Physiological Effects of Training in Elite German Winter Sport Athletes: Sport Specific Remodeling Determined Using Echocardiographic Data and CPET Performance Parameters
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participating Athletes
2.3. Echocardiographic Assessment and CPET Performance Analysis
2.4. Statistical Analysis
2.5. Ethical Consideration
3. Results
3.1. Baseline Characteristics and Anthropometric Data
3.2. Morphological and Functional Cardiac Remodeling
3.3. Sport-Specific Physiological Performance by Laboratory CPET Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Armstrong, R.B.; Laughlin, M.H.; Rome, L.; Taylor, C.R. Metabolism of rats running up and down an incline. J. Appl. Physiol. 1983, 55, 518–521. [Google Scholar] [CrossRef] [PubMed]
- Balducci, P.; Clémençon, M.; Morel, B.; Quiniou, G.; Saboul, D.; Hautier, C.A. Comparison of Level and Graded Treadmill Tests to Evaluate Endurance Mountain Runners. J. Sports Sci. Med. 2016, 15, 239–246. [Google Scholar] [PubMed]
- Buskirk, E.R.; Kollias, J.; Akers, R.F.; Prokop, E.K.; Reategui, E.P. Maximal performance at altitude and on return from altitude in conditioned runners. J. Appl. Physiol. 1976, 23, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Duc, S.; Cassirame, J.; Durand, F. Physiology of Ski Mountaineering Racing. Int. J. Sports Med. 2011, 32, 856–863. [Google Scholar] [CrossRef]
- Menz, V.; Niedermeier, M.; Stehle, R.; Mugele, H.; Faulhaber, M. Assessment of Maximal Aerobic Capacity in Ski Mountaineering: A Laboratory-Based Study. Int. J. Environ. Res. Public Health 2021, 18, 7002. [Google Scholar] [CrossRef]
- Tosi, P.; Leonardi, A.; Schena, F. The energy cost of ski mountaineering: Effects of speed and ankle loading. J. Sport Med. Phys. Fit 2009, 49, 25–29. [Google Scholar]
- Bortolan, L.; Savoldelli, A.; Pellegrini, B.; Modena, R.; Sacchi, M.; Holmberg, H.-C.; Supej, M. Ski Mountaineering: Perspectives on a Novel Sport to Be Introduced at the 2026 Winter Olympic Games. Front. Physiol. 2021, 12, 737249. [Google Scholar] [CrossRef]
- Hoffman, M.D. Physiological comparisons of cross-country skiing techniques. Med. Sci. Sports Exerc. 1992, 24, 1023–1032. [Google Scholar] [CrossRef]
