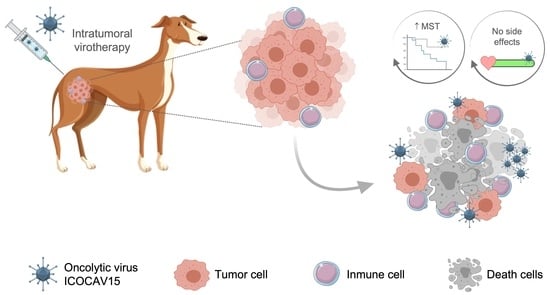
Safety and Efficacy of an Oncolytic Adenovirus as an Immunotherapy for Canine Cancer Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Safety and Efficacy
2.3. ICOCAV15 DNA Extraction and Quantification
2.4. Flow Cytometry
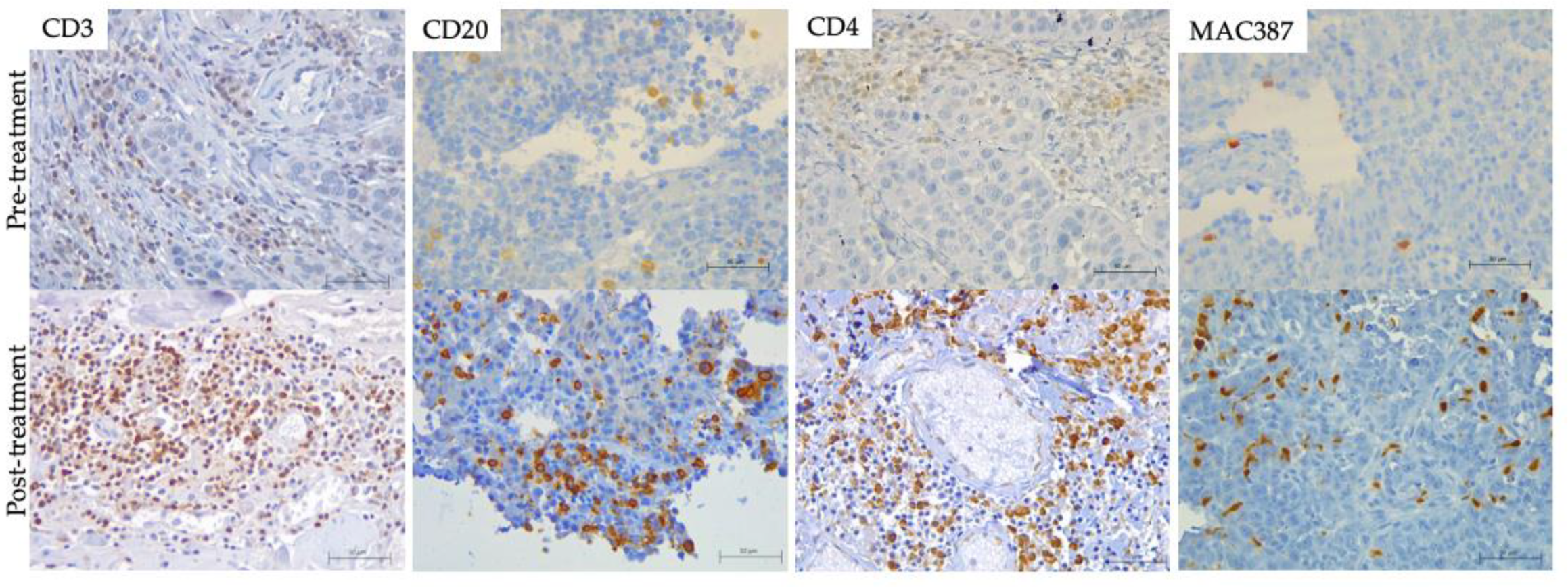
2.5. Immunohistochemistry
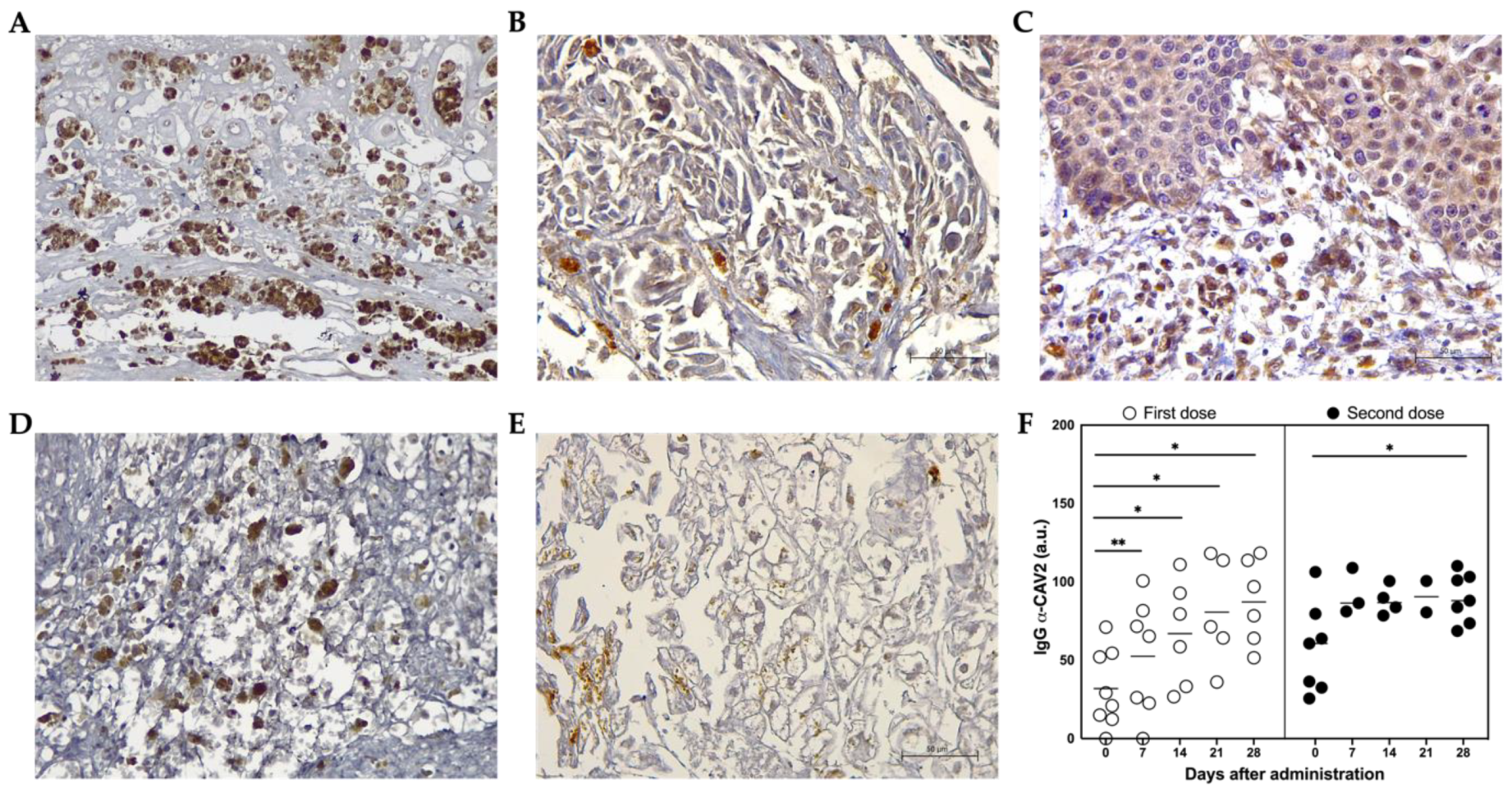
2.6. Virus-Binding Antibodies
2.7. Statistical Analyses
3. Results
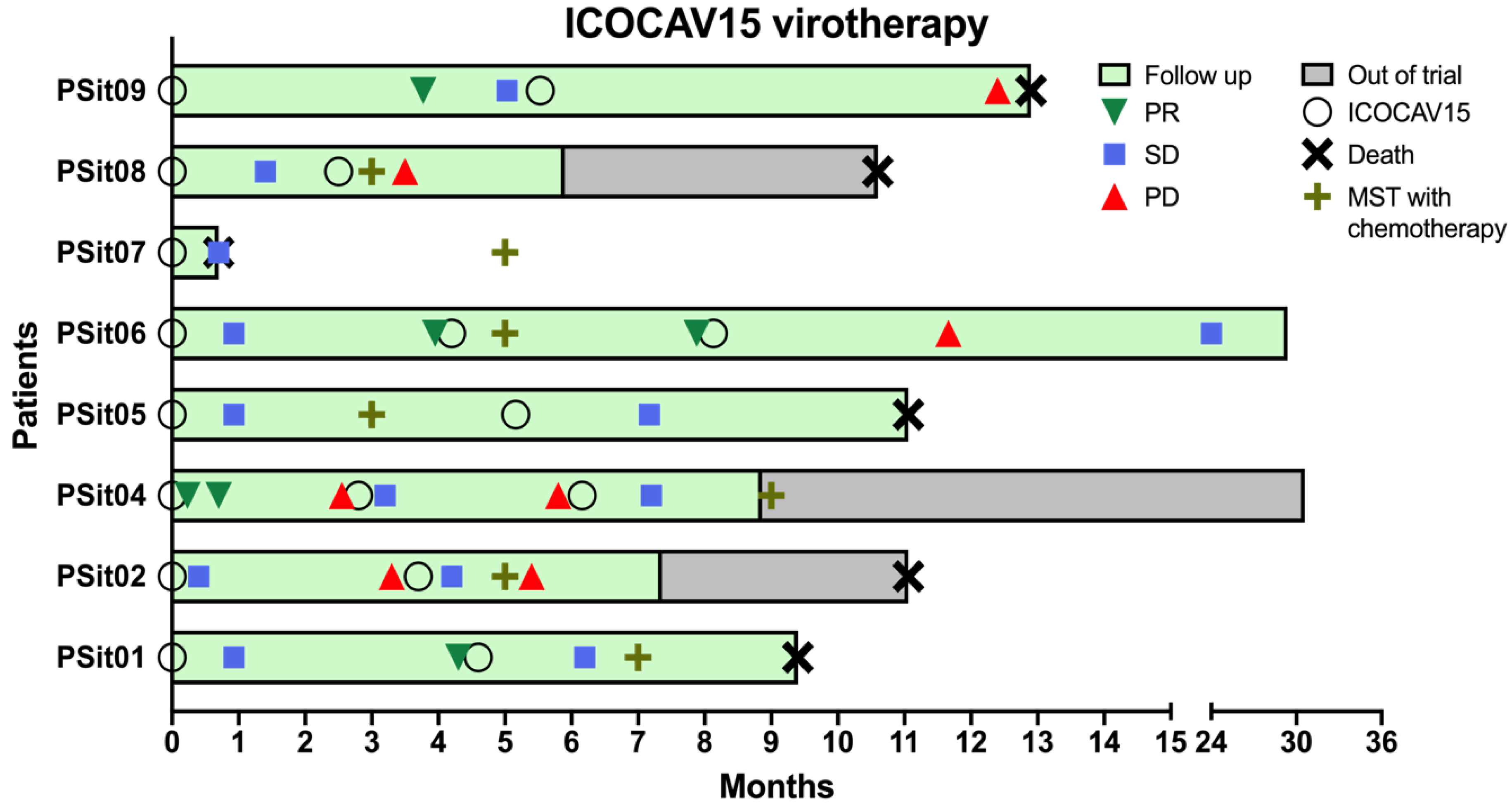
3.1. Canine Patients, Safety, and Quality of Life
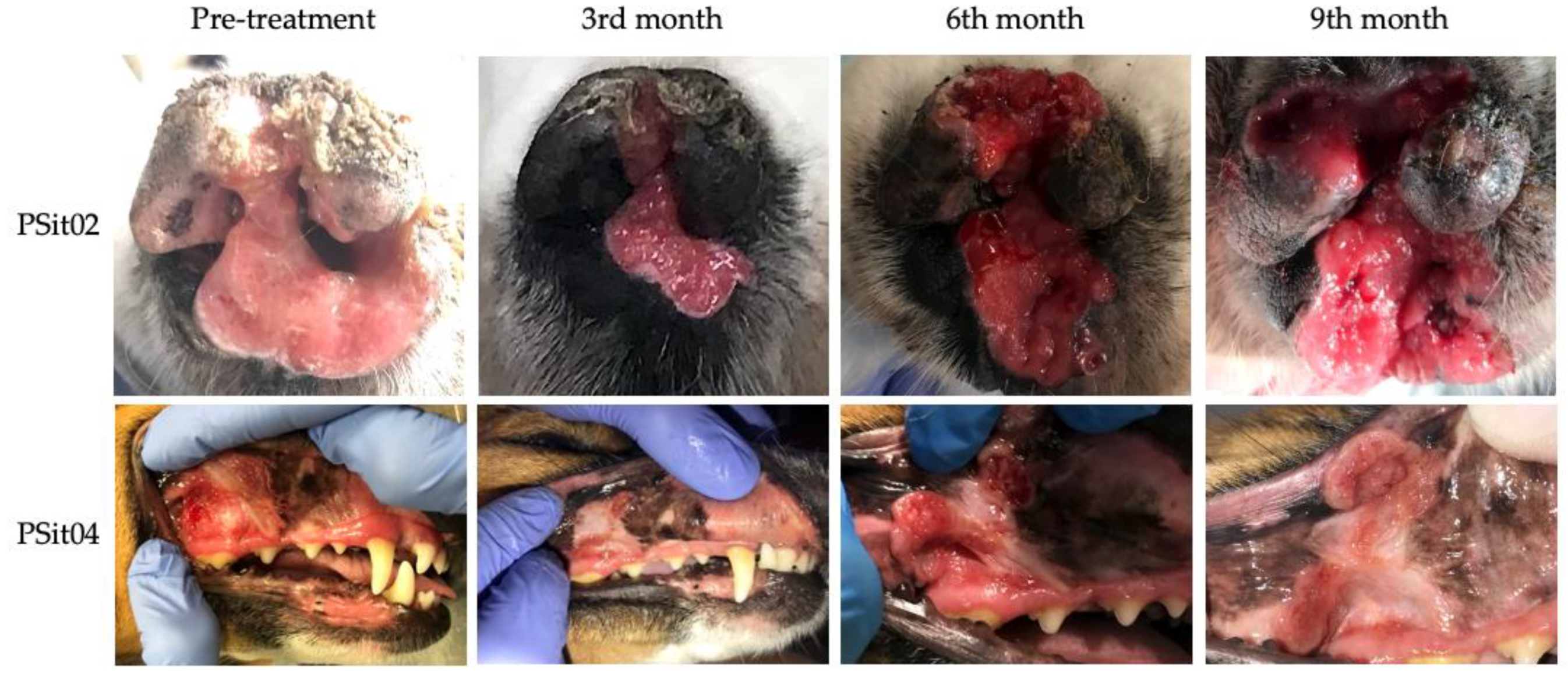
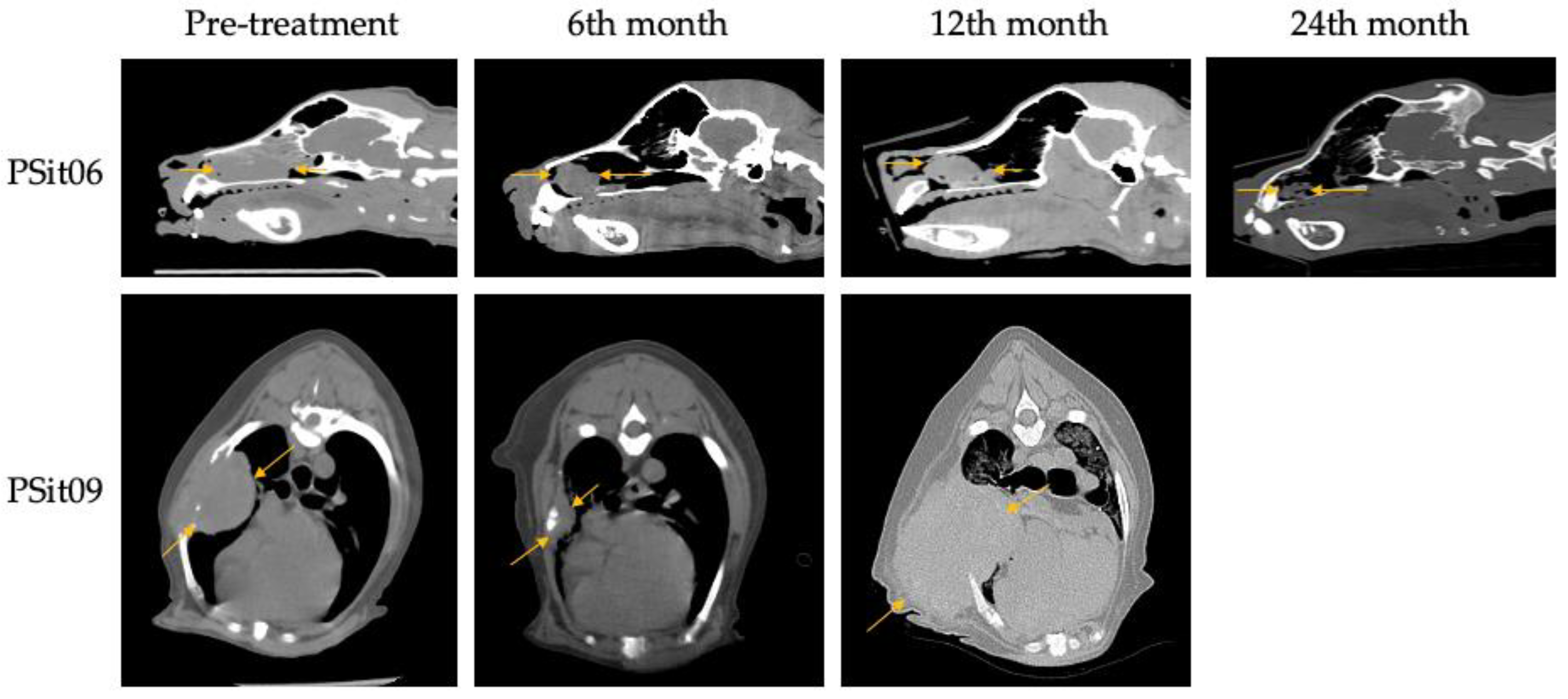
3.2. Outcome Assessment of Virotherapy
3.3. Immune Response Evaluation
3.4. ICOCAV15 and Antiviral Response
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Patil, S.S.; Gentschev, I.; Nolte, I.; Ogilvie, G.; Szalay, A.A. Oncolytic Virotherapy in