Multiple Effects of Egg Weight, in Ovo Carbohydrates, and Sex of Birds on Posthatch Performance in Broilers
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Incubation Protocol
2.2. Treatment Groups
2.3. In Ovo Intervention
2.4. Housing Conditions and Feeding Management
2.5. Statistical Analysis
3. Results
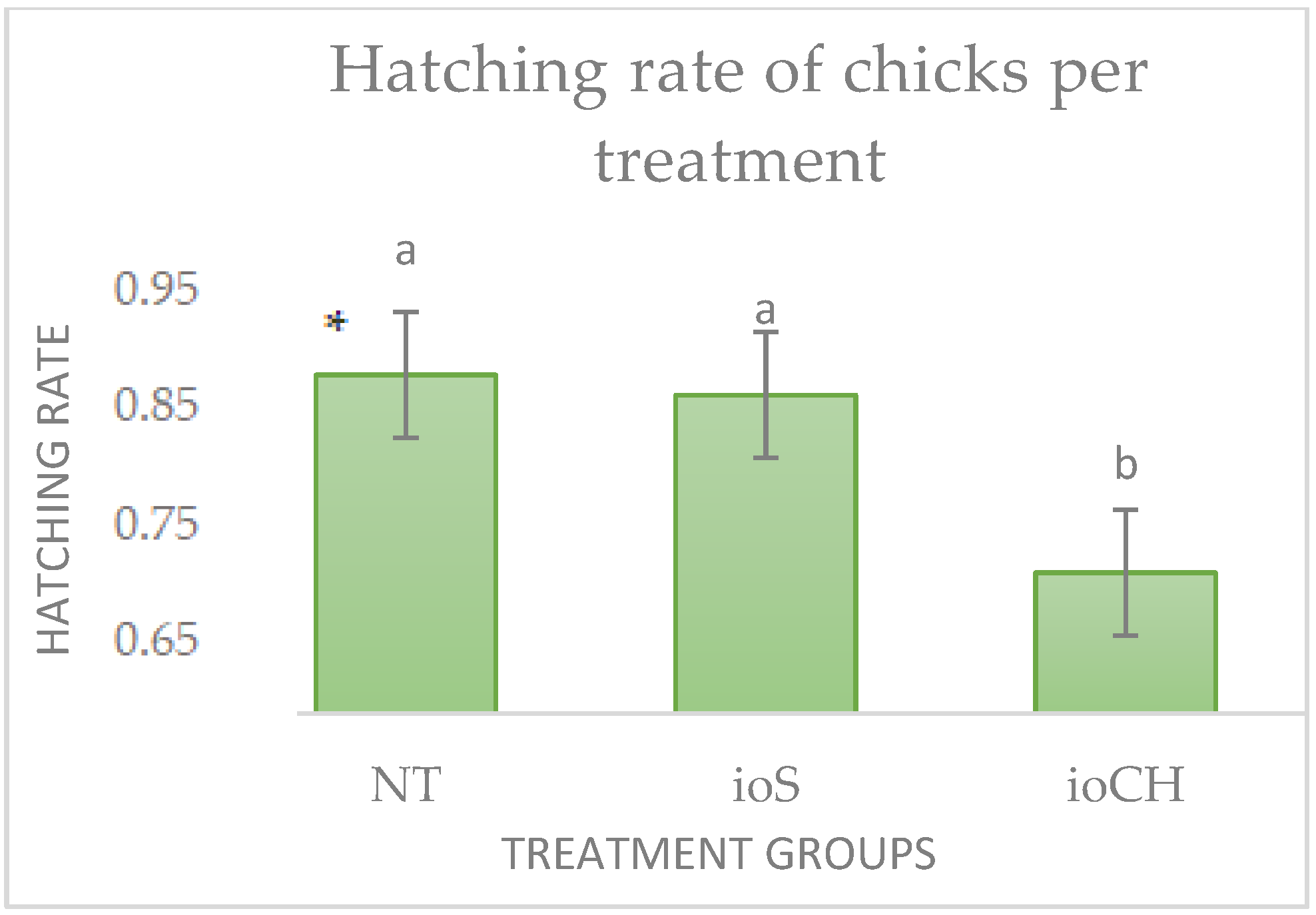
3.1. Hatchability Results
3.1.1. Hatching Rate
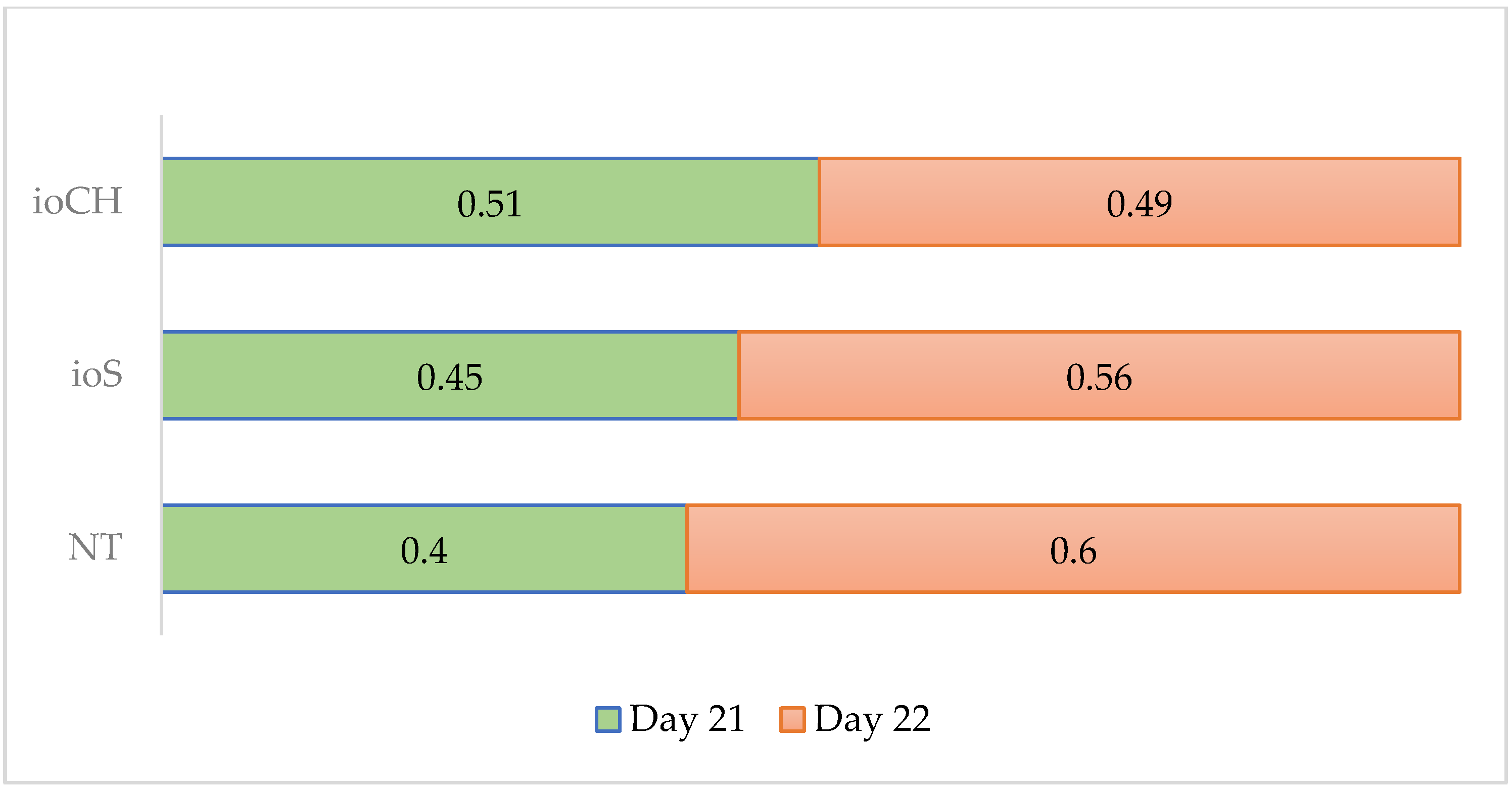
3.1.2. Ratio of Hatched Eggs per Day
3.2. Growth Performance
3.2.1. Liveweight Results
3.2.2. Average Daily Gain Results
3.2.3. Feed Intake and Feed Conversion Ratio
| HW (g) | LW10 (g) | LW21 (g) | LW35 (g) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Body Weight per Sex | NT | ioS | ioCH | NT | ioS | ioCH | NT | ioS | ioCH | NT | ioS | ioCH |
| Female | 43.1 | 44.2 | 44.1 | 185.5 | 184.2 | 185.7 | 676.5 | 651.4 | 689.2 | 1988.7 | 1909.6 | 2033.6 |
| Male | 43.3 | 44.1 | 44.1 | 183.5 | 192.8 | 195.2 | 709.5 | 723.9 | 754.2 | 2108.3 | 2088.8 | 2165.2 |
| Sex p-value | 0.06 | 0.04 | 0.24 | <0.001 | ||||||||
| Treatment p-value | 0.3 | 0.18 | 0.0082 | 0.0004 | ||||||||
| Post hoc Tukey’s test results | a | a | a | a | a | a | ab | a | b | ab | a | b |
| Egg weight | ||||||||||||
| 53–58 g | 42 | 43 | 42 | 178.0 | 179.5 | 184.4 | 671.2 | 667.5 | 707.3 | 2003.2 | 1986.2 | 2058.7 |
| >58 g | 46 | 46 | 46 | 190.8 | 198.8 | 197.6 | 719.7 | 717.4 | 744.3 | 2112.7 | 2041.4 | 2160.9 |
| Egg weight p-value | <0.001 | <0.001 | <0.001 | <0.001 | ||||||||
| Day of hatch | ||||||||||||
| Day 21 | 45 | 45 | 45 | 195.2 | 196.2 | 192 | 691.4 | 700.2 | 716.2 | 2038 | 2005.8 | 2100 |
| Day22 | 43 | 43 | 42 | 176.8 | 183.4 | 190 | 697.7 | 686.2 | 735.4 | 2071 | 2020.1 | 2117.8 |
| Day of hatch p-value | <0.001 | <0.001 | 0.51 | 0.17 | ||||||||
| p-values of interactions | ||||||||||||
| Day of hatch × Egg weight | <0.001 | 0.84 | 0.009 | 0.009 | ||||||||
| Day of hatch × Sex | <0.001 | 0.63 | 0.12 | 0.46 | ||||||||
| Egg weight × Sex | 0.57 | 0.46 | 0.55 | 0.03 | ||||||||
