A Review of the Therapeutic Efficacy and Safety of Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes in Preclinical Models of Subacute and Chronic Myocardial Infarction
Abstract
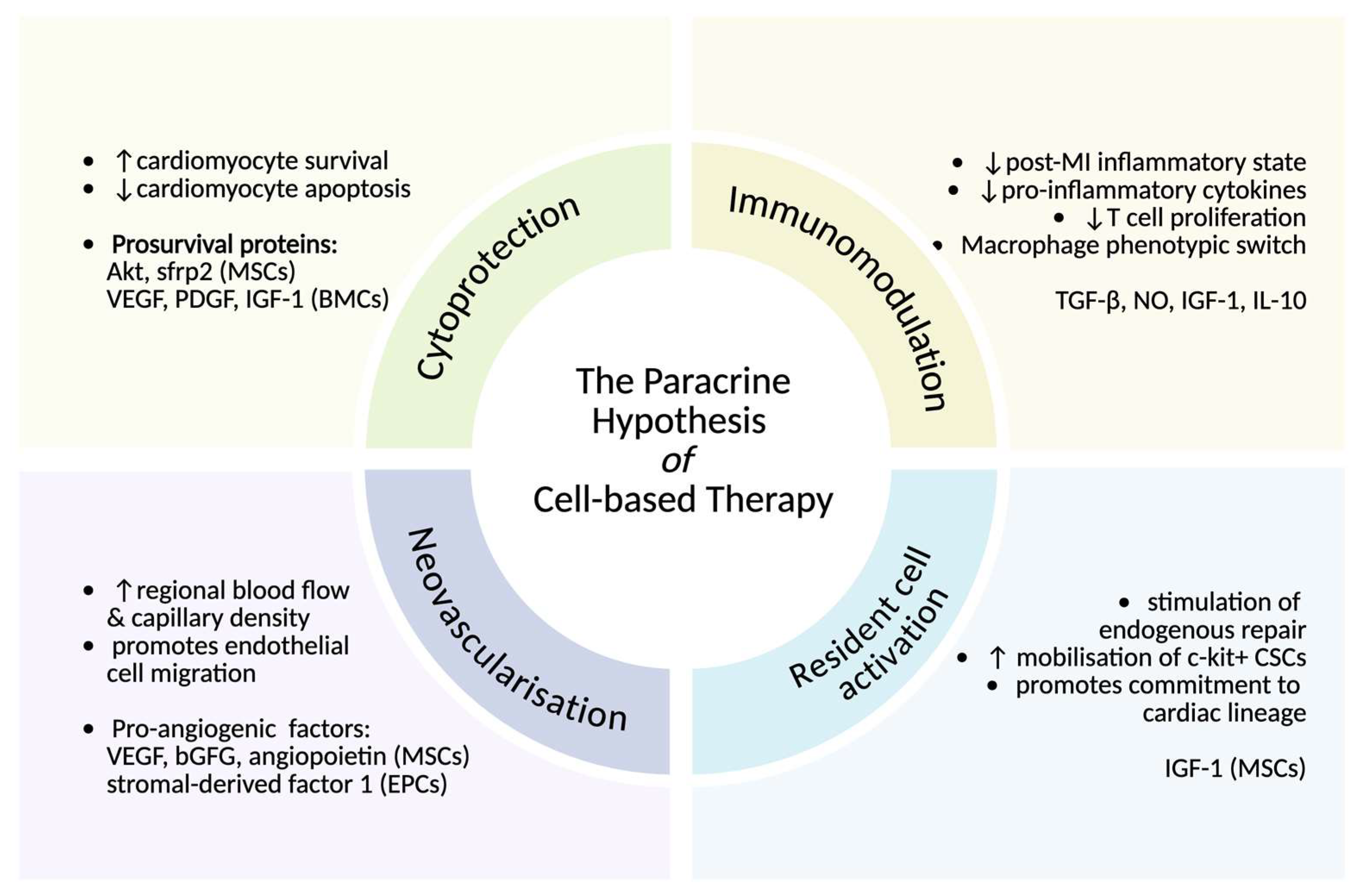
1. Introduction
2. Prior Research on Cell-Based Therapies
3. Studies Investigating the Therapeutic Use of Derived Cardiomyocytes in the Context of Ischemic Heart Disease
3.1. General Characteristics
3.2. Effect of Transplantation of hiPSC-CMs on Myocardial Function and Structure
3.3. Effect of Transplantation of hiPSC-CMs on Infarct and Scar Size
3.4. Effect of Transplanted hiPSC-CMs on Outcomes of Survival, Retention, and Structural and Functional Integration
3.5. Effect of Transplanted hiPSC-CMs on In Vivo Remodeling of Native Myocardium
3.5.1. Changes in Native CM Size and Fibrotic Area
3.5.2. Changes in Vascular Density
3.5.3. Changes in Cytokine Expression
3.6. Safety and Tolerance
3.6.1. Incidence and Characterization of Post-Transplantation Arrhythmia After the Transplantation of hiPSC-CMs
3.6.2. Evidence of Tumor Formation After the Transplantation of hiPSC-CMs
3.6.3. Evidence of an Immune Response, Immune Rejection, and Immunosuppressive Regimens Used
4. General Conclusions
4.1. hiPSC-CMs Cause a Dose-Dependent Long-Term Improvement in Myocardial Contractility Post-MI
4.2. Co-Transplantation with Auxiliary Cells Enhances the Post-Transplantation Therapeutic Effect of hiPSC-CMs
4.3. Pre-Transplantation Vascularity Is Crucial to the Long-Term Engraftment and Survival of hiPSC-CMs In Vivo
4.4. hiPSC-CMs Exert In Vivo Paracrine Effects in the Short Term That Are Sustained in the Longer Term
4.5. hiPSC-CMs Attenuate Post-MI Ventricular Remodeling
4.6. Therapeutic Efficacy and Safety of hiPSC-CMs Are Influenced by the Nature of the Construct
4.7. Transplantation of Pre-Differentiated hiPSC-CMs Is Safe with a Low Incidence of Immune Rejection or Tumor Formation
4.8. Preliminary Findings from Clinical Trials Show Similar Outcomes to Animal Studies, Irrespective of Construct
5. Limitations of the Current Published Studies
6. Future Directions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- GBD 2015 Mortality and Causes of Death Collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef]
- Bergmann, O.; Zdunek, S.; Felker, A.; Salehpour, M.; Alkass, K.; Bernard, S.; Sjostrom, S.L.; Szewczykowska, M.; Jackowska, T.; Dos Remedios, C.; et al. Dynamics of cell generation and turnover in the human heart. Cell 2015, 161, 1566–1575. [Google Scholar] [CrossRef]
- Fu, X.; Khalil, H.; Kanisicak, O.; Boyer, J.G.; Vagnozzi, R.J.; Maliken, B.D.; Sargent, M.A.; Prasad, V.; Valiente-Alandi, I.; Blaxall, B.C.; et al. Specialized fibroblast differentiated states underlie scar formation in the infarcted mouse heart. J. Clin. Investig. 2018, 128, 2127–2143. [Google Scholar] [CrossRef] [PubMed]
- Richardson, W.J.; Clarke, S.A.; Quinn, T.A.; Holmes, J.W. Physiological implications of myocardial scar structure. Compr. Physiol. 2015, 5, 1877–1909. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Weber, K.T. Infarct scar: A dynamic tissue. Cardiovasc. Res. 2000, 46, 250–256. [Google Scholar] [CrossRef]
- Gibb, A.A.; Lazaropoulos, M.P.; Elrod, J.W. Myofibroblasts and fibrosis: Mitochondrial and metabolic control of cellular differentiation. Circ. Res. 2020, 127, 427–447. [Google Scholar] [CrossRef]
- Chaher, N.; Lacerda, S.; Digilio, G.; Padovan, S.; Gao, L.; Lavin, B.; Stefania, R.; Velasco, C.; Cruz, G.; Prieto, C.; et al. Non-invasive in vivo imaging of changes in Collagen III turnover in myocardial fibrosis. npj Imaging 2024, 2, 33. [Google Scholar] [CrossRef]
- Pahlm, U.S.; Ubachs, J.F.; Heiberg, E.; Engblom, H.; Erlinge, D.; Götberg, M.; Arheden, H. Regional wall function before and after acute myocardial infarction; an experimental study in pigs. BMC Cardiovasc. Disord. 2014, 14, 118. [Google Scholar] [CrossRef]
