Clinical Characteristics and Outcomes of Patients Presenting with Acute Coronary Syndromes and Suspected Plaque Erosion Based on Clinical and Laboratory Criteria
Abstract
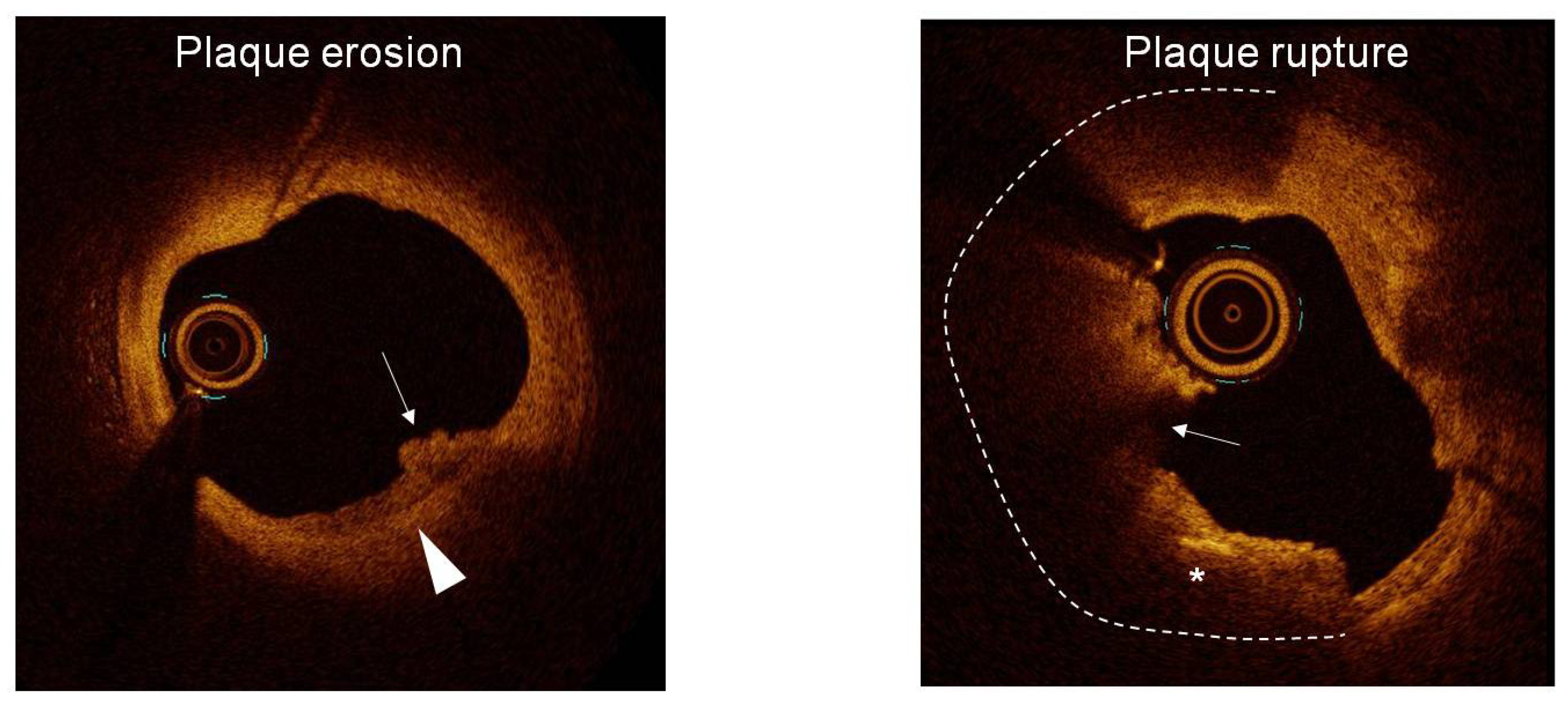
1. Introduction
2. Materials and Methods
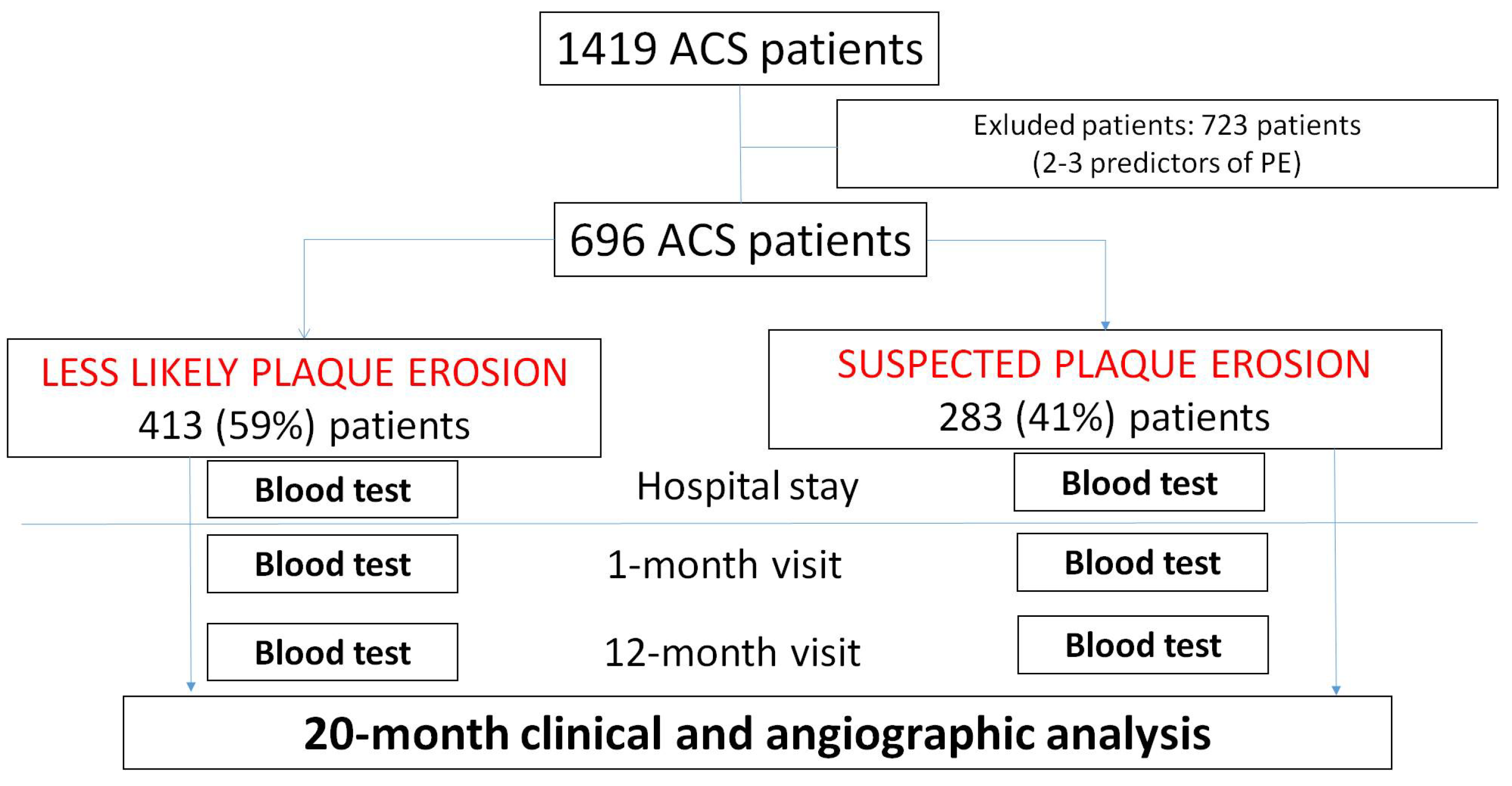
2.1. Study Population
2.2. Clinical and Coronary Angiographic Data
2.3. Laboratory Data
2.4. Clinical and Angiographic Endpoints
2.5. Statistical Analysis
3. Results
3.1. Clinical and Angiographic Characteristics of the Included Population
3.2. Laboratory Data at Hospital Admission and 1-Month, and 12-Month Post-Hospital Discharge Times
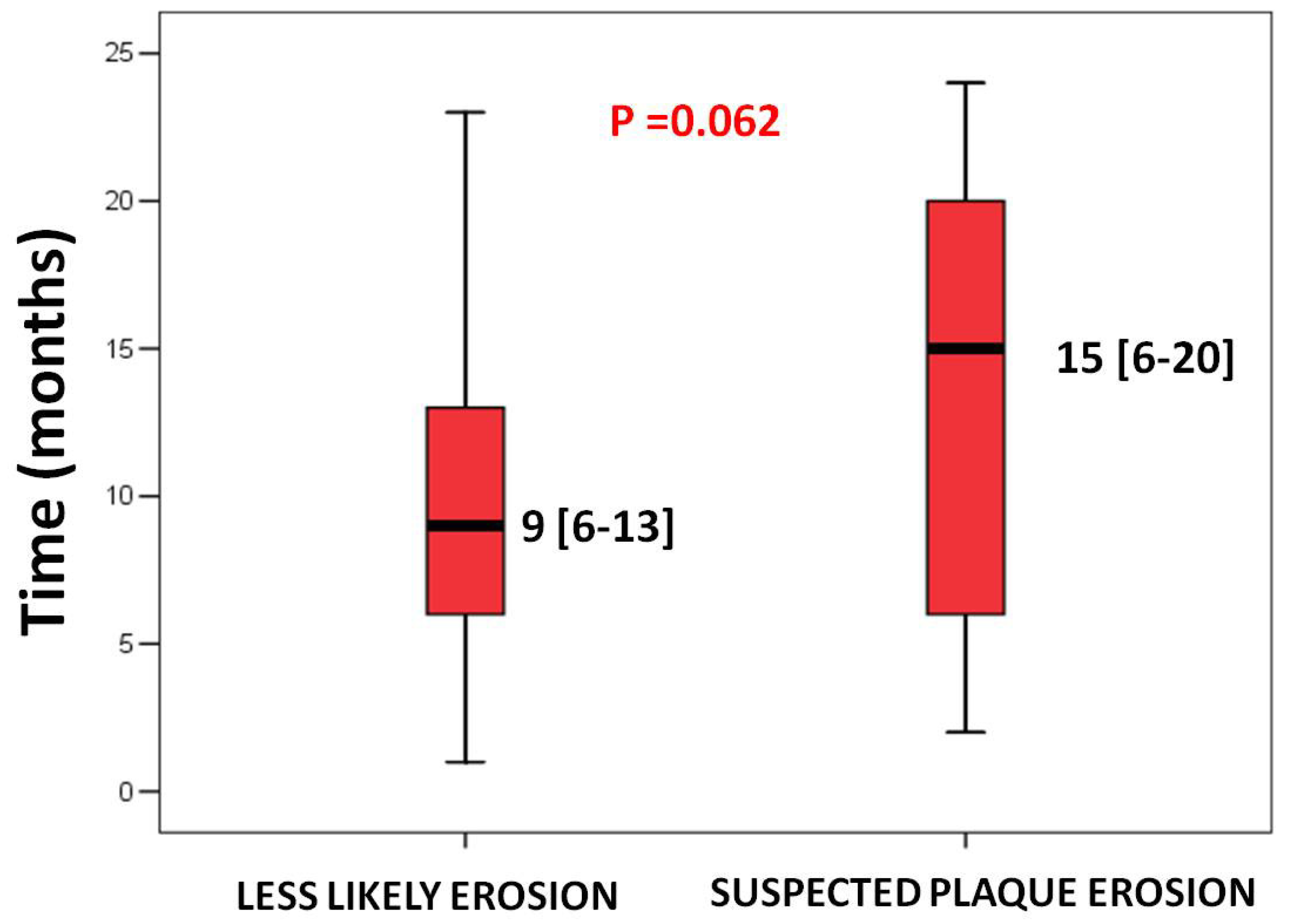
3.3. Major Adverse Cardiovascular Events in the Two Groups at the 20-Month Time
4. Discussion
4.1. Prevalence of Suspected PE Among ACS Patients
4.2. Cardiovascular Risk Factor Distribution in Suspected PE Patients
4.3. Systemic Inflammation and Coronary Atherosclerosis Burden in Suspected PE Group
4.4. Suspected PE Patients Have a Better Prognosis: The Role of Tailored Treatment for PE Management
5. Conclusions
Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACS | acute coronary syndromes |
| BMI | body mass index. |
| CCS | chronic coronary syndrome |
| eGFR | estimated glomerular filtration rate |
| HR | hazard ratios. |
| MACEs | major adverse cardiac events |
| NET | neutrophil extracellular trap |
| non-TVR | non-target vessel revascularization |
| NSTEMI | non-ST-segment elevation myocardial infarction |
| OCT | optical coherence tomography |
| PCI | percutaneous coronary intervention |
| PE | plaque erosion |
| SD | standard deviation |
| STEMI | ST-segment elevation myocardial infarction |
| TVR | target vessel revascularization |
References
- Kolte, D.; Yonetsu, T.; Ye, J.C.; Libby, P.; Fuster, V.; Jang, I.-K. Optical Coherence Tomography of Plaque Erosion: JACC Focus Seminar Part 2/3. J. Am. Coll. Cardiol. 2021, 78, 1266–1274. [Google Scholar] [CrossRef]
- Jia, H.; Abtahian, F.; Aguirre, A.D.; Lee, S.; Chia, S.; Lowe, H.; Kato, K.; Yonetsu, T.; Vergallo, R.; Hu, S.; et al. In Vivo Diagnosis of Plaque Erosion and Calcified Nodule in Patients With Acute Coronary Syndrome by Intravascular Optical Coherence Tomography. J. Am. Coll. Cardiol. 2013, 62, 1748–1758. [Google Scholar] [CrossRef] [PubMed]
- Virmani, R.; Burke, A.P.; Farb, A.; Kolodgie, F.D. Pathology of the Vulnerable Plaque. J. Am. Coll. Cardiol. 2006, 47, C13–C18. [Google Scholar] [CrossRef]
- Partida, R.A.; Libby, P.; Crea, F.; Jang, I.-K. Plaque erosion: A new in vivo diagnosis and a potential major shift in the management of patients with acute coronary syndromes. Eur. Heart J. 2018, 39, 2070–2076. [Google Scholar] [CrossRef]