| Day of hatch × Trt | 0.02 | 0.056 | 0.1 | 0.81 | ||||||||
| Egg weight × Trt | <0.001 | 0.52 | 0.42 | 0.43 | ||||||||
| Sex × Trt | 0.8 | 0.14 | 0.91 | 0.48 | ||||||||
| Day of hatch × Egg weight × Sex | 0.15 | 0.93 | 0.19 | 0.12 | ||||||||
| Day of hatch × Egg weight × Trt | 0.09 | 0.01 | 0.51 | 0.01 | ||||||||
| Egg weight × Sex × Trt | 0.88 | 0.51 | 0.32 | 0.22 | ||||||||
| Day of hatch × Egg weight × Sex × Trt | 0.01 | 0.56 | 0.19 | 0.32 | ||||||||
| ADG 1–10 | ADG 11–21 | ADG 22–35 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| ADG per Sex | NT | ioS | ioCH | NT | ioS | ioCH | NT | ioS | ioCH |
| Female | 14.3 | 14.2 | 14.3 | 44.6 | 42.4 | 45.7 | 91.5 | 88.5 | 94.7 |
| Male | 14.2 | 15 | 15.3 | 47.8 | 48.3 | 50.7 | 98.39 | 97.2 | 100.8 |
| Sex p-value | 0.04 | <0.001 | <0.001 | ||||||
| Treatment p-value | 0.27 | 0.0032 | 0.0013 | ||||||
| Tukey’s test results | a | a | a | a | a | b | ab | b | a |
| Egg weight | |||||||||
| 53–58 g | 13.7 | 13.9 | 14.4 | 44.8 | 44.3 | 47.5 | 93.2 | 93.24 | 95.8 |
| >58 g | 14.7 | 15.4 | 15.3 | 48 | 47.1 | 49.3 | 97.8 | 93.9 | 99.7 |
| Egg weight p-value | <0.001 | <0.001 | <0.001 | ||||||
| Day of hatch | |||||||||
| Day 21 | 15.3 | 15.3 | 14.9 | 45.1 | 45.7 | 47.6 | 94.4 | 92.5 | 97.6 |
| Day22 | 13.5 | 14.1 | 14.7 | 47.4 | 45.7 | 49.5 | 96.2 | 94.4 | 97.9 |
| Day of hatch p-value | <0.001 | 0.02 | 0.09 | ||||||
| Interactions | |||||||||
| Day of hatch × Egg weight | 0.74 | 0.07 | 0.01 | ||||||
| Day of hatch × Sex | 0.67 | 0.4 | 0.41 | ||||||
| Egg weight × Sex | 0.47 | 0.08 | 0.03 | ||||||
| Day of hatch × Trt | 0.07 | 0.34 | 0.67 | ||||||
| Egg weight × Trt | 0.51 | 0.87 | 0.19 | ||||||
| Sex × Trt | 0.13 | 0.27 | 0.46 | ||||||
| Day of hatch × Egg weight × Sex | 0.97 | 0.41 | 0.12 | ||||||
| Day of hatch × Egg weight × Trt | 0.01 | 0.09 | 0.01 | ||||||
| Egg weight × Sex × Trt | 0.5 | 0.24 | 0.35 | ||||||
| Day of hatch × Egg weight × Sex × Trt | 0.8 | 0.36 | 0.35 | ||||||
3.2.4. Regression Model
