Animal Models of Colorectal Cancer: From Spontaneous to Genetically Engineered Models and Their Applications
Abstract
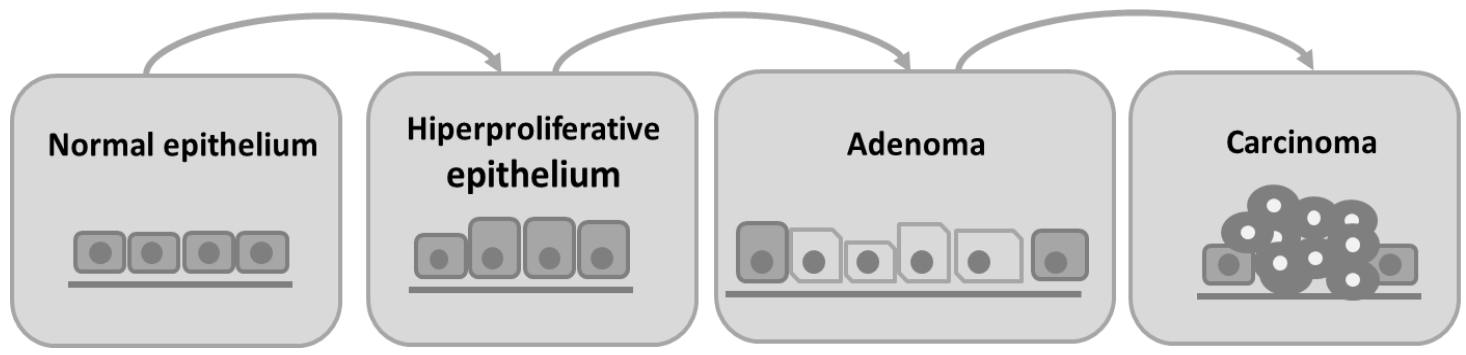
1. Introduction
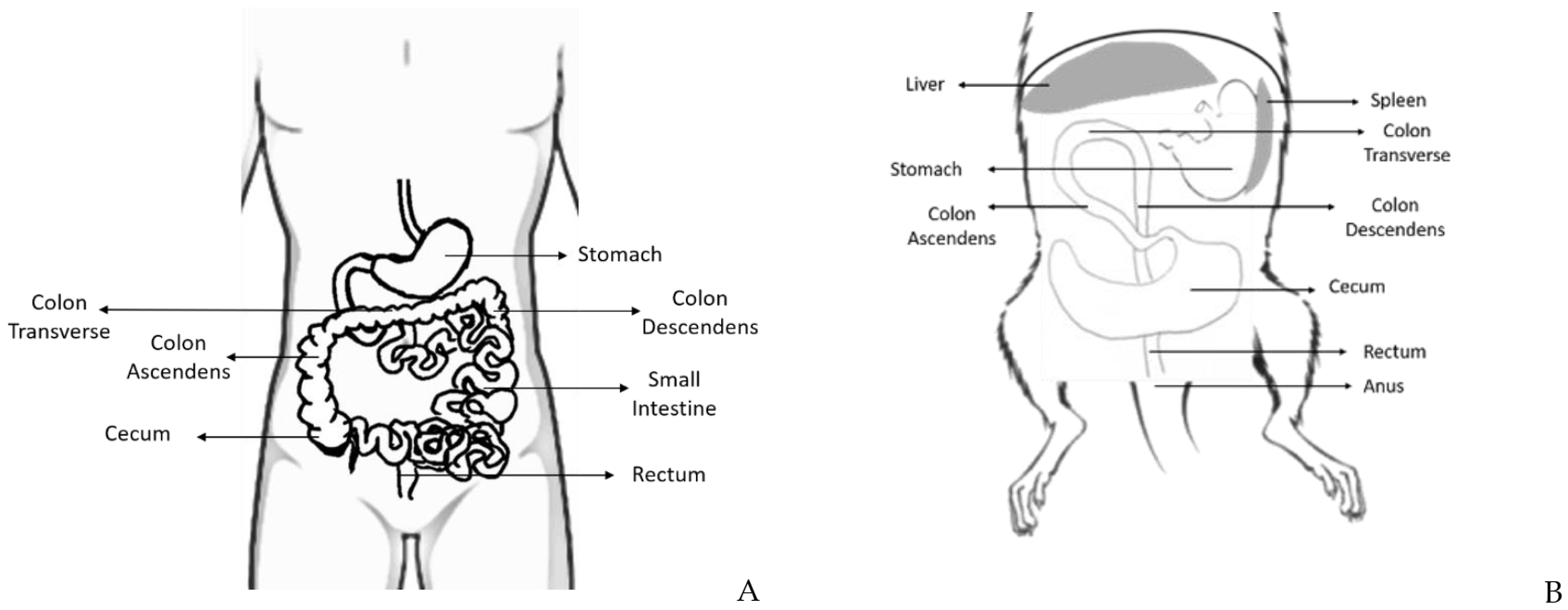
2. Rat and Mouse Colon and Rectum: Anatomy and Histology
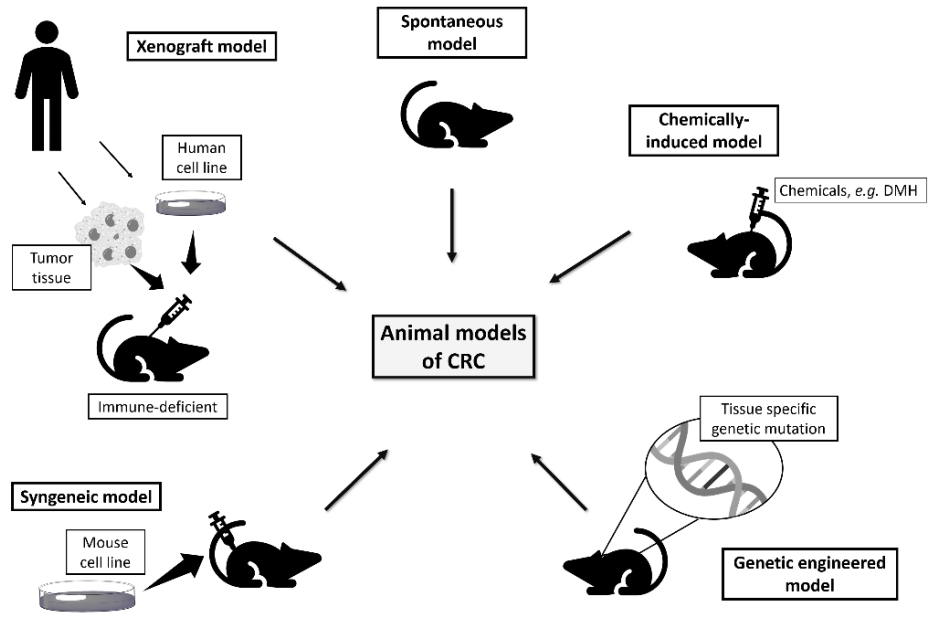
3. Rodents as Models of Colorectal Cancer
3.1. Spontaneous Models
3.2. Induced Models
3.2.1. Chemically Induced Models
3,2′-Dimethyl-4-Aminobiphenyl (DMAB)
N-Methyl-N-Nitrosourea (MNU) and N-Methyl-N-Nitrosoguanidine (MNNG)
1,2-Dimethylhydrazine (DMH)
Azoxymethane (AOM)
| Animal Strain and Gender | Carcinogenic Administration Route | Drugs or Compounds Evaluated (Classification) | Dose/Treatment | Therapeutic Effects (Ref) |
|---|---|---|---|---|
| Wistar male rats | i.p. 40 mg/kg b.w. 2x/wk for 2 wks | Hyperbaric oxygen (HBO2) | HBO2 alone or DMH + HBO2; 15 daily 90 min HBO2 sessions every 24 h at 2.0 atm absolute pressure | HBO2 had a protective effect in colorectal cancer, demonstrated by the decrease in COX-2 [41] |
| s.c. 40 mg/kg b.w. 2x/wk for 2wks | Astaxanthin | p.o. (15 mg/kg b.w.) 1 wk before and after DMH for 16 wks | Positive effects against colorectal cancer [42] | |
| s.c. 30 mg/kg 1x/week for 18 wks | Aspirin (a non-steroidal anti-inflammatory drug) | Gavage (0, 5, 30 or 60 mg/kg diet) daily for 18 wks | Reduced tumor incidence [43] | |
| s.c. 50 mg/kg b.w. 1 wk after diet supplemented and physical activity | Probiotic soy product and physical exercise | Gavage (3 mL/kg b.w./day fermented or unfermented soy products) and t.r. (60 min/day at 3–5% inclination at 355 m/min or 17–20 m/min) alone or in combination for 6 wks | No inhibition of colorectal cancer by the ingestion of fermented soy products or physical activity or by a combination of both [44] | |
| s.c. 40 mg/Kg b.w. for 8 wks | Epigallocatechin gallate (EGCG) | p.o. (50, 100 or 200 mg/Kg b.w.; once daily) for 8 wks | EGCG inhibited the formation of DMH-induced CRC by regulating key pathways, namely p53 and PI3K-Akt signaling pathways and I-kappaB kinase/NF-kappaB signal pathways, apoptosis signal pathways and MAPK cascades, involved in tumorigenesis [29] | |
| Wistar female rats | s.c. 20 mg/kg b.w. for 20 wks | High fiber diet and aspirin | Exp1: gavage (10 or 30 mg/kg/day b.w aspirin) Exp.2: diet supplemented with high fiber (16% crude fiber) from the beginning and for 32 wks | Protective effects of high fiber diet and aspirin. The aspirin effect is dose-related [45] |
| s.c. 20 mg/Kg b.w. for 5 wks | Methanolic extract of Muntingia calabura L. leaves (MEMC) | p.o. (100 or 200 mg/Kg b.w.) all days till the 15 week | MEMC offered a protective role against experimentally induced CRC via suppressing hyperproliferation and inflammation [46] | |
| F344 male rats | Exp.1: s.c.20 mg/kg b.w. daily for 16 wks; Exp.2: s.c. 20 mg/kg b.w. daily for 12 wks | Cellulose, calcium and folic acid | p.o. (Exp.1: 10% cellulose for 30 wks; Exp.2: 250 or 500 mg/100 g diet calcium + 0 or 0.1 mg/100 g folic acid for 22 wks) | Protective effects of cellulose and reduced tumor number and multiplicity of calcium [47] |
| i.p. 40 mg/kg b.w. once weekly for 4 wks | Adlay bran ethanol extract (ABE-Ea) | p.o. (8.64, 17.28 or 34.56 mg/day ABE-Ea) 1 wk before 1st DMH and for 9 or 18 wks | Inhibited preneoplastic lesions [48] | |
| i.p. 30 mg/kg weekly for 10 wks | Calcium and vitamin 3 in low or high-fat diet | p.o. (0.5 or 1%supplemental calcium and 1000 or 2000 IU/kg diet vitamin 3 in combination with a low-fat diet, 0.5% corn oil, or high-fat diet, 20% corn oil) 2 wks before DMH and for 20 wks | Preventive effects only in a high-fat diet [49] | |
| s.c. 100 mg/kg b.w. 2x/wk for 2 wks | Arabinoxylan-oligosaccharides (AXOS) (prebiotic) | p.o. (60 g/kg diet) 10 days before DMH and for 13 wks | Chemopreventive effect [50] | |
| Sprague-Dawley male rats | Gavage 30 mg/kg | Aspirin (non-steroidal anti-inflammatory drug) | Exp.1: s.c.(100 mg/kg/day aspirin) 1 wk before and after DMH and for 1 wk; Exp.2: s.s. (50 mg/kg/day aspirin) 4 wks after DMH and for 36 wks) | Reduced tumor incidence when aspirin was administered 1 wk before or after DMH but no effect when administered 4 wks after [51] |
| s.c. 30 mg/kg b.w. for 6 wks | etoricoxib (selective cycloxygenase-2 inhibitor) and diclofenac (a preferential cycloxygenase-2 inhibitor) | Gavage (0.6 mg/kg b.w. ectoricoxib and 8 mg/kg b.w. diclofenac) daily for 6 wks | Chemopreventive effect of both compounds [52] | |
| s.c. 20 mg/kg b.w. 2x/wk for 4 wks | Soy isoflavones | p.o. (1, 10, 50, 150 or 500 mg/kg diet) 1 wks after DMH and for 12 wks | Inhibited colorectal cancer in dose-independent manner [53] | |
| s.c. 20 mg/kg b.w. weekly from day 3 and for 12 wks | β-carotene (derived from carrots) sodium ascorbate (L-ascorbic acid) and cellulose | Diet supplemented (0.005% β-carotene or 0.02% sodium ascorbate or 1.5% cellulose) 12 wks before and for 28 wks | Only β-carotene showed an inhibitory effect of carcinogenesis [54] | |
| s.c. 20 mg/kg 1x/wk 13 wks | Wheat bran | Diet supplemented (fiber-free diet or 20% wheat bran supplement) for 31 wks | Increased colorectal carcinogenesis [55] | |
| s.c. 20 mg/kg b.w. 6x/wk | Milk and calcium | Diet supplemented (37 g/kg diet of milk and 40 mg/kg rat/day of calcium carbonate) | Protective activity by milk supplementation [56] | |
| Sprague-Dawley male and female rats | s.c. 21 mg/kg 1x/week for 18 wks | Calcium | d.w.(3.2 g/L calcium lactate) daily from the start until 25–34 wks | Inhibited colorectal cancer [57] |
| Animal Strain and Gender | Carcinogenic Administration Route | Drugs or Compounds Evaluated (Classification) | Dose/Treatment | Therapeutic Effects (Ref) |
|---|---|---|---|---|
| F344 male rats | s.c. injections 15 mg/kg b.w./wk once a week for 2 wks | Ursodeoxycholic acid and cholic acid (bile acids) | p.o.(0.2% or 0.4% cholic acid, 0.2% or 0.4% ursodeoxycholic acid, 0.2% cholic acid + 0.2% ursodeoxycholic acid) for 30 wks | Higher dose of ursodeoxycholic acid reduced the incidence of colorectal tumors [58] |
| s.c. 15 mg/kg bw once weekly for 2 wks | Celecoxib (a non-steroidal anti-inflammatory drug) | p.o. (500, 1000 or 1500 ppm) before exposure to AOM, during treatment, and until termination of the study at 52 wks | Chemopreventive activity in all tumor stages [59] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks | iNOS inhibitor L-N6 -(1-iminoethyl) lysine tetrazole-amide (SC-51), celocoxib (nonsteroidal anti-inflammatory) | p.o. (10, 30 or 100 ppm SC-51; 500 ppm celocoxib; 30 or 100 ppm SC-51 + 500 ppm celocoxib) for 8 wks | The combination of SC-51 with celocoxib was more effective in colorectal cancer prevention than the compounds alone [60] | |
| s.c. 15 mg/kg b.w. 1x/wk for 3 wks | Rebaudioside A, oleanolic acid, costunolide and soyasionin A2 (terpenoids), liquiritin (flavonoid), phyllodulcin and hydrangenol (isocumarins) | p.o. (200 ppm of each) for 5 wks | Costunolide is the most effective chemopreventive agent [61] | |
| s.c. 29.6 mg/kg b.w. | Piroxicam (a non-steroidal anti-inflammatory drug) and D, L-α-difluoromethylornithine (DFMO) | p.o. (25, 75 and 150 ppm piroxicam or 400, 1000 and 4000 ppm DFMO) 1 wk after AOM for 26 wks | A combination of piroxicam and DFMO was more effective in the inhibition of colorectal cancer than compounds alone [62] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks | Phenylethyl-3-methylcaffeate (PEMC) | p.o. (750 ppm) 2 wks before AOM for 52 wks | Inhibited colonic tumors [63] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks; start 2 wks after diet | Celocoxib (COX-2 inhibitor) | p.o. (1500 ppm) for 50 wks | Chemopreventive activity [64] | |