- Wight, T.N. A Role for Extracellular Matrix in Atherosclerotic Plaque Erosion. J. Am. Coll. Cardiol. 2018, 72, 1504–1505. [Google Scholar] [CrossRef]
- Pedicino, D.; Vinci, R.; Giglio, A.F.; Pisano, E.; Porto, I.; Vergallo, R.; Russo, G.; Ruggio, A.; D’Aiello, A.; Flego, D.; et al. Alterations of Hyaluronan Metabolism in Acute Coronary Syndrome. J. Am. Coll. Cardiol. 2018, 72, 1490–1503. [Google Scholar] [CrossRef]
- Yamamoto, E.; Yonetsu, T.; Kakuta, T.; Soeda, T.; Saito, Y.; Yan, B.P.; Kurihara, O.; Takano, M.; Niccoli, G.; Higuma, T.; et al. Clinical and Laboratory Predictors for Plaque Erosion in Patients With Acute Coronary Syndromes. J. Am. Heart Assoc. 2019, 8, e012322. [Google Scholar] [CrossRef] [PubMed]
- Prati, F.; Uemura, S.; Souteyrand, G.; Virmani, R.; Motreff, P.; Di Vito, L.; Biondi-Zoccai, G.; Halperin, J.; Fuster, V.; Ozaki, Y.; et al. OCT-Based Diagnosis and Management of STEMI Associated With Intact Fibrous Cap. JACC Cardiovasc. Imaging 2013, 6, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Niccoli, G.; Montone, R.A.; Di Vito, L.; Gramegna, M.; Refaat, H.; Scalone, G.; Leone, A.M.; Trani, C.; Burzotta, F.; Porto, I.; et al. Plaque rupture and intact fibrous cap assessed by optical coherence tomography portend different outcomes in patients with acute coronary syndrome. Eur. Hear. J. 2015, 36, 1377–1384. [Google Scholar] [CrossRef]
- Jia, H.; Dai, J.; Hou, J.; Xing, L.; Ma, L.; Liu, H.; Xu, M.; Yao, Y.; Hu, S.; Yamamoto, E.; et al. Effective anti-thrombotic therapy without stenting: Intravascular optical coherence tomography-based management in plaque erosion (the EROSION study). Eur. Hear. J. 2016, 38, 792–800. [Google Scholar] [CrossRef]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D. Fourth Universal Definition of Myocardial Infarction. Circulation 2018, 138, e618–e651. [Google Scholar] [CrossRef]
- Vrints, C.; Andreotti, F.; Koskinas, K.C.; Rossello, X.; Adamo, M.; Ainslie, J.; Banning, A.P.; Budaj, A.; Buechel, R.R.; Chiariello, G.A.; et al. [2024 ESC Guidelines for the management of chronic coronary syndromes]. G. Ital. Di Cardiol. 2024, 25, 1e–132e. [Google Scholar] [CrossRef]
- Nohara, A. The Worst Combination for Coronary Artery Disease Under Strong-Statin Therapy Is Chronic Kidney Disease and Diabetes ― Non-Lipid Residual Risk Assessment From the REAL-CAD Study. Circ. J. 2024, 88, 1004–1006. [Google Scholar] [CrossRef] [PubMed]
- DeFilippis, E.M.; Collins, B.L.; Singh, A.; Biery, D.W.; Fatima, A.; Qamar, A.; Berman, A.N.; Gupta, A.; Cawley, M.; Wood, M.J.; et al. Women who experience a myocardial infarction at a young age have worse outcomes compared with men: The Mass General Brigham YOUNG-MI registry. Eur. Hear. J. 2020, 41, 4127–4137. [Google Scholar] [CrossRef]