3.3. Carcass Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zuidhof, M.J.; Schneider, B.L.; Carney, V.L.; Korver, D.L.; Robinson, F.E. Growth, Efficiency, and Yield of Commercial Broilers from 1957, 1978, and 2005. Poult. Sci. 2014, 93, 2970–2982. [Google Scholar] [CrossRef] [PubMed]
- Retes, P.L.; Clemente, A.H.S.; Neves, N.G.; Espósito, M.; Makiyama, L.; Alvarenga, R.R. In ovo feeding of carbohydrates for broilers—A systematic review. J. Anim. Phys. Anim. Nut. 2017, 102, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Noble, R.C.; Cocchi, M. Lipid metabolism and the neonatal chicken. Prog. Lipid Res. 1990, 29, 107–140. [Google Scholar] [CrossRef]
- Boersma, S.I.; Robinson, F.E.; Renema, R.A.; Fasenko, G.M. Administering oasis hatching supplement prior to chick placement increases initial growth with no effect on body weight uniformity of female broiler breeders after three weeks of age. J. Appl. Poult. Res. 2003, 12, 428–434. [Google Scholar] [CrossRef]
- De Oliveira, J.E.; Uni, Z.; Ferket, P.R. Important metabolic pathways in poultry embryos prior to hatch. World’s Poult. Sci. J. 2008, 64, 488–499. [Google Scholar] [CrossRef]
- Clark, D.; Sokoloff, L. Basic Neurochemistry: Molecular, Cellular and Medical Aspects; Lippincott Publication: Philadelphia, PA, USA, 1999; pp. 637–670. [Google Scholar]
- Christensen, V.L.; Wineland, M.J.; Fasenko, G.M.; Donaldson, W.E. Egg storage effects on plasma glucose and supply and demand tissue glycogen concentrations of broiler embryos. Poult. Sci. 2001, 80, 1729–1735. [Google Scholar] [CrossRef]
- Pearce, J. Carbohydrate metabolism in the domestic fowl. Proc. Nutr. Soc. 1971, 30, 254–259. [Google Scholar] [CrossRef]
- Klasing, K.C. Comperative Avian Nutrition; CAB International: Wallingford, UK, 1998; p. 350. [Google Scholar]
- Pearce, J.; Brown, W.O. Carbohydrate Metabolism in Physiology and Biochemistry of the Domestic Fowl; Bell, D.J., Freeman, B.M., Eds.; Academic Press: London, UK, 1971; Volume 1, pp. 295–319. [Google Scholar]