- Tromp, J.; Ouwerkerk, W.; van Veldhuisen, D.J.; Hillege, H.L.; Richards, A.M.; van der Meer, P.; Anand, I.S.; Lam, C.S.; Voors, A.A. A systematic review and network meta-analysis of pharmacological treatment of heart failure with reduced ejection fraction. Heart Fail. 2022, 10, 73–84. [Google Scholar] [CrossRef]
- Menasché, P.; Hagège, A.A.; Scorsin, M.; Pouzet, B.; Desnos, M.; Duboc, D.; Schwartz, K.; Vilquin, J.T.; Marolleau, J.P. Myoblast transplantation for heart failure. Lancet 2001, 357, 279–280. [Google Scholar] [CrossRef] [PubMed]
- Reinecke, H.; MacDonald, G.H.; Hauschka, S.D.; Murry, C.E. Electromechanical coupling between skeletal and cardiac muscleImplications for infarct repair. J. Cell Biol. 2000, 149, 731–740. [Google Scholar] [CrossRef]
- Reinecke, H.; Poppa, V.; Murry, C.E. Skeletal muscle stem cells do not transdifferentiate into cardiomyocytes after cardiac grafting. J. Mol. Cell. Cardiol. 2002, 34, 241–249. [Google Scholar] [CrossRef]
- Toma, C.; Pittenger, M.F.; Cahill, K.S.; Byrne, B.J.; Kessler, P.D. Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation 2002, 105, 93–98. [Google Scholar] [CrossRef]
- Williams, A.R.; Hatzistergos, K.E.; Addicott, B.; McCall, F.; Carvalho, D.; Suncion, V.; Morales, A.R.; Da Silva, J.; Sussman, M.A.; Heldman, A.W.; et al. Enhanced effect of combining human cardiac stem cells and bone marrow mesenchymal stem cells to reduce infarct size and to restore cardiac function after myocardial infarction. Circulation 2013, 127, 213–223. [Google Scholar] [CrossRef]
- Shake, J.G.; Gruber, P.J.; Baumgartner, W.A.; Senechal, G.; Meyers, J.; Redmond, J.M.; Pittenger, M.F.; Martin, B.J. Mesenchymal stem cell implantation in a swine myocardial infarct model: Engraftment and functional effects. Ann. Thorac. Surg. 2002, 73, 1919–1926. [Google Scholar] [CrossRef]
- Hodgkinson, C.P.; Bareja, A.; Gomez, J.A.; Dzau, V.J. Emerging concepts in paracrine mechanisms in regenerative cardiovascular medicine and biology. Circ. Res. 2016, 118, 95–107. [Google Scholar] [CrossRef]
- Lee, H.; Cho, H.J.; Han, Y.; Lee, S.H. Mid-to long-term efficacy and safety of stem cell therapy for acute myocardial infarction: A systematic review and meta-analysis. Stem Cell Res. Ther. 2024, 15, 290. [Google Scholar] [CrossRef] [PubMed]
- Kanelidis, A.J.; Premer, C.; Lopez, J.; Balkan, W.; Hare, J.M. Route of delivery modulates the efficacy of mesenchymal stem cell therapy for myocardial infarction: A meta-analysis of preclinical studies and clinical trials. Circ. Res. 2017, 120, 1139–1150. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Yu, Y.; Hu, S.; Chen, Y.; Shen, Z. The therapeutic potential of mesenchymal stem cells for cardiovascular diseases. Cell Death Dis. 2020, 11, 349. [Google Scholar] [CrossRef]
- Li, T.S.; Cheng, K.; Malliaras, K.; Smith, R.R.; Zhang, Y.; Sun, B.; Matsushita, N.; Blusztajn, A.; Terrovitis, J.; Kusuoka, H.; et al. Direct comparison of different stem cell types and subpopulations reveals superior paracrine potency and myocardial repair efficacy with cardiosphere-derived cells. J. Am. Coll. Cardiol. 2012, 59, 942–953. [Google Scholar] [CrossRef] [PubMed]
- Zwetsloot, P.P.; Végh, A.M.; Jansen of Lorkeers, S.J.; Van Hout, G.P.; Currie, G.L.; Sena, E.S.; Gremmels, H.; Buikema, J.W.; Goumans, M.J.; Macleod, M.R.; et al. Cardiac stem cell treatment in myocardial infarction: A systematic review and meta-analysis of preclinical studies. Circ. Res. 2016, 118, 1223–1232. [Google Scholar] [CrossRef]
- Makkar, R.R.; Smith, R.R.; Cheng, K.E.; Malliaras, K.; Thomson, L.E.; Berman, D.; Czer, L.S.; Marbán, L.; Mendizabal, A.; Johnston, P.V.; et al. Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): A prospective, randomised phase 1 trial. Lancet 2012, 379, 895–904. [Google Scholar] [CrossRef]