- Tweet, M.S. Sex differences among young individuals with myocardial infarction. Eur. Hear. J. 2020, 41, 4138–4140. [Google Scholar] [CrossRef]
- Mora, S.; Wenger, N.K.; DeMicco, D.A.; Breazna, A.; Boekholdt, S.M.; Arsenault, B.J.; Deedwania, P.; Kastelein, J.J.; Waters, D.D. Determinants of residual risk in secondary prevention patients treated with high-versus low-dose statin therapy: The Treating to New Targets (TNT) study. Circulation 2012, 125, 1979–1987. [Google Scholar] [CrossRef]
- Ndrepepa, G.; Kufner, S.; Cassese, S.; Joner, M.; Sager, H.B.; Xhepa, E.; Laugwitz, K.L.; Schunkert, H.; Kastrati, A. Impaired Kidney Function and 10-Year Outcome After Percutaneous Coronary Intervention-Interaction with Age, Sex, Diabetic Status and Clinical Presentation. J. Clin. Med. 2024, 13, 6833. [Google Scholar] [CrossRef]
- Gouda, P.; Savu, A.; Bainey, K.R.; Kaul, P.; Welsh, R.C.; Fukumoto, Y. Long-term risk of death and recurrent cardiovascular events following acute coronary syndromes. PLOS ONE 2021, 16, e0254008. [Google Scholar] [CrossRef] [PubMed]
- Bittner, V.A.; Schwartz, G.G.; Bhatt, D.L.; Chua, T.; De Silva, H.A.; Diaz, R.; Goodman, S.G.; Harrington, R.A.; Jukema, J.W.; McGinniss, J.; et al. Alirocumab and cardiovascular outcomes according to sex and lipoprotein(a) after acute coronary syndrome: A report from the ODYSSEY OUTCOMES study. J. Clin. Lipidol. 2024, 18, e548–e561. [Google Scholar] [CrossRef]
- Ostadal, P.; Steg, P.G.; Poulouin, Y.; Bhatt, D.L.; A Bittner, V.; Chua, T.; Diaz, R.; Goodman, S.G.; Huo, Y.; Jukema, J.W.; et al. Metabolic risk factors and effect of alirocumab on cardiovascular events after acute coronary syndrome: A post-hoc analysis of the ODYSSEY OUTCOMES randomised controlled trial. Lancet Diabetes Endocrinol. 2022, 10, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Tearney, G.J.; Regar, E.; Akasaka, T.; Adriaenssens, T.; Barlis, P.; Bezerra, H.G.; Bouma, B.; Bruining, N.; Cho, J.; Chowdhary, S. Consensus standards for acquisition, measurement, and reporting of intravascular optical coherence tomography studies: A report from the International Working Group for Intravascular Optical Coherence Tomography Standardization and Validation. J. Am. Coll. Cardiol. 2012, 59, 1058–1072. [Google Scholar] [CrossRef]
- Dai, J.; Xing, L.; Jia, H.; Zhu, Y.; Zhang, S.; Hu, S.; Lin, L.; Ma, L.; Liu, H.; Xu, M.; et al. In vivo predictors of plaque erosion in patients with ST-segment elevation myocardial infarction: A clinical, angiographical, and intravascular optical coherence tomography study. Eur. Hear. J. 2018, 39, 2077–2085. [Google Scholar] [CrossRef]