Veterinary Medicine: Current Status and Future Prospects for Canine Patients. J. Transl. Med. 2012, 10, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vail, D.M.; Thamm, D.H.; Liptak, J.M. Small Animal Clinical Oncology, 6th ed.; Elsevier Saunders: Saint Louis, MO, US, 2020. [Google Scholar]
- Sharma, S.; Boston, S.E.; Skinner, O.T.; Perry, J.A.; Verstraete, F.J.M.; Lee, D.B.; Van Stee, L.L.L.; Thompson, C.; Boylan, M.; McKee, T.; et al. Survival Time of Juvenile Dogs with Oral Squamous Cell Carcinoma Treated with Surgery Alone: A Veterinary Society of Surgical Oncology Retrospective Study. Vet. Surg. 2021. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, T.B.; Henriques, J.; Marconato, L.; Queiroga, F.L. The Use of Low-Dose Metronomic Chemotherapy in Dogs—Insight into a Modern Cancer Field. Vet. Comp. Oncol. 2018, 16, 2–11. [Google Scholar] [CrossRef] [Green Version]
- Johannes, C.M.; Musser, M.L. Anorexia and the Cancer Patient. Vet. Clin. N. Am.-Small Anim. Pract. 2019, 49, 837–854. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.A.D.; Burnside, S.; Helm, J.R.; Morris, J.S. Owner Perceptions of Radiotherapy Treatment for Veterinary Patients with Cancer. Vet. Comp. Oncol. 2019, 17, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Meier, V.; Polton, G.; Cancedda, S.; Roos, M.; Laganga, P.; Emmerson, T.; Rohrer Bley, C. Outcome in Dogs with Advanced (Stage 3b) Anal Sac Gland Carcinoma Treated with Surgery or Hypofractionated Radiation Therapy. Vet. Comp. Oncol. 2017, 15, 1073–1086. [Google Scholar] [CrossRef]
- Shahid, K.; Khalife, M.; Dabney, R.; Phan, A.T. Immunotherapy and Targeted Therapy—The New Roadmap in Cancer Treatment. Ann. Transl. Med. 2019, 7, 595. [Google Scholar] [CrossRef]
