Using Naturalistic Driving Data to Predict Mild Cognitive Impairment and Dementia: Preliminary Findings from the Longitudinal Research on Aging Drivers (LongROAD) Study
Abstract
1. Introduction
2. Materials and Methods
2.1. LongROAD Study
2.2. Monthly Record Classification
2.3. Five Classification Models
2.4. Random Forests
2.4.1. Performance Metrics
2.4.2. Test Data Selection
2.4.3. Training Data Rebalance
2.4.4. Parameter Tuning
3. Results
3.1. Model Comparison
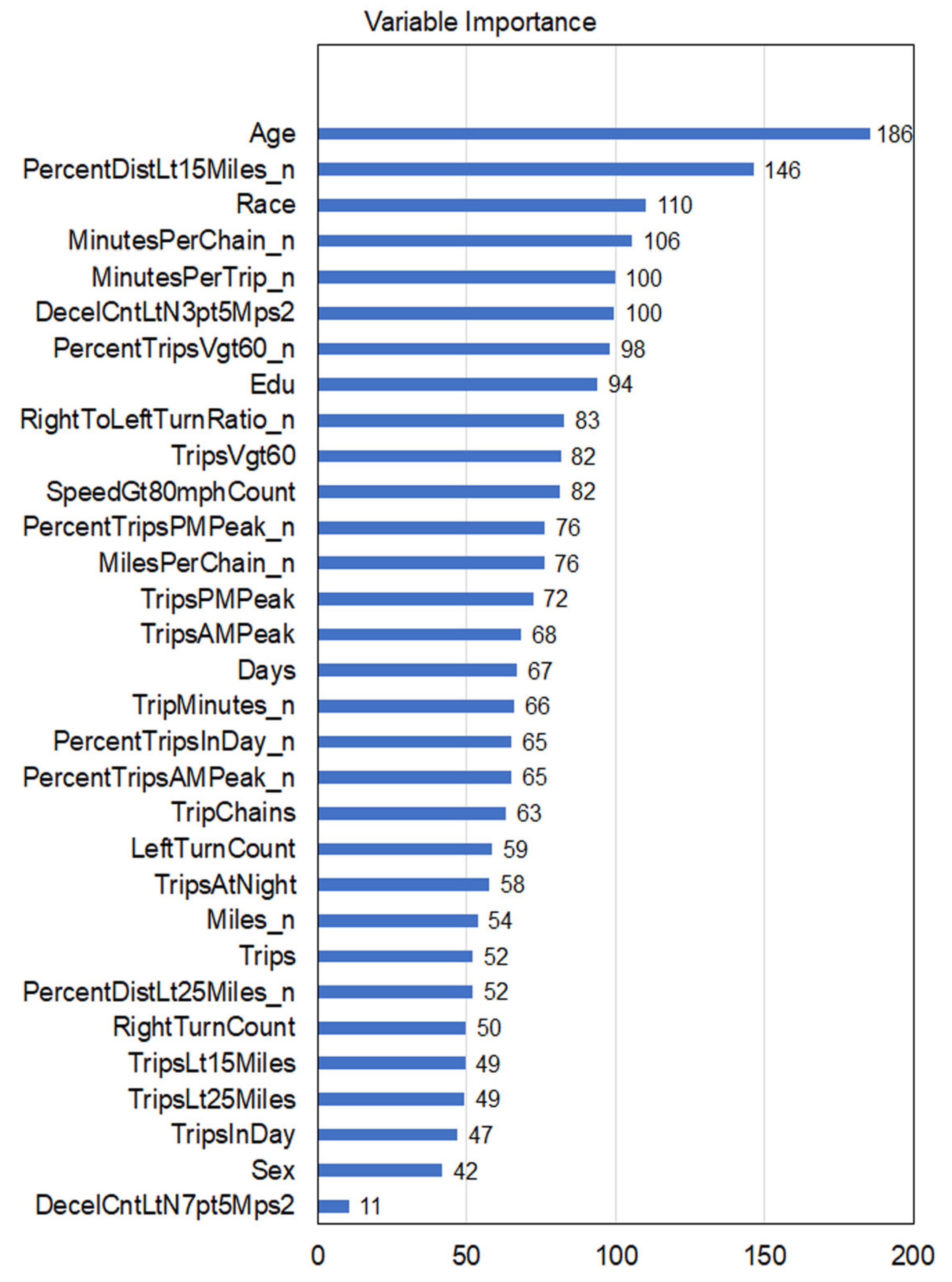
3.2. Feature Importance Ranking
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Roberts, A.W.; Ogunwole, S.U.; Blakeslee, L.; Rabe, M.A. The Population 65 Years and Older in the United States: 2016. US Census Bureau: Washington, DC, USA, 2018; American Community Survey Reports, ACS-38. Available online: https://www.census.gov/content/dam/Census/library/publications/2018/acs/ACS-38.pdf (accessed on 20 March 2021).