| s.c. 15 mg/kg b.w. at 7 and 8 wks of rat age | S-methylmethane thiosulfonate (S-MMTS) (isolate from cauliflower) and sulindac | p.o. (80 ppm S-MMTS, 160 ppm sulindac or 40 ppm S-MMTS + 160 ppm sulindac) 14 wks after AOM for | A combination of S-MMTS and sulindac was more effective in the inhibition of colorectal cancer than compounds alone [65] | |
| s.c. 15 mg/kg 1x/wk for 2 wks | Naproxen and NO-naproxen (nonspecific nonsteroidal anti-inflammatory drugs) | p.o. (200 or 400 ppm naproxen and 300 or 600 ppm nitric oxide-naproxen) 3 days after AOM for 8 wks | Chemopreventive effects [66] | |
| 15 mg/kg i.p. 1x/wk for 2 wks | Lovastatin (statin) and exisulind (selective apoptotic antineoplastic drug) | p.o. (50 ppm lovastatin, 100, 250 or 1000 ppm exisulind alone or in combination with 50 ppm lovastatin) for for 4 wks | Chemopreventive effects of lovastatin but not exisulind [67] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks | CP-31398 (p53-modulating agent) and celocoxib (non-steroidal anti-inflammatory drug) | Diet supplemented (1, 150 or 300 ppm CP-31398, 300 ppm celecoxib or 1500 ppm CP-31398 + 300 ppm celecoxib) 2 wks after AOM and for 48 wks | A combination of compounds enhanced colorectal cancer chemopreventive efficacy [68] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks | Aspirin (a non-steroidal anti-inflammatory drug) | p.o. (0, 200 or 400 ppm) daily 2 wks before AOM and for 52 wks | Inhibited incidence and multiplicity of colorectal carcinomas [69] | |
| s.c. injection 15 mg/kg b.w. 1x/wk for 2 wks | Prebiotic germinated barley foodstuff (a mixture of insoluble protein and dietary fiber) | Diet supplemented with prebiotic germinated barley foodstuff for 4 wks | Anti-tumorigenicity activity [70] | |
| i.p. 15 mg/kg b.w. | Aspirin (a non-steroidal anti-inflammatory drug) and α-Difluoromethylornithine (DFMO) (ornithine decarboxylase inhibitor) | p.o. (Exp1.: 0, 200, 600 or 1800 mg/kg/diet of aspirin or 1000 mg/kg diet of DFMO; 8 days before 1st AOM; Exp.2: 200, 600, 1800 mg/kg/diet aspirin or 1000 or 3000 mg/kg/diet of DFMO or 1000 mg/kg/diet DFMO + 200 or 600 mg/kg/diet aspirin; 8 days before 1st) for 43 wks after last AOM | The combination of aspirin and DFMO after AOM reduced colorectal tumors [71] | |
| s.c. 15 mg/kg b.w 1x/wk for 2 wks | Vitamin D, acetylsalicylic acid (a non-steroid anti-inflammatory drug) and calcium | Diet supplemented (0, 2500, 5000 or 7500 ppm calcium; 0 or 300 ppm acetylsalicylic acid alone or combination with 0 or 0.02 µg/kg diet vitamin D) 20 days before AOM and for 18 wks | Increased incidence of tumors with high levels of calcium alone or in combination with vitamin D; Vitamin D with acetylsalicylic acid also increased tumor incidence [72] | |
| s.c. 8 mg/kg b.w./wk for 10 wks | Dietary wheat bran and dehydrated citrus fiber (in form of orange peel) | Diet supplemented (0 or 15% wheat bran or citrus fiber) for 20 wks | Reduced the risk of colorectal tumors [24] | |
| s.c. 15 mg/kg 1x/wk for 2 wks) | Tea extracts, Polyphenols and epigallocatechin gallate (EGCG) | d.w. (360 or 3600 ppm black and green tea extracts; 360 or 1800 ppm EGCG; 360 or 1800 black tea polyphenols and 360 or 3600 green tea polyphenols) at 6 wks and for 43 wks | No effect in tumor incidence [73] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks) | Aspirin, celecoxib, (cyclooxygenase-2 inhibitor), and atorvastatin (3-hydroxy-3-methylglutaryl CoA reductase inhibitors) | Diet supplemented (150 ppm atorvastatin, 600 pp celecoxib, 400 ppm aspirin, 100 ppm atorvastatin + 300 ppm celecoxib or 100 ppm atorvastatin + 200 ppm aspirin) one day after AOM and for 42 wks | Inhibited the incidence and multiplicity of colorectal carcinomas alone or in combination [74] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks | Grape seed extract (GSE) | Diet supplemented (0.25 or 0.5% (w/w) GSE) 1 wk before AOM, 4 wks last AOM or during all study and for 16 wks | Chemopreventive efficacy against early steps of colorectal carcinogenesis [75] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks) | Celecoxib (cyclooxygenase-2 inhibitor) in diets high in mixed lipids (HFML) or fish oil (HFFO) | Diet supplemented (0, 250, 500, or 1000 ppm celecoxib with HFML or HFFO diet) one day after AOM and for 26 wks | Preventive effect of low doses of celecoxib in HFFO diet [76] | |
| F344 female rats | i.p. 20 mg/kg b.w. | Polyethylene-glycol (PEG) (non-fermented polymer) | Diet supplemented (3 g/kg b.w/day) 7 days after AOM and for 105 days | Chemopreventive effects [77] |
| i.p. 20 mg/kg b.w. | Heme in food (in form of chicken, beef, black pudding) | Diet supplemented (600 g/kg diet chicken, beef and black pudding) 7 days after AOM and for 100 days | Increased colorectal carcinogenesis for all compounds [78] | |
| s.c. 8 mg/kg b.w./wk for 10 wks | Alfalfa, pectin and wheat bran | Diet supplemented (0 or 15% alfalfa, pectin and wheat bran) for 40 wks after 1st AOM | Inhibited colorectal tumor incidence, especially by pectin or wheat bran [79] | |
| BALB-c female mice | i.p. 15 mg/kg1x/wk for 2 wks | Kefir (a probiotic fermented milk product) | p.o. (5 mL/kg b.w. fermented kefir milk) for 8 wks | Decreased and prevented the growth of colorectal tumors [80] |
| Sprague-Dawley male rats | s.c. 15 mg/kg 1x/wk for 2 wks, 28 days after diet supplementation | Amylose maize starch and butyrylated high-amylose maize starch | Diet supplemented (10% of high-amylose maize starch or 10% butyrylated alone or in combination) start at day 0 until euthanasia | The compound combination reduced the risk of developing colorectal cancer [81] |
| i.p. 15 mg/kg 1x/wk for 4 wks | indomethacin and copper-indomethacin (non-steroidal anti-inflammatory drug) | i.p. (3.0 mg/kg indomethacin or 3.8 mg/kg copper-indomethacin) daily | Both compounds showed chemopreventive activity, but indomethacin was more effective [82] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks | R-Flurbiprofen (non-steroidal anti-inflammatory drug) | Gavage (30 mg/kg b.w./per day) 6 days a week, 1 wk before AOM and for 30 wks | Protective effects against colorectal cancer development [83] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks, at day 45 of rat’s life | Soy isoflavones | p.o. (0, 40 100 mg/kg diet) from birth, including pregnancy and lactation, until 26 wks of life. AOM at day 45 | Lifetime exposure suppressed colon tumors growth [84] | |
| s.c. 15 mg/kg b.w. 1x/wk for 2 wks | Probiotic bacteria “bifidobacterium lactis” (B. lactis) and carbohydrate “resistant starch” (from a commercial source called Hi-maize 958 or Hi-maize S260) | Diet supplemented (100 g/kg/diet of Hi-maize 958 or Hi-miaze 260 and 1% lyophilized culture of B. lactis) | Protective effects by the combination of the two products [85] | |
| s.c. 15 mg/kg b.w. for three weekly doses | Xanthohumol (a polyphenol isolated from Humulus lupulus L.) | Gavage (5 mg/kg b.w.) every alternate day for 8 wks | Inhibited cell proliferation and induced apoptosis [86] | |
| Wistar rats | i.p. 15 mg/kg | L-lysine, propolis, or gum arabic | Gavage water (150 mg/kg L.-lysine, 100 mg/5 mL/kg propolis or 5 mL/kg gum arabic) daily for 16 wks | Gum arabic and propolis reduced the total number of aberrant crypt foci, L-lysine neither protected against nor enhanced colorectal cancer [87] |
Azoxymethane (AOM) and Dextran-Sodium Sulfate Model (DSS)
2-Amino-1-Methyl-6-Phenylimidazo (4,5-b) Pyridine (PhIP)
