Diastolic and Systolic Longitudinal Myocardial Velocities of Healthy Racing Pigeons (Columba livia f. domestica) Measured by Tissue Doppler Imaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Animals
2.2. Doppler Sonographic Examination
2.3. Statistical Analysis
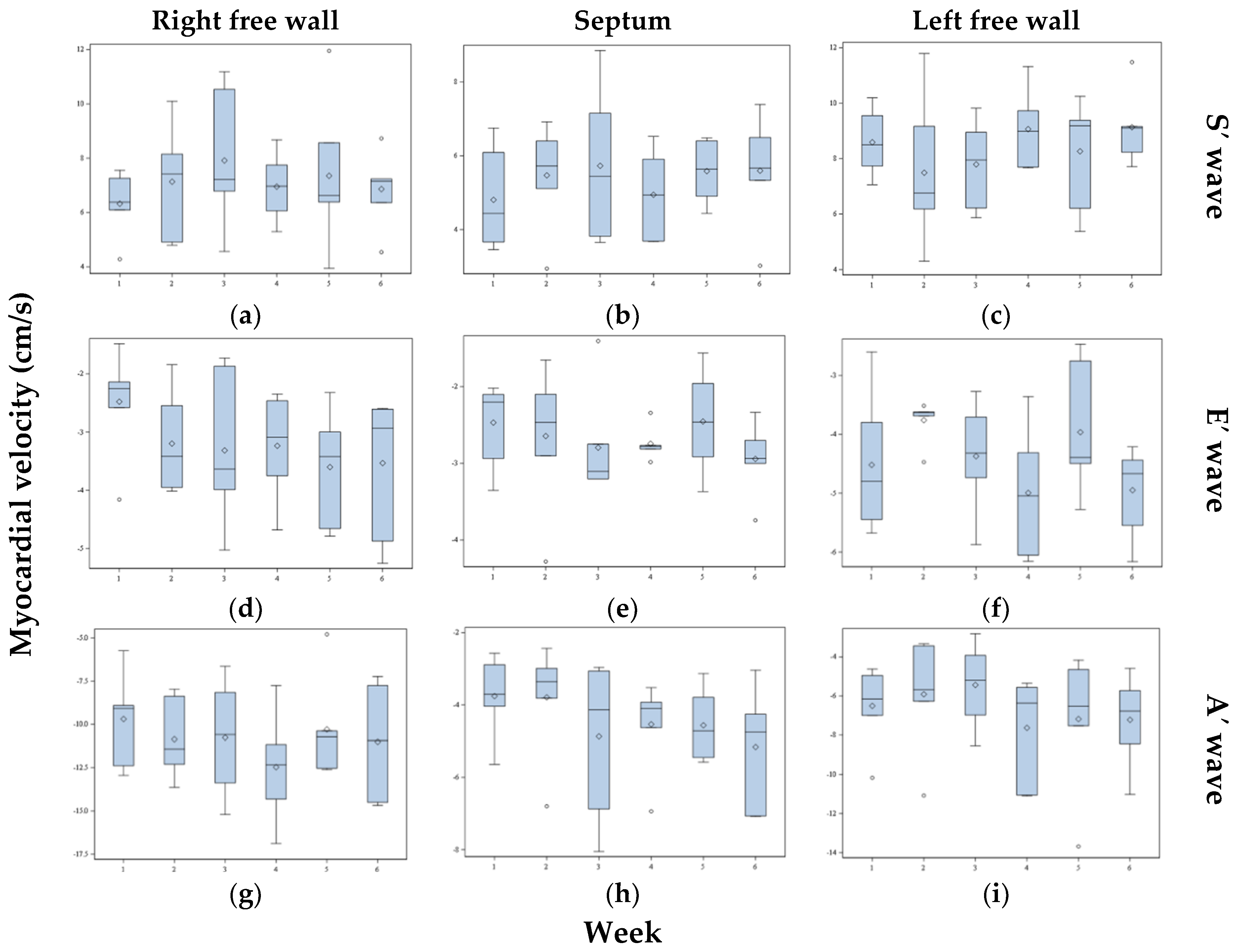
3. Results
3.1. Myocardial Velocities
3.2. The Influence of Heart Rate
3.3. Influence of Age, Sex, and Body Weight
3.4. Day-Dependent Variability in Myocardial Velocity
3.5. Influence of the Sonographic Angle and the Sonographic Window on the Myocardial Velocities
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pees, M.; Krautwald-Junghanns, M.-E.; Straub, J. Evaluation and treating the cardio-vascular system. In Clinical Avian Medicine; Harrison, G.J., Lightfoot, T., Eds.; Spix Publishing: Palm Beach, FL, USA, 2006; pp. 379–394. [Google Scholar]
- Pees, M.; Krautwald-Junghanns, M.-E. Cardiovascular physiology and diseases of pet birds. Vet. Clin. N. Am. Exot. Anim. Pract. 2009, 12, 81–97. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, B.C.; Beaufrère, H. Cardiology. In Current Therapy in Avian Medicine and Surgery, 1st ed.; Speer, B.L., Ed.; ELSVIER: St Louis Missouri, MO, USA, 2016; pp. 252–328. [Google Scholar]
- Boskovic, M.; Krautwald-Junghanns, M.-E.; Failing, K.; Schneider, M. Möglichkeiten und Grenzen echokardiographischer Untersuchungen bei Tag- und Nachtgreifvögeln (Accipitriformes, Falconiformes, Strigiformes). Tierarztl. Prax. 1999, 27, 334–341. [Google Scholar]
- Pees, M. Echokardiographische Untersuchungen bei Psittaciformes unter Besonderer Berücksichtigung des Kongo-Graupapageis (Psittacus erithacus erithacus). Ph.D. Thesis, University of Leipzig, Leipzig, Germany, 2001. [Google Scholar]
- Pees, M.; Straub, J.; Krautwald-Junghanns, M.-E. Echocardiographic examinations of 60 African grey parrots and 30 other psittacine birds. Vet. Rec. 2004, 155, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Straub, J.; Pees, M.; Schumacher, J.; Gompf, R.E.; Basade, H.; Kautwald-Junghanns, M.-E. Dopplerechocardiography in birds. In Proceedings of the 6th Main Conference of the European Association of Avian Veterinarians, Munich, Germany, 8–10 March 2001; pp. 92–94. [Google Scholar]