- Federal Highway Administration (FHWA). Distribution of Licensed Drivers—2016, by Sex and Percentage in each Age Group and Relation to Population; US Department of Transportation, Federal Highway Administration: Washington, DC, USA, 2017. Available online: https://www.fhwa.dot.gov/policyinformation/statistics/2016/dl20.cfm (accessed on 20 March 2021).
- Ott, B.R.; Jones, R.N.; Noto, R.B.; Yoo, D.C.; Snyder, P.J.; Bernier, J.N.; Carr, D.B.; Roe, C.M. Brain Amyloid in Preclinical Alzheimer’s Disease Is Associated with Increased Driving Risk. Alzheimers Dement. 2017, 6, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Roe, C.M.; Babulal, G.M.; Head, D.M.; Stout, S.H.; Vernon, E.K.; Ghoshal, N.; Garland, B.; Barco, P.P.; Williams, M.M.; Johnson, A.; et al. Preclinical Alzheimer’s Disease and Longitudinal Driving Decline. Alzheimers Dement. 2017, 3, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Roe, C.M.; Barco, P.P.; Head, D.M.; Ghoshal, N.; Selsor, N.; Babulal, G.M.; Fierberg, R.; Vernon, E.K.; Shulman, N.; Johnson, A.; et al. Amyloid Imaging, Cerebrospinal Fluid Biomarkers Predict Driving Performance Among Cognitively Normal Individuals. Alzheimer Dis. Assoc. Disord. 2017, 31, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Roe, C.M.; Babulal, G.M.; Mishra, S.; Gordon, B.A.; Stout, S.H.; Ott, B.R.; Carr, D.B.; Ances, B.M.; Morris, J.C.; Benzinger, T.L.S. Tau and Amyloid Positron Emission Tomography Imaging Predict Driving Performance Among Older Adults with and without Preclinical Alzheimer’s Disease. J. Alzheimers Dis. 2018, 61, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Roe, C.M.; Stout, S.H.; Rajasekar, G.; Ances, B.M.; Jones, J.M.; Head, D.; Benzinger, T.L.S.; Williams, M.M.; Davis, J.D.; Ott, B.R.; et al. A 2.5-Year Longitudinal Assessment of Naturalistic Driving in Preclinical Alzheimer’s Disease. J. Alzheimers Dis. 2019, 68, 1625–1633. [Google Scholar] [CrossRef] [PubMed]
- Babulal, G.M.; Stout, S.H.; Head, D.; Holtzman, D.M.; Fagan, A.M.; Morris, J.C.; Roe, C.M. Neuropsychiatric Symptoms and Alzheimer’s Disease Biomarkers Predict Driving Decline: Brief Report. J. Alzheimers Dis. 2017, 58, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Babulal, G.M.; Chen, S.; Williams, M.M.; Trani, J.-F.; Bakhshi, P.; Chao, G.L.; Stout, S.H.; Fagan, A.M.; Benzinger, T.L.S.; Holtzman, D.M.; et al. Depression and Alzheimer’s Disease Biomarkers Predict Driving Decline. J. Alzheimers Dis. 2018, 66, 1213–1221. [Google Scholar] [CrossRef] [PubMed]
- Babulal, G.M.; Stout, S.H.; Benzinger, T.L.S.; Ott, B.R.; Carr, D.B.; Webb, M.; Traub, C.M.; Addison, A.; Morris, J.C.; Warren, D.K.; et al. A Naturalistic Study of Driving Behavior in Older Adults and Preclinical Alzheimer Disease: A Pilot Study. J. Appl. Gerontol. 2019, 38, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Eby, D.W.; Silverstein, N.M.; Molnar, L.J.; LeBlanc, D.; Adler, G. Driving Behaviors in Early Stage Dementia: A Study Using in-Vehicle Technology. Accid. Anal. Prev. 2012, 49, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.L.; Ohman, J.M. Driving in Early-Stage Alzheimer’s Disease: An Integrative Review of the Literature. Res. Gerontol. Nurs. 2017, 10, 86–100. [Google Scholar] [CrossRef] [PubMed]