| Animal Strain and Gender | Carcinogenic Administration Route | Drugs or Compounds Evaluated (Classification) | Dose/Treatment | Therapeutic Effects (Ref) |
|---|---|---|---|---|
| CF-1 male mice | s.c. AOM 10 mg/kg body wt + 1 wk later d.w. 1.2% DSS for 7 days | Aspirin (acetylsalicylic acid) | Diet supplemented (0.02% aspirin) 1 wk before AOM and for 20 wks | Suppressed inflammatory colitis symptoms and tumor multiplicity [96] |
| C57BL/6 male mice | i.p. AOM 10 mg/kg + 1 wk later d.w. 2% DSS for 1 wk | Asther glehni Franchet et Sckmidt (common Korean dietary edible herb) | p.o. (25 mg/kg/day) 1 wk after AOM + DSS and for 1 wk | Inhibited colitis-associated colon carcinogenesis [97] |
| i.p. AOM 10 mg/kg b.w. + d.w. 2% DSS for 5 days | DA-6034 (7-Carboxymethyloxy-39,49,5-trimethoxyflavone) (synthetic derivative of flavonoid eupatilin) | Gavage (30 mg/kg) from day 7 to the end | Reduced the number of colon tumors [98] | |
| i.p. AOM 10 mg/kg b.w. + 5 days later d.w. 2.5% DSS followed by 14 days of normal water x 3 cycles | Pristimerin (a naturally triterpenoid) | i.p. (125 ng/kg) every 2 days for 80 days | Reduced the number and size of the tumors [99] | |
| i.p. AOM 10 mg/kg + 1 wk after d.w. 2% DSS for 1 wk | Chitooligosaccharides (oligomers that are depolymerized from chitosan) | i.g. (300 mg/kg) once a day and 6x/wk | Prevented colorectal cancer through regulating the gut microbiota and mycobiota [100] | |
| C57BL/6 female mice | i.p. AOM 10 mg/kg + 1wk later d.w. 2% DSS for 1x/wk for 3 wks | Conjugated linoleic acid (CLA) | Diet supplemented (1% CLA) 3 wk before AOM + DSS and for 13 wks | Increased colorectal cancer [101] |
| i.p. AOM 10 mg/kg b.w. + d.w. DSS 2.5% 1 wk after AOM for two cycle of 7 days | Licorice flavonoids (LFs) (Chinese herbal medicine) | Gavage (0, 50 or 100 mg/kg) once a day for 10 wks | Reduced tumorigenesis [102] | |
| BALB/c female mice | i.p. AOM 10 mg/kg + 1 wk after d.w.DSS 2.5% 2.5% of 3 cycles of 1 wk | Aloe vera gel | p.o. (200 or 400 mg/kg/day) 1 wk before AOM and for 13 wks | Reduced the multiplicity of colorectal adenomas and adenocarcinomas [103] |
| BALB/c male mice | i.p. AOM 12.5 mg/kg + 1 wk after dDSS 2.5% in d.w. for 3 cycles of 5 days | Triticum aestivum sprouts ethanol extract (TAEE) | Gavage (100 or 200 mg/kg/day) for 40 days | Inhibited colon inflammation and neoplasm formation [104] |
| CD-1 (ICR) male mice | i.p. AOM 10 mg/kg b.w. + 1 wk after d.w. DSS 1.5% for 7 days | Zerumbone (tropical ginger sesquiterpene) | Diet supplemented (100, 250, or 500 ppm) for 17 wks | Zerumbone suppresses mouse colon carcinogenesis through mechanisms of growth, apoptosis, inflammation that are involved in carcinogenesis in the colon [105] |
| i.p. AOM 10 mg/kg b.w. + 1 wk after d.w. DSS 1% for 7 days | Prenyloxycoumarins, auraptene and collinin nonsteroidal anti-inflammatory drugs) | Diet supplemented (0.01 and 0.05% of all compounds) 1 wk after DSS and for 17 wks | Chemopreventive activity [106] | |
| i.p. AOM 10 mg/kg b.w. + 1 wk after d.w. DSS 1% for 7 days | Ursodeoxycholic Acid (UDCA) and Sulfasalazine (anti-inflammatory agents) | Diet supplemented (0.016, 0.08 or 0.4% UDCA, 0.05% sulfasalazine or 0.5% UDCA + 0.05% sulfasalazine) 1 wk after DSS and for 20 wks | UDCA showed more suppressing effects on colorectal cancer [107] | |
| i.p. AOM 10 mg/kg b.w. + d.w. DSS 2.5% 1 wk after AOM and for 7 days | Dried açaí berry powder | Diet supplemented (2.5 or 5%) for 14 wks | Reduced the incidence of colorectal cancer [108] | |
| i.p. AOM (10 mg/kg b.w.) + d.w. DSS 1.5% 1 wk after AOM and for 7 days | Fucoxanthin (a xanthophyll present in marine brown algae) | Gavage (6 or 30 mg/kg) | Reduced the number of colorectal polyps [109] | |
| CD-1 (ICR) female mice | i.p. AOM 10 mg/kg b.w. + 1 wk after d.w. DSS 2% for 7 days | Nimesulide (a cyclooxygenase-2 inhibitor), troglitazone and bezafibrate (ligands for peroxisome proliferator-activated receptors) | Diet supplemented (0.04% nimesulide, 0.05% troglitazone and 0.05% bezafibrate) 1 wk after DSS and for 14 wk | Suppressed development of colorectal cancer [110] |
| 129SvJxC57BL6 male and female mice | i.p. AOM 12.5 mg/kg + 5 days later d.w. DSS 2% for 5 days followed by a 2-wk rest period and again 5 days of DSS | Chalcone lonchocarpin isolated from Lonchocarpus sericeus | i.p. (2.5 mg/mL) 4 wks after the last DSS cycle and for 4 days | Reduced tumor proliferation [111] |
| Animal Strain and Gender | Carcinogenic Administration Route | Drugs or Compounds Evaluated (Classification) | Dose/Treatment | Therapeutic Effects (Ref) |
|---|---|---|---|---|
| F344 male rats | p.o. 200 ppm for the first 20 wks | Tomato + broccoli powder in AIN93G diet | control, PhIP alone, PhIP + diet with 10% of tomato and broccoli powder for 20 wks and without PhIP for 32 wks | A diet rich in tomato + broccoli can reduce or prevent dietary carcinogens-induced cancer. Tomato + broccoli group reduced incidence and/or severity of cancer lesions [112] |
| Gavage 75 mL/kg b.w. 5 times a week for 2 wks | Yogurt powder (milk fermented by Lactobacillus delbrueckii subsp. Bulgaricus strain 2038 and Streptococcus salivariu subsp. thermophilus strain 1131) | Diet supplemented (10.4646% yogurt powder) 14 days before PhIP and for 14 days | Yogurt appears to have tumor-suppressing properties [113] | |
| Gavage daily 100 mg/kg b.w. for 2 wks | White tea, green tea, epigallocatechin-3-gallate (EGCG) and caffeine | d.w. (2% white tea, 2% green tea, 0.5 mg/mL EGCG or 9.5 mg/mL caffeine) 1 wk after last PhiP and for 16 wks | Inhibition of tumor initiation mostly by white tea, caffeine and EGCG [114] | |
| i.g. 100 mg/kg2x/wk for 10 wks | Nobiletin (5,6,7,8,3,4 -hexamethoxy flavone) (polymerthoxy-flavonoid extracted from citrus fruits) | Diet supplemented (0.05% nobiletin) for 50 wks | Chemopreventive activity of early carcinogenesis changes [115] | |
| Gavage 150 mg/kg for 5 alternate days | White tea | d.w. (2% wt/vol white tea) for 2 wks | Inhibition of preneoplastic lesion development [116] | |
| i.g. 200 mg/kg 2x/wk for 10 wks) | Fujiflavone (a commercial isoflavone supplement) | Diet supplemented (0.25% fujiflavone) for 50 wks | Preventive effects on colorectal cancer [117] | |
| Gavage 50 mg/kg b.w. | Clorophyllin (CHL) indole-3-carbinol (I3C) | p.o. (0.1% I3C and 0.1% CHL), before and during PhiP exposure or 1 wk after PhIP and for 16 wks | Protective effects for CHL and I3C on colorectal carcinogenesis [118] | |
| F344 female rats | Diet supplemented 0.02% | caffeine, α-tocopherol (lipophilic antioxidant), and n-tritriacontane-16,18-dione (TTAD) (β-diketone derivative) | p.o. (0.1% caffeine, 0.5% α-tocopherol or 0.1% TTAD) for 54 wks | Increase the incidence of colorectal tumors by caffeine; α-tocopherol and TTAD had no effect on colorectal tumors [119] |
| Sprangue-Dawley male rats | Gavage 10 mg/kg b.w. | Chinese cabbage (Brassica chinensis) | p.o. (20% freeze-dried cabbage powder) 10 days before PhIP and for 20 h | Preventive effect on initiated colorectal tumors [120] |
3.3. Genetically Engineered Models
3.3.1. Adenomatous Polyposis Mouse Models (APC)
3.3.2. F344-Pirc Rat Model
3.3.3. Hereditary Nonpolyposis Colon Cancer Mouse Models (HNPCC)
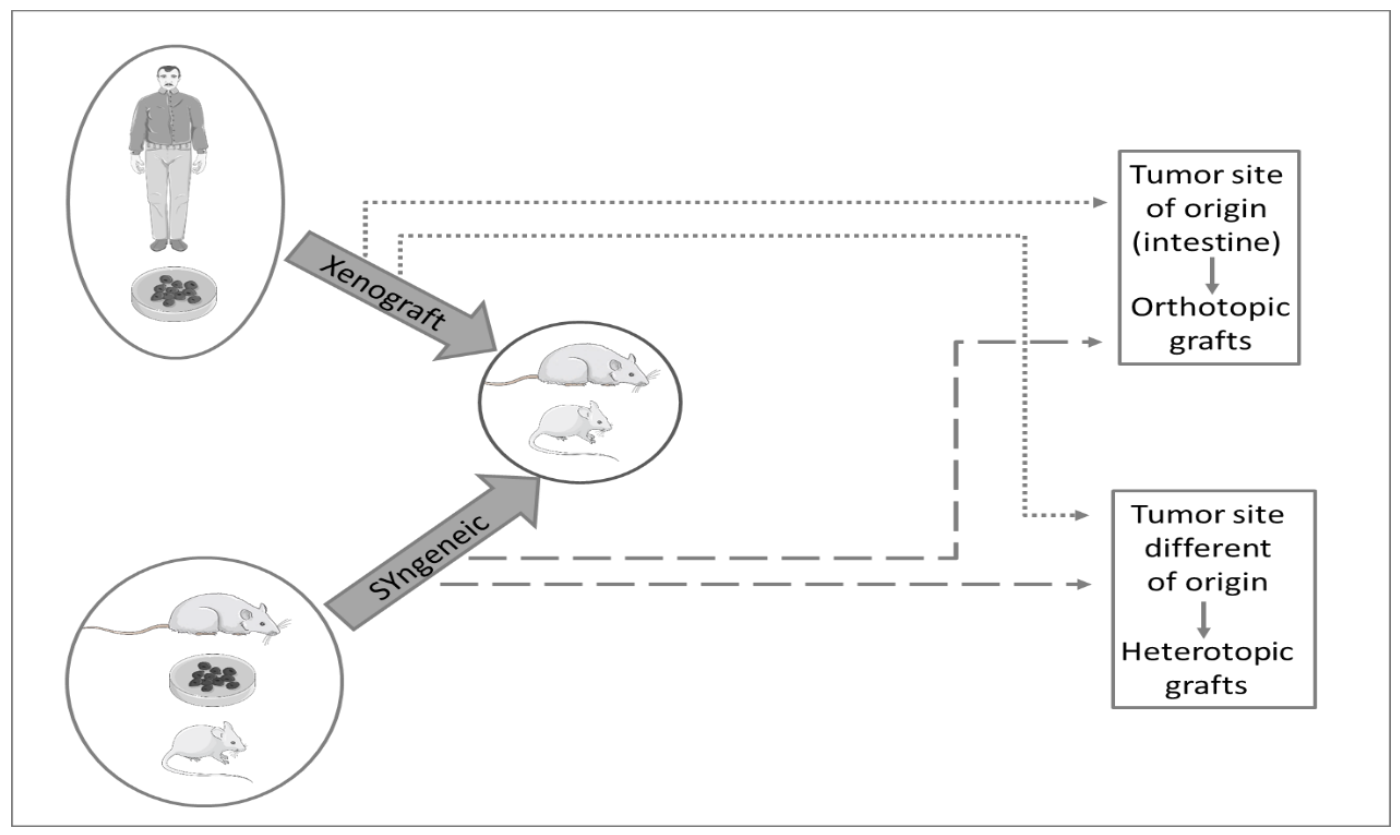
3.4. Xenograft and Syngeneic Models
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.; Torre, L.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Diet, Nutrition, Physical Activity and Colorectal Cancer; World Cancer Research Fund: London, UK; American Institute for Cancer Research: Washington, DC, USA, 2018.