- Chida, K.; Nakanishi, K.; Shomura, H.; Homma, S.; Hattori, A.; Kazui, K.; Taketomi, A. Spontaneous Regression of Transverse Colon Cancer: A Case Report. Surg. Case Rep. 2017, 3, 4–8. [Google Scholar] [CrossRef] [Green Version]
- Van Barren, N.; Chambost, H.; Ferrant, A.; Michaux, L.; Ikeda, H.; Millard, I.; Olive, D.; Boon, T.; Coulie, P.G. A Gene Encoding an Antigen Recognized on a Human Melanoma by Cytolytic T Cells, Is Expressed in Acute Leukaemia Cells. Br. J. Haematol. 1998, 102, 1376–1379. [Google Scholar] [CrossRef]
- Bergman, P.J. Immunotherapy in Veterinary Oncology. Vet. Clin. N. Am.-Small Anim. Pract. 2014, 44, 925–939. [Google Scholar] [CrossRef]
- Oiseth, S.J.; Aziz, M.S. Cancer Immunotherapy: A Brief Review of the History, Possibilities, and Challenges Ahead. J. Cancer Metastasis Treat. 2017, 3, 250. [Google Scholar] [CrossRef]
- Abbott, M.; Aocnp, Ò.; Ustoyev, Y. Cancer and the Immune System: The History and Background of Immunotherapy. Semin. Oncol. Nurs. 2019, 35, 150923. [Google Scholar] [CrossRef] [PubMed]
- Dunn, G.P.; Bruce, A.T.; Ikeda, H.; Old, L.J.; Schreiber, R.D. Cancer Immunoediting: From Immunosurveillance to Tumor Escape. Nat. Immunol. 2002, 3, 991–998. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farkona, S.; Diamandis, E.P.; Blasutig, I.M. Cancer Immunotherapy: The Beginning of the End of Cancer? BMC Med. 2016, 14, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klingemann, H. Immunotherapy for Dogs: Still Running Behind Humans. Front. Immunol. 2021, 12, 665784. [Google Scholar] [CrossRef]
- International Clinical Trials Registry Platform (ICTRP), World Health Organization. Available online: https://www.who.int/clinical-trials-registry-platform (accessed on 29 May 2022).