- Allison, S.; Babulal, G.M.; Stout, S.H.; Barco, P.P.; Carr, D.B.; Fagan, A.M.; Morris, J.C.; Roe, C.M.; Head, D. Alzheimer Disease Biomarkers and Driving in Clinically Normal Older Adults: Role of Spatial Navigation Abilities. Alzheimer Dis. Assoc. Disord. 2018, 32, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Eby, D.W.; Santos, R.; Mielenz, T.J.; Molnar, L.J.; Strogatz, D.; Betz, M.E.; DiGuiseppi, C.; Ryan, L.H.; Jones, V.; et al. Longitudinal Research on Aging Drivers (LongROAD): Study design and methods. Inj. Epidemiol. 2017, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Babulal, G.M.; Johnson, A.; Fagan, A.M.; Morris, J.C.; Roe, C.M. Identifying Preclinical Alzheimer’s Disease Using Everyday Driving Behavior: Proof of Concept. J. Alzheimers Dis. 2021, 79, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Campbell, N.L.; Unverzagt, F.; LaMantia, M.A.; Khan, B.A.; Boustani, M.A. Risk Factors for the Progression of Mild Cognitive Impairment to Dementia. Clin. Geriatr. Med. 2013, 29, 873–893. [Google Scholar] [CrossRef] [PubMed]
| Variable | Name | Definition | Statistics | ||||
|---|---|---|---|---|---|---|---|
| Min | Max | Mean | SD | ||||
| Diagnosis_labels | MCI/Dementia | One’s disease level in a month (0-Healthy; 1-Mild Cognitive Impairment (MCI)/Dementia/Alzheimer’s | 0 | 1 | - | - | |
| Demographic characteristics | |||||||
| Age in years | Age | Age at enrollment | 65 | 79 | 71.1 | 4.1 | |
| Sex | Sex | Male; Female | NA | NA | NA | NA | |
| Race/Ethnicity | Race | Alaska Native, Native Hawaiian, Pacific Islander; American Indian, Asian; Black (non-Hispanic); White (non-Hispanic); Hispanic; Other | NA | NA | NA | NA | |
| Education | Education | Associate degree; Bachelor degree; Master, professional, or doctoral degree; Some college but no degree; Vocational, technical, business, or trade school (beyond high school level); Other | NA | NA | NA | NA | |
| Driving variables | |||||||
| 1 | Miles_n | Miles | Total number of miles driven in month | 0 | 15,783 | 762.2 | 587.8 |
| 2 | Trips | Trips | Total number of trips in month | 1 | 2341 | 115.8 | 64.6 |
| 3 | TripsLt15Miles | No. trips < 15 miles of home | Number of trips traveled in month within 15 miles of home | 0 | 1953 | 95.5 | 58.4 |
| 4 | PercentDistLt15Miles_n | % trip < 15 miles of home | Percent of trips traveled in month within 15 miles of home | 0.0 | 100.0 | 64.9 | 28.9 |
| 5 | TripsLt25Miles | No. trips < 25 miles of home | Number of trips traveled in month within 25 miles of home | 0 | 1953 | 101.7 | 59.7 |
| 6 | PercentDistLt25Miles_n | % trip < 25 miles of home | Percent of trips traveled in month within 25 miles of home | 0.0 | 100.0 | 76.5 | 26.4 |
| 7 | MilesPerTrip_n | Miles per trip | Total number of miles driven in month divided by total number of trips in month | 0.0 | 74.5 | 6.7 | 4.1 |