- Jonsson Kårström, M.; McGawley, K.; Laaksonen, M.S. Physiological Responses to Rifle Carriage During Roller-Skiing in Elite Biathletes. Front. Physiol. 2019, 10, 1519. [Google Scholar] [CrossRef]
- Laaksonen, M.S.; Andersson, E.; Jonsson Kårström, M.; Lindblom, H.; McGawley, K. Laboratory-Based Factors Predicting Skiing Performance in Female and Male Biathletes. Front. Sports Act. Living 2020, 2, 99. [Google Scholar] [CrossRef]
- Schöffl, V.; Pöppelmeier, O.; Emmler, J.; Schöffl, I.; Küpper, T.; Lutter, C. Ski Mountaineering–Evaluation of a Sports Specific Performance Diagnosis. Sportverletz. Sportschaden 2018, 32, 233–242. [Google Scholar] [CrossRef]
- Zimmermann, P.; Moser, O.; Eckstein, M.L.; Wüstenfeld, J.; Schöffl, V.; Zimmermann, L.; Braun, M.; Schöffl, I. Athlete’s Heart in Elite Biathlon, Nordic Cross—Country and Ski-Mountaineering Athletes: Cardiac Adaptions Determined Using Echocardiographic Data. J. Cardiovasc. Dev. Dis. 2021, 9, 8. [Google Scholar] [CrossRef]
- Schöffl, V.R.; Bösl, T.; Lutter, C. Ski mountaineering: Sports medical considerations for this new Olympic sport. Br. J. Sports Med. 2022, 56, 2–3. [Google Scholar] [CrossRef]
- Galanti, G.; Stefani, L.; Mascherini, G.; di Tante, V.; Toncelli, L. Left ventricular remodeling and the athlete’s heart, irrespective of quality load training. Cardiovasc. Ultrasound 2016, 14, 46. [Google Scholar] [CrossRef]
- Larsson, P.; Olofsson, P.; Jakobsson, E.; Burlin, L.; Henriksson-Larsén, K. Physiological predictors of performance in cross-country skiing from treadmill tests in male and female subjects. Scand. J. Med. Sci. Sports 2002, 12, 347–353. [Google Scholar] [CrossRef]
- Major Zs Csajági, E.; Kneffel Zs Kováts, T.; Szauder, I.; Sidó, Z.; Pavlik, G. Comparison of left and right ventricular adaptation in endurance-trained male athletes. Acta Physiol. Hung. 2015, 102, 23–33. [Google Scholar] [CrossRef]
- Pavlik, G.; Major Zs Varga-Pintér, B.; Jeserich, M.; Kneffel, Z.S. The athlete’s heart Part I (Review). Acta Physiol. Hung. 2010, 97, 337–353. [Google Scholar] [CrossRef]
- Schöffl, I.; Wüstenfeld, J.; Jones, G.; Dittrich, S.; Lutter, C.; Schöffl, V. Athlete’s Heart in Elite Sport Climbers: Cardiac Adaptations Determined Using ECG and Echocardiography Data. Wilderness Environ. Med. 2020, 31, 418–425. [Google Scholar] [CrossRef]