- Verganti, S.; Berlato, D.; Blackwood, L.; Amores-Fuster, I.; Polton, G.A.; Elders, R.; Doyle, R.; Taylor, A.; Murphy, S. Use of Oncept Melanoma Vaccine in 69 Canine Oral Malignant Melanomas in the UK. J. Small Anim. Pract. 2017, 58, 10–16. [Google Scholar] [CrossRef]
- Cejalvo, T.; Perisé-Barrios, A.J.; Del Portillo, I.; Laborda, E.; Rodriguez-Milla, M.A.; Cubillo, I.; Azquez, F.V.; Sard On, D.; Ramirez, M.; Alemany, R.; et al. Tumor Biology and Immunology Remission of Spontaneous Canine Tumors after Systemic Cellular Viroimmunotherapy. Cancer Res. 2018. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Liu, Z.; Li, L.; Wu, J.; Zhang, H.; Zhang, H.; Lei, T.; Xu, B. Oncolytic Adenovirus: Prospects for Cancer Immunotherapy. Front. Microbiol. 2021, 12, 1–16. [Google Scholar] [CrossRef]
- Reale, A.; Calistri, A. Giving Oncolytic Viruses a Free Ride: Carrier Cells for Oncolytic Virotherapy. Pharmaceutics 2021, 13, 2192. [Google Scholar] [CrossRef]
- Kooti, W.; Esmaeili Gouvarchin Ghaleh, H.; Farzanehpour, M.; Dorostkar, R.; Jalali Kondori, B.; Bolandian, M. Oncolytic Viruses and Cancer, Do You Know the Main Mechanism? Front. Oncol. 2021, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Feola, S.; Russo, S.; Ylösmäki, E.; Cerullo, V. Oncolytic ImmunoViroTherapy: A Long History of Crosstalk between Viruses and Immune System for Cancer Treatment. Pharmacol. Ther. 2021, 236, 108103. [Google Scholar] [CrossRef] [PubMed]
- Goradel, N.H.; Mohajel, N.; Malekshahi, Z.V.; Jahangiri, S.; Najafi, M.; Farhood, B.; Mortezaee, K.; Negahdari, B.; Arashkia, A. Oncolytic Adenovirus: A Tool for Cancer Therapy in Combination with Other Therapeutic Approaches. J. Cell. Physiol. 2019, 234, 8636–8646. [Google Scholar] [CrossRef] [PubMed]
- Lichty, B.D.; Breitbach, C.J.; Stojdl, D.F.; Bell, J.C. Going Viral with Cancer Immunotherapy. Nat. Rev. Cancer 2014, 14, 559–567. [Google Scholar] [CrossRef]
- Lawler, S.E.; Speranza, M.C.; Cho, C.F.; Chiocca, E.A. Oncolytic Viruses in Cancer Treatment a Review. JAMA Oncol. 2017, 3, 841–849. [Google Scholar] [CrossRef] [Green Version]
- Gujar, S.; Bell, J.; Diallo, J.S. SnapShot: Cancer Immunotherapy with Oncolytic Viruses. Cell 2019, 176, 1240–1240.e1. [Google Scholar] [CrossRef]
- Iizuka, K.; Shoji, K.; Fujiyuki, T.; Moritoh, K.; Tamura, K.; Yoshida, A.; Sato, H.; Yoneda, M.; Asano, K.; Kai, C. Antitumor Activity of an Oncolytic Measles Virus against Canine Urinary Bladder Transitional Cell Carcinoma Cells. Res. Vet. Sci. 2020, 133, 313–317. [Google Scholar] [CrossRef]
- Li, P.; Wang, J.; Chen, G.; Zhang, X.; Lin, D.; Zhou, Y.; Yu, Y.; Liu, W.; Zhang, D. Oncolytic Activity of Canine Distemper Virus in Canine Mammary Tubular Adenocarcinoma Cells. Vet. Comp. Oncol. 2019, 17, 174–183. [Google Scholar] [CrossRef]
- Chambers, M.R.; Foote, J.B.; Bentley, R.T.; Botta, D.; Crossman, D.K.; Della Manna, D.L.; Estevez-Ordonez, D.; Koehler, J.W.; Langford, C.P.; Miller, M.A.; et al. Evaluation of Immunologic Parameters in Canine Glioma Patients Treated with an Oncolytic Herpes Virus. J. Transl. Genet. Genomics 2021, 5, 423–442. [Google Scholar] [CrossRef]
- Santos, M.R.; Xavier, P.L.P.; Pires, P.R.L.; Rochetti, A.L.; Rosim, D.F.; Scagion, G.P.; de Campos Zuccari, D.A.P.; Munir, M.; Ferreira, H.L.; Fukumasu, H. Oncolytic Effect of Newcastle Disease Virus Is Attributed to Interferon Regulation in Canine Mammary Cancer Cell Lines. Vet. Comp. Oncol. 2021, 19, 593–601. [Google Scholar] [CrossRef]
