Evaluation of the Fluorescence Polarization Assay for the Diagnosis of Brucellosis in Goat Milk
Abstract
1. Introduction
2. Materials and Methods
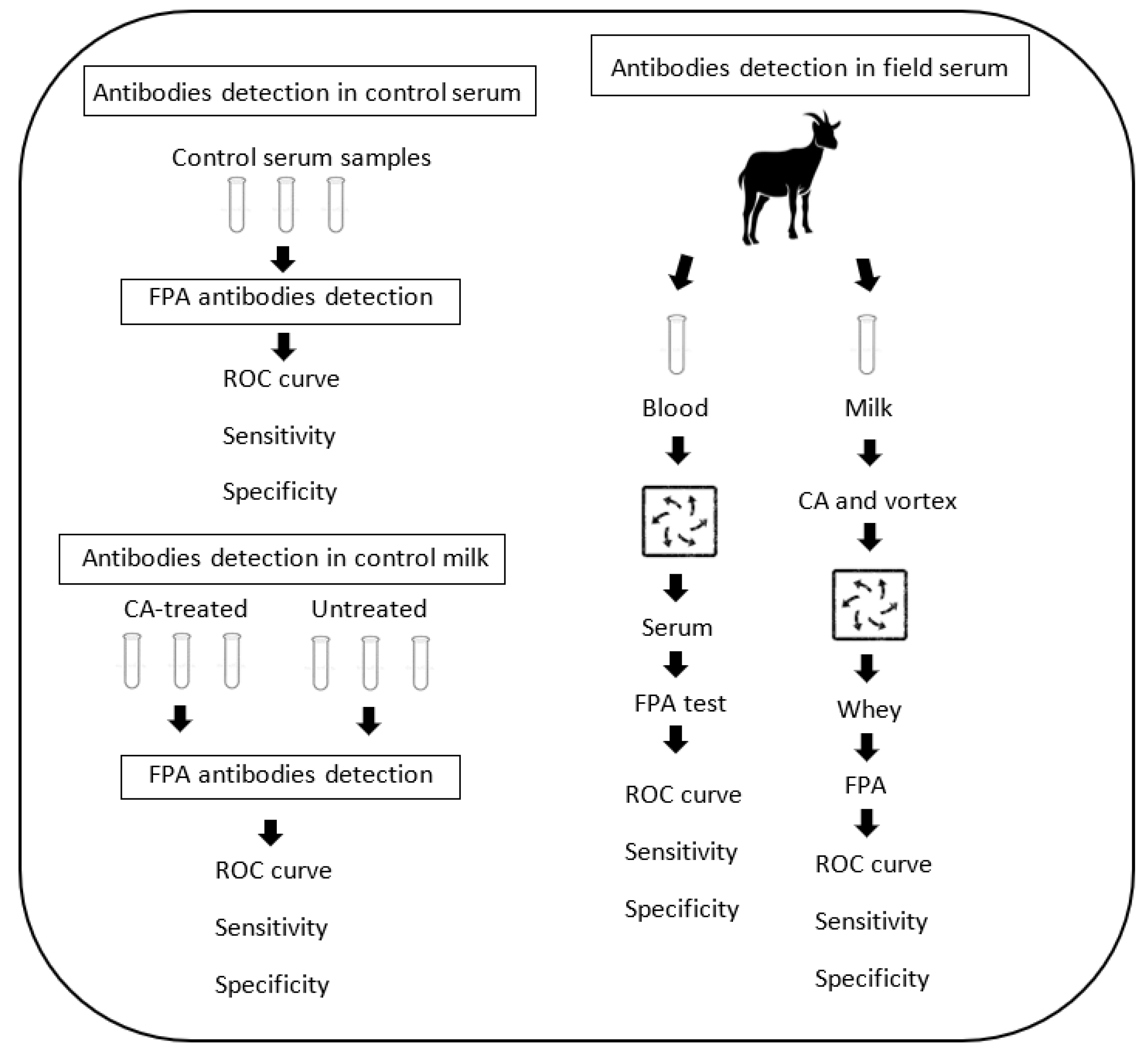
2.1. Schematic Overview of the Experimental Program
2.2. Serum and Milk Controls
2.3. Serum and Milk Field Samples
2.4. Milk Treatment
2.5. Serological Tests
2.6. Fluorescence Polarization Assay
2.7. Statistical Analysis
3. Results
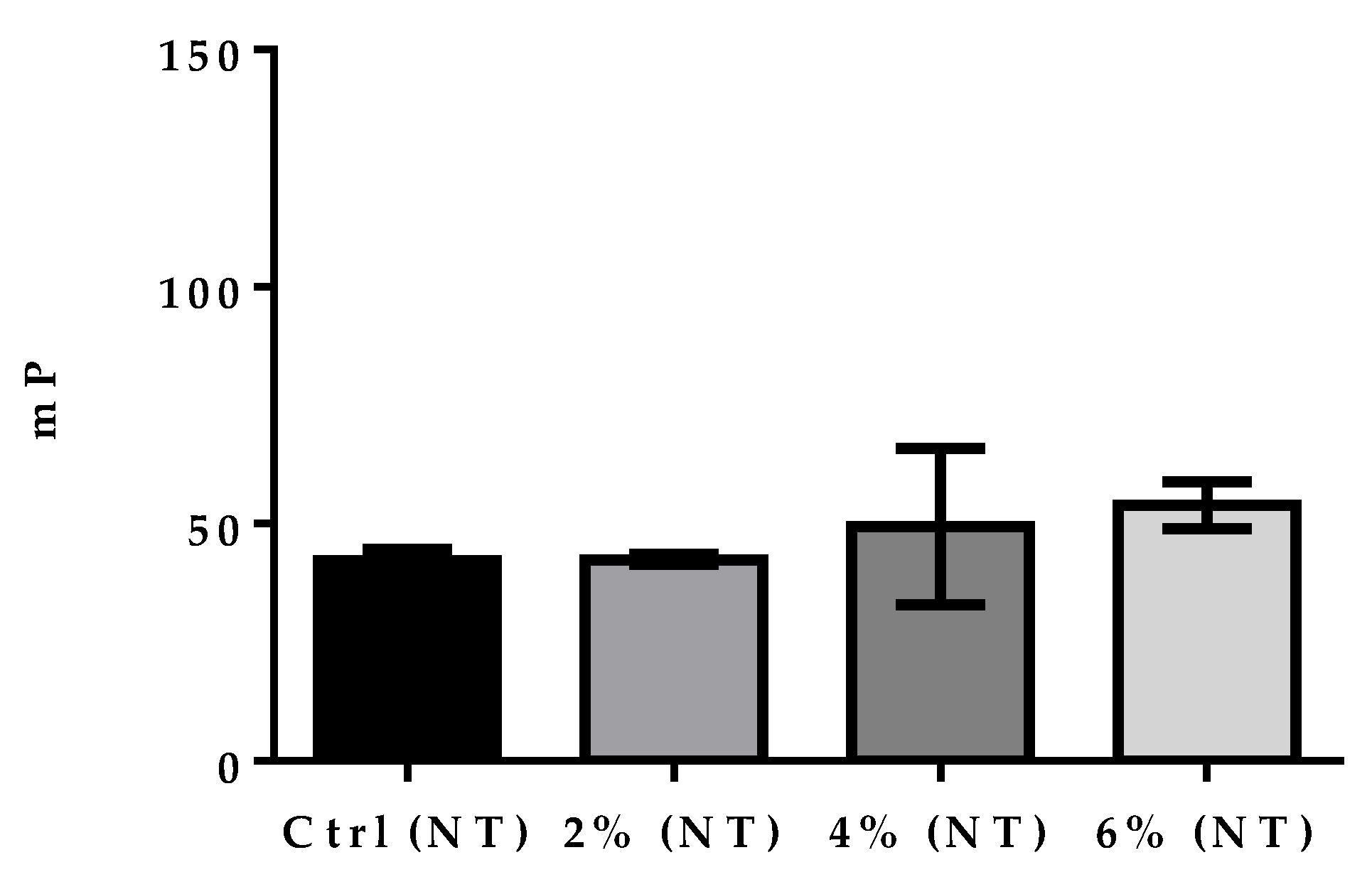