- Straub, J.; Forbes, N.A.; Thielebein, J.; Pees, M.; Krautwald-Junghanns, M.-E. The effects of isoflurane anaesthesia on some Doppler-derived cardiac parameters in the Common Buzzard (Buteo buteo). Vet. J. 2003, 166, 273–276. [Google Scholar] [CrossRef]
- Legler, M.; Koy, L.; Kummerfeld, N.; Fehr, M. Evaluation of blood flow velocity in the ascending aorta and pulmonary artery of clinically healthy racing pigeons (Columba livia f. dom.) by pulsed-wave Doppler echocardiography. Wiener Tierärztliche Mon. 2019, 106, 179–187. [Google Scholar]
- Legler, M.; Koy, L.; Kummerfeld, N.; Fehr, M. The influence of anesthesia with isoflurane on the pulmonary and aortic blood flow of Racing Pigeons (Columba livia f. domestica) measured by pulsed wave Doppler echocardiography. Open Vet. J. 2019, 9, 18–26. [Google Scholar] [CrossRef]
- Legler, M.; Koy, L.; Kummerfeld, N.; Fehr, M. Differences between the filling velocities of the left and right heart ventricle in racing pigeons (Columba livia f. domestica) and the influence of anesthesia with isoflurane. Vet. Sci. 2019, 6, 79. [Google Scholar] [CrossRef]
- Legler, M.; Koy, L.; Kummerfeld, N.; Fehr, M. Color Flow Doppler echocardiography in healthy Racing Pigeons (Columba livia f. domestica) and the evidence of physiological blood flow vortex formations. Vet. Sci. 2020, 7, 60. [Google Scholar] [CrossRef]
- Pees, M.; Straub, J.; Schumacher, J.; Gompf, R.; Krautwald-Junghanns, M.-E. Pilot study for normal colour-flow and pulsed-wave spectral Doppler echocardiography in Hispaniolan amazons (Amazona ventralis) and blue-fronted amazons (Amazona a. aestiva). Dtsch. Tierärztliche Wochenschr. 2005, 112, 39–43. [Google Scholar]
- Marwick, T.H.; Yu, C.-M.; Sun, J.P. Myocardial Imaging: Tissue Doppler and Speckle Tracking, 1st ed.; Blackwell Publishing: Oxford, UK, 2007. [Google Scholar]
- Killich, M.; Hartmann, K.; Wess, G. Messung der myokardialen Geschwindigkeit mittels Gewebe-Doppler beim gesunden Hund. Tierärztl. Prax. 2011, 39, 69–78. [Google Scholar] [CrossRef]
- Serres, F.; Chetboul, V.; Gouni, V.; Tissier, R.; Sampedrano, C.; Pouchelon, J.L. Diagnostic value of echo-Doppler and tissue Doppler imaging in dogs with pulmonary arterial hypertension. J. Vet. Intern. Med. 2007, 21, 1280–1289. [Google Scholar] [CrossRef] [PubMed]