- Burley, R.W.; Vadehra, D.V. The Avian Egg. Chemistry and Biology; John Wiley and Sons: New York, NY, USA, 1989. [Google Scholar]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Biochemistry, 5th ed.; W. H. Freeman and Company: New York, NY, USA, 2002. [Google Scholar]
- Moran, E.T., Jr. Nutrition of the developing embryo and hatchling. Poult. Sci. 2007, 86, 1043–1049. [Google Scholar] [CrossRef]
- Sklan, D. Development of the digestive tract of poultry. Worlds Poult. Sci. J. 2001, 57, 415–428. [Google Scholar] [CrossRef]
- Sharma, J.; Burmester, B. Resistance of Marek's disease at hatching in chickens vaccinated as embryos with the turkey herpesvirus. Avian Dis. 1982, 26, 134–149. [Google Scholar] [CrossRef]
- Uni, Z.; Ferket, P.R. Enhancement of Development of Oviparous Species by in Ovo Feeding. U.S. Regular Patent US6592878B2, 15 July 2003. [Google Scholar]
- EI-Husseiny, O.M.; EI-Wafa, S.A.; EI-Komy, H.M.A. Influence of fasting or early feeding on broiler performance. Int. J. Poult. Sci. 2008, 7, 263–271. [Google Scholar]
- Alasahan, S.; Copur, A.G. Hatching characteristics and growth performance of eggs with different shapes. Braz. J. Poult. Sci. 2016, 18, 1–14. [Google Scholar] [CrossRef]
- Official Methods of Analysis of AOAC International, 19th ed.; Official Method 2008.01.; AOAC International: Gaithersburg, MD, USA, 2013.
- SAS Institute Inc. SAS/ACCESS® 9.4 Interface to ADABAS: Reference; SAS Institute Inc.: Cary, NC, USA, 2013. [Google Scholar]
- Pedroso, A.A.; Chave, L.S.; Lopes, K.L.A.M.; Leandro, N.S.M.; Café, M.B.; Stringhini, J.H. Inoculação de nutrientes em ovos de matrizes pesadas. Rev. Bras. Zootec. 2006, 35, 2018–2026. [Google Scholar] [CrossRef]
- Leitão, R.A.; Leandro, N.S.M.; Café, M.B.; Stringhini, J.H.; Pedroso, A.A.; Chaves, L.d.S. Inoculação de glicose em ovos embrionados de frango de corte: Parâmetros de incubação e desempenho inicial. Cienc. Anim. Bras. 2008, 9, 847–855. [Google Scholar]
- Zhai, W.; Rowe, D.E.; Peebles, E.D. Effects of commercial in ovo injection of carbohydrates on broiler embryogenesis. Poult. Sci. 2011, 90, 1295–1301. [Google Scholar] [CrossRef]