- Nagamine, T.; Hoshino, M.; Yonetsu, T.; Sugiyama, T.; Kanaji, Y.; Matsuda, K.; Sayama, K.; Ueno, H.; Nogami, K.; Hanyu, Y.; et al. Identification of Optical Coherence Tomography-Defined Coronary Plaque Erosion by Preprocedural Computed Tomography Angiography. J. Am. Hear. Assoc. 2023, 12, e029239. [Google Scholar] [CrossRef]
- Virmani, R.; Burke, A.; Farb, A. Coronary risk factors and plaque morphology in men with coronary disease who died suddenly. Eur Heart J. 1998, 19, 678–680. [Google Scholar]
- Burke, A.P.; Farb, A.; Malcom, G.T.; Liang, Y.-H.; Smialek, J.; Virmani, R. Effect of Risk Factors on the Mechanism of Acute Thrombosis and Sudden Coronary Death in Women. Circulation 1998, 97, 2110–2116. [Google Scholar] [CrossRef]
- Ferrante, G.; Nakano, M.; Prati, F.; Niccoli, G.; Mallus, M.T.; Ramazzotti, V.; Montone, R.A.; Kolodgie, F.D.; Virmani, R.; Crea, F. High levels of systemic myeloperoxidase are associated with coronary plaque erosion in patients with acute coronary syndromes: A clinicopathological study. Circulation 2010, 122, 2505–2513. [Google Scholar] [CrossRef]
- Seegers, L.M.; Araki, M.; Nakajima, A.; Yonetsu, T.; Minami, Y.; Ako, J.; Soeda, T.; Kurihara, O.; Higuma, T.; Kimura, S.; et al. Sex Differences in Culprit Plaque Characteristics Among Different Age Groups in Patients With Acute Coronary Syndromes. Circ. Cardiovasc. Interv. 2022, 15, e011612. [Google Scholar] [CrossRef] [PubMed]
- Abtahian, F.; Yonetsu, T.; Kato, K.; Jia, H.; Vergallo, R.; Tian, J.; Hu, S.; McNulty, I.; Lee, H.; Yu, B.; et al. Comparison by Optical Coherence Tomography of the Frequency of Lipid Coronary Plaques in Current Smokers, Former Smokers, and Nonsmokers. Am. J. Cardiol. 2014, 114, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Higuma, T.; Soeda, T.; Abe, N.; Yamada, M.; Yokoyama, H.; Shibutani, S.; Vergallo, R.; Minami, Y.; Ong, D.S.; Lee, H.; et al. A Combined Optical Coherence Tomography and Intravascular Ultrasound Study on Plaque Rupture, Plaque Erosion, and Calcified Nodule in Patients With ST-Segment Elevation Myocardial Infarction. JACC Cardiovasc. Interv. 2015, 8, 1166–1176. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Zhu, Y.; Zhang, Y.; Dai, J.; Li, L.; Dauerman, H.; Soeda, T.; Wang, Z.; Lee, H.; Wang, C.; et al. Management and Outcome of Patients With Acute Coronary Syndrome Caused by Plaque Rupture Versus Plaque Erosion: An Intravascular Optical Coherence Tomography Study. J. Am. Hear. Assoc. 2017, 6, e004730. [Google Scholar] [CrossRef]