- Thanikachalam, K.; Khan, G. Colorectal Cancer and Nutrition. Nutrients 2019, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Kobaek-Larsen, M.; Thorup, I.; Diederichsen, A.; Fenger, C.; Hoitinga, M.R. Review of Colorectal Cancer and Its Metastases in Rodent Models: Comparative Aspects with Those in Humans. Comp. Med. 2000, 50, 16–26. [Google Scholar] [PubMed]
- Tanaka, T. Colorectal Carcinogenesis: Review of Human and Experimental Animal Studies. J. Carcinog. 2009, 8, 5. [Google Scholar] [CrossRef]
- Machado, V.F.; Feitosa, M.R.; da Rocha, J.J.R.; Féres, O. A Review of Experimental Models in Colorectal Carcinogenesis. J. Coloproctology 2016, 36, 53–57. [Google Scholar] [CrossRef][Green Version]
- Raskov, H.; Pommergaard, H.-C.; Burcharth, J.; Rosenberg, J. Colorectal Carcinogenesis-Update and Perspectives. World J. Gastroenterol. 2014, 20, 18151. [Google Scholar] [CrossRef]
- Fagundes, D.J.; Taha, M.O. Modelo Animal de Doença: Critérios de Escolha e Espécies de Animais de Uso Corrente. Acta Cirúrgica Bras. 2004, 19, 59–65. [Google Scholar] [CrossRef]
- Johnson, R.L.; Fleet, J.C. Animal Models of Colorectal Cancer. Cancer Metastasis Rev. 2013, 32, 39–61. [Google Scholar] [CrossRef]
- Kwong, L.N.; Dove, W.F. APC and Its Modifiers in Colon Cancer. In APC Proteins; Näthke, I.S., McCartney, B.M., Eds.; Springer: New York, NY, 2009; Volume 656, pp. 85–106. ISBN 978-1-4419-1144-5. [Google Scholar]
- Oliveira, R.C.; Abrantes, A.M.; Tralhão, J.G.; Botelho, M.F. The role of mouse models in colorectal cancer research—The need and the importance of the orthotopic models. Anim. Model Exp. Med. 2020, 3, 1–8. [Google Scholar] [CrossRef]
- Treuting, P.M.; Dintzis, S.M.; Montine, K.S. Comparative Anatomy and Histology: A Mouse, Rat, and Human Atlas; Academic Press: Cambridge, MA, USA, 2017; ISBN 978-0-12-802919-0. [Google Scholar]
- Vdoviaková, K.; Petrovova, E.; Maloveska, M.; Krešáková, L.; Teleky, J.; Elias, M.; Petrášová, D. Surgical Anatomy of the Gastrointestinal Tract and Its Vasculature in the Laboratory Rat. Gastroenterol. Res. Pract. 2016, 2016, 2632368. [Google Scholar] [CrossRef]
- Evans, J.P.; Sutton, P.A.; Winiarski, B.K.; Fenwick, S.W.; Malik, H.Z.; Vimalachandran, D.; Tweedle, E.M.; Costello, E.; Palmer, D.H.; Park, B.K.; et al. From Mice to Men: Murine Models of Colorectal Cancer for Use in Translational Research. Crit. Rev. Oncol. Hematol. 2016, 98, 94–105. [Google Scholar] [CrossRef]
- Suman, S.; Datta, K. Animal Models of Colorectal Cancer in Chemoprevention and Therapeutics Development. In Colorectal Cancer: From Prevention to Patient Care; Intech Open: London, UK, 2012; pp. 277–300. [Google Scholar] [CrossRef]
- Newmark, H.; Yang, K.; Kurihara, N.; Fan, K.; Augenlicht, L. Western-Style Diet-Induced Colonic Tumors and Their Modulation by Calcium and Vitamin D in C57Bl/6 Mice: A Preclinical Model for Human Sporadic Colon Cancer. Carcinogenesis 2008, 30, 88–92. [Google Scholar] [CrossRef]
- Miyamoto, M.; Tani, Y. A Study on Colon Cancer-Prone Rats of WF-Osaka Strain. Med. J. Osaka Univ. 1989, 38, 1–12. [Google Scholar]
- Lorenz, E.; Stewart, H.L. Intestinal Carcinoma and Other Lesions in Mice Following Oral Administration of 1, 2, 5, 6-Dibenzanthracene and 20-Methylcholanthrene. J. Natl. Cancer Inst. 1940, 1, 17–40. [Google Scholar]
- Rosenberg, D.W.; Giardina, C.; Tanaka, T. Mouse Models for the Study of Colon Carcinogenesis. Carcinogenesis 2009, 30, 183–196. [Google Scholar] [CrossRef]
- Greene, F.L.; Lamb, L.S.; Barwick, M. Colorectal Cancer in Animal Models-A Review. J. Surg. Res. 1987, 43, 476–487. [Google Scholar] [CrossRef]
- Reddy, B.S. Colon Carcinogenesis Models for Chemoprevention Studies. Cancer Chemoprev. 1998, 12, 963–973. [Google Scholar] [CrossRef]
- Walpole, A.L.; Williams, M.H.C.; Roberts, D.C. The Carcinogenic Action of 4-Aminodiphenyl and 3:2′-Dimethyl-4-Amino-Diphenyl. Br. J. Ind. Med. 1952, 9, 255–263. [Google Scholar]
- Reddy, B.S.; Mori, H. Effect of Dietary Wheat Bran and Dehydrated Citrus Fiber on 3,2′-Dimethyl-4-Aminobiphenyl-Induced Intestinal Carcinogenesis in F344 Rats. Carcinogenesis 1981, 2, 21–25. [Google Scholar] [CrossRef]
- Davis, C.D.; Feng, Y. Dietary Copper, Manganese and Iron Affect the Formation of Aberrant Crypts in Colon of Rats Administered 3,2′-Dimethyl-4-Aminobiphenyl. J. Nutri. 1999, 129, 1060–1067. [Google Scholar] [CrossRef][Green Version]
- Feng, Y.; Finley, J.W.; Davis, C.D.; Becker, W.K.; Fretland, A.J.; Hein, D.W. Dietary Selenium Reduces the Formation of Aberrant Crypts in Rats Administered 3,2′-Dimethyl-4-Aminobiphenyl. Toxicol. Appl. Pharmacol. 1999, 157, 36–42. [Google Scholar] [CrossRef]
- Ravoori, S.; Feng, Y.; Neale, J.R.; Jeyabalan, J.; Hein, D.W.; Gupta, R.C. Dose-Dependent Reduction of 3,2′-Dimethyl-4-Aminobiphenyl- Derived DNA Adducts in Colon and Liver of Rats Administered Celecoxib. Mutat. Res. 2008, 638, 103–109. [Google Scholar] [CrossRef][Green Version]
- Williams, G.M.; Iatropoulos, M.J.; Jeffrey, A.M.; Shirai, T. Protective Effect of Acetaminophen against Colon Cancer Initiation Effects of 3,2′-Dimethyl-4-Aminobiphenyl in Rats. Eur. J. Cancer Prev. 2002, 11, 39–48. [Google Scholar] [CrossRef]
- Narisawa, T.; Weisburger, J.H. Colon Cancer Induction in Mice by Intrarectal Instillation of N-Methylnitrosourea (38498). Proc. Soc. Exp. Biol. Med. 1075, 148, 166–169. [Google Scholar] [CrossRef]
- Narisawa, T.; Fukaura, Y.; Takeba, N.; Nakai, K. Chemoprevention of N-Methylnitrosourea-Induced Colon Carcinogenesis by Ursodeoxycholic Acid-5-Aminosalicylic Acid Conjugate in F344 Rats. Jpn. J. Cancer Res. 2002, 93, 143–150. [Google Scholar] [CrossRef]
- Wang, Y.; Jin, H.-Y.; Fang, M.-Z.; Wang, X.-F.; Chen, H.; Huang, S.-L.; Kong, D.-S.; Li, M.; Zhang, X.; Sun, Y.; et al. Epigallocatechin Gallate Inhibits Dimethylhydrazine-Induced Colorectal Cancer in Rats. World J. Gastroenterol. 2020, 26, 2064–2081. [Google Scholar] [CrossRef]
- Nakayama, Y.; Inoue, Y.; Minagawa, N.; Onitsuka, K.; Nagata, J.; Shibao, K.; Hirata, K.; Sako, T.; Nagata, N.; Yamaguchi, K. Chemopreventive Effect of 4-[3,5-Bis(Trimethylsilyl) Benzamido] Benzoic Acid (TAC-101) on MNU-Induced Colon Carcinogenesis in a Rat Model. Anticancer Res. 2009, 29, 2059–2065. [Google Scholar]
- McSherry, C.K.; Cohen, B.I.; Bokkenheuser, V.D.; Mosbach, E.H.; Winter, J.; Matoba, N.; Scholes, J. Effects of Calcium and Bile Acid Feeding on Colon Tumors in the Rat. Cancer Res. 1989, 49, 6039–6043. [Google Scholar]
- Hawks, A.; Magee, P.N. The Alkylation of Nucleic Acids of Rat and Mouse in Vivo by the Carcinogen 1,2-Dimethylhydrazine. Br. J. Cancer 1974, 30, 440–447. [Google Scholar] [CrossRef][Green Version]
- Druckrey, H.; Preussmann, R.; Matzkies, F.; Ivankovic, S. Selektive Erzeugung von Darmkrebs bei Ratten durch 1,2-Dimethyl-hydrazin. Naturwissenschaften 1967, 54, 285–286. [Google Scholar] [CrossRef]
- Thorup, I.; Meyer, O.; Kristiansen, E. Effect of a Dietary Fiber (Beet Fiber) on Dimethylhydrazine-Induced Colon Cancer in Wistar Rats. Nutr. Cancer 1992, 17, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Markert, C.; Rogers, L.W.; Chiu, J.-F. Induction of Colonic Adenocarcinomas by 1, 2-Dimethylhiydrazine Intrarectally Administered in Rats. Digestion 1978, 18, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Bissahoyo, A.; Pearsall, R.S.; Hanlon, K.; Amann, V.; Hicks, D.; Godfrey, V.L.; Threadgill, D.W. Azoxymethane Is a Genetic Background-Dependent Colorectal Tumor Initiator and Promoter in Mice: Effects of Dose, Route, and Diet. Toxicol. Sci. 2005, 88, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.S. Studies with the Azoxymethane-Rat Preclinical Model for Assessing Colon Tumor Development and Chemoprevention. Environ. Mol. Mutagenesis 2004, 44, 26–35. [Google Scholar] [CrossRef]
- Sohn, O.S.; Fiala, E.S.; Puz, C.; Hamilton, S.R.; Williams, G.M. Enhancement of Rat Liver Microsomal Metabolism of Azoxymethane to Methylazoxymethanol by Chronic Ethanol Administration: Similarity to the Microsomal Metabolism of N-Nitrosodimethylamine. Cancer Res. 1987, 47, 3123–3129. [Google Scholar]
- Derry, M.; Raina, K.; Agarwal, R.; Agarwal, C. Characterization of Azoxymethane-Induced Colon Tumor Metastasis to Lung in a Mouse Model Relevant to Human Sporadic Colorectal Cancer and Evaluation of Grape Seed Extract Efficacy. Exp. Toxic Pathol. 2014, 66, 2166–2171. [Google Scholar] [CrossRef]
- Gois, E.; Daniel, R.; Parra, R.; Almeida, A.; Rocha, J.; Garcia, S.; Féres, O. Hyperbaric Oxygen Therapy Reduces COX-2 Expression in a Dimethylhydrazine-Induced Rat Model of Colorectal Carcinogenesis. Undersea Hyperb. Med. 2012, 39, 693–698. [Google Scholar]
- Prabhu, P.N.; Ashokkumar, P.; Sudhandiran, G. Antioxidative and Antiproliferative Effects of Astaxanthin during the Initiation Stages of 1,2-Dimethyl Hydrazine-Induced Experimental Colon Carcinogenesis. Fundam. Clin. Pharmacol. 2009, 23, 225–234. [Google Scholar] [CrossRef]
- Davis, A.E.; Patterson, F. Aspirin Reduces the Incidence of Colonic Carcinoma in the Dimethylhydrazine Rat Animal Model. Aust. N. Z. J. Med. 1994, 24, 301–303. [Google Scholar] [CrossRef]
- Silva, M.F.; Sivieri, K.; Rossi, E.A. Effects of a Probiotic Soy Product and Physical Exercise on Formation of Pre-Neoplastic Lesions in Rat Colons in a Short-Term Model of Carcinogenic. J. Int. Soc. Sports Nutr. 2009, 6, 17. [Google Scholar] [CrossRef]