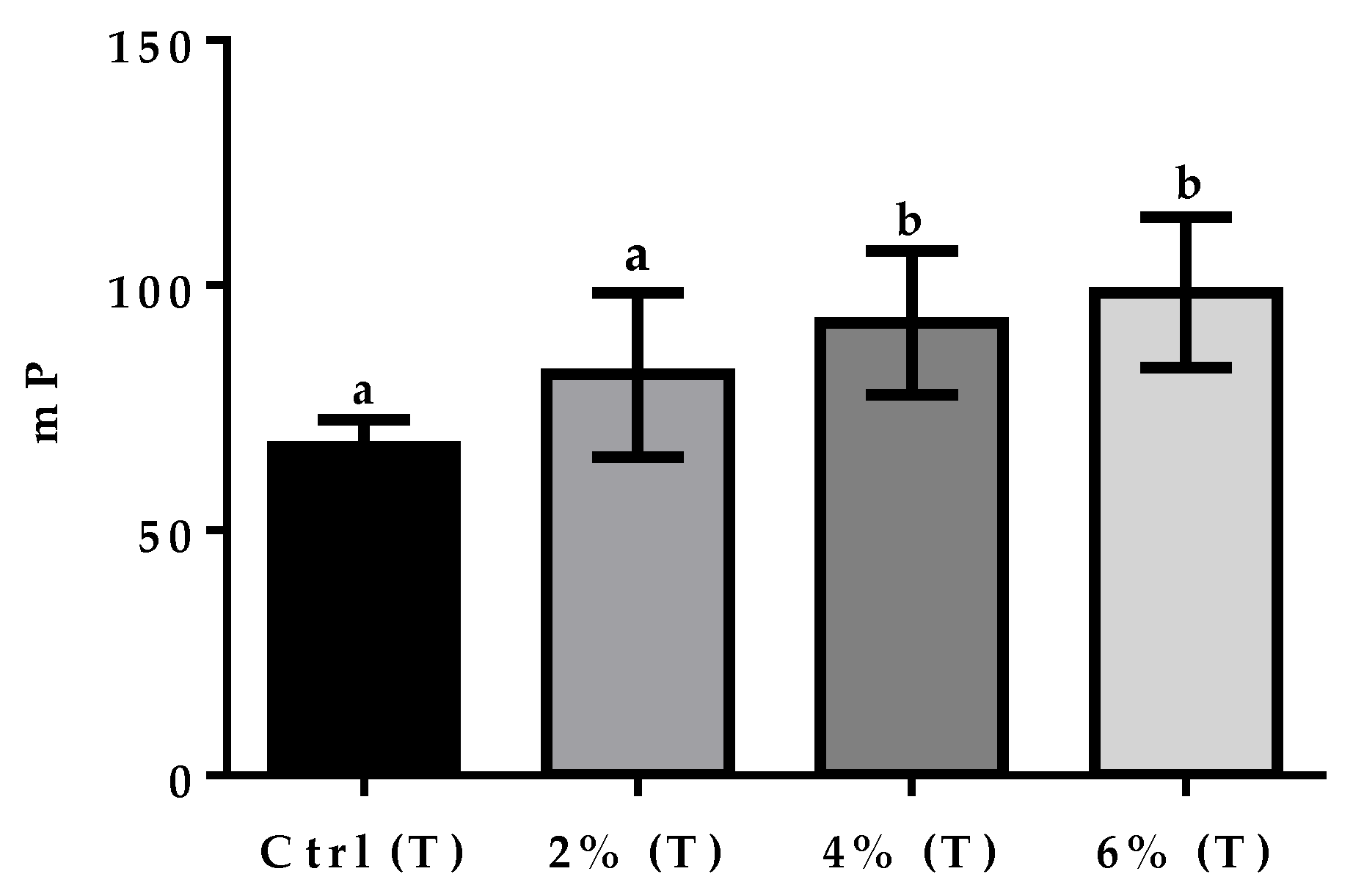
3.1. Fluorescence Polarization Assay on Treated and Untreated Control Milk Samples
3.2. The FPA in Control Milk Samples
3.3. The FPA on Milk Samples
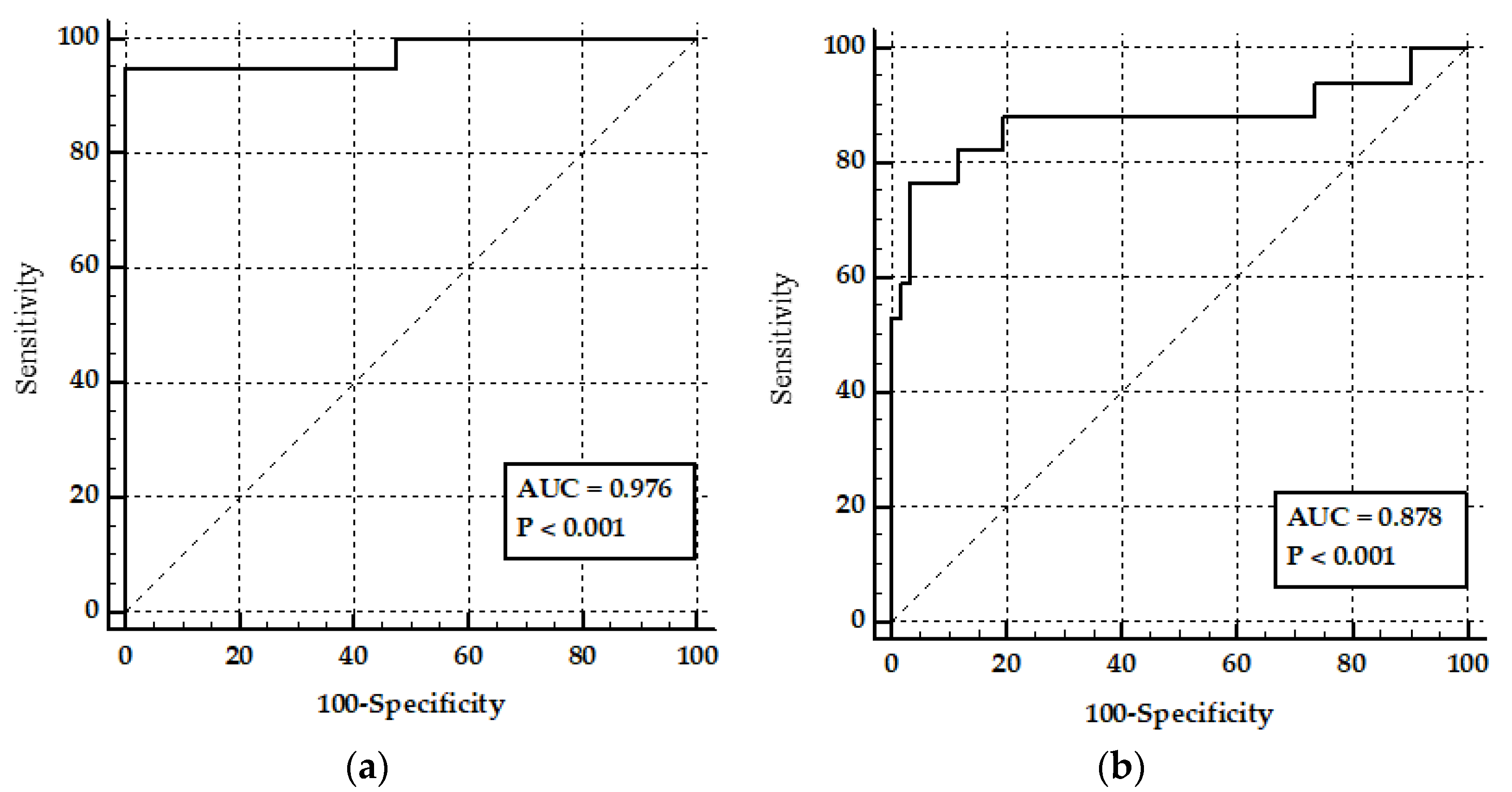
3.4. Comparison of FPA and PT3 Test Performance on Milk Samples, Using the Serum FPA as the Reference Test
3.5. Comparison of FPA in Serum Performance with FPA in Milk and RBT
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Seleem, M.N.; Boyle, S.M.; Sriranganathan, N. Brucellosis: A re-emerging zoonosis. Vet. Microbiol. 2010, 140, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, Z.I.; Pascual, D.W. Brucellosis vaccines for livestock. Vet. Imm. 2016, 18, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Mantur, B.G.; Amarnath, S.K.; Shinde, R.S. Review of clinical and laboratory features of human brucellosis. Indian J. Med. Microbiol. 2007, 25, 188–202. [Google Scholar] [CrossRef]
- Dadar, M.; Shahali, Y.; Whatmore, A.M. Human brucellosis caused by raw dairy products: A review on the occurrence, major risk factors and prevention. Int. J. Food Microbiol. 2019, 292, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations. Statistics Division (FAOSTAT) Food and Agriculture Data. 2014. Available online: http://faostat3.fao.org/home/S (accessed on 1 March 2022).