- Igase, M.; Shousu, K.; Fujiki, N.; Sakurai, M.; Bonkobara, M.; Hwang, C.C.; Coffey, M.; Noguchi, S.; Nemoto, Y.; Mizuno, T. Anti-Tumour Activity of Oncolytic Reovirus against Canine Histiocytic Sarcoma Cells. Vet. Comp. Oncol. 2019, 17, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Béguin, J.; Foloppe, J.; Maurey, C.; Laloy, E.; Hortelano, J.; Nourtier, V.; Pichon, C.; Cochin, S.; Cordier, P.; Huet, H.; et al. Preclinical Evaluation of the Oncolytic Vaccinia Virus TG6002 by Translational Research on Canine Breast Cancer. Mol. Ther.-Oncolytics 2020, 19, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Petrov, I.; Gentschev, I.; Vyalkova, A.; Elashry, M.I.; Klymiuk, M.C.; Arnhold, S.; Szalay, A.A. Canine Adipose-Derived Mesenchymal Stem Cells (CAdMSCs) as a “Trojan Horse” in Vaccinia Virus Mediated Oncolytic Therapy against Canine Soft Tissue Sarcomas. Viruses 2020, 12, 750. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.H.; Wechman, S.L.; McMasters, K.M.; Zhou, H.S. Oncolytic Replication of E1b-Deleted Adenoviruses. Viruses 2015, 7, 5767–5779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fukuhara, H.; Ino, Y.; Todo, T. Oncolytic Virus Therapy: A New Era of Cancer Treatment at Dawn. Cancer Sci. 2016, 107, 1373–1379. [Google Scholar] [CrossRef]
- Laborda, E.; Puig-Saus, C.; Rodriguez-García, A.; Moreno, R.; Cascalló, M.; Pastor, J.; Alemany, R. A PRb-Responsive, RGD-Modified, and Hyaluronidase-Armed Canine Oncolytic Adenovirus for Application in Veterinary Oncology. Mol. Ther. 2014, 22, 986–998. [Google Scholar] [CrossRef] [Green Version]
- Minev, B.R.; Lander, E.; Feller, J.F.; Berman, M.; Greenwood, B.M.; Minev, I.; Santidrian, A.F.; Nguyen, D.; Draganov, D.; Killinc, M.O.; et al. First-in-Human Study of TK-Positive Oncolytic Vaccinia Virus Delivered by Adipose Stromal Vascular Fraction Cells. J. Transl. Med. 2019, 17, 1–15. [Google Scholar] [CrossRef]
- Hamada, M.; Yura, Y. Efficient Delivery and Replication of Oncolytic Virus for Successful Treatment of Head and Neck Cancer. Int. J. Mol. Sci. 2020, 21, 7073. [Google Scholar] [CrossRef]
- Cervera-Carrascon, V.; Quixabeira, D.C.A.; Havunen, R.; Santos, J.M.; Kutvonen, E.; Clubb, J.H.A.; Siurala, M.; Heiniö, C.; Zafar, S.; Koivula, T.; et al. Comparison of Clinically Relevant Oncolytic Virus Platforms for Enhancing T Cell Therapy of Solid Tumors. Mol. Ther.-Oncolytics 2020, 17, 47–60. [Google Scholar] [CrossRef]
- Nguyen, S.M.; Thamm, D.H.; Vail, D.M.; London, C.A. Response Evaluation Criteria for Solid Tumours in Dogs (v1.0): A Veterinary Cooperative Oncology Group (VCOG) Consensus Document. Vet. Comp. Oncol. 2015, 13, 176–183. [Google Scholar] [CrossRef]
- Yazbek, K.V.B.; Fantoni, D.T. Validity of a Health-Related Quality-of-Life Scale for Dogs with Signs of Pain Secondary to Cancer. J. Am. Vet. Med. Assoc. 2005, 226, 1354–1358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 Years of Image Analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Mantwill, K.; Klein, F.G.; Wang, D.; Hindupur, S.V.; Ehrenfeld, M.; Holm, P.S.; Nawroth, R. Concepts in Oncolytic Adenovirus Therapy. Int. J. Mol. Sci. 2021, 22, 10522. [Google Scholar] [CrossRef] [PubMed]
- Worgall, S.; Wolff, G.; Falck-Pedersen, E.; Crystal, R.G. Innate Immune Mechanisms Dominate Elimination of Adenoviral Vectors Following in Vivo Administration. Hum. Gene Ther. 1997, 8, 37–44. [Google Scholar] [CrossRef]
- Zhou, J.; He, Z.; Ma, S.; Liu, R. AST/ALT Ratio as a Significant Predictor of the Incidence Risk of Prostate Cancer. Cancer Med. 2020, 9, 5672–5677. [Google Scholar] [CrossRef]
- Zhang, L.X.; Lv, Y.; Xu, A.M.; Wang, H.Z. The Prognostic Significance of Serum Gamma-Glutamyltransferase Levels and AST/ALT in Primary Hepatic Carcinoma. BMC Cancer 2019, 19, 841. [Google Scholar] [CrossRef] [Green Version]
- Steffl, J.L.; Bennett, W.; Olyaei, A.J. The Old and New Methods of Assessing Kidney Function. J. Clin. Pharmacol. 2012, 52, 63S–71S. [Google Scholar] [CrossRef]
- Saleh, R.; Elkord, E. Acquired Resistance to Cancer Immunotherapy: Role of Tumor-Mediated Immunosuppression. Semin. Cancer Biol. 2020, 65, 13–27. [Google Scholar] [CrossRef]
- Rogers, K.S.; Helman, R.G.; Walker, M.A. Squamous Cell Carcinoma of the Canine Nasal Planum: Eight Cases (1988-1994). J. Am. Anim. Hosp. Assoc. 1995, 31, 373–378. [Google Scholar] [CrossRef]