- Miliaras, S.; Miliaras, D.; Vrettou, E.; Zavitsanakis, A.; Kiskinis, D. The Effect of Aspirin and High Fibre Diet on Colorectal Carcinoma: A Comparative Experimental Study. Tech. Coloproctology 2004, 8, s59–s61. [Google Scholar] [CrossRef]
- Jisha, N.; Vysakh, A.; Vijeesh, V.; Anand, P.S.; Latha, M.S. Methanolic Extract of Muntingia Calabura L. Mitigates 1,2-Dimethyl Hydrazine Induced Colon Carcinogenesis in Wistar Rats. Nutr. Cancer 2020, 1–13. [Google Scholar] [CrossRef]
- Ranhotra, G.S.; Gelroth, J.A.; Glaser, B.K.; Schoening, P.; Brown, S.E. Cellulose and Calcium Lower the Incidence of Chemically-Induced Colon Tumors in Rats. Plant Foods Hum. Nutr. 1999, 54, 295–303. [Google Scholar] [CrossRef]
- Chung, C.-P.; Hsu, H.-Y.; Huang, D.-W.; Hsu, H.-H.; Lin, J.-T.; Shih, C.-K.; Chiang, W. Ethyl Acetate Fraction of Adlay Bran Ethanolic Extract Inhibits Oncogene Expression and Suppresses DMH-Induced Preneoplastic Lesions of the Colon in F344 Rats through an Anti-Inflammatory Pathway. J. Agric. Food Chem. 2010, 58, 7616–7623. [Google Scholar] [CrossRef]
- Pence, B.C.; Buddingh, F. Inhibition of Dietary Fat-Promoted Colon Carcinogenesis in Rats by Supplemental Calcium or Vitamin D3. Carcinogenesis 1988, 9, 187–190. [Google Scholar] [CrossRef]
- Femia, A.P.; Salvadori, M.; Broekaert, W.F.; François, I.E.J.A.; Delcour, J.A.; Courtin, C.M.; Caderni, G. Arabinoxylan-Oligosaccharides (AXOS) Reduce Preneoplastic Lesions in the Colon of Rats Treated with 1,2-Dimethylhydrazine (DMH). Eur. J. Nutr. 2010, 49, 127–132. [Google Scholar] [CrossRef]
- Craven, P.A.; DeRubertis, F.R. Effects of Aspirin on 1, 2-Dimethylhydrazine-Induced Colonic Carcinogenesis. Carcinogenesis 1992, 13, 541–546. [Google Scholar] [CrossRef]
- Kaur Saini, M.; Nath Sanyal, S. Evaluation of Chemopreventive Response of Two Cycloxygenase-2 Inhibitors, Etoricoxib and Diclofenac in Rat Colon Cancer Using FTIR and NMR Spectroscopic Techniques. Nutr. Hosp. 2010, 25, 577–585. [Google Scholar]
- Min, W.-K.; Sung, H.-Y.; Choi, Y.-S. Suppression of Colonic Aberrant Crypt Foci by Soy Isoflavones Is Dose-Independent in Dimethylhydrazine-Treated Rats. J. Med. Food 2010, 13, 495–502. [Google Scholar] [CrossRef]
- Yamamoto, I.; Maruyama, H.; Moriguchi, M. Effect of P-Carotene, Sodium Ascorbate and Cellulose on 1,2-Dimethylhydrazine-Induced Intestinal Carcinogenesis in Rats. Cancer Lett. 1994, 86, 5–9. [Google Scholar] [CrossRef]
- Jacobs, L.R. Enhancement of Rat Colon Carcinogenesis by Wheat Bran Consumption during the Stage of 1,2-Dimethylhydrazine Administration. Cancer Res. 1983, 43, 6. [Google Scholar]
- Nelson, R.L.; Tanure, J.C.; Andrianopoulos, G. The Effect of Dietary Milk and Calcium on Experimental Colorectal Carcinogenesis. Dis. Colon Rectum 1987, 30, 947–949. [Google Scholar] [CrossRef]
- Viñas-Salas, J.; Biendicho-Palau, P.; Piñol-Felis, C.; Miguelsanz-Garcia, S.; Perez-Holanda, S. Calcium Inhibits Colon Carcinogenesis in an Experimental Model in the Rat. Eur. J. Cancer 1998, 34, 1941–1945. [Google Scholar] [CrossRef]
- Earnest, L.; Wall, K.; Jolley, S.; Bhattacharyya, K. Chemoprevention of Azoxymethane-Induced Colonic Carcinogenesis by Supplemental Dietary Ursodeoxycholic Acid. Cancer Res. 1994, 54, 5071–5074. [Google Scholar]
- Reddy, B.S.; Hirose, Y.; Lubet, R.; Steele, V.; Kelloff, G.; Paulson, S.; Seibert, K.; Rao, C.V. Chemoprevention of Colon Cancer by Specific Cyclooxygenase-2 Inhibitor, Celecoxib, Administered during Different Stages of Carcinogenesis. Cancer Res. 2000, 60, 293–297. [Google Scholar]
- Rao, C.V.; Indranie, C.; Simi, B.; Manning, P.T.; Connor, J.R.; Reddy, B.S. Chemopreventive Properties of a Selective Inducible Nitric Oxide Synthase Inhibitor in Colon Carcinogenesis, Administered Alone or in Combination with Celecoxib, a Selective Cyclooxygenase-2 Inhibitor. Cancer Res. 2002, 62, 165–170. [Google Scholar]
- Kawamori, T.; Tanaka, T.; HarÃ, A.; Yamahara, J.; Mori, H. Modifying Effects of Naturally Occurring Products on the Development of Colonie Aberrant Crypt Foci Induced by Azoxymethane in F344 Rats. Cancer Res. 1995, 55, 1277–1282. [Google Scholar]
- Reddy, B.S.; Nayini, J.; Tokumo, K.; Rigotty, J.; Zang, E.; Kelloff, G. Chemoprevention of Colon Carcinogenesis by Concurrent Administration of Piroxicam, a Nonsteroidal Antiinflammatory Drug with d,l-α-Difluoromethylornithine, an Ornithine Decarboxylase Inhibitor, in Diet. Cancer Res. 1990, 50, 2562–2568. [Google Scholar]
- Chinthalapally, R.; Desai, D.; Rivenson, A.; Simi, B.; Amin, S.; Reddy, B. Chemoprevention of Colon Carcinogenesis by Phenylethyl-3-Methylcaffeate1. Cancer Res. 1995, 55, 2310–2315. [Google Scholar]
- Kawamori, T.; Rao, V.; Seibert, K.; Reddy, B.S. Chemopreventive Activity of Celecoxib, a Specific Cyclooxygenase-2 Inhibitor, against Colon Carcinogenesis. Cancer Res. 1998, 58, 409–412. [Google Scholar]
- Reddy, B.S. Chemopreventive Effect of S-Methylmethane Thiosulfonate and Sulindac Administered Together during the Promotion/Progression Stages of Colon Carcinogenesis. Carcinogenesis 1999, 20, 1645–1648. [Google Scholar] [CrossRef] [PubMed]
- Steele, V.E.; Rao, C.V.; Zhang, Y.; Patlolla, J.; Boring, D.; Kopelovich, L.; Juliana, M.M.; Grubbs, C.J.; Lubet, R.A. Chemopreventive Efficacy of Naproxen and Nitric Oxide-Naproxen in Rodent Models of Colon, Urinary Bladder, and Mammary Cancers. Cancer Prev. Res. 2009, 2, 951–956. [Google Scholar] [CrossRef]
- Kim, K.P.; Whitehead, C.; Piazza, G.; Wargovich, M.J. Combinatorial Chemoprevention: Efficacy of Lovostatin and Exisulind on the Formation and Progression of Aberrant Crypt Foci. Anticancer Res. 2004, 24, 1805–1812. [Google Scholar] [PubMed]
- Rao, C.V.; Steele, V.E.; Swamy, M.V.; Patlolla, J.M.R.; Guruswamy, S.; Kopelovich, L. Inhibition of Azoxymethane-Induced Colorectal Cancer by CP-31398, a TP53 Modulator, Alone or in Combination with Low Doses of Celecoxib in Male F344 Rats. Cancer Res. 2009, 69, 8175–8182. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.S.; Rao, C.V.; Rivenson, A.; Kelloff, G. Inhibitory Effect of Aspirin on Azoxymethane-Induced Colon Carcinogenesis in F344 Rats. Carcinogenesis 1993, 14, 1493–1497. [Google Scholar] [CrossRef] [PubMed]
- Kanauchi, O.; Mitsuyama, K.; Andoh, A.; Iwanaga, T. Modulation of Intestinal Environment by Prebiotic Germinated Barley Foodstuff Prevents Chemo-Induced Colonic Carcinogenesis in Rats. Oncol. Rep. 2008, 20, 793–801. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, H.; Schut, H.A.J.; Conran, P.; Kramer, P.M.; Lubet, R.A.; Steele, V.E.; Hawk, E.E.; Kelloff, G.J.; Pereira, M.A. Prevention by Aspirin and Its Combination with α-Difluoromethylornithine of Azoxymethane-Induced Tumors, Aberrant Crypt Foci and Prostaglandin E2 Levels in Rat Colon. Carcinogenesis 1999, 20, 425–430. [Google Scholar] [CrossRef]
- Mølck, A.-M.; Poulsen, M.; Meyer, O. The Combination of 1a,25(OH)2–Vitamin D3, Calcium and Acetylsalicylic Acid Affects Azoxymethane-Induced Aberrant Crypt Foci and Colorectal Tumours in Rats. Cancer Lett. 2002, 10, 19–28. [Google Scholar] [CrossRef]
- Weisburger, J.H.; Rivenson, A.; Aliaga, C.; Reinhardt, J.; Kelloff, G.J.; Boone, C.W.; Steele, V.E.; Balentine, D.A.; Pittman, B.; Zang, E. Effect of Tea Extracts, Polyphenols, and Epigallocatechin Gallate on Azoxymethane-Induced Colon Cancer. Exp. Biol. Med. 1998, 217, 104–108. [Google Scholar] [CrossRef]
- Reddy, B.S.; Wang, C.X.; Kong, A.-N.; Khor, T.O.; Zheng, X.; Steele, V.E.; Kopelovich, L.; Rao, C.V. Prevention of Azoxymethane-Induced Colon Cancer by Combination of Low Doses of Atorvastatin, Aspirin, and Celecoxib in F 344 Rats. Cancer Res. 2006, 66, 4542–4546. [Google Scholar] [CrossRef]
- Velmurugan, B.; Singh, R.P.; Agarwal, R.; Agarwal, C. Dietary-Feeding of Grape Seed Extract Prevents Azoxymethane-Induced Colonic Aberrant Crypt Foci Formation in Fischer 344 Rats. Mol. Carcinog. 2010, 49, 641–652. [Google Scholar] [CrossRef]
- Reddy, B.S.; Patlolla, J.M.; Simi, B.; Wang, S.H.; Rao, C.V. Prevention of Colon Cancer by Low Doses of Celecoxib, a Cyclooxygenase Inhibitor, Administered in Diet Rich in ω-3 Polyunsaturated Fatty Acids. Cancer Res. 2005, 65, 8022–8027. [Google Scholar] [CrossRef]
- Corpet, D.E.; Parnaud, G. Polyethylene-Glycol, a Potent Suppressor of Azoxymethane-Induced Colonic Aberrant Crypt Foci in Rats. Carcinogenesis 1999, 20, 915–918. [Google Scholar] [CrossRef][Green Version]
- Pierre, F.; Freeman, A.; Taché, S.; Van der Meer, R.; Corpet, D.E. Beef Meat and Blood Sausage Promote the Formation of Azoxymethane-Induced Mucin-Depleted Foci and Aberrant Crypt Foci in Rat Colons. J. Nutr. 2004, 134, 2711–2716. [Google Scholar] [CrossRef]
- Watanabe, K.; Reddy, B.S.; Weisburger, J.H.; Kritchevsky, D. Effect of Dietary Alfalfa, Pectin, and Wheat Bran on Azoxymethane-or Methylnitrosourea-Induced Colon Carcinogenesis in F344 Rats. J. Natl. Cancer Inst. 1979, 63, 141–145. [Google Scholar]
- Melo, A.; Mendonça, M.; Rosa-Castro, R. The Protective Effects of Fermented Kefir Milk on Azoxymethane-Induced Aberrant Crypt Formation in Mice Colon. Tissue Cell 2018, 52, 51–56. [Google Scholar] [CrossRef]