| 8 | MinutesPerTrip_n | Minutes per trip | Total driving minutes in month divided by total number of trips in month | 0.1 | 137.7 | 14.9 | 5.9 |
| 9 | TripMinutes_n | Total trip minutes | Total minutes of driving in month | 0.1 | 16,645.0 | 1633.4 | 1083.6 |
| 10 | TripsInDay | No. trips during day | Number of trips in month not classified as nighttime | 0 | 1279 | 107.2 | 57.4 |
| 11 | PercentTripsInDay_n | % trips during day | Percent of trips in month not classified as nighttime | 0.0 | 100.0 | 93.1 | 8.0 |
| 12 | TripsAMPeak | No. trips in AM peak | Number of trips in month during 7–9 AM on weekdays | 0 | 167 | 8.6 | 9.4 |
| 13 | PercentTripsAMPeak_n | % trips in AM peak | Percent of trips in month during 7–9 AM on weekdays | 0.0 | 100.0 | 7.3 | 6.9 |
| 14 | TripsAtNight | No. trips at night | Number of trips during which at least 80% of a trip was during nightime in month (Nightime was defined as civil twilight or a solar angle greater than 96 deg) | 0 | 1143 | 8.7 | 16.6 |
| 15 | PercentTripsAtNight_n | % trips at night | Percent of trips during which at least 80% of a trip was during nightime in month (Nightime was defined as civil twilight or a solar angle greater than 96 deg) | 0.0 | 100.0 | 6.9 | 8.0 |
| 16 | TripsPMPeak | No. trips in PM peak | Number of trips in month during 4–6PM on weekdays | 0 | 150 | 10.9 | 9.5 |
| 17 | PercentTripsPMPeak_n | % trips in PM peak | Percent of trips in month during 4–6PM on weekdays | 0.0 | 100.0 | 9.3 | 6.7 |
| 18 | LeftTurnCount | No. left turns | Number of left turns made in month | 0 | 2592 | 261.6 | 159.9 |
| 19 | RightTurnCount | No.right turns | Number of right turns made in month | 0 | 2751 | 242.6 | 150.1 |
| 20 | RightToLeftTurnRatio_n | Right to left turn ratio | Ratio of all right-hand to left-hand turning events for a driver in a month | 0.0 | 7.0 | 0.9 | 0.2 |
| 21 | TripsVgt60 | No. trips on high speed roads | Number of trips in month where 20% of distance travelled was at a speed of 60 MPH or greater | 0 | 226 | 13.9 | 15.0 |
| 22 | PercentTripsVgt60_n | % trip on high speed roads | Percent of trips in month where 20% of distance travelled was at a speed of 60 MPH or greater | 0.0 | 12.6 | 100.0 | 12.3 |
| 23 | SpeedGt80mphCount | No. speeding events | Number speeding events in month (speed > 80 MPH sustained for at least 8 s) | 0 | 3300 | 7.3 | 31.3 |
| 24 | DecelCntLtN3pt5Mps2 | No. hard braking events with deceleration rates ≥ 0.35 g | Number of events with a deceleration rate ≥ 0.35 g in a month | 0 | 1112 | 3.8 | 8.2 |
| 25 | DecelCntLtN4pt0Mps2 | No. hard braking events with deceleration rates ≥ 0.40 g | Number of events with a deceleration rate ≥ 0.4 g in a month | 0 | 734 | 0.9 | 4.2 |
| 26 | DecelCntLtN7pt5Mps2 | No. hard braking events ≥ 0.75 g | Number of events with a deceleration rate ≥ 0.75 g in a month | 0 | 24 | 0.005 | 0.2 |
| 27 | TripChains | Trip chains | Number of trip chains in month (Note: chain is a series of trips starting and ending at home) | 0 | 180 | 8.2 | 7.8 |