- Chetboul, V. Tissue Doppler Imaging: A promising technique for quantifying regional myocardial function. J. Vet. Cardiol. 2002, 4, 7–12. [Google Scholar] [CrossRef]
- Nicolle, A.P.; Sampedrano, C.; Fontaine, J.J.; Tessier-Vetzel, D.; Goumi, V.; Pelligand, L.; Pouchelon, J.L.; Chetboul, V. Longitudinal left ventricular myocardial dysfunction assessed by 2D colour tissue Doppler imaging in a dog with systemic hypertension and severe arteriosclerosis. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2005, 52, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Wagner, T. Tissue-Doppler-Imaging (TDI): Erstellung von Referenzwerten für Tissue Velocity, Strain und Strain-Rate bei der Katze. Ph.D. Thesis, Ludwig-Maximilians-Universität München, München, Germany, 2005. [Google Scholar]
- Ho, C.Y.; Solomon, S.D. A clinician‘s guide to tissue Doppler imaging. Circulation 2006, 113, e396–e398. [Google Scholar] [CrossRef] [PubMed]
- Sampedrano, C.; Chetboul, V.; Gouni, V.; Nicolle, A.P.; Pouchelon, J.L.; Tissier, R. Systolic and diastolic myocardial dysfunction in cats with hypertrophic cardiomyopathy or systemic hypertension. J. Vet. Int. Med. 2006, 20, 1106–1115. [Google Scholar] [CrossRef]
- Chetboul, V.; Sampedrano, C.; Blot, S.; Thibaud, J.L.; Escriou, C.; Tissier, R.; Retortillo, J.L.; Pouchelon, J.L. Tissue Doppler assessment of diastolic and systolic alterations of radial and longitudinal left ventricular motions in Golden Retrievers during the preclinical phase of cardiomyopathy associated with muscular dystrophy. Am. J. Vet. Res. 2004, 65, 1335–1341. [Google Scholar] [CrossRef]
- Schulz, U.; Krautwald-Junghanns, M.E.; Pees, M. Use of the spectral Doppler Ultrasonography for the diagnosis of atherosclerosis in African Grey Parrots. In Proceedings of the 2nd International Conference on Avian Herpethological and Exotic Mammal Medicine, Paris, France, 18–23 April 2015; p. 271. [Google Scholar]
- Fudge, A.M. Laboratory Medicine Avian and Exotic Pets; W.B. Saunders Company: Philadelphia, PA, USA, 2000; p. 377. [Google Scholar]