- Stefani, L.; Toncelli, L.; Gianassi, M.; Manetti, P.; di Tante, V.; Vono, M.R.C.; Moretti, A.; Cappelli, B.; Pedrizzetti, G.; Galanti, G. Two-dimensional tracking and TDI are consistent methods for evaluating myocardial longitudinal peak strain in left and right ventricle basal segments in athletes. Cardiovasc. Ultrasound 2007, 5, 7. [Google Scholar] [CrossRef]
- Szauder, I.; Kovács, A.; Pavlik, G. Comparison of left ventricular mechanics in runners versus bodybuilders using speckle tracking echocardiography. Cardiovasc. Ultrasound 2015, 13, 7. [Google Scholar] [CrossRef]
- Zimmermann, P.; Moser, O.; Edelmann, F.; Schöffl, V.; Eckstein, M.L.; Braun, M. Electrical and Structural Adaption of Athlete’s Heart and the Impact on Training and Recovery Management in Professional Basketball Players: A Retrospective Observational Study. Front. Physiol. 2022, 13, 142. [Google Scholar] [CrossRef]
- Zimmermann, P.; Wüstenfeld, J.; Zimmermann, L.; Schöffl, V.; Schöffl, I. Physiological Aspects of World Elite Competitive German Winter Sport Athletes. Int. J. Environ. Res. Public Health 2022, 19, 5620. [Google Scholar] [CrossRef]
- Evangelista, A.; Flachskampf, F.; Lancellotti, P.; Badano, L.; Aguilar, R.; Monaghan, M.; Zamorano, J.; Nihoyannopoulos, P. European Association of Echocardiography recommendations for standardization of performance, digital storage and reporting of echocardiographic studies. Eur. J. Echocardiogr. 2008, 9, 438–448. [Google Scholar] [CrossRef]
- Hagendorff, A.; Fehske, W.; Flachskampf, F.A.; Helfen, A.; Kreidel, F.; Kruck, S.; la Rosée, K.; Tiemann, K.; Voigt, J.-U.; von Bardeleben, R.S.; et al. Manual zur Indikation und Durchführung der Echokardiographie–Update 2020 der Deutschen Gesellschaft für Kardiologie. Kardiologe 2020, 14, 396–431. [Google Scholar] [CrossRef]
- Lang, R.; Bierig, M.; Devereux, R.; Flachskampf, F.; Foster, E.; Pellikka, P.; Picard, M.; Roman, M.; Seward, J.; Shanewise, J. Recommendations for chamber quantification. Eur. J. Echocardiogr. 2006, 7, 79–108. [Google Scholar] [CrossRef]