- Isidro-Requejo, L.M.; Meza-Herrera, C.A.; Pastor-López, F.J.; Maldonado, J.A.; Salinas-González, H. Physicochemical characterization of goat milk produced in the Comarca Lagunera, Mexico. Anim. Sci. J. 2019, 90, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Morales-Pablo, R.; Avalos de la Cruz, D.A.; Leyva-Ruelas, G.; Ybarra-Moncada, M.C. Bacteriological quality goat raw milk produced in Miravalles, Puebla. Rev. Mex. Ing. Quim. 2012, 11, 45–54. [Google Scholar]
- Rossetti, C.A.; Arenas-Gamboa, A.M.; Maurizio, E. Caprine brucellosis: A historically neglected disease with significant impact on public health. PLoS Negl. Trop. Dis. 2017, 11, e0005692. [Google Scholar] [CrossRef]
- Behera, S.K.; Das, D.; Balasubramani, K.; Chellappan, S.; Rajaram, K.; Mohanta, H.K.; Nina, P.B. Seroprevalence and risk factors of brucellosis in livestock in the wildlife and livestock interface area of Similipal Biosphere Reserve, India. Vet. World 2020, 13, 465–470. [Google Scholar] [CrossRef]
- Stear, M.J. OIE manual of diagnostic tests and vaccines for terrestrial animals (mammals, birds and bees). Parasitology 2005, 130, 727. [Google Scholar] [CrossRef]
- Pal, M.; Kerorsa, G.B.; Desalegn, C.; Kandi, V. Human and Animal Brucellosis: A Comprehensive Review of Biology, Pathogenesis, Epidemiology, Risk Factors, Clinical Signs, Laboratory Diagnosis, Public Health Significance, Economic Importance, Prevention and Control. Am. J. Infect. Dis. 2020, 8, 118–126. [Google Scholar]
- Gall, D.; Nielsen, K.; Bermudez, M.R.; Moreno, F.; Smith, P. Fluorescence Polarization Assay for Detection of Brucella abortus Antibodies in Bulk Tank Bovine Milk Samples. Clin. Diagn. Lab. Immunol. 2002, 9, 1356–1360. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Deb, R.; Chakraborty, S.; Veeregowda, B.; Verma, A.K.; Tiwari, R.; Dhama, K. Monoclonal antibody and its use in the diagnosis of livestock diseases. Adv. Biosci. Biotechnol. 2013, 4, 50–62. [Google Scholar] [CrossRef]
- Khan, T.I.; Ehtisham-ul-Haque, S.; Waheed, U.; Khan, I.; Younus, M.; Ali, S. Milk Indirect-ELISA and Milk Ring Test for Screening of Brucellosis in Buffaloes, Goats and Bulk Tank Milk Samples Collected from Two Districts of Punjab, Pakistan. Pak. Vet. J. 2018, 38, 105–108. [Google Scholar] [CrossRef]
- Kolar, J. Diagnosis and control of brucellosis in small ruminants. Prev. Vet. Med. 1984, 2, 215–225. [Google Scholar] [CrossRef]
- Haenlein, G.F.W.; Caccese, R. Goat milk versus cow milk. In Extension Goat Handbook; Haenlein, G.F.W., Ace, D.L., Eds.; USDA Publication: Washington, DC, USA, 1984; pp. 1–4. [Google Scholar]
- Park, Y.W.; Nam, M.S. Bioactive Peptides in Milk and Dairy Products: A Review. Korean J. Food Sci. Anim. Resour. 2015, 35, 831–840. [Google Scholar] [CrossRef]
- Sanz, L.; Ramos, E.; De la Torre, G.; Díaz, J.; Pérez, L.; Sanz, M. Composition of goat and cow milk produced under similar conditions and analyzed by identical methodology. J. Food Compost. Anal. 2009, 22, 322–332. [Google Scholar]