- Clarke, J.M.; Topping, D.L.; Bird, A.R.; Young, G.P.; Cobiac, L. Effects of High-Amylose Maize Starch and Butyrylated High-Amylose Maize Starch on Azoxymethane-Induced Intestinal Cancer in Rats. Carcinogenesis 2008, 29, 2190–2194. [Google Scholar] [CrossRef]
- Bonin, A.M.; Yáñez, J.A.; Fukuda, C.; Teng, X.W.; Dillon, C.T.; Hambley, T.W.; Lay, P.A.; Davies, N.M. Inhibition of Experimental Colorectal Cancer and Reduction in Renal and Gastrointestinal Toxicities by Copper–Indomethacin in Rats. Cancer Chemother. Pharmacol. 2010, 66, 755–764. [Google Scholar] [CrossRef]
- Martin, J.E.; Le Leu, R.K.; Hu, Y.; Young, G.P. R-Flurbiprofen Suppresses Distal Nonmucin-Producing Colorectal Tumors in Azoxymethane-Treated Rats, without Suppressing Eicosanoid Production. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G860–G864. [Google Scholar] [CrossRef]
- Raju, J.; Bielecki, A.; Caldwell, D.; Lok, E.; Taylor, M.; Kapal, K.; Curran, I.; Cooke, G.M.; Bird, R.P.; Mehta, R. Soy Isoflavones Modulate Azoxymethane-Induced Rat Colon Carcinogenesis Exposed Pre- and Postnatally and Inhibit Growth of DLD-1 Human Colon Adenocarcinoma Cells by Increasing the Expression of Estrogen Receptor-β. J. Nutr. 2009, 139, 474–481. [Google Scholar] [CrossRef]
- Le Leu, R.K.; Hu, Y.; Brown, I.L.; Woodman, R.J.; Young, G.P. Synbiotic Intervention of Bifidobacterium Lactis and Resistant Starch Protects against Colorectal Cancer Development in Rats. Carcinogenesis 2010, 31, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, L.; Li, G.; Gao, Z. Xanthohumol Protects against Azoxymethane-Induced Colorectal Cancer in Sprague-Dawley Rats. Environ. Toxicol. 2020, 35, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Braga, V.N.L.; Juanes, C.D.C.; Peres Júnior, H.D.S.; Sousa, J.R.D.; Cavalcanti, B.C.; Jamacaru, F.V.F.; Lemos, T.L.G.D.; Dornelas, C.A. Gum Arabic and Red Propolis Protecteting Colorectal Preneoplastic Lesions in a Rat Model of Azoxymethane. Acta Cir. Bras. 2019, 34, e201900207. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kohno, H.; Suzuki, R.; Yamada, Y.; Sugie, S.; Mori, H. A Novel Inflammation-Related Mouse Colon Carcinogenesis Model Induced by Azoxymethane and Dextran Sodium Sulfate. Cancer Sci. 2003, 94, 965–973. [Google Scholar] [CrossRef]
- Tanaka, T.; Suzuki, R.; Kohno, H.; Sugie, S.; Takahashi, M.; Wakabayashi, K. Colonic Adenocarcinomas Rapidly Induced by the Combined Treatment with 2-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine and Dextran Sodium Sulfate in Male ICR Mice Possess β-Catenin Gene Mutations and Increases Immunoreactivity for β-Catenin, Cyclooxygenas. Carcinogenesis 2005, 26, 229–238. [Google Scholar] [CrossRef]
- Kohno, H.; Suzuki, R.; Sugie, S.; Tanaka, T. β-Catenin Mutations in a Mouse Model of Inflammation-Related Colon Carcinogenesis Induced by 1,2-Dimethylhydrazine and Dextran Sodium Sulfate. Cancer Sci. 2005, 96, 69–76. [Google Scholar] [CrossRef]
- Suzuki, R.; Kohno, H.; Sugie, S.; Nakagama, H.; Tanaka, T. Strain Differences in the Susceptibility to Azoxymethane and Dextran Sodium Sulfate-Induced Colon Carcinogenesis in Mice. Carcinogenesis 2006, 27, 162–169. [Google Scholar] [CrossRef]
- Robertis, M.; Massi, E.; Poeta, M.L.; Carotti, S.; Cecchetelli, L.; Signori, E.; Fazio, V.M. The AOM/DSS Murine Model for the Study of Colon Carcinogenesis: From Pathways to Diagnosis and Therapy Studies. J. Carcinog. 2011, 10. [Google Scholar] [CrossRef]
- Takayama, K.; Yamashita, K.; Wakabayashi, K.; Sugimura, T.; Nagao, M. DNA Modification by 2-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine in Rats. Jpn. J. Cancer Res. 1989, 80, 1145–1148. [Google Scholar] [CrossRef]
- Nakagama, H.; Nakanishi, M.; Ochiai, M. Modeling Human Colon Cancer in Rodents Using a Food-Borne Carcinogen, PhIP. Cancer Sci. 2005, 96, 627–636. [Google Scholar] [CrossRef]
- Ito, N.; Hasegawa, R.; Sano, M.; Tamano, S.; Esumi, H.; Takayama, S.; Sugimura, T. A New Colon and Mammary Carinogen in Cooked Food, 1-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine (PhIP). Carcinogenesis 1991, 12, 1503–1506. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, Y.; Zhang, C.; Su, Z.-Y.; Li, W.; Huang, M.-T.; Kong, A.-N. The Epigenetic Effects of Aspirin: The Modification of Histone H3 Lysine 27 Acetylation in the Prevention of Colon Carcinogenesis in Azoxymethane- and Dextran Sulfate Sodium-Treated CF-1 Mice. Carcinogenesis 2016, 37, 616–624. [Google Scholar] [CrossRef]
- Chung, K.-S.; Cheon, S.-Y.; Roh, S.-S.; Lee, M.; An, H.-J. Chemopreventive Effect of Aster Glehni on Inflammation-Induced Colorectal Carcinogenesis in Mice. Nutrients 2018, 10, 202. [Google Scholar] [CrossRef]
- Nam, S.Y.; Kim, J.S.; Kim, J.M.; Lee, J.Y.; Kim, N.; Jung, H.C.; Song, I.S. DA-6034, a Derivative of Flavonoid, Prevents and Ameliorates Dextran Sulfate Sodium-Induced Colitis and Inhibits Colon Carcinogenesis. Exp. Biol. Med. 2008, 233, 180–191. [Google Scholar] [CrossRef]
- Zhao, Q.; Bi, Y.; Zhong, J.; Ren, Z.; Liu, Y.; Jia, J.; Yu, M.; Tan, Y.; Zhang, Q.; Yu, X. Pristimerin Suppresses Colorectal Cancer through Inhibiting Inflammatory Responses and Wnt/β-Catenin Signaling. Toxicol. Appl. Pharmacol. 2020, 386, 114813. [Google Scholar] [CrossRef]
- Wu, M.; Li, J.; An, Y.; Li, P.; Xiong, W.; Li, J.; Yan, D.; Wang, M.; Zhong, G. Chitooligosaccharides Prevents the Development of Colitis-Associated Colorectal Cancer by Modulating the Intestinal Microbiota and Mycobiota. Front. Microbiol. 2019, 10, 2101. [Google Scholar] [CrossRef]
- Moreira, T.G.; Horta, L.S.; Gomes-Santos, A.C.; Oliveira, R.P.; Queiroz, N.M.G.P.; Mangani, D.; Daniel, B.; Vieira, A.T.; Liu, S.; Rodrigues, A.M.; et al. CLA-Supplemented Diet Accelerates Experimental Colorectal Cancer by Inducing TGF-β-Producing Macrophages and T Cells. Mucosal Immunol. 2018, 12, 188–199. [Google Scholar] [CrossRef]
- Huo, X.; Liu, D.; Gao, L.; Li, L.; Cao, L. Flavonoids Extracted from Licorice Prevents Colitis-Associated Carcinogenesis in AOM/DSS Mouse Model. Int. J. Mol. Sci. 2016, 17, 1343. [Google Scholar] [CrossRef]
- Im, S.-A.; Kim, J.-W.; Kim, H.-S.; Park, C.-S.; Shin, E.; Do, S.-G.; Park, Y.I.; Lee, C.-K. Prevention of Azoxymethane/Dextran Sodium Sulfate-Induced Mouse Colon Carcinogenesis by Processed Aloe Vera Gel. Int. Immunopharmacol. 2016, 40, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Ki, H.-H.; Lee, J.-H.; Lee, H.-Y.; Lee, Y.-M.; Kim, D.-K. Triticum Aestivum Sprouts Extract Inhibits Azoymethane (AOM)/Dextran Sodium Sulfate (DSS)-Induced Colon Carcinogenesis in Mice. Nutr. Cancer 2018, 70, 928–937. [Google Scholar] [CrossRef]
- Kim, M.; Miyamoto, S.; Yasui, Y.; Oyama, T.; Murakami, A.; Tanaka, T. Zerumbone, a Tropical Ginger Sesquiterpene, Inhibits Colon and Lung Carcinogenesis in Mice. Int. J. Cancer 2009, 124, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Kohno, H.; Suzuki, R.; Curini, M.; Epifano, F.; Maltese, F.; Gonzales, S.P.; Tanaka, T. Dietary Administration with Prenyloxycoumarins, Auraptene and Collinin, Inhibits Colitis-Related Colon Carcinogenesis in Mice. Int. J. Cancer 2006, 118, 2936–2942. [Google Scholar] [CrossRef] [PubMed]
- Kohno, H.; Suzuki, R.; Yasui, Y.; Miyamoto, S.; Wakabayashi, K.; Tanaka, T. Ursodeoxycholic Acid versus Sulfasalazine in Colitis-Related Colon Carcinogenesis in Mice. Clin. Cancer Res. 2007, 13, 2519–2525. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.J.; Choi, Y.J.; Kim, N.; Nam, R.H.; Lee, S.; Lee, H.S.; Lee, H.-N.; Surh, Y.-J.; Lee, D.H. Açaí Berries Inhibit Colon Tumorigenesis in Azoxymethane/Dextran Sulfate Sodium-Treated Mice. Gut Liver 2017, 11, 243–252. [Google Scholar] [CrossRef]
- Terasaki, M.; Masaka, S.; Fukada, C.; Houzaki, M.; Endo, T.; Tanaka, T.; Maeda, H.; Miyashita, K.; Mutoh, M. Salivary Glycine Is a Significant Predictor for the Attenuation of Polyp and Tumor Microenvironment Formation by Fucoxanthin in AOM/DSS Mice. In Vivo 2019, 33, 365–374. [Google Scholar] [CrossRef]
- Kohno, H.; Suzuki, R.; Sugie, S.; Tanaka, T. Suppression of Colitis-Related Mouse Colon Carcinogenesis by a COX-2 Inhibitor and PPAR Ligands. BMC Cancer 2005, 5, 46. [Google Scholar] [CrossRef]
- Predes, D.; Oliveira, L.F.S.; Ferreira, L.S.S.; Maia, L.A.; Delou, J.M.A.; Faletti, A.; Oliveira, I.; Amado, N.G.; Reis, A.H.; Fraga, C.A.M.; et al. The Chalcone Lonchocarpin Inhibits Wnt/β-Catenin Signaling and Suppresses Colorectal Cancer Proliferation. Cancers 2019, 11, 1968. [Google Scholar] [CrossRef]
- Canene-Adams, K.; Sfanos, K.S.; Liang, C.-T.; Yegnasubramanian, S.; Nelson, W.G.; Brayton, C.; De Marzo, A.M. Dietary Chemoprevention of PhIP Induced Carcinogenesis in Male Fischer 344 Rats with Tomato and Broccoli. PLoS ONE 2013, 8, e79842. [Google Scholar] [CrossRef]
- Narushima, S.; Sakata, T.; Hioki, K.; Itoh, T.; Nomura, T.; Itoh, K. Inhibitory Effect of Yogurt on Aberrant Crypt Foci Formation in the Rat Colon and Colorectal Tumorigenesis in RasH2 Mice. Exp. Anim. 2010, 59, 487–494. [Google Scholar] [CrossRef]
- Carter, O.; Wang, R.; Dashwood, W.M.; Orner, G.A.; Fischer, K.A.; Löhr, C.V.; Pereira, C.B.; Bailey, G.S.; Williams, D.E.; Dashwood, R.H. Comparison of White Tea, Green Tea, Epigallocatechin-3-Gallate, and Caffeine as Inhibitors of PhIP-Induced Colonic Aberrant Crypts. Nutr. Cancer 2007, 58, 60–65. [Google Scholar] [CrossRef]