- Hashem, M.-S.; Kalashyan, H.; Choy, J.; Chiew, S.K.; Shawki, A.-H.; Dawood, A.H.; Becher, H. Left Ventricular Relative Wall Thickness Versus Left Ventricular Mass Index in Non-Cardioembolic Stroke Patients. Medicine 2015, 94, e872. [Google Scholar] [CrossRef]
- Fletcher, G.F.; Balady, G.J.; Amsterdam, E.A.; Chaitman, B.; Eckel, R.; Fleg, J.; Froelicher, V.F.; Leon, A.S.; Piña, I.L.; Rodney, R.; et al. Exercise Standards for Testing and Training. Circulation 2001, 104, 1694–1740. [Google Scholar] [CrossRef]
- Mezzani, A. Cardiopulmonary Exercise Testing: Basics of Methodology and Measurements. Ann. Am. Thorac. Soc. 2017, 14, S3–S11. [Google Scholar] [CrossRef]
- Harriss, D.J.; MacSween, A.; Atkinson, G. Ethical Standards in Sport and Exercise Science Research: 2020 Update. Int. J. Sports Med. 2019, 40, 813–817. [Google Scholar] [CrossRef]
- Tønnessen, E.; Haugen, T.A.; Hem, E.; Leirstein, S.; Seiler, S. Maximal aerobic capacity in the winter-Olympics endurance disciplines: Olympic-medal benchmarks for the time period 1990–2013. Int. J. Sports Physiol. Perform. 2015, 10, 835–839. [Google Scholar] [CrossRef]
- Hébert-Losier, K.; Zinner, C.; Platt, S.; Stöggl, T.; Holmberg, H.-C. Factors that Influence the Performance of Elite Sprint Cross-Country Skiers. Sports Med. 2017, 47, 319–342. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.D.; Clifford, P.S. Physiological aspects of competitive cross-country skiing. J. Sports Sci. 1992, 10, 3–27. [Google Scholar] [CrossRef] [PubMed]
- Losnegard, T. Energy system contribution during competitive cross-country skiing. Eur. J. Appl. Physiol. 2019, 119, 1675–1690. [Google Scholar] [CrossRef] [PubMed]
- Talsnes, R.K.; Solli, G.S.; Kocbach, J.; Torvik, P.-Ø.; Sandbakk, Ø. Laboratory- and field-based performance-predictions in cross-country skiing and roller-skiing. PLoS ONE 2021, 16, e0256662. [Google Scholar] [CrossRef] [PubMed]
- Utomi, V.; Oxborough, D.; Whyte, G.P.; Somauroo, J.; Sharma, S.; Shave, R.; Atkinson, G.; George, K. Systematic review and meta-analysis of training mode, imaging modality and body size influences on the morphology and function of the male athlete’s heart. Heart 2013, 99, 1727–1733. [Google Scholar] [CrossRef] [PubMed]
- Fagard, R. Athlete’s heart. Heart 2003, 89, 1455–1461. [Google Scholar] [CrossRef]