- Perrin, F. The polarisation of flourescence light. Average life of molecules in their excited state. J. Phys. 1926, 7, 390–401. [Google Scholar]
- Hall, M.D.; Yasgar, A.; Peryea, T.; Braisted, J.C.; Jadhav, A.; Simeonov, A.; Coussens, N.P. Fluorescence polarization assays in high-throughput screening and drug discovery: A review. Methods Appl. Fluoresc. 2016, 4, 022001. [Google Scholar] [CrossRef]
- Weber, G. Polarization of the fluorescence of macromolecules. I. Theory and experimental method. Biochem. J. 1952, 51, 145–155. [Google Scholar] [CrossRef]
- Dandliker, W.B.; Feigen, G.A. Quantification of the antigen-antibody reaction by the polarization of fluorescence. Biochem. Biophys. Res. Commun. 1961, 5, 299–304. [Google Scholar] [CrossRef]
- Nielsen, K.; Gall, D.; Smith, P.; Kelly, W.; Yeo, J.; Kenny, K.; Heneghan, T.; McNamara, S.; Maher, P.; O’Connor, J.; et al. Fluorescence polarization assay for the diagnosis of bovine brucellosis: Adaptation to field use. Vet. Microbiol. 2001, 80, 163–170. [Google Scholar] [CrossRef]
- Ramírez-Pfeiffer, C.; Díaz-Aparicio, E.; Rodríguez-Padilla, C.; Morales-Loredo, A.; Álvarez-Ojeda, G.; Gómez-Flores, R. Improved performance of B. melitensis native hapten over B. abortus OPS tracer on goat antibody detection by the fluorescence polarization assay. Vet. Immunol. Immunopathol. 2008, 123, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Kalleshamurthy, T.; Skariah, S.; Rathore, Y.; Ramanjinappa, K.D.; Nagaraj, C.; Shome, B.R.; Rahman, H.; Barmand, N.N.; Shome, R. Comparative evaluation of fluorescence polarization assay and competitive ELISA for the diagnosis of bovine brucellosis vis-a-vis sero-monitoring. J. Microbiol. Methods 2020, 170, 105858. [Google Scholar] [CrossRef] [PubMed]
- Gul, S.T.; Khan, A. Epidemiology and epizootology of brucellosis: A review. Pak. Vet. J. 2007, 27, 145–151. [Google Scholar]
- Alton, G.G.; Jones, L.M.; Angus, R.D.; Verger, J.M. Techniques for the Brucellosis Laboratory; INRA: Paris, France, 1988; ISBN 978-2738000422. [Google Scholar]
- Diaz, R.; Casanova, A.; Ariza, J.; Moriyón, I. The Rose Bengal test in human brucellosis: A neglected test for the diagnosis of a neglected disease. PLoS Negl. Trop. Dis. 2011, 5, e950. [Google Scholar] [CrossRef]
- OIE. Brucella abortus, B. melitensis and B. suis (infections with B. abortus, B. melitensis and B. suis). Manual of Diagnostic Tests and Vaccines for Terrestrial Animals; OIE World Organization for Animal Health: Paris, France, 2016; Available online: http://www.oie.int/manual-of-diagnostic-tests-and-vaccines-for-terrestrial-animals/ (accessed on 1 March 2022).
- Nielsen, K.; Gall, D. Fluorescence polarization assay for the diagnosis of brucellosis: A review. J. Immunoass. Immunochem. 2001, 22, 183–201. [Google Scholar] [CrossRef]