- Caspi, O.; Huber, I.; Kehat, I.; Habib, M.; Arbel, G.; Gepstein, A.; Yankelson, L.; Aronson, D.; Beyar, R.; Gepstein, L. Transplantation of human embryonic stem cell-derived cardiomyocytes improves myocardial performance in infarcted rat hearts. J. Am. Coll. Cardiol. 2007, 50, 1884–1893. [Google Scholar] [CrossRef] [PubMed]
- Foo, K.S.; Lehtinen, M.L.; Leung, C.Y.; Lian, X.; Xu, J.; Keung, W.; Geng, L.; Kolstad, T.R.; Thams, S.; Wong, A.O.; et al. Human ISL1+ ventricular progenitors self-assemble into an in vivo functional heart patch and preserve cardiac function post infarction. Mol. Ther. 2018, 26, 1644–1659. [Google Scholar] [CrossRef]
- Menasché, P.; Vanneaux, V.; Hagège, A.; Bel, A.; Cholley, B.; Parouchev, A.; Cacciapuoti, I.; Al-Daccak, R.; Benhamouda, N.; Blons, H.; et al. Transplantation of human embryonic stem cell–derived cardiovascular progenitors for severe ischemic left ventricular dysfunction. J. Am. Coll. Cardiol. 2018, 71, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef]
- Ben-David, U.; Benvenisty, N. The tumorigenicity of human embryonic and induced pluripotent stem cells. Nat. Rev. Cancer 2011, 11, 268–277. [Google Scholar] [CrossRef]
- Yang, X.; Pabon, L.; Murry, C.E. Engineering adolescence: Maturation of human pluripotent stem cell–derived cardiomyocytes. Circ. Res. 2014, 114, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Balsam, L.B.; Wagers, A.J.; Christensen, J.L.; Kofidis, T.; Weissman, I.L.; Robbins, R.C. Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature 2004, 428, 668–673. [Google Scholar] [CrossRef]
- Snyder, C.A.; Dwyer, K.D.; Coulombe, K.L. Advancing Human iPSC-Derived Cardiomyocyte Hypoxia Resistance for Cardiac Regenerative Therapies through a Systematic Assessment of In Vitro Conditioning. Int. J. Mol. Sci. 2024, 25, 9627. [Google Scholar] [CrossRef]
- Nelson, T.J.; Martinez-Fernandez, A.; Yamada, S.; Perez-Terzic, C.; Ikeda, Y.; Terzic, A. Repair of acute myocardial infarction by human stemness factors induced pluripotent stem cells. Circulation 2009, 120, 408–416. [Google Scholar] [CrossRef]
- Cheng, Y.C.; Hsieh, M.L.; Lin, C.J.; Chang, C.M.C.; Huang, C.Y.; Puntney, R.; Wu Moy, A.; Ting, C.Y.; Herr Chan, D.Z.; Nicholson, M.W.; et al. Combined Treatment of Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes and Endothelial Cells Regenerate the Infarcted Heart in Mice and Non-Human Primates. Circulation 2023, 148, 1395–1409. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kobayashi, H.; Tohyama, S.; Ichimura, H.; Ohashi, N.; Chino, S.; Soma, Y.; Tani, H.; Tanaka, Y.; Yang, X.; Shiba, N.; et al. Regeneration of Nonhuman Primate Hearts with Human Induced Pluripotent Stem Cell-Derived Cardiac Spheroids. Circulation 2024, 150, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, J.J.; Sanchez, P.; Repetti, G.G.; Juneman, E.; Pandey, A.C.; Chinyere, I.R.; Moukabary, T.; LaHood, N.; Daugherty, S.L.; Goldman, S. Human Induced Pluripotent Stem Cell-Derived Cardiomyocyte Patch in Rats with Heart Failure. Ann. Thorac. Surg. 2019, 108, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, J.; Miyagawa, S.; Akagi, T.; Akashi, M.; Sawa, Y. Human induced pluripotent stem cell-derived three-dimensional cardiomyocyte tissues ameliorate the rat ischemic myocardium by remodeling the extracellular matrix and cardiac protein phenotype. PLoS ONE 2021, 16, e0245571. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Guan, X.; Xu, W.; Zhang, H.; Wang, Q.; Yu, J.; Zhang, R.; Chen, Y.; Xia, Y.; Wang, J.; Wang, D. Transplantation of human induced pluripotent stem cell-derived cardiomyocytes improves myocardial function and reverses ventricular remodeling in infarcted rat hearts. Stem Cell Res. Ther. 2020, 11, 73, Erratum in Stem Cell Res. Ther. 2020, 11, 201. [Google Scholar] [CrossRef]