- Sugiyama, T.; Yamamoto, E.; Bryniarski, K.; Xing, L.; Lee, H.; Isobe, M.; Libby, P.; Jang, I.K. Nonculprit Plaque Characteristics in Patients with Acute Coronary Syndrome Caused by Plaque Erosion vs Plaque Rupture: A 3-Vessel Optical Coherence Tomography Study. JAMA Cardiol. 2018, 3, 207–214. [Google Scholar] [CrossRef]
- Kim, H.O.; Kim, C.-J.; Kurihara, O.; Thondapu, V.; Russo, M.; Yamamoto, E.; Sugiyama, T.; Fracassi, F.; Lee, H.; Yonetsu, T.; et al. Angiographic features of patients with coronary plaque erosion. Int. J. Cardiol. 2019, 288, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Klebanoff, S.J. Myeloperoxidase. Proc. Assoc. Am. Physicians 1999, 111, 383–389. [Google Scholar] [CrossRef]
- Scalone, G.; Niccoli, G.; Refaat, H.; Vergallo, R.; Porto, I.; Leone, A.M.; Burzotta, F.; D’amario, D.; Liuzzo, G.; Fracassi, F.; et al. Not all plaque ruptures are born equal: An optical coherence tomography study. Eur. Hear. J.—Cardiovasc. Imaging 2016, 18, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Yonetsu, T.; Lee, T.; Murai, T.; Suzuki, M.; Matsumura, A.; Hashimoto, Y.; Kakuta, T. Plaque morphologies and the clinical prognosis of acute coronary syndrome caused by lesions with intact fibrous cap diagnosed by optical coherence tomography. Int. J. Cardiol. 2016, 203, 766–774. [Google Scholar] [CrossRef] [PubMed]
- Saada, M.; Kobo, O.; Kauer, F.; Sakhov, O.; Laanmets, P.; Abhaichand, R.; Lozano, I.; Crowley, J.; Wander, G.S.; Mamas, M.A.; et al. Prognosis of PCI in the Older Adult Population: Outcomes From the Multicenter Prospective e-ULTIMASTER Registry. J Soc Cardiovasc Angiogr Interv. 2022, 1, 100442. [Google Scholar] [CrossRef]
- Di Vito, L.; Di Giusto, F.; Bruscoli, F.; Scalone, G.; Silenzi, S.; Mariani, L.; Selimi, A.; Delfino, D.; Grossi, P. Recurrent events after acute ST-segment elevation myocardial infarction: Predictors and features of plaque progression and stent failure. Coron. Artery Dis. 2024, 35, 277–285. [Google Scholar] [CrossRef]
- Yang, X.; Yang, J.; Kashima, Y.; Hachinohe, D.; Sugie, T.; Xu, S.; Guo, X.; Li, X.; Hu, X.; Sun, B.; et al. The influence between plaque rupture and non-plaque rupture on clinical outcomes in patients with ST-segment elevation myocardial infarction after primary percutaneous coronary intervention: A prospective cohort study. J. Thorac. Dis. 2024, 16, 7771–7786. [Google Scholar] [CrossRef]
- Libby, P.; Pasterkamp, G.; Crea, F.; Jang, I.-K. Reassessing the Mechanisms of Acute Coronary Syndromes. Circ. Res. 2019, 124, 150–160. [Google Scholar] [CrossRef]