- Campos, A.M.A.; Rostagno, H.S.; Gomes, P.C.; Silva, E.A.; Albino, L.F.T.; Nogueira, E.T. Efeito da inoculação de soluções nutritivas in ovo sobre a eclodibilidade e o desempenho de frangos de corte. Rev. Bras. Zootec. 2011, 40, 1712–1717. [Google Scholar] [CrossRef]
- Chotinsky, D.; Toncheva, E.; Profirov, Y. Development of dissacharidases activity in the small intestine of broiler chickens. Br. Poult. Sci. 2001, 42, 389–393. [Google Scholar] [CrossRef]
- Pedroso, A.A.; Chaves, L.S.; de Almeida Martinez Lopes, K.L.; Leandro, N.S.M.; Café, M.B.; Stringhini, J.H. Nutrient inoculation in eggs from heavy breeders. Rev. Bras. Zootec. 2006, 5, 2018–2026. [Google Scholar] [CrossRef]
- Zhai, W.; Gerard, P.D.; Pulikanti, R.; Peebles, E. Effects of in ovo injection of carbohydrates on embryonic metabolism, hatchability, and subsequent somatic characteristics of broiler hatchlings. Poult. Sci. 2011, 90, 2134–2143. [Google Scholar] [CrossRef]
- Romanoff, A.L. Biochemistry and biophysics of the developing hen’s egg. I. Influence of humidity. Mem. Cornell Univ. Agric. Exp. Stn. 1930, 132, 1–27. [Google Scholar]
- Shafey, T.M.; Alodan, M.A.; Al-Ruqaie, I.M.; Abouheif, M.A. In ovo feeding of carbohydrates and incubated at a high incubation temperature on hatchability and glycogen status of chicks. S. Afr. J. Anim. Sci. 2012, 42, 210–220. [Google Scholar] [CrossRef]
- Salmanzadeh, M. The effects of in-ovo injection of glucose on hatchability, hatching weight and subsequent performance of newly-hatched chicks. Rev. Bras. Ciência Avícola 2012, 14, 137–140. [Google Scholar] [CrossRef]
- Salmanzadeh, M.; Ebrahimnejad, Y.; Shahryar, H.A.; Gorbani, A.; Oskuei, H.R. The effects of in ovo glucose administration on hatching results and subsequent blood glucose concentration in newly-hatched chicks. J. Appl. Biol. Sci. 2011, 5, 21–22. [Google Scholar]
- Enting, H.; Kruip, T.A.M.; Verstegen, M.W.A.; van Der Aar, P.J. The effect of low-density diets on broiler breeder performance during the laying period and on embryonic development of their offspring. Poult. Sci. 2007, 86, 850–885. [Google Scholar] [CrossRef]