- Ishida, M.; Miyagawa, S.; Saito, A.; Fukushima, S.; Harada, A.; Ito, E.; Ohashi, F.; Watabe, T.; Hatazawa, J.; Matsuura, K.; et al. Transplantation of Human-induced Pluripotent Stem Cell-derived Cardiomyocytes is Superior to Somatic Stem Cell Therapy for Restoring Cardiac Function and Oxygen Consumption in a Porcine Model of Myocardial Infarction. Transplantation 2019, 103, 291–298. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Samura, T.; Miyagawa, S.; Kawamura, T.; Fukushima, S.; Yokoyama, J.Y.; Takeda, M.; Harada, A.; Ohashi, F.; Sato-Nishiuchi, R.; Toyofuku, T.; et al. Laminin-221 Enhances Therapeutic Effects of Human-Induced Pluripotent Stem Cell-Derived 3-Dimensional Engineered Cardiac Tissue Transplantation in a Rat Ischemic Cardiomyopathy Model. J. Am. Heart Assoc. 2020, 9, e015841. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, J.; Li, J.; Qu, X.; Liu, Y.; Sun, L.; Harada, A.; Hua, Y.; Sougawa, N.; Tabata, A.; Liu, L.; et al. Development of composite functional tissue sheets using hiPSC-CMs and hADSCs to improve the cardiac function after myocardial infarction. Bioact. Mater. 2024, 37, 533–548. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kawamura, M.; Miyagawa, S.; Miki, K.; Saito, A.; Fukushima, S.; Higuchi, T.; Kawamura, T.; Kuratani, T.; Daimon, T.; Shimizu, T.; et al. Feasibility, safety, and therapeutic efficacy of human induced pluripotent stem cell-derived cardiomyocyte sheets in a porcine ischemic cardiomyopathy model. Circulation 2012, 126, S29–S37. [Google Scholar] [CrossRef] [PubMed]
- Ishigami, M.; Masumoto, H.; Ikuno, T.; Aoki, T.; Kawatou, M.; Minakata, K.; Ikeda, T.; Sakata, R.; Yamashita, J.K.; Minatoya, K. Human iPS cell-derived cardiac tissue sheets for functional restoration of infarcted porcine hearts. PLoS ONE 2018, 13, e0201650. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Iida, J.; Kotani, K.; Murata, K.; Hakamada, K.; Maihemuti, W.; Mandai, Y.; Hiraoka, Y.; Minatoya, K.; Masumoto, H. Retention of locally injected human iPS cell-derived cardiomyocytes into the myocardium using hydrolyzed gelatin. Sci. Rep. 2025, 15, 4635. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kawamura, M.; Miyagawa, S.; Fukushima, S.; Saito, A.; Miki, K.; Funakoshi, S.; Yoshida, Y.; Yamanaka, S.; Shimizu, T.; Okano, T.; et al. Enhanced Therapeutic Effects of Human iPS Cell Derived-Cardiomyocyte by Combined Cell-Sheets with Omental Flap Technique in Porcine Ischemic Cardiomyopathy Model. Sci. Rep. 2017, 7, 8824. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nakazato, T.; Kawamura, T.; Uemura, T.; Liu, L.; Li, J.; Sasai, M.; Harada, A.; Ito, E.; Iseoka, H.; Toda, K.; et al. Engineered three-dimensional cardiac tissues maturing in a rotating wall vessel bioreactor remodel diseased hearts in rats with myocardial infarction. Stem Cell Rep. 2022, 17, 1170–1182. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sun, X.; Wu, J.; Mourad, O.; Li, R.; Nunes, S.S. Microvessel co-transplantation improves poor remuscularisation by hiPSC-cardiomyocytes in a complex disease model of myocardial infarction and type 2 diabetes. Stem Cell Rep. 2025, 20, 102394. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Miyagawa, S.; Kawamura, T.; Ito, E.; Takeda, M.; Iseoka, H.; Yokoyama, J.; Harada, A.; Mochizuki-Oda, N.; Imanishi-Ochi, Y.; Li, J.; et al. Pre-clinical evaluation of the efficacy and safety of human induced pluripotent stem cell-derived cardiomyocyte patch. Stem Cell Res. Ther. 2024, 15, 73. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chinyere, I.R.; Bradley, P.; Uhlorn, J.; Eason, J.; Mohran, S.; Repetti, G.G.; Daugherty, S.; Koevary, J.W.; Goldman, S.; Lancaster, J.J. Epicardially Placed Bioengineered Cardiomyocyte Xenograft in Immune-Competent Rat Model of Heart Failure. Stem Cells Int. 2021, 2021, 9935679. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Biagi, D.; Fantozzi, E.T.; Campos-Oliveira, J.C.; Naghetini, M.V.; Ribeiro, A.F., Jr.; Rodrigues, S.; Ogusuku, I.; Vanderlinde, R.; Christie, M.L.A.; Mello, D.B.; et al. In Situ Maturated Early-Stage Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes Improve Cardiac Function by Enhancing Segmental Contraction in Infarcted Rats. J. Pers. Med. 2021, 11, 374. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kawaguchi, S.; Soma, Y.; Nakajima, K.; Kanazawa, H.; Tohyama, S.; Tabei, R.; Hirano, A.; Handa, N.; Yamada, Y.; Okuda, S.; et al. Intramyocardial Transplantation of Human iPS Cell-Derived Cardiac Spheroids Improves Cardiac Function in Heart Failure Animals. JACC Basic Transl. Sci. 2021, 6, 239–254. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Weinberger, F.; Breckwoldt, K.; Pecha, S.; Kelly, A.; Geertz, B.; Starbatty, J.; Yorgan, T.; Cheng, K.H.; Lessmann, K.; Stolen, T.; et al. Cardiac repair in guinea pigs with human engineered heart tissue from induced pluripotent stem cells. Sci. Transl. Med. 2016, 8, 363ra148. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, J.J.; Grijalva, A.; Fink, J.; Ref, J.; Daugherty, S.; Whitman, S.; Fox, K.; Gorman, G.; Lancaster, L.D.; Avery, R.; et al. Biologically derived epicardial patch induces macrophage mediated pathophysiologic repair in chronically infarcted swine hearts. Commun. Biol. 2023, 6, 1203. [Google Scholar] [CrossRef]
- Querdel, E.; Reinsch, M.; Castro, L.; Köse, D.; Bähr, A.; Reich, S.; Geertz, B.; Ulmer, B.; Schulze, M.; Lemoine, M.D.; et al. Human engineered heart tissue patches remuscularize the injured heart in a dose-dependent manner. Circulation 2021, 143, 1991–2006. [Google Scholar] [CrossRef]
- Masumoto, H.; Ikuno, T.; Takeda, M.; Fukushima, H.; Marui, A.; Katayama, S.; Shimizu, T.; Ikeda, T.; Okano, T.; Sakata, R.; et al. Human iPS cell-engineered cardiac tissue sheets with cardiomyocytes and vascular cells for cardiac regeneration. Sci. Rep. 2014, 4, 6716. [Google Scholar] [CrossRef]
- Li, S.; Tao, B.; Liu, J.; Liu, D.; Wang, Y.; Zhang, J.; Tian, L.; Xu, B.; Fang, Y.; Wang, Y.; et al. Minimally Invasive Transthoracic Intramyocardial Cellular Transplantation Under Echocardiographic Guidance for Myocardial Impairment. Cardiol. Discov. 2024, 4, 260–273. [Google Scholar] [CrossRef]
- Morrissette-McAlmon, J.; Xu, W.R.; Teuben, R.; Boheler, K.R.; Tung, L. Adipocyte-mediated electrophysiological remodeling of human stem cell-derived cardiomyocytes. J. Mol. Cell. Cardiol. 2024, 189, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Shang, T.; Li, S.; Zhang, Y.; Lu, L.; Cui, L.; Guo, F.F. Hypoxia promotes differentiation of adipose-derived stem cells into endothelial cells through demethylation of ephrinB2. Stem Cell Res. Ther. 2019, 10, 133. [Google Scholar] [CrossRef]
- Barone, L.; Palano, M.T.; Gallazzi, M.; Cucchiara, M.; Rossi, F.; Borgese, M.; Raspanti, M.; Zecca, P.A.; Mortara, L.; Papait, R.; et al. Adipose mesenchymal stem cell-derived soluble factors, produced under hypoxic condition, efficiently support in vivo angiogenesis. Cell Death Discov. 2023, 9, 174. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Gao, E.; Jiao, X.; Yuan, Y.; Li, S.; Christopher, T.A.; Lopez, B.L.; Koch, W.; Chan, L.; Goldstein, B.J.; et al. Adiponectin cardioprotection after myocardial ischemia/reperfusion involves the reduction of oxidative/nitrative stress. Circulation 2007, 115, 1408–1416. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yu, H.; Dong, Y.; Xie, W. Omentum transplantation for malignant tumors: A narrative review of emerging techniques and clinical applications. Eur. J. Med. Res. 2025, 30, 322. [Google Scholar] [CrossRef]