- Jia, H.; Dai, J.; He, L.; Xu, Y.; Shi, Y.; Zhao, L.; Sun, Z.; Liu, Y.; Weng, Z.; Feng, X.; et al. EROSION III: A Multicenter RCT of OCT-Guided Reperfusion in STEMI With Early Infarct Artery Patency. JACC Cardiovasc. Interv. 2022, 15, 846–856. [Google Scholar] [CrossRef]
- Madsen, J.M.; Kelbæk, H.; Nepper-Christensen, L.; Jacobsen, M.J.; Ahtarovski, K.A.; Høfsten, D.H.; Holmvang, L.; Pedersen, F.; Tilsted, H.-H.; Aarøe, J.; et al. Clinical outcomes of no stenting in patients with ST-segment elevation myocardial infarction undergoing deferred primary percutaneous coronary intervention. EuroIntervention 2022, 18, 482–491. [Google Scholar] [CrossRef] [PubMed]

| Overall (696 Patients) | Less Likely Plaque Erosion (413 Patients) | Suspected Plaque Erosion (283 Patients) | p Value | |
|---|---|---|---|---|
| Age, years | 70.1 ± 13.4 | 79.0 ± 7.5 | 57.2 ± 8.8 | 0.0001 |
| Male sex, n (%) | 501 (72) | 241 (58) | 260 (91.9) | 0.0001 |
| Weight, Kg | 76.4 ± 15.3 | 71.9 ± 13.9 | 83.0 ± 15.0 | 0.0001 |
| Height, cm | 168 ± 8.9 | 164.6 ± 8.3 | 172.9 ± 7.2 | 0.0001 |
| BMI, Kg/m2 | 26.9 ± 4.4 | 26.4 ± 4.4 | 27.6 ± 4.2 | 0.0001 |
| Smoker | ||||
| Never, n (%) | 418 (60) | 272 (66) | 146 (52) | 0.0001 |
| Past, n (%) | 153 (22) | 95 (23) | 58 (21) | |
| Current, n (%) | 125 (18) | 46 (11) | 79 (28) | |
| Dyslipidemia, n (%) | 343 (49) | 199 (48) | 144 (51) | 0.488 |
| Diabetes mellitus, n (%) | 345 (49.5) | 296 (71.6) | 49 (17.3) | 0.0001 |
| Chronic kidney disease, n (%) | 264 (37.9) | 219 (53) | 45 (15.9) | 0.0001 |
| STEMI, n (%) | 401 (57) | 235 (57) | 166 (59) | 0.696 |
| NSTEMI, n (%) | 295 (43) | 178 (43) | 117 (41) |
| Overall (696 Patients) | Less Likely Plaque Erosion (413 Patients) | Suspected Plaque Erosion (283 Patients) | p Value | |
|---|---|---|---|---|
| Culprit vessel | 0.0001 | |||
| Left main, n (%) | 14 (2) | 12 (3) | 2 (1) | |
| Left anterior descending artery, n (%) | 314 (45) | 99 (24) | 215 (76) | |
| Circumflex, n (%) | 154 (22) | 127 (31) | 27 (9) | |
| Right coronary artery, n (%) | 214 (31) | 172 (42) | 42 (15) | |
| One-vessel disease, n (%) | 264 (38) | 123 (30) | 141 (50) | 0.0001 |
| Three-vessel disease, n (%) | 139 (20) | 103 (25) | 36 (13) | 0.004 |
| Coronary bifurcation involvement, n (%) | 193 (28) | 77 (19) | 116 (41) | 0.0001 |
| Overall (698 Patients) | Less Likely Plaque Erosion (413 Patients) | Suspected Plaque Erosion (283 Patients) | p Value | |
|---|---|---|---|---|
| Clopidogrel, n (%) | 318 (45.7) | 188 (45.5) | 130 (45.9) | 0.938 |
| Potent P2Y12 inhibitor, n (%) | 417 (59.9) | 255 (61.7) | 162 (57.2) | 0.239 |
| DAPT > 1 year, n (%) | 113 (16.2) | 74 (17.9) | 39 (13.8) | 0.174 |
| Ezetimibe, n (%) | 197 (28.3) | 79 (19.1) | 118 (41.7) | 0.0001 |
| High-intensity statin, n (%) | 638 (91.4) | 377 (90.8) | 261 (92.2) | 0.583 |