- Tang, M.X.; Ogawa, K.; Asamoto, M.; Chewonarin, T.; Suzuki, S.; Tanaka, T.; Shirai, T. Effects of Nobiletin on PhIP-Induced Prostate and Colon Carcinogenesis in F344 Rats. Nutr. Cancer 2011, 63, 227–233. [Google Scholar] [CrossRef]
- Santana-Rios, G.; Orner, G.A.; Xu, M.; Izquierdo-Pulido, M. Inhibition by White Tea of 2-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine-Induced Colonic Aberrant Crypts in the F344 Rat. Nutr. Cancer 2001, 41. [Google Scholar] [CrossRef]
- Hikosaka, A. Inhibitory Effects of Soy Isoflavones on Rat Prostate Carcinogenesis Induced by 2-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine (PhIP). Carcinogenesis 2003, 25, 381–387. [Google Scholar] [CrossRef]
- Guo, D.; Schut, H.A.; Davis, C.D.; Snyderwine, E.G.; Bailey, G.S.; Dashwood, R.H. Protection by Chlorophyllin and Indole-3-Carbinol against 2-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine (PhIP)-Induced DNA Adducts and Colonic Aberrant Crypts in the F344 Rat. Carcinogenesis 1995, 16, 2931–2937. [Google Scholar] [CrossRef]
- Hagiwara, A.; Boonyaphiphat, P.; Tanaka, H.; Kawabe, M.; Tamano, S.; Kaneko, H.; Matsui, M.; Hirose, M.; Ito, N.; Shirai, T. Organ-Dependent Modifying Effects of Caffeine, and Two Naturally Occurring Antioxidants Alpha-Tocopherol and n-Tritriacontane-16,18-Dione, on 2-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine (PhIP)-Induced Mammary and Colonic Carcinogenesis in Female F344 Rats. Jpn. J. Cancer Res. 1999, 90, 399–405. [Google Scholar] [CrossRef]
- Tan, W. Chemoprevention of 2-Amino-1-Methyl-6-Phenyli-Midazo [4,5-b] Pyridine-Induced Carcinogen-DNA Adducts by Chinese Cabbage in Rats. World J. Gastroenterol. 1999, 5, 138. [Google Scholar] [CrossRef]
- Yamada, Y.; Mori, H. Multistep Carcinogenesis of the Colon in Apc Min/+ Mouse. Cancer Sci. 2007, 98, 6–10. [Google Scholar] [CrossRef]
- Moser, A.; Gledhill, S.; Dove, W. Loss of Apc in Intestinal Adenomas from Min Mice. Cancer Res. 1994, 54, 5947–5952. [Google Scholar]
- Floyd, R.A.; Towner, R.A.; Wu, D.; Abbott, A.; Cranford, R.; Branch, D.; Guo, W.-X.; Foster, S.B.; Jones, I.; Alam, R.; et al. Anti-Cancer Activity of Nitrones in the APCMin/+ Model of Colorectal Cancer. Free Radic. Res. 2010, 44, 108–117. [Google Scholar] [CrossRef]
- Mutanen, M.; Pajari, A.-M.; Päivärinta, E.; Misikangas, M.; Rajakangas, J.; Marttinen, M.; Oikarinen, S. Berries as Chemopreventive Dietary Constituents—A Mechanistic Approach with the ApcMin/+ Mouse. Asia Pac. J. Clin. Nutr. 2008, 17 (Suppl. S1), 123–125. [Google Scholar]
- Swamy, M.V.; Patlolla, J.M.R.; Steele, V.E.; Kopelovich, L.; Reddy, B.S.; Rao, C.V. Chemoprevention of Familial Adenomatous Polyposis by Low Doses of Atorvastatin and Celecoxib Given Individually and in Combination to APCMin Mice. Cancer Res. 2006, 66, 7370–7377. [Google Scholar] [CrossRef]
- Ritland, S.R.; Gendler, S.J. Chemoprevention of Intestinal Adenomas in the ApcMin Mouse by Piroxicam: Kinetics, Strain Effects and Resistance to Chemosuppression. Carcinogenesis 1999, 20, 51–58. [Google Scholar] [CrossRef][Green Version]
- Fini, L.; Piazzi, G.; Daoud, Y.; Selgrad, M.; Maegawa, S.; Garcia, M.; Fogliano, V.; Romano, M.; Graziani, G.; Vitaglione, P.; et al. Chemoprevention of Intestinal Polyps in ApcMin/+ Mice Fed with Western or Balanced Diets by Drinking Annurca Apple Polyphenol Extract. Cancer Prev. Res. 2011, 4, 907–915. [Google Scholar] [CrossRef]
- Phutthaphadoong, S.; Yamada, Y.; Hirata, A.; Tomita, H.; Hara, A.; Limtrakul, P.; Iwasaki, T.; Kobayashi, H.; Mori, H. Chemopreventive Effect of Fermented Brown Rice and Rice Bran (FBRA) on the Inflammation-Related Colorectal Carcinogenesis in ApcMin/+ Mice. Oncol. Rep. 2010, 23, 53–59. [Google Scholar] [CrossRef][Green Version]
- Khor, T.O.; Hu, R.; Shen, G.; Jeong, W.-S.; Hebbar, V.; Chen, C.; Xu, C.; Nair, S.; Reddy, B.; Chada, K.; et al. Pharmacogenomics of Cancer Chemopreventive Isothiocyanate Compound Sulforaphane in the Intestinal Polyps of ApcMin/+ Mice. Biopharm. Drug Dispos. 2006, 27, 407–420. [Google Scholar] [CrossRef]
- Smith, D.L.H.; Keshavan, P.; Avissar, U.; Ahmed, K.; Zucker, S.D. Sodium Taurocholate Inhibits Intestinal Adenoma Formation in APCMin/+ Mice, Potentially through Activation of the Farnesoid X Receptor. Carcinogenesis 2010, 31, 1100–1109. [Google Scholar] [CrossRef]
- Tomimoto, A.; Endo, H.; Sugiyama, M.; Fujisawa, T.; Hosono, K.; Takahashi, H.; Nakajima, N.; Nagashima, Y.; Wada, K.; Nakagama, H.; et al. Metformin Suppresses Intestinal Polyp Growth in ApcMin/+ Mice. Cancer Sci. 2008, 99, 2136–2141. [Google Scholar] [CrossRef]
- Rajamanickam, S.; Velmurugan, B.; Kaur, M.; Singh, R.P.; Agarwal, R. Chemoprevention of Intestinal Tumorigenesis in APCmin/+ Mice by Silibinin. Cancer Res. 2010, 70, 2368–2378. [Google Scholar] [CrossRef]
- Barnes, C.J.; Lee, M. Chemoprevention of Spontaneous Intestinal Adenomas in the Adenomatous Polyposis Coli Min Mouse Model with Aspirin. Gastroenterology 1998, 114, 873–877. [Google Scholar] [CrossRef]
- Murphy, E.A.; Davis, J.M.; McClellan, J.L.; Gordon, B.T.; Carmichael, M.D. Curcumin’s Effect on Intestinal Inflammation and Tumorigenesis in the ApcMin/+ Mouse. J. Interferon Cytokine Res. 2011, 31, 219–226. [Google Scholar] [CrossRef]
- Bobe, G.; Wang, B.; Seeram, N.P.; Nair, M.G.; Bourquin, L.D. Dietary Anthocyanin-Rich Tart Cherry Extract Inhibits Intestinal Tumorigenesis in APCMin Mice Fed Suboptimal Levels of Sulindac. J. Agric. Food Chem. 2006, 54, 9322–9328. [Google Scholar] [CrossRef] [PubMed]
- Mehl, K.A.; Davis, J.M.; Clements, J.M.; Berger, F.G.; Pena, M.M.; Carson, J.A. Decreased Intestinal Polyp Multiplicity Is Related to Exercise Mode and Gender in ApcMin/+ Mice. J. Appl. Physiol. 2005, 98, 2219–2225. [Google Scholar] [CrossRef] [PubMed]
- Sale, S.; Tunstall, R.G.; Ruparelia, K.C.; Butler, P.C.; Potter, G.A.; Steward, W.P.; Gescher, A.J. Effects of the Potential Chemopreventive Agent DMU-135 on Adenoma Development in the ApcMin+ Mouse. Investig. New Drugs 2006, 24, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Cekanova, M.; McEntee, M.F.; Yoon, J.-H.; Fischer, S.M.; Renes, I.B.; Van Seuningen, I.; Baek, S.J. Peroxisome Proliferator-Activated Receptor Ligand MCC-555 Suppresses Intestinal Polyps in ApcMin/+ Mice via Extracellular Signal-Regulated Kinase and Peroxisome Proliferator-Activated Receptor-Dependent Pathways. Mol. Cancer Ther. 2008, 7, 2779–2787. [Google Scholar] [CrossRef]
- Sørensen, I.K.; Kristiansen, E.; Mortensen, A.; Nicolaisen, G.M.; Wijnands, J.A.H.; van Kranen, H.J.; van Kreijl, C.F. The Effect of Soy Isoflavones on the Development of Intestinal Neoplasia in ApcMin Mouse. Cancer Lett. 1998, 130, 217–225. [Google Scholar] [CrossRef]
- Fan, K.; Kurihara, N.; Abe, S.; Ho, C.-T.; Ghai, G.; Yang, K. Chemopreventive Effects of Orange Peel Extract (OPE) I. OPE Inhibits Intestinal Tumor Growth in ApcMin/+ Mice. J. Med. Food 2007, 10, 11–17. [Google Scholar] [CrossRef]
- Baltgalvis, K.A.; Berger, F.G.; Peña, M.M.O.; Davis, J.M.; Carson, J.A. Effect of Exercise on Biological Pathways in ApcMin/+ Mouse Intestinal Polyps. J. Appl. Physiol. 2008, 104, 1137–1143. [Google Scholar] [CrossRef]
- Mabley, J.G.; Pacher, P.; Bai, P.; Wallace, R.; Goonesekera, S.; Virag, L.; Southan, G.J.; Szabó, C. Suppression of Intestinal Polyposis in Apcmin/+ Mice by Targeting the Nitric Oxide or Poly(ADP-Ribose) Pathways. Mutat. Res. Fundam. Mol. Mech. Mutagenesis 2004, 548, 107–116. [Google Scholar] [CrossRef]
- Bose, M.; Hao, X.; Ju, J.; Husain, A.; Park, S.; Lambert, J.D.; Yang, C.S. Inhibition Of Tumorigenesis in ApcMin/+ Mice by a Combination of (–)-Epigallocatechin-3-Gallate and Fish Oil. J. Agric. Food Chem. 2007, 55, 7695–7700. [Google Scholar] [CrossRef]
- Williamson, S.L.H.; Kartheuser, A.; Coaker, J.; Kooshkghazi, M.D.; Fodde, R.; Burn, J.; Mathers, J.C. Intestinal Tumorigenesis in the Apc1638N Mouse Treated with Aspirin and Resistant Starch for up to 5 Months. Carcinogenesis 1999, 20, 805–810. [Google Scholar] [CrossRef][Green Version]
- Amos-Landgraf, J.M.; Kwong, L.N.; Kendziorski, C.M.; Reichelderfer, M.; Torrealba, J.; Weichert, J.; Haag, J.D.; Chen, K.-S.; Waller, J.L.; Gould, M.N.; et al. A Target-Selected Apc-Mutant Rat Kindred Enhances the Modeling of Familial Human Colon Cancer. Proc. Natl. Acad. Sci. USA 2007, 104, 4036–4041. [Google Scholar] [CrossRef]
- Edelmann, L.; Edelmann, W. Loss of DNA Mismatch Repair Function and Cancer Predisposition in the Mouse: Animal Models for Human Hereditary Nonpolyposis Colorectal Cancer. Am. J. Med Genet. 2004, 129C, 91–99. [Google Scholar] [CrossRef]
- Chang, Y.-J.; Hsu, W.-H.; Chang, C.-H.; Lan, K.-L.; Ting, G.; Lee, T.-W. Combined Therapeutic Efficacy of 188Re-Liposomes and Sorafenib in an Experimental Colorectal Cancer Liver Metastasis Model by Intrasplenic Injection of C26-Luc Murine Colon Cancer Cells. Mol. Clin. Oncol. 2014, 2, 380–384. [Google Scholar] [CrossRef]
- Dunnington, D.J.; Buscarino, C.; Gennaro, D.; Greig, R.; Poste, G. Characterization of an Animal Model of Metastatic Colon Carcinoma. Int. J. Cancer 1987, 39, 248–254. [Google Scholar] [CrossRef]
- Xie, X.; Briinnert, N.; Jensen, G.; Albrectsen, J.; Gotthardsen, B.; Rygaard, J. Comparative Studies between Nude and Scid Mice on the Growth and Metastatic Behavior of Xenografted Human Tumors. Clin. Exp. Metastasis 1992, 10, 201–210. [Google Scholar] [CrossRef]