- Iskandar, A.; Mujtaba, M.T.; Thompson, P.D. Left Atrium Size in Elite Athletes. JACC Cardiovasc. Imaging 2015, 8, 753–762. [Google Scholar] [CrossRef]
- Wonisch, M.; Fruwald, F.M.; Hofmann, P.; Hödl, R.; Klein, W.; Kraxner, W.; Maier, R.; Pokan, R.; Smekal, G.; Watzinger, N. Spiroergometrie in der Kardiologie–Grundlagen der Physiologie und Terminologie. J. Kardiol.-Austrian J. Cardiol. 2003, 10, 383–390. [Google Scholar]
- Mujika, I.; Padilla, S.; Pyne, D.; Busso, T. Physiological changes associated with the pre-event taper in athletes. Sports Med. 2004, 34, 891–927. [Google Scholar] [CrossRef]
- Kasikcioglu, E.; Oflaz, H.; Akhan, H.; Kayserilioglu, A.; Umman, B.; Bugra, Z.; Erzengin, F. Left Atrial Geometric and Functional Remodeling in Athletes. Int. J. Sports Med. 2006, 27, 267–271. [Google Scholar] [CrossRef]
- Klasnja, A.V.; Jakovljevic, D.G.; Barak, O.F.; Popadic Gacesa, J.Z.; Lukac, D.D.; Grujic, N.G. Cardiac power output and its response to exercise in athletes and non-athletes. Clin. Physiol. Funct. Imaging 2013, 33, 201–205. [Google Scholar] [CrossRef]
- Mandraffino, G.; Imbalzano, E.; lo Gullo, A.; Zito, C.; Morace, C.; Cinquegrani, M.; Savarino, F.; Oreto, L.; Giuffrida, C.; Carerj, S.; et al. Abnormal left ventricular global strain during exercise-test in young healthy smokers. Sci. Rep. 2020, 10, 5700. [Google Scholar] [CrossRef]
- Pagourelias, E.D.; Mirea, O.; Duchenne, J.; van Cleemput, J.; Delforge, M.; Bogaert, J.; Kuznetsova, T.; Voigt, J.-U. Echo Parameters for Differential Diagnosis in Cardiac Amyloidosis. Circ. Cardiovasc. Imaging 2017, 10, e005588. [Google Scholar] [CrossRef]
- Yang, C.G.; Ma, C.S.; Fan, L.; Su, B.; Wang, Y.X.; Jiang, G.D.; Zhou, B.Y. The value of left ventricular longitudinal strain in the diagnosis and differential diagnosis of myocardial amyloidosis. Zhonghua Yi Xue Za Zhi 2020, 100, 3431–3436. [Google Scholar]
- Cassirame, J.; Tordi, N.; Fabre, N.; Duc, S.; Durand, F.; Mourot, L. Heart rate variability to assess ventilatory threshold in ski-mountaineering. Eur. J. Sport Sci. 2015, 15, 615–622. [Google Scholar] [CrossRef]
- Schupfner, R.; Pecher, S.; Pfeifer, E.; Stumpf, C. Physiological factors which influence the performance potential of athletes: Analysis of sports medicine performance testing in Nordic combined. Physician Sportsmed. 2021, 49, 106–115. [Google Scholar] [CrossRef]