- Polton, G.; Finotello, R.; Sabattini, S.; Rossi, F.; Laganga, P.; Vasconi, M.E.; Barbanera, A.; Stiborova, K.; Rohrer Bley, C.; Marconato, L. Survival Analysis of Dogs with Advanced Primary Lung Carcinoma Treated by Metronomic Cyclophosphamide, Piroxicam and Thalidomide. Vet. Comp. Oncol. 2018, 16, 399–408. [Google Scholar] [CrossRef]
- Wylleman, B.; Brancato, L.; Gorbaslieva, I.; Van Zwol, E.; Mori Da Cunha, M.G.M.C.; Benoit, J.; Tierny, D.; Vueghs, P.; Van Den Bossche, J.; Rudenko, O.; et al. Tolerability of Long-Term Temperature Controlled Whole-Body Thermal Treatment in Advanced Cancer-Bearing Dogs. Int. J. Hyperth. 2021, 39, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Moreno, R.; Fajardo, C.A.; Farrera-Sal, M.; Perise-Barrios, A.J.; Morales-Molina, A.; Al-Zaher, A.A.; García-Castro, J.; Alemany, R. Enhanced Antitumor Efficacy of Oncolytic Adenovirus–Loaded Menstrual Blood–Derived Mesenchymal Stem Cells in Combination with Peripheral Blood Mononuclear Cells. Mol. Cancer Ther. 2019, 18, 127–138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frelaut, M.; du Rusquec, P.; de Moura, A.; Le Tourneau, C.; Borcoman, E. Pseudoprogression and Hyperprogression as New Forms of Response to Immunotherapy. BioDrugs 2020, 34, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Boutilier, A.J.; Elsawa, S.F. Macrophage Polarization States in the Tumor Microenvironment. Int. J. Mol. Sci. 2021, 22, 6995. [Google Scholar] [CrossRef]
- Krane, G.A.; O’Dea, C.A.; Malarkey, D.E.; Miller, A.D.; Miller, C.R.; Tokarz, D.A.; Jensen, H.L.; Janardhan, K.S.; Shockley, K.R.; Flagler, N.; et al. Immunohistochemical Evaluation of Immune Cell Infiltration in Canine Gliomas. Vet. Pathol. 2021, 58, 952–963. [Google Scholar] [CrossRef]

| Patient | Breed | Age (years) | Sex | Diagnosis | ICOCAV15 Doese | Survival Time (days) |
|---|---|---|---|---|---|---|
| PSit01 | Miniature Schnauzer | 13 | Female | Apocrine Gland Anal Sac Adenocarcinoma | 2 | 282 |
| PSit02 | Golden Retriever | 11 | Male | Squamous Cell Carcinoma (nasal plane) | 2 | 332 |
| PSit04 | Mixed | 7 | Female | Squamous Cell Carcinoma (oral) | 3 | To date (2 years 7months) |
| PSit05 | German Sheperd | 10 | Female | Pulmonary Carcinoma | 2 | 332 |
| PSit06 | Mixed | 11 | Male | Nasal Adenocarcinoma | 3 | To date (2 years 7months) |
| PSit07 | Labrador Retriever | 13 | Female | Squamous Cell Carcinoma (nasal plane) | 1 | 21 |
| PSit08 | Mixed | 10 | Female | Pulmonary Carcinoma | 2 | 319 |
| PSit09 | Cocker Spaniel | 13 | Female | Squamous Cell Carcinoma (rib wall) | 2 | 389 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín-Carrasco, C.; Delgado-Bonet, P.; Tomeo-Martín, B.D.; Pastor, J.; de la Riva, C.; Palau-Concejo, P.; del Castillo, N.; García-Castro, J.; Perisé-Barrios, A.J. Safety and Efficacy of an Oncolytic Adenovirus as an Immunotherapy for Canine Cancer Patients. Vet. Sci. 2022, 9, 327. https://doi.org/10.3390/vetsci9070327
Martín-Carrasco C, Delgado-Bonet P, Tomeo-Martín BD, Pastor J, de la Riva C, Palau-Concejo P, del Castillo N, García-Castro J, Perisé-Barrios AJ. Safety and Efficacy of an Oncolytic Adenovirus as an Immunotherapy for Canine Cancer Patients. Veterinary Sciences. 2022; 9(7):327. https://doi.org/10.3390/vetsci9070327
Chicago/Turabian StyleMartín-Carrasco, Clara, Pablo Delgado-Bonet, Beatriz Davinia Tomeo-Martín, Josep Pastor, Claudia de la Riva, Paula Palau-Concejo, Noemí del Castillo, Javier García-Castro, and Ana Judith Perisé-Barrios. 2022. "Safety and Efficacy of an Oncolytic Adenovirus as an Immunotherapy for Canine Cancer Patients" Veterinary Sciences 9, no. 7: 327. https://doi.org/10.3390/vetsci9070327
APA StyleMartín-Carrasco, C., Delgado-Bonet, P., Tomeo-Martín, B. D., Pastor, J., de la Riva, C., Palau-Concejo, P., del Castillo, N., García-Castro, J., & Perisé-Barrios, A. J. (2022). Safety and Efficacy of an Oncolytic Adenovirus as an Immunotherapy for Canine Cancer Patients. Veterinary Sciences, 9(7), 327. https://doi.org/10.3390/vetsci9070327