- Ai, X.; Yan, B.; Witman, N.; Gong, Y.; Yang, L.; Tan, Y.; Chen, Y.; Liu, M.; Lu, T.; Luo, R.; et al. Transient secretion of VEGF protein from transplanted hiPSC-CMs enhances engraftment and improves rat heart function post MI. Mol. Ther. 2023, 31, 211–229. [Google Scholar] [CrossRef]
- Carlsson, L.; Clarke, J.C.; Yen, C.; Gregoire, F.; Albery, T.; Billger, M.; Egnell, A.C.; Gan, L.M.; Jennbacken, K.; Johansson, E.; et al. Biocompatible, purified VEGF-A mRNA improves cardiac function after intracardiac injection 1 week post-myocardial infarction in swine. Mol. Ther. Methods Clin. Dev. 2018, 9, 330–346. [Google Scholar] [CrossRef]
- Rottbauer, W.; Just, S.; Wessels, G.; Trano, N.; Most, P.; Katus, H.A.; Fishman, M.C. VEGF–PLCγ1 pathway controls cardiac contractility in the embryonic heart. Genes Dev. 2005, 19, 1624–1634. [Google Scholar] [CrossRef]
- Weiss, J.L.; Marino, P.N.; Shapiro, E.P. Myocardial infarct expansion: Recognition, significance and pathology. Am. J. Cardiol. 1991, 68, 35–40. [Google Scholar] [CrossRef]
- Idais, D.; Roche, C.D.; Kalogianni, G.; Polonchuk, L.; Gentile, C. Generation and applications of cardiac spheroids. npj Biomed. Innov. 2025, 2, 17. [Google Scholar] [CrossRef]
- Griffin, K.H.; Fok, S.W.; Kent Leach, J. Strategies to capitalize on cell spheroid therapeutic potential for tissue repair and disease modeling. npj Regen. Med. 2022, 7, 70. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xue, Y.; Pan, T.; Zhu, X.; Chong, H.; Xu, C.; Fan, F.; Cao, H.; Zhang, B.; Pan, J.; et al. Epicardial injection of allogeneic human-induced-pluripotent stem cell-derived cardiomyocytes in patients with advanced heart failure: Protocol for a phase I/IIa dose-escalation clinical trial. BMJ Open 2022, 12, e056264. [Google Scholar] [CrossRef]
- Kawamura, T.; Ito, Y.; Ito, E.; Takeda, M.; Mikami, T.; Taguchi, T.; Mochizuki-Oda, N.; Sasai, M.; Shimamoto, T.; Nitta, Y.; et al. Safety confirmation of induced pluripotent stem cell-derived cardiomyocyte patch transplantation for ischemic cardiomyopathy: First three case reports. Front. Cardiovasc. Med. 2023, 10, 1182209. [Google Scholar] [CrossRef] [PubMed]
- Heartseed Inc. Heartseed Announces Positive 26-Week Data for the First Patient in the Phase ½ Clinical Tral (LAPiS Study) Dosed with HS-001, an Investigational Stem Cell-Derived Therapy for the Treatment of Advanced Heart Failure. 6 July 2023. Available online: https://heartseed.jp/en/news/assets/2023/07/aa5fdd0940390720c758960ae066298f1c35d66c.pdf (accessed on 13 August 2025).
- Jebran, A.F.; Seidler, T.; Tiburcy, M.; Daskalaki, M.; Kutschka, I.; Fujita, B.; Ensminger, S.; Bremmer, F.; Moussavi, A.; Yang, H.; et al. Engineered heart muscle allografts for heart repair in primates and humans. Nature 2025, 639, 503–511. [Google Scholar] [CrossRef]
- Laflamme, M.A.; Chen, K.Y.; Naumova, A.V.; Muskheli, V.; Fugate, J.A.; Dupras, S.K.; Reinecke, H.; Xu, C.; Hassanipour, M.; Police, S.; et al. Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat. Biotechnol. 2007, 25, 1015–1024. [Google Scholar] [CrossRef]
- Shin, H.S.; Shin, H.H.; Shudo, Y. Current status and limitations of myocardial infarction large animal models in cardiovascular translational research. Front. Bioeng. Biotechnol. 2021, 9, 673683. [Google Scholar] [CrossRef]
- Ma, N.; Cheng, H.; Lu, M.; Liu, Q.; Chen, X.; Yin, G.; Zhu, H.; Zhang, L.; Meng, X.; Tang, Y.; et al. Magnetic resonance imaging with superparamagnetic iron oxide fails to track the long-term fate of mesenchymal stem cells transplanted into heart. Sci. Rep. 2015, 5, 9058. [Google Scholar] [CrossRef] [PubMed]
- Amsalem, Y.; Mardor, Y.; Feinberg, M.S.; Landa, N.; Miller, L.; Daniels, D.; Ocherashvilli, A.; Holbova, R.; Yosef, O.; Barbash, I.M.; et al. Iron-oxide labeling and outcome of transplanted mesenchymal stem cells in the infarcted myocardium. Circulation 2007, 116, I38–I45. [Google Scholar] [CrossRef] [PubMed]