- Rusko, H.K. Development of aerobic power in relation to age and training in cross-country skiers. Med. Sci. Sports Exerc. 1992, 24, 1040–1047. [Google Scholar] [CrossRef]

| Athlete | Average Years of Training | Pre-Season | In-Season | ||||
|---|---|---|---|---|---|---|---|
| 10 Training Hours per Week | 20–25 Training Hours per Week | ||||||
| Endurance | Strength | Movement Specific/Flexibility | Endurance | Strength | Movement Specific/Flexibility | ||
| Ski-Mo | 5 ± 3 | 90% | 5% | 5% | 90% | 7% | 3% |
| NCC | 15 ± 5.3 | 84% | 10% | 6% | 89% | 8% | 3% |
| Bia | 14 ± 4.5 | 76% | 11% | 13% | 87% | 9% | 4% |
| Ski-Mo n = 9 | NCC n = 10 | Biathletes n = 12 | ||||
|---|---|---|---|---|---|---|
| Male | Female | Male | Female | Male | Female | |
| n = 5 | n = 4 | n = 6 | n = 4 | n = 6 | n = 6 | |
| Age (y) | 21.4 ± 1.8 | 20.8 ± 2.4 | 26.3 ± 4.1 | 25.5 ± 0.5 | 27.3 ± 3.6 | 29.0 ± 3.2 |
| Height (cm) | 178.0 ± 3.9 | 163.5 ± 8.8 | 181.3 ± 4.7 | 171.2 ± 5.8 | 180.9 ± 5.1 | 172.8 ± 3.7 |
| Weight (kg) | 66.5 ± 0.8 | 53.2 ± 6.5 | 72.0 ± 3.0 | 63.4 ± 5.9 | 77.1 ± 3.7 | 62.5 ± 4.1 |
| Resting blood pressure | 118 ± 5.4 | 100 ± 8.2 | 125 ± 8.3 | 105 ± 7.2 | 117 ± 7.6 | 108 ± 6.2 |
| systolic/diastolic (mmHg) | 78 ± 4.0 | 72 ± 1.5 | 78 ± 2.9 | 71 ± 3.8 | 77 ± 2.2 | 70 ± 3.3 |
| Resting heart rate (bpm) | 41 ± 4.6 | 44 ± 4.5 | 42 ± 3.6 | 46 ± 5.1 | 41 ± 4.2 | 45 ± 5.1 |
| Heart rate VT2 (bpm) | 133 ± 22 | 132.3 ± 1.9 | 136.3 ± 11.6 | 128.3 ± 9.1 | 148.5 ± 20.9 | 134.5 ± 8.1 |
| Maximum heart rate (bpm) | 185.6 ± 6.3 | 171.8 ± 2.5 | 183 ± 14.3 | 173.8 ± 4.0 | 179.5 ± 10.3 | 181.0 ± 12.9 |
| BMI (body mass index in kg/m2) | 19.9 ± 1.4 | 19.8 ± 0.4 | 22.0 ± 1.1 | 22.0 ± 1.1 | 23.6 ± 0.9 | 20.9 ± 1.0 |
| BSA (body surface area in m2) | 1.70 ± 0.06 | 1.61 ± 0.12 | 1.88 ± 0.04 | 1.81 ± 0.07 | 1.92 ± 0.04 | 1.77 ± 0.05 |
| Ski-Mo (I) n = 10 | NCC (II) n = 10 | Biathletes (III) n = 12 | p-Value | ||||
|---|---|---|---|---|---|---|---|
| Male Female | Male Female | Male Female | |||||
| n = 6 | n = 4 | n = 6 | n = 4 | n = 6 | n = 6 | ||
| LV edd (mm) | 50.83 ± 4.22 | 45.25 ± 5.96 | 55.50 ± 3.83 | 50.75 ± 3.50 | 55.50 ± 5.24 | 49.50 ± 1.52 | ns |