- Lumeij, J.T.; Ritchie, B.W. Cardiology. In Avian Medicine, Principles and Application; Ritchie, B.W., Harrison, G.J., Harrison, L.R., Eds.; HBD Intl Inc.: Brentwood, TN, USA, 1994; pp. 694–722. [Google Scholar]
- Lumeij, J.T.; Stokhof, A.A. Electrocardiogram of the Racing Pigeon (Columba livia forma domestica). Res. Vet. Sci. 1985, 38, 275–278. [Google Scholar] [CrossRef]
- Murcia, L.M.M.; Bernal, L.J.; Montes, A.M.; Ayala, I. The normal electrocardiogram of the unanaesthetized competition ‘Spanish Pouler’ pigeon (Columba livia gutturosa). J. Vet. Med. A Physiol. Pathol. Clin. Med. 2005, 52, 347–349. [Google Scholar] [CrossRef]
- Riedel, U. Die Ultraschalluntersuchung des Vogels am Beispiel der Brieftaube (Columba Livia Forma Domestica) mit Hilfe der Schnittbildechokardiographie. Ph.D. Thesis, Justus-Liebig-Universität Gießen, Giessen, Germany, 1995. [Google Scholar]
- Schulz, M. Morphologische und Funktionelle Messungen am Herzen von Brieftauben (Columba livia forma domestica) mit Hilfe der Schnittbildechokardiographie. Ph.D. Thesis, Justus-Liebig-Universität Gießen, Giessen, Germany, 1995. [Google Scholar]
- Masoudifard, M.; Bidgoli, V.R.; Madani, S.A.; Vajhi, A.; Davoodipoor, S.; Vali, Y. Normal echocardiographic findings in healthy pigeons. Iran. J. Vet. Surg. 2016, 11, 7–13. [Google Scholar]
- Waibl, H.; Sinowatz, F. Kreislaufapparat und Lymphatisches System. In Lehrbuch der Anatomie der Haustiere, Band V, Anatomie der Vögel, 2nd ed.; Schummer, A., Vollmerhaus, B., Eds.; Verlag Paul Parey: Berlin/Hamburg, Germany, 1992; pp. 282–330. [Google Scholar]
- Bezuidenhout, A.J. The valva atrioventricularis dextra of the avian heart. Anat. Histol. Embryol. 1983, 12, 104–108. [Google Scholar] [CrossRef]
- Codreanu, I.; Robson, M.D.; Golding, S.J.; Jung, B.A.; Clarke, K.; Holloway, C.J. Longitudinally and circumferentially directed movements of the left ventricle studied by cardiovascular magnetic resonance phase contrast velocity mapping. J. Cardiovasc. Magn. Reson. 2010, 12, 48. [Google Scholar] [CrossRef] [PubMed]
- Kovács, A.; Lakatos, B.; Tokodi, M.; Merkely, B. Right ventricular mechanical pattern in health and disease: Beyond longitudinal shortening. Heart. Fail. Rev. 2019, 24, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Tadjalli, M.; Ghazi, S.R.; Parto, P. Gross anatomy of the heart in Ostrich. Iran. J. Vet. Res. 2009, 10, 21–27. [Google Scholar]