- Terrovitis, J.; Stuber, M.; Youssef, A.; Preece, S.; Leppo, M.; Kizana, E.; Schar, M.; Gerstenblith, G.; Weiss, R.G.; Marbán, E.; et al. Magnetic resonance imaging overestimates ferumoxide-labeled stem cell survival after transplantation in the heart. Circulation 2008, 117, 1555–1562. [Google Scholar] [CrossRef]
- Bois, A.; Grandela, C.; Gallant, J.; Mummery, C.; Menasché, P. Revitalizing the heart: Strategies and tools for cardiomyocyte regeneration post-myocardial infarction. npj Regen. Med. 2025, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Heineke, J.; Auger-Messier, M.; Xu, J.; Oka, T.; Sargent, M.A.; York, A.; Klevitsky, R.; Vaikunth, S.; Duncan, S.A.; Aronow, B.J.; et al. Cardiomyocyte GATA4 functions as a stress-responsive regulator of angiogenesis in the murine heart. J. Clin. Investig. 2007, 117, 3198–3210. [Google Scholar] [CrossRef]
- Tang, Y.; Aryal, S.; Geng, X.; Zhou, X.; Fast, V.G.; Zhang, J.; Lu, R.; Zhou, Y. TBX20 improves contractility and mitochondrial function during direct human cardiac reprogramming. Circulation 2022, 146, 1518–1536. [Google Scholar] [CrossRef]
- Boopathy, G.T.; Hong, W. Role of hippo pathway-YAP/TAZ signaling in angiogenesis. Front. Cell Dev. Biol. 2019, 7, 49. [Google Scholar] [CrossRef]


| Author | Animal Model | Cell Type | Construct | Delivery | Days after MI | Follow-Up |
|---|---|---|---|---|---|---|
| Cheng et al. [32] | NHP | CMs and ECs | Cells | Intramyocardial | 8 days | 28 days |
| Kobayashi et al. [33] | NHP | CMs | Spheroids | Intramyocardial | 14 days | ~84 days |
| Lancaster J et al. [34] | Rodent | CMs and hNDFs | Patch | Epicardial | 21 days | 21 days |
| Yokoyama et al. [35] | Rodent | CMs | Patch | Epicardial | 14 days | 84 days |
| Guan X et al. [36] | Rodent | CMs | Cells | Intramyocardial | 10 days | 28 days |
| Ishida et al. [37] | Porcine | CMs | Sheets | Epicardial | 30 days | 60 days |
| Samura T et al. [38] | Rodent | CMs | Sheets | Epicardial | 28 days | 28 days |
| Zhang et al. [39] | Rodent | CMs and hADSCs | Sheets | Epicardial | 14 days | 28 days |
| Kawamura et al. [40] | Porcine | CMs | Sheets | Epicardial | 28 days | 56 days |
| Ishigami et al. [41] | Porcine | CMs/ECs/VMCs | Sheets | Epicardial | 14 days | 28 days |
| Lida J et al. [42] | Rodent | CMS | Hydrogel | Intramyocardial | 7 days | 7 days |
| Kawamura et al. [43] | Porcine | CMs | Sheets | Epicardial | 28 days | ~91 days |
| Nakazato et al. [44] | Rodent | CMs | Sheets | Epicardial | 14 days | 28 days |
| Sun X et al. [45] | Rodent | CMs | Cells | Intramyocardial | 14 days | 28 days |
| Miyagawa et al. [46] | Porcine | CMs | Patch | Epicardial | 28 days | 84 days |
| Chinyere et al. [47] | Rodents | CMs and hNDFs | Patch | Epicardial | 21 days | 49 days |
| Biagi et al. [48] | Rodent | CMs | Cells | Intramyocardial | 7 days | 37 days |
| Kawaguchi et al. [49] | Rodent/porcine | CMs | Spheroids | Intramyocardial | 7/28 days | 56 days |
| Weinberger et al. [50] | Rodent | CMs/ECs | Patch | Epicardial | 7 days | 28 days |
| Lancaster J et al. [51] | Porcine | CMs and hNDFs | Patch | Epicardial | 28 days | ~182 days |
| Querdel et al. [52] | Rodent | CMs | Patch | Epicardial | 7 days | 28 days |
| Masumoto et al. [53] | Rodent | CMs/MCs/ECs | Sheets | Epicardial | 7 days | 28 days |
| Li et al. [54] | Porcine | CMTK+ and CMs | Cells | Intramyocardial | 84 days | 84 days |
| Efficacy | Safety | |||||||
|---|---|---|---|---|---|---|---|---|
| Study |  Systolic function Systolic function | Reduced infarct/scar size | Survival |  Vascular density Vascular density | Arrhythmias | Immune rejection | Tumor formation | |
| Spheroids | [33] | ✓ | NM | NR | NM | ✓ |  | NR |
| [49] | ✓ | ✓ |  | ✓ | ✓ |  |  | |
| Cells | [54] |  |  |  | NM | ✓ | NR | NR |
| [48] | ✓ | NM | ✓ | NM | NR |  | NR | |