- Kornasio, R.; Halevy, O.; Kedar, O.; Uni, Z. Effect of in ovo feeding and its interaction with timing of first feed on glycogen reserves, muscle growth, and body weight. Poult. Sci. 2011, 90, 1467–1477. [Google Scholar] [CrossRef]
- Uni, Z.; Ferket, P.R.; Tako, E.; Kedar, O. In ovo feeding improves energy status of late-term chicken embryos. Poult. Sci. 2005, 84, 764–770. [Google Scholar] [CrossRef]
- Chen, W.; Wang, R.; Wan, H.F.; Xiong, X.L.; Peng, P.; Peng, J. Influence of in ovo injection of glutamine and carbohydrates on digestive organs and pectoralis muscle mass in the duck. Brit. Poult. Sci. 2009, 50, 436–442. [Google Scholar] [CrossRef]
- Amitav, B.; Majumdar, S.; Bhanja, S.K.; Mandal, A.B.; Dash, B.B.; Agarwal, S.K. Effect of in ovo injection of glucoseon growth, immunocompetence and development of diges-tive organs in turkey poults. In Proceedings of the 16th European Symposium on Poultry Nutrition, Strasbourg, France, 26–30 August 2007; pp. 147–150. [Google Scholar]
- Bhanja, S.K.; Mandal, A.B.; Agarwal, S.K.; Majumdar, S. Effect of in ovo glucose injection on the post-hatch growth, digestive organ development and blood biochemical profilesin broiler chickens. Indian J. Anim. Sci. 2008, 78, 869–872. [Google Scholar]
- Tako, E.; Ferket, P.R.; Uni, Z. Effects of in ovo feeding of carbohydrates and beta-hydroxy-beta-methylbutyrate on the development of chicken intestine. Poult. Sci. 2004, 83, 2023–2028. [Google Scholar] [CrossRef] [PubMed]
- Kucharska-Gaca, J.; Kowalska, E.; Dębowska, M. In ovo Feeding-Technology of the Future—A Review. Ann. Anim. Sci. 2017, 17, 979–992. [Google Scholar] [CrossRef] [Green Version]



| Tray Levels | Egg Weight | Treatment Groups | Number of Eggs |
|---|---|---|---|
| 1 | Light | ioCH | 66 |
| Heavy | 67 | ||
| 2 | Light | ioS | 67 |
| Heavy | 66 | ||
| 3 | Light | NT | 67 |
| Heavy | 67 | ||
| 4 | Light | ioCH | 67 |
| Heavy | 67 | ||
| 5 | Light | ioS | 66 |
| Heavy | 67 | ||
| 6 | Light | NT | 66 |
| Heavy | 67 | ||
| 7 | Light | ioCH | 67 |