| 28 | MilesPerChain_n | Miles per chain | Total miles of chains in month divided by total number of trip chains in month | 0.00 | 4273.3 | 100.7 | 121.1 |
| 29 | MinutesPerChain_n | Minutes per chain | Total driving minutes for chains divided by total number of trip chains in month | 0.0 | 6606.3 | 222.0 | 219.8 |
| Model | Covariates | Accuracy | Precision or PPV | Recall or Sensitivity | Specificity | NPV | F1 Score | AUC | Out-of-Bag Error Rate | Confusion Matrix | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Predicted | Observed | ||||||||||||
| % | SD | 0 | 1 | ||||||||||
| 1 | Age only | 0.46 | 1.00 | 0.06 | 1.00 | 0.45 | 0.11 | 0.56 | 6.00 | 0.01 | 0 | 77 | 96 |
| 1 | 0 | 6 | |||||||||||
| 2 | Age, sex, race/ethnicity, and education | 0.53 | 1.00 | 0.17 | 1.00 | 0.48 | 0.29 | 0.64 | 0 | 77 | 85 | ||
| 4.27 | 0.01 | 1 | 0 | 17 | |||||||||
| 3 | Driving variables only | 0.66 | 0.79 | 0.56 | 0.81 | 0.58 | 0.66 | 0.76 | 2.60 | 0.75 | 0 | 62 | 45 |
| 1 | 12 | 57 | |||||||||||
| 4 | Age and driving variables | 0.80 | 0.89 | 0.74 | 0.88 | 0.72 | 0.81 | 0.91 | 2.14 | 0.50 | 0 | 68 | 27 |
| 1 | 9 | 75 | |||||||||||
| 5 | Age, sex, race/ethnicity, education and driving variables | 0.86 | 0.86 | 0.90 | 0.81 | 0.86 | 0.88 | 0.90 | 2.07 | 0.57 | 0 | 62 | 10 |
| 1 | 15 | 92 | |||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di, X.; Shi, R.; DiGuiseppi, C.; Eby, D.W.; Hill, L.L.; Mielenz, T.J.; Molnar, L.J.; Strogatz, D.; Andrews, H.F.; Goldberg, T.E.; et al. Using Naturalistic Driving Data to Predict Mild Cognitive Impairment and Dementia: Preliminary Findings from the Longitudinal Research on Aging Drivers (LongROAD) Study. Geriatrics 2021, 6, 45. https://doi.org/10.3390/geriatrics6020045
Di X, Shi R, DiGuiseppi C, Eby DW, Hill LL, Mielenz TJ, Molnar LJ, Strogatz D, Andrews HF, Goldberg TE, et al. Using Naturalistic Driving Data to Predict Mild Cognitive Impairment and Dementia: Preliminary Findings from the Longitudinal Research on Aging Drivers (LongROAD) Study. Geriatrics. 2021; 6(2):45. https://doi.org/10.3390/geriatrics6020045
Chicago/Turabian StyleDi, Xuan, Rongye Shi, Carolyn DiGuiseppi, David W. Eby, Linda L. Hill, Thelma J. Mielenz, Lisa J. Molnar, David Strogatz, Howard F. Andrews, Terry E. Goldberg, and et al. 2021. "Using Naturalistic Driving Data to Predict Mild Cognitive Impairment and Dementia: Preliminary Findings from the Longitudinal Research on Aging Drivers (LongROAD) Study" Geriatrics 6, no. 2: 45. https://doi.org/10.3390/geriatrics6020045
APA StyleDi, X., Shi, R., DiGuiseppi, C., Eby, D. W., Hill, L. L., Mielenz, T. J., Molnar, L. J., Strogatz, D., Andrews, H. F., Goldberg, T. E., Lang, B. H., Kim, M., & Li, G. (2021). Using Naturalistic Driving Data to Predict Mild Cognitive Impairment and Dementia: Preliminary Findings from the Longitudinal Research on Aging Drivers (LongROAD) Study. Geriatrics, 6(2), 45. https://doi.org/10.3390/geriatrics6020045