- Mittal, V.K.; Bhullar, J.S.; Jayant, K. Animal models of human colorectal cancer: Current status, uses and limitations. World J. Gastroenterol. 2015, 21, 11854–11861. [Google Scholar] [CrossRef]
- Goldrosen, M.H. Murine Colon Adenocarcinoma. Immunobiology of Metastases. Cancer 1980, 45, 1223–1228. [Google Scholar] [CrossRef]
- Donigan, M.; Loh, B.D.; Norcross, L.S.; Li, S.; Williamson, P.R.; DeJesus, S.; Ferrara, A.; Gallagher, J.T.; Baker, C.H. A Metastatic Colon Cancer Model Using Nonoperative Transanal Rectal Injection. Surg. Endosc. 2010, 24, 642–647. [Google Scholar] [CrossRef]
- Tao, L.; Yang, J.-K.; Gu, Y.; Zhou, X.; Zhao, A.-G.; Zheng, J.; Zhu, Y.-J. Weichang’an and 5-Fluorouracil Suppresses Colorectal Cancer in a Mouse Model. World J. Gastroenterol. 2015, 21, 1125–1139. [Google Scholar] [CrossRef]
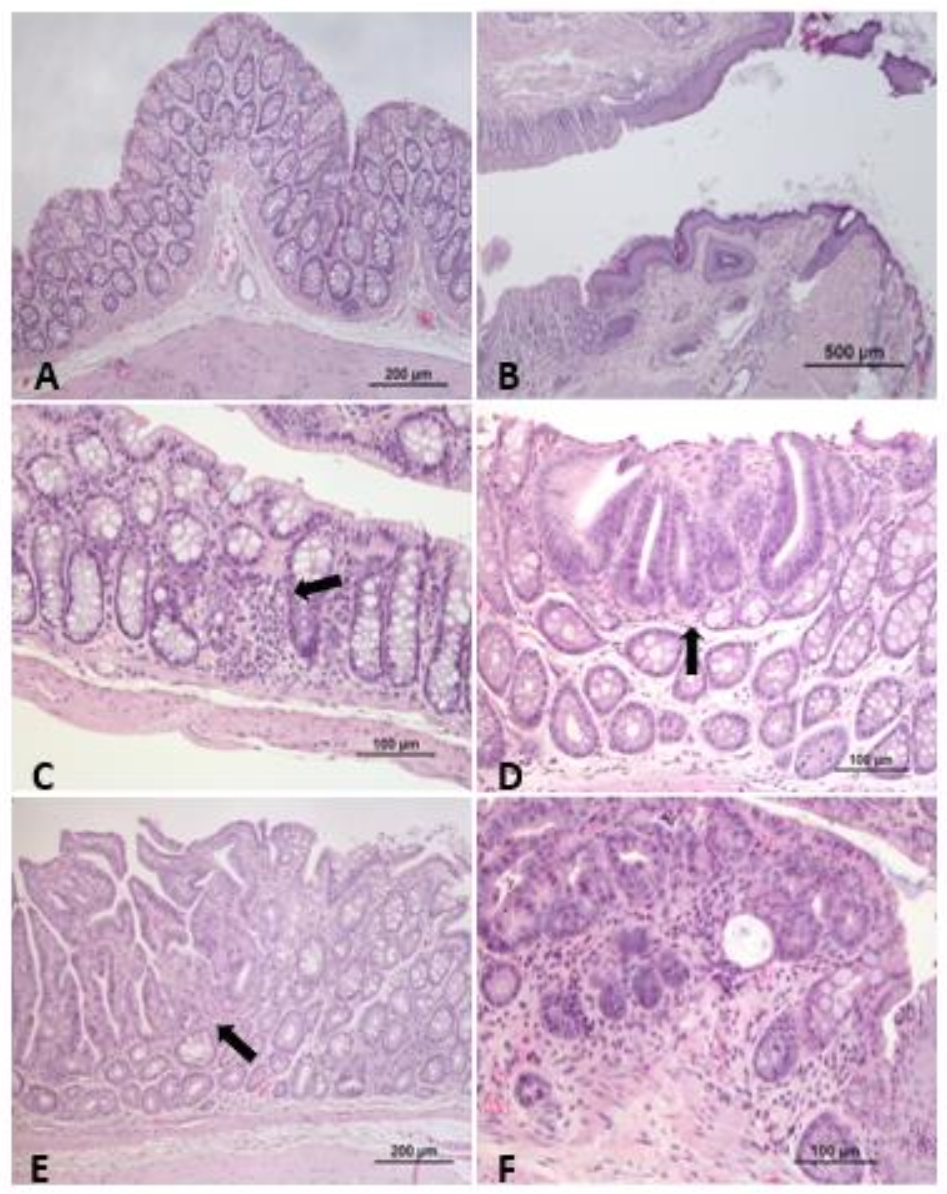
| Animal Strain and Gender | Carcinogenic Administration Route | Drugs or Compounds Evaluated (Classification) | Dose/Treatment | Therapeutic Effects (Ref) |
|---|---|---|---|---|
| F344 male rats | s.c. 100 mg/kg b.w. | Copper-zinc (CU), manganese (Mn), and iron (Fe) | p.o. (0.8 or 5.1 µg CU/g diet; 0.6 or 17 µg Mn/g diet and 37 or 140 µg Fe/g diet) 3.5 wks before DMAB and for 8 wks | Increased neoplastic lesions by low doses of copper and manganese relative to iron [25] |
| s.c. 100 mg/kg b.w. 1/wk for 2 wks | Selenium (nutritionally essential trace element) | p.o. (0, 0.1 or 2.0 mg selenium/kg diet as selenite, selenate or selenomethionine) 4 wks before DAMBP for 12 wks | Dietary administration of selenium in the form of the inorganic salts selenite and selenate reduced colon ACF [26] | |
| s.c. 100 mg/kg b.w. | Celecoxib (selective cyclooxygenase-2 inhibitor) | Diet supplemented (0, 500, 1000, or 1500 ppm celecoxib) 2 wks before DMABP and for 2 days | Chemopreventive effect for colorectal cancer in a dose-response manner [27] | |
| Gavage 50 or 5 mg/kg b.w. 1/wk for 4 wks | Acetaminophen | Diet supplemented (1000 ppm) 2 wks before DMAB and for 6 wks | Protective effect on the development of colorectal carcinogenesis [28] |
| Animal Strain and Gender | Carcinogenic Administration Route | Drugs or Compounds Evaluated (Classification) | Dose/Treatment | Therapeutic Effects [Ref] |
|---|---|---|---|---|
| F344/NSlc female rats | i.r. 2 mg 3x/wk for 3 wks | Ursodeoxycholic acid (UDCA) and 5-aminosalicylic acid (5-ASA) (non-steroid anti-inflammatory drug) | p.o. (0%, 0.11 or 0.02%) for 30 wks | Inhibited colorectal cancer development [29] |
| Sprague Dawley female rats | i.r. 10 mg/Kg 3x/wk for 4 wks | Omega 3 polyunsaturated fatty acid (Omega-3PUFA) | i.g. (2 g/kg) daily for 4 wks | Attenuated CRC by blocking PI3K/AKT/Bcl-2 [30] |
| F344 male rats | i.r. 2 mg/rat 3x/wk for 4 wks | 4-[3,5-Bis(trimethylsilyl)benzamido]benzoic acid (TAC-101) (retinobenzoic acid derivative) | p.o. (0.8 or 8 mg/kg for 1 or 4 wks) for 20 wks | Inhibited colorectal cancer development [31] |
| i.r. 4 mg on days 1 and 4 | Calcium and cholic (bile acid) | d.t. (0.2% cholic acid or 1.6% calcium) for 28 wks | Increased colorectal tumor development by bile acid and no protective effect of calcium [32] |
| Animal Strain/Gender | Drugs or Compounds Evaluated (Classification) | Dose/Treatment | Therapeutic Effects (Ref) |
|---|---|---|---|
| Min mice/n.d. | α-phenyl-tert-butyl nitrone (PBN) and 4-hydroxyl-PBN (4-O-PBN) (nitrones) | d.w. (100 ng/kg/day PBN or 4-O-PBN) for 3–4 months | Anti-cancer activity of PBN more significant than 4-O-PBN [123] |
| Bilberry (Vaccinium myrtillus), lingonberry (Vaccinium vitis-idaea), cloudberry (Rubus chamaemorus), cloudberry seeds or cloudberry pulp, or pure ellagic acid | p.o. (1564 mg/kg of each) for 10 wks. | Chemopreventive potential [124] | |
| Atorvastatin (hydroxy-3- methylglutaryl CoA reductase (HMGR) inhibitor) and celecoxib (cyclooxygenase-2 (COX-2) inhibitors) | p.o. (0 or 100 ppm atorvastatin or 300 celecoxib alone or in combination) for 80 days | Inhibited intestinal tumorigenesis by atorvastatin and increased chemopreventive activity in combination with celecoxib [125] | |
| Piroxicam (a non-steroidal anti-inflammatory drug) | p.o. (200 ppm piroxicam) for 100 or 200 days of rat age | Reduced tumor multiplicity [126] | |
| Annurca Apple polyphenol extract | d.w. (60 µmol/L) in combination with a western diet or balanced diet for 12 wks | Chemopreventive potential [127] | |
| Fermented brown rice and rice bran (FBRA) | Exp.1: diet supplemented (5 or 10% FBRA) for 20 wks; Exp2: diet supplemented (5 or 10% FBRA and 2% DSS in d.w. for 1 wk) for 12 wks; Exp3: diet supplemented (10% FBRA and 1.5% DSS in d.w. for 1 wk) for 7 wks | No effect on tumor development by FBRA alone but in combination with DSS suppressed the multiplicity of colon tumors [128] | |
| Sulforaphane (SFN) (isothiocyanate) | Diet supplemented (600 ppm SFN) for 1, 3, or 5 days | Chemopreventive potential [129] | |
| Bilirubin, bovine serum albumin (BSA) and sodium taurocholate (NaTC) | p.o (0.5 mM BSA alone or in combination with 0.25 mM bilirubin or 5 mM NaTC) for 8 wks | Reduced intestinal adenomas by NaTC [130] | |
| Metformin (biguanide derivative) | p.o. (250 mg/kg/day) for 10 wks | Chemopreventive potential [131] | |
| Silibinin | Gavage (750 mg/kg b.w.) for 5 days a wk for 13 wks | Chemopreventive potential [132] | |
| Min mice/female and male | Aspirin | Diet supplemented (250 or 500 ppm) for 7 wks | Chemopreventive potential [133] |
| Curcumin | Diet supplemented (2% curcumin) from 4 to 18 wks of age | Chemopreventive potential [134] | |
| Anthocyanin-rich tart cherry extract and sulindac (a nonsteroidal anti-inflammatory drug) | p.o. (0, 375, 750, 1500 or 3000 mg anthocyanin-rich tar cherry extract/kg if diet with 100 mg sulindac/kg diet) for 19 wks | The combination of both compounds had a more protective effect than compounds alone [135] | |
| Physical activity | t.r. (18 m/min, 60 min/day, 6 days/wk or voluntary wheel running) for 9 wks | Reduced number and size of intestinal polyps, dependent on exercise mode and gender [136] | |
| DMU-135 (3,4-Methylenedioxy-3,4,5 -trimethoxy chalcone) (anticancer prodrug) | Diet supplemented (0.2% w:w) from 4–18 wks | Chemopreventive activity [137] | |
| MCC-555 (peroxisome proliferator-activated receptor (PPAR) ligand) | Gavage (30 mg/kg/day 5 days/wk) for 4 wks | Suppressed activity [138] | |
| Soy isoflavones | Diet supplemented (low-isoflavone: 11.5 genistein, 2.3 daidzein and 2.3 mg of glycitein/kg diet, rich isoflavone diet: 280.6 genistein, 147.2 daidzein and 48.3 mg of glycitein/kg diet) for 107 days | No inhibition of colorectal tumor development [139] | |
| Min mice/male | Orange peel extract (OPE) | Diet supplemented (0.25 or 0.5% OPE) for 9 wks | Inhibited colorectal tumorigenesis [140] |
| Physical activity | t.r.(18 m/min, 60 min, 6 days/wk, 5% grade) for 9 wks | Reduced the overall tumor burden (size and number) [141] | |
| Guanidinoethyldisulfide (GED) [14,15,16,17], peroxynitrite decomposition catalyst, FP 15 and poly(ADPribose) synthetase (PARP) inhibitor, N-(6-oxo-5,6-dihydrophenanthridin- 2-yl)-N, N-dimethylacetamide hydrochloride (PJ 34) (specific inhibitors of inducible nitric oxide synthase) | Gavage (10 or 30 mg/kg/day GED, 1 or 3 mg/kg/day FP15 and 3 or 10 mg/kg/day PJ34) twice a day from 5 wks of age until 12 wks | Chemopreventive activity of all compounds [142] | |
| Min mice/female | (–)-epigallocatechin-3-gallate (EGCG) and fish oil | Diet supplemented (0.16% EGCG alone or in combination with high-fat fish oil diet, 20% w:w) for 9 wks | Inhibited tumor multiplicity by a combination of low doses of EGCG and fish oil [143] |
| Apc1638N mice/male and female | Aspirin and α-amylase resistant starch (RS) | Diet supplemented (125 g/kg diet RS or 0.3 g/kg aspirin alone or in combination) from 6 wks | The combination of two compounds showed more preventive activity than compounds alone [144] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nascimento-Gonçalves, E.; Mendes, B.A.L.; Silva-Reis, R.; Faustino-Rocha, A.I.; Gama, A.; Oliveira, P.A. Animal Models of Colorectal Cancer: From Spontaneous to Genetically Engineered Models and Their Applications. Vet. Sci. 2021, 8, 59. https://doi.org/10.3390/vetsci8040059
Nascimento-Gonçalves E, Mendes BAL, Silva-Reis R, Faustino-Rocha AI, Gama A, Oliveira PA. Animal Models of Colorectal Cancer: From Spontaneous to Genetically Engineered Models and Their Applications. Veterinary Sciences. 2021; 8(4):59. https://doi.org/10.3390/vetsci8040059
Chicago/Turabian StyleNascimento-Gonçalves, Elisabete, Bruno A.L. Mendes, Rita Silva-Reis, Ana I. Faustino-Rocha, Adelina Gama, and Paula A. Oliveira. 2021. "Animal Models of Colorectal Cancer: From Spontaneous to Genetically Engineered Models and Their Applications" Veterinary Sciences 8, no. 4: 59. https://doi.org/10.3390/vetsci8040059
APA StyleNascimento-Gonçalves, E., Mendes, B. A. L., Silva-Reis, R., Faustino-Rocha, A. I., Gama, A., & Oliveira, P. A. (2021). Animal Models of Colorectal Cancer: From Spontaneous to Genetically Engineered Models and Their Applications. Veterinary Sciences, 8(4), 59. https://doi.org/10.3390/vetsci8040059