| Heavy | 66 | ||
| 8 | Light | ioS | 67 |
| Heavy | 67 | ||
| 9 | Light | NT | 66 |
| Heavy | 66 |
| Hatching Day | °C | % CO2 |
|---|---|---|
| 1 | 37.9 | 0.60 |
| 2 | 37.9 | 0.60 |
| 3 | 37.9 | 0.60 |
| 4 | 37.9 | 0.60 |
| 5 | 37.9 | 0.60 |
| 6 | 37.9 | 0.60 |
| 7 | 37.8 | 0.60 |
| 8 | 37.8 | 0.60 |
| 9 | 37.6 | 0.60 |
| 10 Candling | 37.6 | 0.60 |
| 11 | 37.5 | 0.35 |
| 12 | 37.5 | 0.35 |
| 13 | 37.4 | 0.35 |
| 14 | 37.3 | 0.35 |
| 15 | 37.3 | 0.35 |
| 16 | 37.2 | 0.35 |
| 17 Candling, in ovo intervention, placing into the incubator | 37.1 | 0.35 |
| 18 | 37.0/36.7 | 0.35/0.60 |
| 19 | 36.7 | 0.60 |
| 20 | 36.5 | 0.60 |
| 21 | 36.2 | 0.60 |
| 22 | 36.2/35.8 | 0.35 |
| Ingredients | Starter (1–10) | Grower (11–21) | Finisher (22–35) |
|---|---|---|---|
| Corn (grain) | 551 | 577 | 601 |
| Corn gluten (60%) | 32 | 32 | 32 |
| Sunflower meal | 53.5 | 53.5 | 75 |
| Soybean meal (CP 44.2%) | 262 | 230 | 175 |
| Fat, vegetable | 44.7 | 55 | 67.00 |
| MCP | 18.7 | 17.5 | 15 |
| Limestone | 15 | 13.5 | 12.2 |
| NaCl | 2.7 | 2.7 | 2.7 |
| L-Lysin HCl | 5.2 | 4.6 | 4.3 |
| DL-Methionin | 4.5 | 3.9 | 3.2 |
| L-Treonin | 2.6 | 2.3 | 1.8 |
| Premix 1 | 5.00 | 5.00 | 5.00 |
| Total | 1000.00 | 1000.00 | 1000.00 |
| Nutrient content (g/kg) | |||
| AMEn (MJ/kg) | 12.5 | 12.9 | 13.4 |
| DM % | 90 | 91.3 | 91.1 |
| Crude protein | 204.2 | 190.7 | 174.9 |
| Crude fat | 71.87 | 82.3 | 94.4 |
| Crude fiber | 41.5 | 41.1 | 44.8 |
| Lysine | 13.5 | 12,1 | 10,8 |
| M + C | 10.8 | 9.9 | 9.0 |
| Threonin | 9.7 | 8,8 | 7,8 |
| Tryptophan | 2.4 | 2.3 | 1.7 |
| Ca | 9.6 | 8.7 | 7.8 |
| Pavailable | 4.7 | 4.5 | 3.9 |
| Na | 1.7 | 1.7 | 1.7 |
| Trt | FI 1–10 | FI 11–21 | FI 22–35 | FI Total | FCR 1–10 | FCR 11–21 | FCR 22–35 | FCR Total |
|---|---|---|---|---|---|---|---|---|
| NT | 19.2 a | 67.9 | 138.4 | 81.8 | 1.56 | 1.68 | 1.70 | 1.68 |
| ioS | 20.4 b | 66.0 | 139.7 | 82.2 | 1.6 | 1.66 | 1.73 | 1.70 |
| ioCH | 19.7 ab | 66.9 | 141.7 | 83.2 | 1.57 | 1.63 | 1.71 | 1.68 |
| p-value | 0.04 | 0.54 | 0.65 | 0.74 | 0.54 | 0.74 | 0.34 | 0.54 |