- Nielsen, K.; Lin, M.; Gall, D.; Jolley, M. Fluorescence polarization immunoassay: Detection of antibody to Brucella abortus. Methods 2000, 22, 71–76. [Google Scholar] [CrossRef]
- Nielsen, K.; Smith, P.; Gall, D.; Perez, B.; Cosma, C.; Mueller, P.; Trottier, J.; Cote, G.; Boag, L.; Bosse, J. Development and validation of an indirect enzyme immunoassay for detection of antibody to Brucella abortus in milk. Vet. Microbiol. 1996, 52, 165–173. [Google Scholar] [CrossRef]
- González-Espinoza, G.; Arce-Gorvel, V.; Mémet, S.; Gorvel, J.P. Brucella: Reservoirs and niches in animals and humans. Pathogens 2021, 10, 186. [Google Scholar] [CrossRef]
- Mazlina, M.; Khairani-Bejo, S.; Hazilawati, H.; Shaqinah, N.N.; Zamri-Saad, M. Antigenic distribution, pathological changes, antibody response and serological detection in non-pregnant goats following experimental infection by Brucella melitensis. Transbound. Emerg. Dis. 2021, 68, 2028–2038. [Google Scholar] [CrossRef]
- Ducrotoy, M.J.; Muñoz, P.M.; Conde-Álvarez, R.; Blasco, J.M.; Moriyón, I. A systematic review of current immunological tests for the diagnosis of cattle brucellosis. Prev. Vet. Med. 2018, 151, 57–72. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, P. Further evaluations of serologic test procedures used to diagnose brucellosis. Am. J. Vet. Res. 1969, 30, 1811–1816. [Google Scholar] [PubMed]
- Al-Mariri, A.; Ramadan, L.; Akel, R. Assessment of milk ring test and some serological tests in the detection of Brucella melitensis in Syrian female sheep. Trop. Anim. Health Prod. 2011, 43, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Matope, G.; Muma, J.B.; Toft, N.; Gori, E.; Lund, A.; Nielsen, K.H.; Skjerve, E. Evaluation of sensitivity and specificity of RBT, c-ELISA and fluorescence polarisation assay for diagnosis of brucellosis in cattle using latent class analysis. Vet. Immunol. Immunopathol. 2011, 141, 58–63. [Google Scholar] [CrossRef]
- Muma, J.B.; Lund, A.; Nielsen, K.H.; Matope, G.; Munyeme, M.; Mwacalimba, K.; Skjerve, E. Effectiveness of Rose Bengal test and fluorescence polarization assay in the diagnosis of Brucella spp. infections in free range cattle reared in endemic areas in Zambia. Trop. Anim. Health Prod. 2009, 41, 723–729. [Google Scholar] [CrossRef]
- Vanzini, V.R.; Aguirre, N.P.; Valentini, B.S.; Torioni de Echaide, S.; Lugaresi, C.I.; Machesino, M.D.; Nielsen, K. Comparison of an indirect ELISA with the Brucella milk ring test for detection of antibodies to Brucella abortus in bulk milk samples. Vet. Microbiol. 2001, 82, 55–60. [Google Scholar] [CrossRef]
- Padilla, F.; Nielsen, K.; Samartino, L.E.; Yu, W.L. Diagnosis of Brucellosis. Open. Vet. Sci. J. 2010, 4, 46–60. [Google Scholar] [CrossRef]
- Tilocca, B.; Soggiu, A.; Greco, V.; Piras, C.; Arrigoni, N.; Ricchi, M.; Britti, D.; Urbani, A.; Roncada, P. Immunoinformatic-based prediction of candidate epitopes for the diagnosis and control of paratuberculosis (Johne’s disease). Pathogens 2020, 9, 705. [Google Scholar] [CrossRef]