| 48.6 ± 5.48 | 53.6 ± 4.27 | 52.5 ± 4.83 | |||||
| LV Mass Index (g/m) | 97.2 ± 25.2 | 76.3 ± 26.7 | 130.7 ± 16.5 | 106 ± 16.4 | 133.5 ± 20.6 | 102.3 ± 14.8 | 0.0078 * |
| −88.8 ± 26.6 * | 120.8 ± 20.1 * | 117.9 ± 23.6 * | |||||
| Relative wall Thickness RWT | 0.38 ± 0.03 | 0.34 ± 0.06 | 0.40 ± 0.04 | 0.41 ± 0.04 | 0.40 ± 0.04 | 0.42 ± 0.04 | Ski-Mo vs. NCC 0.0230 * Ski-Mo vs. Bia 0.0230 * |
| 0.37 ± 0.05 | 0.41 ± 0.03 | 0.41 ± 0.04 | |||||
| IVSd (mm) | 8.67 ± 1.97 | 8.25 ± 2.50 | 11.00 ± 0.63 | 10.50 ± 0.58 | 10.83 ± 0.98 | 9.67 ± 1.37 | Ski-Mo vs. NC C 0.0266 * Ski-Mo vs. Bia 0.0337 * |
| 8.5 ± 2.07 | 10.4 ± 1.17 | 10.3 ± 1.29 | |||||
| LVPWs (mm) | 3.97 ± 11.03 | 7.75 ± 1.50 | 11.17 ± 0.41 | 10.50 ± 0.58 | 12.33 ± 2.07 | 10.17 ± 1.17 | Ski-Mo vs. NC C 0.0161 * Ski-Mo vs. Bia 0.0030 * |
| 8.9 ± 1.52 | 10.9 ± 0.57 | 11.3 ± 1.96 | |||||
| E/A | 2.18 ± 0.58 | 1.98 ± 0.17 | 2.48 ± 0.26 | 2.40 ± 0.77 | 1.97 ± 0.52 | 1.75 ± 0.40 | NCC vs. Bia 0.0166 * |
| 2.1 ± 0.45 | 2.5 ± 0.49 | 19 ± 0.46 | |||||
| E/E’ | 6.75 ± 1.71 | 7 ± 1.79 | 6.80 ± 0.86 | 6.13 ± 1.22 | 7 ± 0.86 | 6.37 ± 1.04 | ns |
| 6.9 ± 1.66 | 6.4 ± 1.09 | 6.7 ± 0.97 | |||||
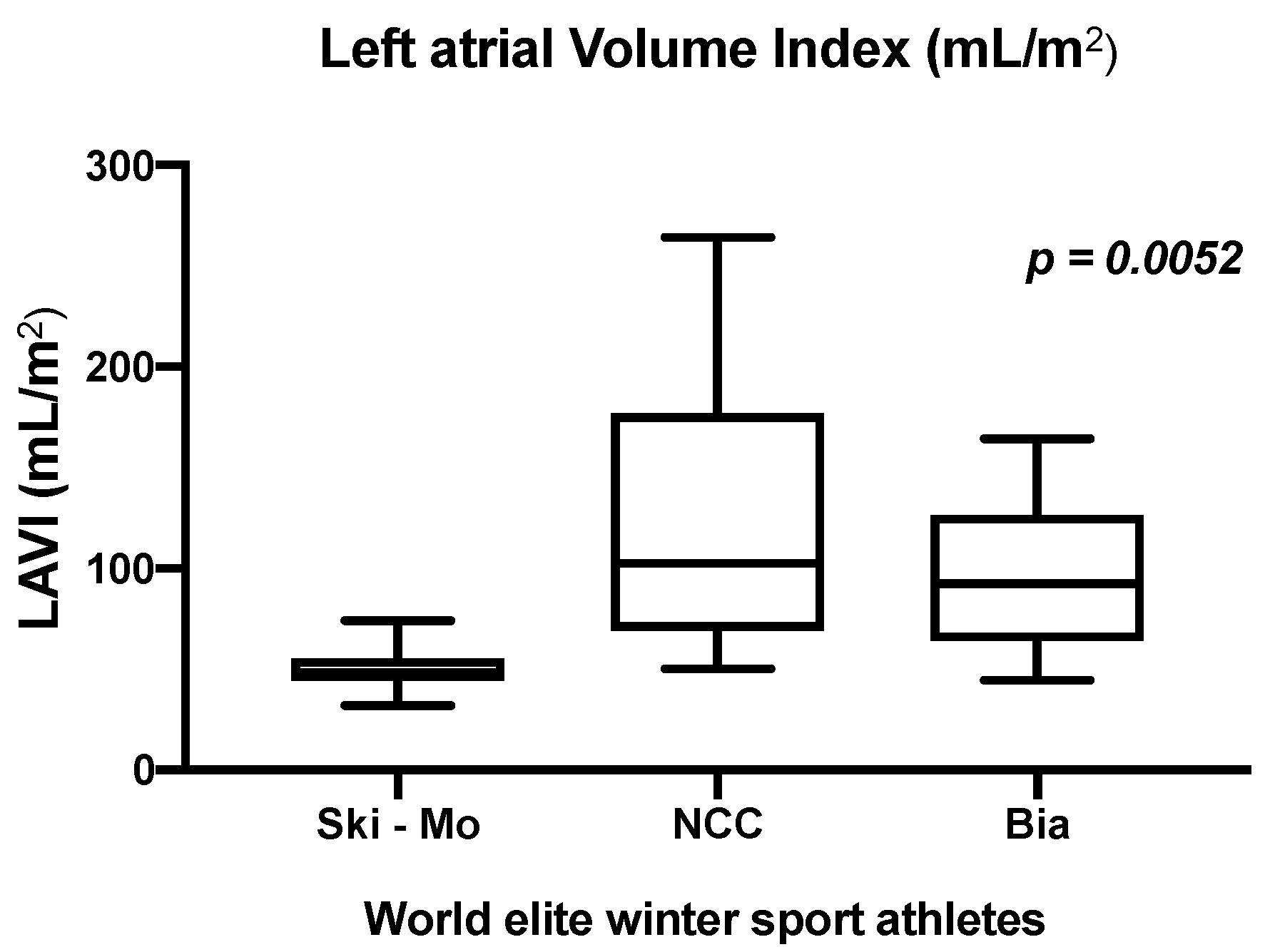
| LAVI (mL/m2) | 51.83 ± 12.1 | 46.25 ± 11.1 | 150 ± 84.58 | 89.3 ± 45.7 | 117.5 ± 37.7 | 72.8 ± 19.6 | 0.0052 * |
| 49.6 ± 11.4 | 125.7 ± 75.2 | 95.2 ± 36.9 | |||||
| RA (cm2) | 19.17 ± 3.87 | 16.75 ± 2.87 | 24.83 ± 3.73 | 18.28 ± 4.72 | 20.78 ± 3.64 | 15.50 ± 2.40 | ns |
| 18.2 ± 3.55 | 22.2 ± 5.16 | 18.1 ± 4.03 | |||||
| GLS | −18.26 ± 2.21 | −18.83 ± 2.93 | 21.21 ± 1.99 | −23.25 ± 3.23 | 22.62 ± 1.26 | 22.34 ± 1.42 | 0.0003 * |
| −18.5 ± 2.38 | −22.0 ± 2.61 | −22.5 ± 1.29 | |||||
| Ski-Mo (I) | NCC (II) | Biathletes (II) | p-Value Male | p-Value Female | Overall p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|
| Male | Female | Male | Female | Male | Female | ||||
| VE maximum (L) | 134.9 ± 24.6 | 109.2 ± 20.6 | 166.2 ± 28.4 | 118 2 ± 23.8 | 175.8 ± 11.7 | 125.4 ± 9.1 | Ski-Mo vs. Bia 0.0087 * | ns | Ski-Mo vs. Bia 0.0409 * |
| 123.5 ± 25.4 | 147.0 ± 3 5.4 | 150.6 ± 28.1 | |||||||
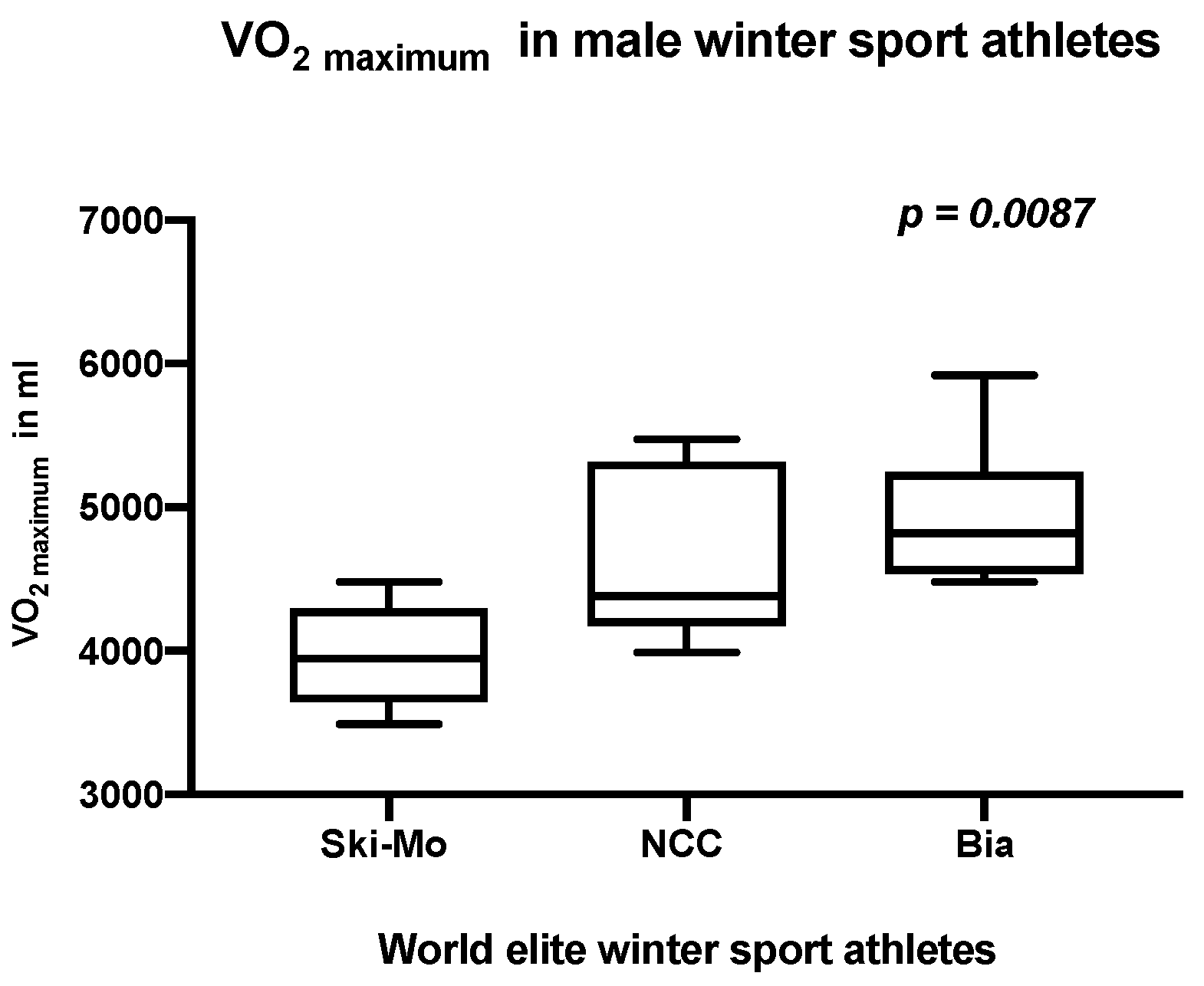