| RMSE | 1.42 | 5.03 | 8.86 | 4.55 | 0.12 | 0.15 | 0.07 | 0.06 |
| Variable | Parameter Estimate | SE | t-Value | p-Value |
|---|---|---|---|---|
| Intercept | 1791.78 | 219.00 | 8.18 | <0.0001 |
| Treatment | 26.03 | 12.23 | 2.13 | 0.03 |
| Egg weight | 64.83 | 26.08 | 2.49 | 0.01 |
| Day of hatch | 39.87 | 23.08 | 1.73 | 0.08 |
| Sex | −142.22 | 19.85 | −7.16 | <0.0001 |
| Hatching weight | 6.50 | 5.01 | 1.3 | 0.19 |
| RMSE | 276.24 | |||
| R2 | 0.91 | |||
| Adjusted R2 | 0.86 | |||
| Thigh (g) | Breast (g) | |||||
|---|---|---|---|---|---|---|
| Weight of Breast and Thigh per Sex | NT | ioS | ioCH | NT | ioS | ioCH |
| Male | 400.8 | 408.7 | 410.5 | 492.4 | 452.9 | 522.8 |
| Female | 405.1 | 388.0 | 422.0 | 505.0 | 465.8 | 500.3 |
| Trt p-value | 0.004 | 0.03 | ||||
| Tukey’s test results | ab | a | b | ab | a | b |
| Sex p-value | 0.88 | 0.13 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ács, V.; Áprily, S.; Nagy, J.; Kacsala, L.; Tossenberger, J.; Szeli, N.K.; Halas, V. Multiple Effects of Egg Weight, in Ovo Carbohydrates, and Sex of Birds on Posthatch Performance in Broilers. Vet. Sci. 2022, 9, 491. https://doi.org/10.3390/vetsci9090491
Ács V, Áprily S, Nagy J, Kacsala L, Tossenberger J, Szeli NK, Halas V. Multiple Effects of Egg Weight, in Ovo Carbohydrates, and Sex of Birds on Posthatch Performance in Broilers. Veterinary Sciences. 2022; 9(9):491. https://doi.org/10.3390/vetsci9090491
Chicago/Turabian StyleÁcs, Virág, Szilvia Áprily, József Nagy, László Kacsala, János Tossenberger, Nóra Katalin Szeli, and Veronika Halas. 2022. "Multiple Effects of Egg Weight, in Ovo Carbohydrates, and Sex of Birds on Posthatch Performance in Broilers" Veterinary Sciences 9, no. 9: 491. https://doi.org/10.3390/vetsci9090491
APA StyleÁcs, V., Áprily, S., Nagy, J., Kacsala, L., Tossenberger, J., Szeli, N. K., & Halas, V. (2022). Multiple Effects of Egg Weight, in Ovo Carbohydrates, and Sex of Birds on Posthatch Performance in Broilers. Veterinary Sciences, 9(9), 491. https://doi.org/10.3390/vetsci9090491