| Overall (696 Patients) | Less Likely Plaque Erosion (413 Patients) | Suspected Plaque Erosion (283 Patients) | Overall (696 Patients) | |
|---|---|---|---|---|
| Hemoglobin, gr/dL | 13.6 ± 2.1 | 12.4 ± 1.7 | 15.2 ± 1.3 | 0.0001 |
| White blood cells, 1000 xmm3 | 10.2 ± 4.6 | 9.8 ± 4.0 | 10.8 ± 5.3 | 0.012 |
| C-Reactive Protein, mg/dL | 3.6 ± 5.8 | 4.4 ± 6.4 | 2.7 ± 4.7 | 0.017 |
| Fibrinogen, mg/dL | 486.9 ± 172.2 | 516.4 ± 166.1 | 446.6 ± 159.7 | 0.024 |
| High sensitivity troponin I, ng/L | 2482.6 ± 2863.4 | 2473.0 ± 2825.1 | 2489.2 ± 2894.5 | 0.953 |
| Platelets, 1000/µl | 227.4 ± 72.6 | 223.5 ± 79.7 | 232 ± 61.4 | 0.110 |
| Uric acid, mg/dL | 6.2 ± 1.7 | 6.3 ± 1.9 | 6.1 ± 1.5 | 0.234 |
| LDL cholesterol, mg/dL | 122.1 ± 38.1 | 108.1 ± 33.3 | 139.7 ± 36.6 | 0.0001 |
| HDL cholesterol, mg/dL | 42.1 ± 9.7 | 42.2 ± 8.8 | 42.2 ± 8.8 | 0.766 |
| Triglyceride, mg/dL | 134.7 ± 72.3 | 126.6 ± 58.8 | 145.2 ± 85.8 | 0.002 |
| Creatinine, mg/dL | 1.2 ± 0.9 | 1.4 ± 1.1 | 0.9 ± 0.2 | 0.0001 |
| Glycemia, mg/dL | 138.3 ± 64.5 | 161.7 ± 68.6 | 117.3 ± 52.3 | 0.0001 |
| Overall (696 Patients) | Less Likely Plaque Erosion (413 Patients) | Suspected Plaque Erosion (283 Patients) | p-Value | |
|---|---|---|---|---|
| At 1-month follow-up | ||||
| Creatinine, mg/dL | 1.1 ± 0.7 | 1.3 ± 0.9 | 0.9 ± 0.1 | 0.0001 |
| Glycemia, mg/dL | 113.1 ± 35.3 | 123.4 ± 42.9 | 101.9 ± 19.1 | 0.0001 |
| HbA1c, % | 6.8 ± 1.2 | 7.2 ± 1.1 | 5.9 ± 0.9 | 0.0001 |
| Hemoglobin, gr/dL | 13.5 ± 1.9 | 12.4 ± 1.7 | 14.7 ± 1.2 | 0.0001 |
| LDL cholesterol, mg/dL | 79.1 ± 27.7 | 79.0 ± 30.2 | 79.2 ± 25.1 | 0.961 |
| HDL cholesterol, mg/dL | 42.9 ± 11.3 | 44.4 ± 9.5 | 43.3 ± 12.9 | 0.500 |
| Triglycerides, mg/dL | 126.0 ± 60.4 | 122.5 ± 54.1 | 129.3 ± 65.9 | 0.380 |
| At 12-month follow-up | ||||
| Creatinine, mg/dL | 1.3 ± 3.1 | 1.7 ± 4.4 | 0.9 ± 0.2 | 0.048 |
| Glycemia, mg/dL | 118.4 ± 41.5 | 133.4 ± 52.0 | 102.5 ± 14.3 | 0.0001 |
| HbA1c, % | 6.6 ± 1.1 | 7.2 ± 1.0 | 5.7 ± 0.4 | 0.0001 |
| Hemoglobin, gr/dL | 13.5 ± 1.9 | 12.4 ± 1.7 | 14.7 ± 1.2 | 0.0001 |
| LDL cholesterol, mg/dL | 75.2 ± 26.9 | 74.4 ± 26.8 | 76.0 ± 27.1 | 0.636 |
| HDL cholesterol, mg/dL | 43.8 ± 10.3 | 43.9 ± 10.3 | 43.7 ± 10.3 | 0.899 |
| Triglycerides, mg/dL | 129.6 ± 66.8 | 127.1 ± 68.5 | 132.3 ± 65.2 | 0.616 |
| Overall (696 Patients) | Less Likely Plaque Erosion (413 Patients) | Suspected Plaque Erosion (283 Patients) | p Value | |
|---|---|---|---|---|
| MACE, n (%) | 140 (20.1) | 119(28.8) | 21 (7.4) | 0.0001 |
| Death from any cause | 109 (15.7) | 103(24.9) | 6 (2.1) | 0.0001 |
| Non-fatal recurrent coronary event, n (%) | 41 (5.9) | 26 (6.3) | 15 (5.3) | 0.626 |