| [32] | ✓ | ✓ | NR | ✓ | ✓ | NR | NR | |
| [36] | ✓ | NM | ✓ | NM |  |  | NR | |
| [45] | ✓ | ✓ | ✓ | ✓ | NR | NR | NR | |
| Hydrogel | [42] | ✓ | NM | ✓ | NM | NR | NR | NR |
| Efficacy | Safety | |||||||
|---|---|---|---|---|---|---|---|---|
| Study |  Systolic function Systolic function | Reduced infarct/scar size | Survival |  Vascular density Vascular density | Arrhythmias | Immune rejection | Tumor formation | |
| Patches | [34] |  | NR |  | NR |  | NR | NR |
| [35] | ✓ | NM | NR | ✓ | NR | NR | NR | |
| [46] | ✓ |  | NR | ✓ |  |  |  | |
| [47] | ✓ | NM |  | NM | NR |  | NR | |
| [50] | ✓ | UD | ✓ | ✓ | NR | NR |  | |
| [51] |  | NR |  | ✓ | NR |  | NR | |
| [52] | ✓ | NM | ✓ |  | NR | NR | NR | |
| Sheets | [38] | ✓ | NM |  | ✓ | NR | NR | NR |
| [37] | ✓ | NM |  | ✓ | NR | NR |  | |
| [41] | ✓ | ✓ | ✓ | ✓ | NR | NR | NR | |
| [40] | ✓ | NM | ✓ | ✓ | NR | NR | NR | |
| [43] | ✓ | NM | ✓ | ✓ | NR | NR | NR | |
| [44] | ✓ | NM | ✓ | ✓ | NR | NR | NR | |
| [53] | ✓ | NR | ✓ | ✓ | NR | NR |  | |
| Case Number | Efficacy Outcomes | Baseline | Months Post-Transplantation | |||
|---|---|---|---|---|---|---|
| 3 | 4 | 6 | 12 | |||
| Case 1 (LAPiS) | LVEF (%) | 26 (Echo) 15 (CMR) | 28 19 | |||
| LVEDV (mL) Echo MRI | 345 431 | 252 389 | ||||
| NYHA Class | III | II | ||||
| NT-proBNP (pg/mL) | 11,471 | 5733 | ||||
| Case 2 (LAPiS) | LVEF (%) | 26 | 26 | |||
| LVEDV (mL) Echo | 324 | 362 | ||||
| NYHA Class | III | III | ||||
| Case 3 (LAPiS) | LVEF (%) | 17 | 36 | |||
| LVEDV (mL) | 196 | 172 | ||||
| NYHA Class | III | II | ||||
| Case 4 (NCT04696328) | LVEF (%) | <30% | ||||
| NYHA Class | III | I | I | |||
| LVIDs (mm) | n.a | |||||
| LVIDd (mm) | n.a | |||||
| NT-proBNP (pg/dL) | n.a | |||||
| Peak VO2 (mL/min/kg) | n.a | |||||
| Myocardial blood flow (mL/min/g) | n.m | 1.67 | 3.12 | |||
| Case 5 (NCT04696328) | LVEF (%) | 21% | - | - | ||
| NYHA Class | III | II | II | |||
| LVIDs (mm) | n.a | - | - | |||
| LVIDd (mm) | n.a | - | - | |||
| NT-proBNP (pg/dL) | n.a | |||||
| Peak VO2 (mL/min/kg) | n.a | - | n.a | |||
| Myocardial blood flow (mL/min/g) | 0.51 | 0.57 | 0.47 | |||
| Case 6 (NCT04696328) | LVEF (%) | 35% | ||||
| NYHA Class | III | I | I | |||
| LVIDs (mm) | n.a | - | ||||
| LVIDd (mm) | n.a | - | ||||
| NT-proBNP (pg/dL) | n.a | |||||
| Peak VO2 (mL/min/kg) | n.a | |||||
| Myocardial blood flow (mL/min/g) | 1.01 | 0.69 | 0.67 | |||
| Case 7 (BioVAT-HF) 46 y/o F | LVEF (%) | 35% | 39% | |||
| LVEDV (mL) | 104 | 78 | ||||
| LVESV (mL) | 68 | 48 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Callender, K.; Smith, G. A Review of the Therapeutic Efficacy and Safety of Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes in Preclinical Models of Subacute and Chronic Myocardial Infarction. J. Cardiovasc. Dev. Dis. 2026, 13, 42. https://doi.org/10.3390/jcdd13010042
Callender K, Smith G. A Review of the Therapeutic Efficacy and Safety of Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes in Preclinical Models of Subacute and Chronic Myocardial Infarction. Journal of Cardiovascular Development and Disease. 2026; 13(1):42. https://doi.org/10.3390/jcdd13010042
Chicago/Turabian StyleCallender, Kristen, and Godfrey Smith. 2026. "A Review of the Therapeutic Efficacy and Safety of Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes in Preclinical Models of Subacute and Chronic Myocardial Infarction" Journal of Cardiovascular Development and Disease 13, no. 1: 42. https://doi.org/10.3390/jcdd13010042
APA StyleCallender, K., & Smith, G. (2026). A Review of the Therapeutic Efficacy and Safety of Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes in Preclinical Models of Subacute and Chronic Myocardial Infarction. Journal of Cardiovascular Development and Disease, 13(1), 42. https://doi.org/10.3390/jcdd13010042







