin attenuates angiotensin II-induced cardiac fibrosis via Nrf2 mediated inhibition of ROS/ TGFβ1/Smad2/3 signaling axis. Chem. Biol. Interact. 2019, 302, 11–21. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, C.; Guo, J.; Chen, Y.; Meng, C. The Neuroprotective Effect of Irisin in Ischemic Stroke. Front. Aging Neurosci. 2020, 12, 588958. [Google Scholar] [CrossRef]
- Hirayama, K.; Ishii, H.; Kikuchi, R.; Suzuki, S.; Aoki, T.; Harada, K.; Sumi, T.; Negishi, Y.; Shibata, Y.; Tatami, Y.; et al. Clinical Impact of Circulating Irisin on Classified Coronary Plaque Characteristics. J. Appl. Lab. Med. 2018, 3, 79–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.-Q.; Huang, Y.-Y.; Gusdon, A.M.; Qu, S. Irisin: A new molecular marker and target in metabolic disorder. Lipids Health Dis. 2015, 14, 2. [Google Scholar] [CrossRef] [PubMed]
- Varela-Rodríguez, B.M.; Pena-Bello, L.; Juiz-Valiña, P.; Vidal-Bretal, B.; Cordido, F.; Sangiao-Alvarellos, S. FNDC5 expression and circulating irisin levels are modified by diet and hormonal conditions in hypothalamus, adipose tissue and muscle. Sci. Rep. 2016, 6, 29898. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Q. Mechanisms of the Effects of lcariin on Irisin/FNDC5 Expression in Skeletal Muscle. Master’s Thesis, Guangzhou University of Chinese Medicine, Guangzhou, China, 2019. [Google Scholar] [CrossRef]
- Chen, X.B. Effect of Balanophora polysaccharide on expression of PPARy, irisin and glucolipid metabolism in experimental diabetic rats. Chin. J. Pharmacol. Toxicol. 2018, 32, 400–406. [Google Scholar] [CrossRef]
- Leng, L.N. Changes and Significance of Serum Irisin and Leptin in Ghd Children with Recombinant Human Growth Hormone Therapy. Master’s Thesis, Hebei North University, Zhangjiakou, China, 2021. [Google Scholar] [CrossRef]
- Deng, W.J.; Ning, G.J.; Wang, J.; Zhuo, L. Effect of Levothyroxine Sodium on Serum Irisin Level, Blood Lipid, and Coagulation Function of Pregnant Metaphase Patients with Subclinical Hypothyroidism. China Med. Her. 2021, 18. Available online: https://www.yiyaodaobao.com.cn/EN/ (accessed on 29 June 2022).
- Nan, J.L.; Xue, L.Y.; He, W.S.; Han, X.F.; Fan, Y. The effect of orlistat on serum irisin level in male adults with abdominal obesity. Chin. J. Geriatr. Care 2015, 13. [Google Scholar] [CrossRef]
| Intervention Mode | Exercise Frequency | Exercise Duration | Tissue | Detection Method | Change | Reference |
|---|---|---|---|---|---|---|
| Aerobic exercise | 45 min/time | 8 weeks | Blood | ELISA | irisin↑; Compared with pre-exercise | [39] |
| Aerobic exercise | 40 min/time | 16 weeks | Blood | ELISA | irisin↑; Compared with pre-exercise | [40] |
| Resistance exercise | 60 min/time | 12 weeks | Blood | ELISA | irisin↑; Compared with pre-exercise | [41] |
| Resistance exercise | 55 min/time | 13 weeks | Blood | ELISA | irisin↑; Compared with pre-training | [42] |
| Low-impact AE | 1 h | / | Muscle | ELISA | irisin (no significance); Compared with pre- exercise | [35] |
| HIIT and moderate intensity continuous training | 33, 41 min/time 3 times/week, | 12 weeks | Blood | ELISA | irisin (no significance); Compared with pre- exercise | [43] |
| Resistance exercise (Progressive training) | / | 12 weeks | Blood | ELISA | irisin (no significance); Compared with pre- exercise | [44] |
| A single strenuous endurance exercise | 60 min/d | / | Blood | ELISA | irisin↑; Compared with pre- exercise | [45] |
| Resistance exercise | >60 min/d, 5 >times/week | 8 weeks | Blood | ELISA | irisin↑; Compared with pre- exercise | [46] |
| Model | Intervention | Detection Method | Change | Reference |
|---|---|---|---|---|
| Mouse | Leptin | PCR, ELISA | irisin↓FNDC5↑FNDC5 mRNA↑; compared to no injection | [93] |
| Mouse | Insulin | PCR, ELISA | irisin↓FNDC5 mRNA↓(muscle); compared to no injection | [93] |
| Mouse | Metformin | PCR | FNDC5 mRNA↓(muscle); compared to no injection | [93] |
| Mouse (Obesity) | Lcariin | PCR, WB, ELISA | PGC1-α↑ FNDC5mRNA↑ irisin↑; compared to no injection | [94] |
| Rat (DM) | BPS | ELISA | irisin↓; compared to no injection | [95] |
| Human (GHD) | rhGH | ELISA | irisin↑↑; compared to no injection | [96] |
| Human (SHT) | Levothyroxine sodium | ELISA | irisin↓; compared with those before treatment | [97] |
| Human (obesity) | Orlistat | ELISA | irisin↑; compared with those before treatment | [98] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, C.; Wei, A.; Wang, T. Irisin, an Effective Treatment for Cardiovascular Diseases? J. Cardiovasc. Dev. Dis. 2022, 9, 305. https://doi.org/10.3390/jcdd9090305
Liu C, Wei A, Wang T. Irisin, an Effective Treatment for Cardiovascular Diseases? Journal of Cardiovascular Development and Disease. 2022; 9(9):305. https://doi.org/10.3390/jcdd9090305
Chicago/Turabian StyleLiu, Chen, Aili Wei, and Tianhui Wang. 2022. "Irisin, an Effective Treatment for Cardiovascular Diseases?" Journal of Cardiovascular Development and Disease 9, no. 9: 305. https://doi.org/10.3390/jcdd9090305
APA StyleLiu, C., Wei, A., & Wang, T. (2022). Irisin, an Effective Treatment for Cardiovascular Diseases? Journal of Cardiovascular Development and Disease, 9(9), 305. https://doi.org/10.3390/jcdd9090305