| HR (95% CI) | p Value | |
|---|---|---|
| Suspected plaque erosion | 0.37 (0.18–0.76) | 0.007 |
| NSTEMI as a type of ACS presentation | 1.32 (0.89–1.96) | 0.162 |
| Chronic kidney disease | 0.79 (0.54–1.17) | 0.253 |
| Diabetes mellitus | 0.95 (0.65–1.38) | 0.782 |
| Male sex | 0.91 (0.63–1.32) | 0.645 |
| Age | 1.02 (1.01–1.05) | 0.031 |
| Overall (696 Patients) | Less Likely Plaque Erosion (413 Patients) | Suspected Plaque Erosion (283 Patients) | p-Value | |
|---|---|---|---|---|
| Non-TVR, n (%) | 12 (1.7) | 9 (2.1) | 3 (1.0) | 0.608 |
| TVR, n (%) | 19 (2.7) | 11 (2.6) | 8 (2.8) | |
| Small vessel disease, n (%) | 10 (1.4) | 6 (1.4) | 4 (1.4) |
| Overall (41 RCES) | Less Likely Plaque Erosion (26 RCEs) | Suspected Plaque Erosion (15 RCEs) | p-Value | |
|---|---|---|---|---|
| Chronic coronary syndrome, n (%) | 9 (22) | 4 (15.4) | 5(33.3) | 0.248 |
| Acute coronary syndrome, n (%) | 32 (78) | 22 (84.6) | 10 (66.7) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Vito, L.; Scalone, G.; Di Giusto, F.; Bruscoli, F.; Alfieri, M.; Delfino, D.; Panzella, F.; Silenzi, S.; Jang, I.-K.; Grossi, P. Clinical Characteristics and Outcomes of Patients Presenting with Acute Coronary Syndromes and Suspected Plaque Erosion Based on Clinical and Laboratory Criteria. J. Cardiovasc. Dev. Dis. 2025, 12, 335. https://doi.org/10.3390/jcdd12090335
Di Vito L, Scalone G, Di Giusto F, Bruscoli F, Alfieri M, Delfino D, Panzella F, Silenzi S, Jang I-K, Grossi P. Clinical Characteristics and Outcomes of Patients Presenting with Acute Coronary Syndromes and Suspected Plaque Erosion Based on Clinical and Laboratory Criteria. Journal of Cardiovascular Development and Disease. 2025; 12(9):335. https://doi.org/10.3390/jcdd12090335
Chicago/Turabian StyleDi Vito, Luca, Giancarla Scalone, Federico Di Giusto, Filippo Bruscoli, Michele Alfieri, Domenico Delfino, Federico Panzella, Simona Silenzi, Ik-Kyung Jang, and Pierfrancesco Grossi. 2025. "Clinical Characteristics and Outcomes of Patients Presenting with Acute Coronary Syndromes and Suspected Plaque Erosion Based on Clinical and Laboratory Criteria" Journal of Cardiovascular Development and Disease 12, no. 9: 335. https://doi.org/10.3390/jcdd12090335
APA StyleDi Vito, L., Scalone, G., Di Giusto, F., Bruscoli, F., Alfieri, M., Delfino, D., Panzella, F., Silenzi, S., Jang, I.-K., & Grossi, P. (2025). Clinical Characteristics and Outcomes of Patients Presenting with Acute Coronary Syndromes and Suspected Plaque Erosion Based on Clinical and Laboratory Criteria. Journal of Cardiovascular Development and Disease, 12(9), 335. https://doi.org/10.3390/jcdd12090335