| VO2 maximum (mL) | 3964.8 ± 1367.8 | 3021.3 ± 515.1 | 4620.8 ± 603.8 | 3315. 3 ± 576.0 | 4935.2 ± 525.1 | 3555. 7 ± 274.7 | Ski-Mo vs. Bia 0.0087 * | Ski-Mo vs. Bia 0.0381 * | ns |
| 3545.4 ± 643.7 | 4098.6 ± 876.2 | 4245. 4 ± 823.8 | |||||||
| VO2/kg maximum (mL/kg) | 65.0 ± 7.9 | 57.4 ± 4.5 | 64.5 ± 7.1 | 52.7 ± 4.9 | 64.6 ± 4.4 | 57.4 ± 2.3 | ns | ns | ns |
| 61.6 ± 7.5 | 59.7 ± 8.6 | 61.0 ± 5.0 | |||||||
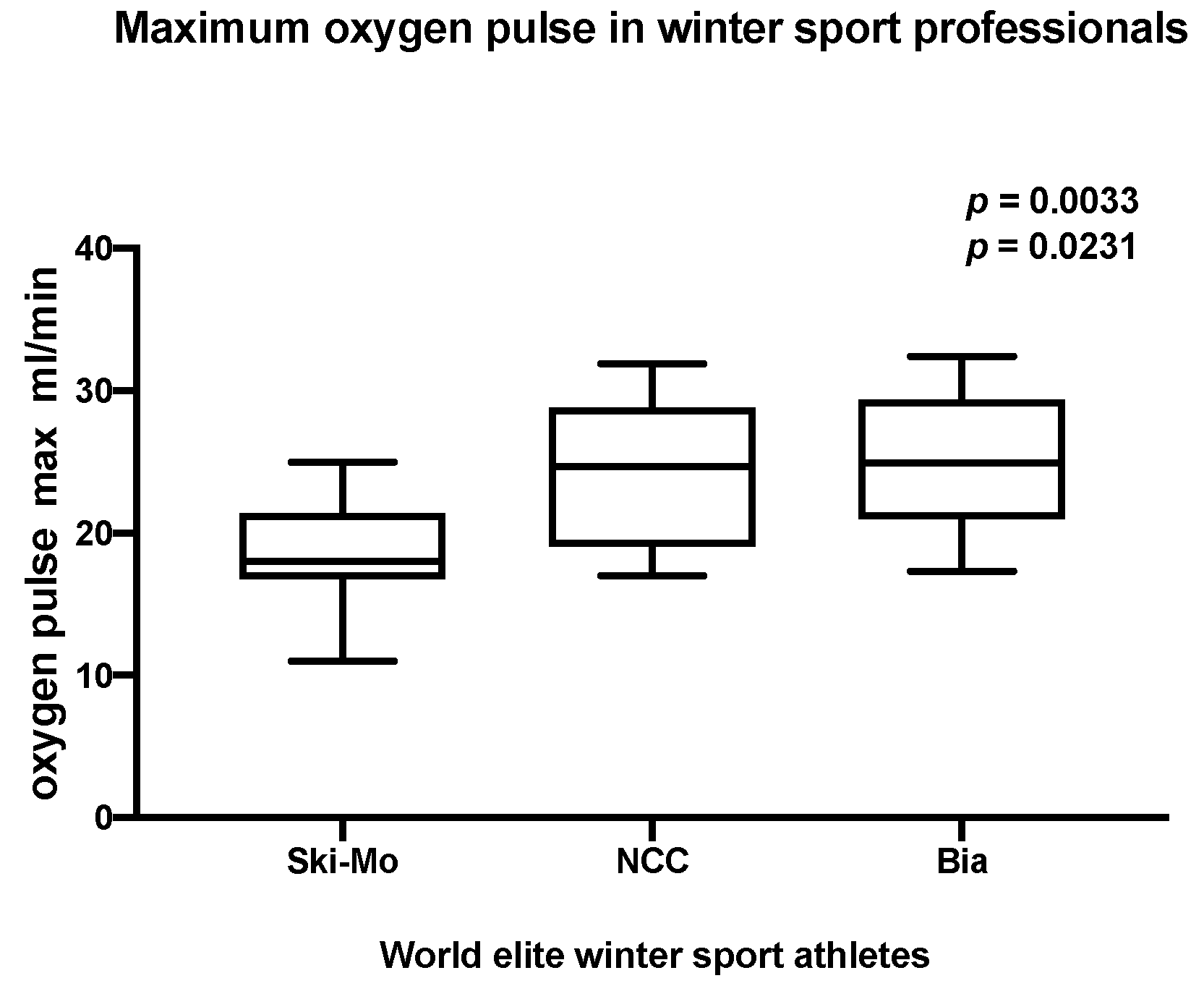
| Oxygen pulse maximum (mL/min) | 20.8 ± 30 | 15.6 ± 31 | 26.9 ± 4.2 | 19.4 ± 31 | 27.8 ± 3.2 | 22. 8 ± 5.4 | Ski-Mo vs. NCC 0.0303 * Ski-Mo vs. Bia 0.0260 * | Ski-Mo vs. Bia 0.0190 * | Ski-Mo vs. NCC 0.0231 * Ski-Mo vs. Bia 0.0033 * |
| 18.5 ± 4.0 | 23.9 ± 5.3 | 25. 3 ± 4.9 | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zimmermann, P.; Schöffl, I.; Schöffl, V.; Zimmermann, L.; Eckstein, M.L.; Moser, O.; Wüstenfeld, J. Physiological Effects of Training in Elite German Winter Sport Athletes: Sport Specific Remodeling Determined Using Echocardiographic Data and CPET Performance Parameters. J. Cardiovasc. Dev. Dis. 2022, 9, 235. https://doi.org/10.3390/jcdd9080235
Zimmermann P, Schöffl I, Schöffl V, Zimmermann L, Eckstein ML, Moser O, Wüstenfeld J. Physiological Effects of Training in Elite German Winter Sport Athletes: Sport Specific Remodeling Determined Using Echocardiographic Data and CPET Performance Parameters. Journal of Cardiovascular Development and Disease. 2022; 9(8):235. https://doi.org/10.3390/jcdd9080235
Chicago/Turabian StyleZimmermann, Paul, Isabelle Schöffl, Volker Schöffl, Lukas Zimmermann, Max L. Eckstein, Othmar Moser, and Jan Wüstenfeld. 2022. "Physiological Effects of Training in Elite German Winter Sport Athletes: Sport Specific Remodeling Determined Using Echocardiographic Data and CPET Performance Parameters" Journal of Cardiovascular Development and Disease 9, no. 8: 235. https://doi.org/10.3390/jcdd9080235
APA StyleZimmermann, P., Schöffl, I., Schöffl, V., Zimmermann, L., Eckstein, M. L., Moser, O., & Wüstenfeld, J. (2022). Physiological Effects of Training in Elite German Winter Sport Athletes: Sport Specific Remodeling Determined Using Echocardiographic Data and CPET Performance Parameters. Journal of Cardiovascular Development and Disease, 9(8), 235. https://doi.org/10.3390/jcdd9080235