- Im, Y.B.; Park, W.B.; Jung, M.; Kim, S.; Yoo, H.S. Comparative analysis of immune responses by outer membrane antigens OMP10, OMP19 and OMP28 of Brucella abortus. Jpn. J. Infect. Dis. 2018, 71, 197–204. [Google Scholar] [CrossRef]
- Bulashev, A.; Akibekov, O.; Syzdykova, A.; Suranshiyev, Z.; Ingirbay, B. Use of recombinant Brucella outer membrane proteins 19, 25, and 31 for serodiagnosis of bovine brucellosis. Vet. World 2020, 13, 1439–1447. [Google Scholar] [CrossRef]
- Bai, Q.; Li, H.; Wu, X.; Shao, J.; Sun, M.; Yin, D. Comparative analysis of the main outer membrane proteins of Brucella in the diagnosis of brucellosis. Biochem. Biophys. Res. Commun. 2021, 560, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Meirelles-Bartoli, R.B.; de Alvarenga Cruz, C.; de Moraes, R.S.; Oliveira, R.A.; de Paula, E.M.N.; de Sousa, D.B.; Assis, N.A.; Mathias, L.A. Comparison of four serological tests for the diagnosis of swine brucellosis. Res. Soc. Dev. 2020, 9, e493974418. [Google Scholar] [CrossRef]
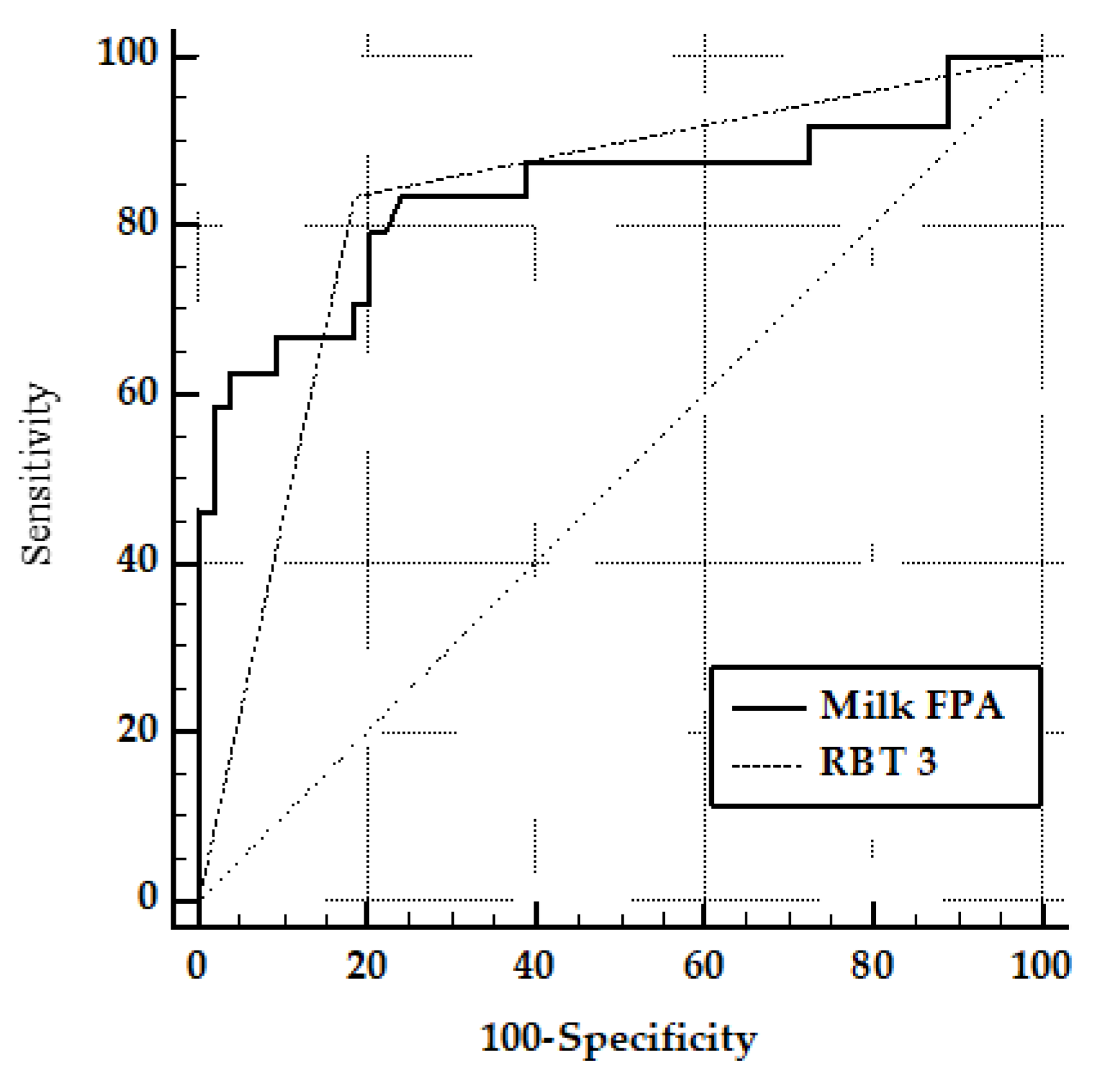
| Milk FPA versus RBT 3 | |
|---|---|
| Difference between areas | 0.0135 |
| SEM | 0.0766 |
| 95% Confidence interval | −0.137 to 0.164 |
| z Statistic | 0.176 |
| Significance level | p = 0.8601 |
| TEST | SE | 95% CI | SP | 95% CI | AUC |
|---|---|---|---|---|---|
| RBT 3 | 99.26 | 95.9–100.0 | 83.33 | 51.6–97.9 | 0.913 |
| SERUM FPA | 88.89 | 73.9–96.9 | 100.0 | 93.6–100.0 | 99.26 |
| MILK FPA | 83.33 | 62.6–95.3 | 75.93 | 62.4–86.5 | 0.878 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sotolongo-Rodríguez, D.; Gomez-Flores, R.; Navarro-Soto, M.C.; Arellano-Reynoso, B.; Tamez-Guerra, P.; Ramírez-Pfeiffer, C. Evaluation of the Fluorescence Polarization Assay for the Diagnosis of Brucellosis in Goat Milk. Vet. Sci. 2022, 9, 303. https://doi.org/10.3390/vetsci9060303
Sotolongo-Rodríguez D, Gomez-Flores R, Navarro-Soto MC, Arellano-Reynoso B, Tamez-Guerra P, Ramírez-Pfeiffer C. Evaluation of the Fluorescence Polarization Assay for the Diagnosis of Brucellosis in Goat Milk. Veterinary Sciences. 2022; 9(6):303. https://doi.org/10.3390/vetsci9060303
Chicago/Turabian StyleSotolongo-Rodríguez, Dianelys, Ricardo Gomez-Flores, Magda Celina Navarro-Soto, Beatriz Arellano-Reynoso, Patricia Tamez-Guerra, and Carlos Ramírez-Pfeiffer. 2022. "Evaluation of the Fluorescence Polarization Assay for the Diagnosis of Brucellosis in Goat Milk" Veterinary Sciences 9, no. 6: 303. https://doi.org/10.3390/vetsci9060303
APA StyleSotolongo-Rodríguez, D., Gomez-Flores, R., Navarro-Soto, M. C., Arellano-Reynoso, B., Tamez-Guerra, P., & Ramírez-Pfeiffer, C. (2022). Evaluation of the Fluorescence Polarization Assay for the Diagnosis of Brucellosis in Goat Milk. Veterinary Sciences, 9(6), 303. https://doi.org/10.3390/vetsci9060303