- Hummel, G. Anatomie und Physiologie der Vögel; Verlag Eugen Ulmer GmbH & Co.: Stuttgart, Germany, 2000; pp. 83–103. [Google Scholar]
- Dzialowski, E.M.; Crossley, D.A. The Cardiovascular System. In Sturkie’s Avian Physiology, 6th ed.; Scanes, C.G., Ed.; Elsevier: London, UK, 2015; pp. 193–299. [Google Scholar]
- Chetboul, V.; Sampedrano, C.; Gouni, V.; Concordet, D.; Lamour, T.; Ginesta, J.; Nicolle, A.P.; Pouchelon, J.L.; Lefebvre, H.P. Quantitative assessment of regional right ventricular myocardial velocities in awake dogs by Doppler tissue imaging: Repeatability, reproducibility, effect of body weight and breed, and comparison with left ventricular myocardial velocities. J. Vet. Intern. Med. 2005, 19, 837–844. [Google Scholar] [CrossRef]
- Schmidt, R.E.; Reavill, D.R.; Phalen, D.N. Pathology of Pet and Aviary Birds, 2nd ed.; Wiley Blackwell: Iowa, IA, USA, 2015; pp. 1–19. [Google Scholar]
- Legler, M.; Kummerfeld, N. Tissue Doppler imaging (TDI): A new technique in avian cardiology. In Proceedings of the 1st International Conference on Avian, Herpetological and Exotic Mammal Medicine, Wiesbaden, Germany, 20–26 April 2013; VVB LAUFERSWEILER Verlag: Giessen, Germany, 2013; pp. 491–493. [Google Scholar]
- Chetboul, V.; Serres, F.; Gouni, V.; Tissier, R.; Pouchelon, J.L. Noninvasive assessment of systolic left ventricular torsion by 2-dimensional speckle tracking imaging in the awake dog: Repeatability, reproducibility, and comparison with tissue Doppler imaging variables. J. Vet. Intern. Med. 2008, 22, 342–350. [Google Scholar] [CrossRef]
- Legler, M.; Koy, L.; Fehr, M.; Kummerfeld, N. Untersuchungen zur Myocardbewegung und –geschwindigkeiten bei Vögeln mit Hilfe der Gewebedoppler-Echokadiographie. In Proceedings of the Tagungsband der 2. Jahrestagung der DVG-Fachgruppe Zier-, Zoo- und Wildvögel, Reptilien, Amphibien & Fische, Giessen, Germany, 14–16 September 2016; Verlag der DVG Service GmbH: Giessen, Germany, 2016; p. 9. [Google Scholar]
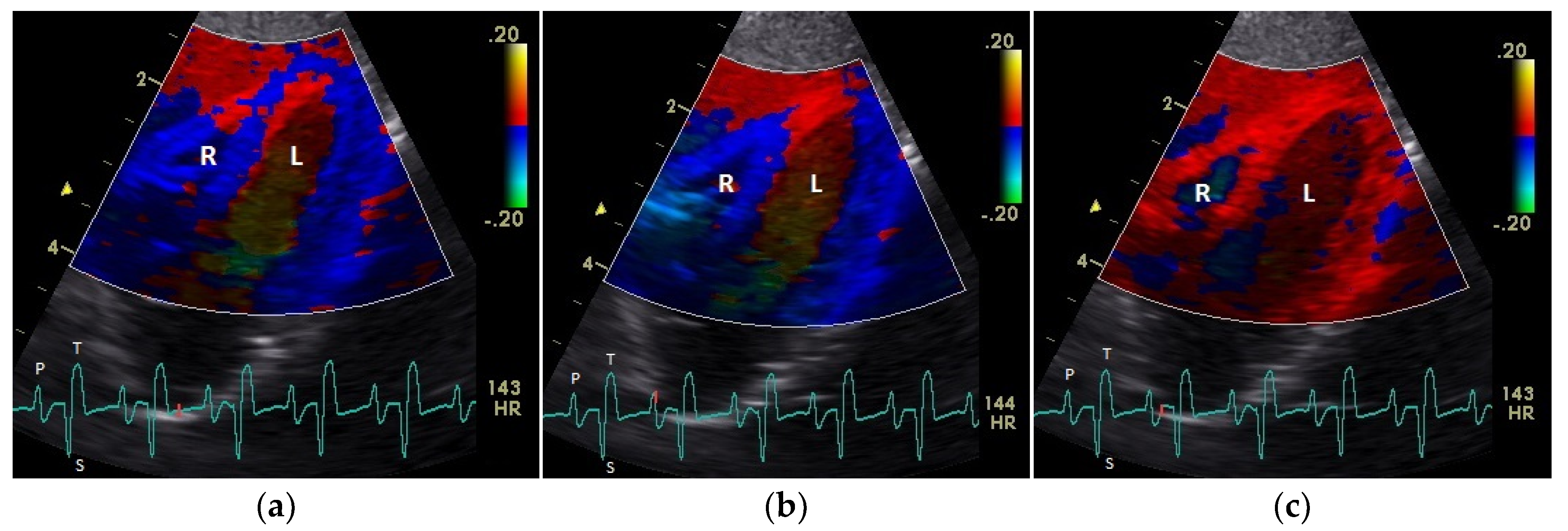
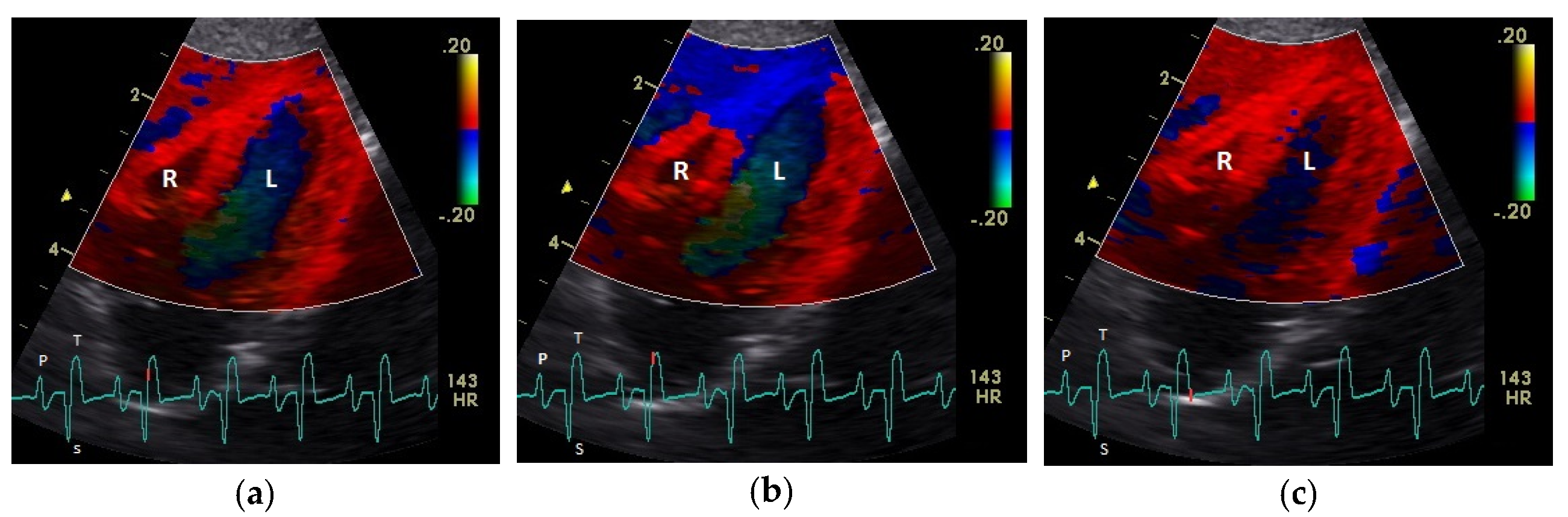
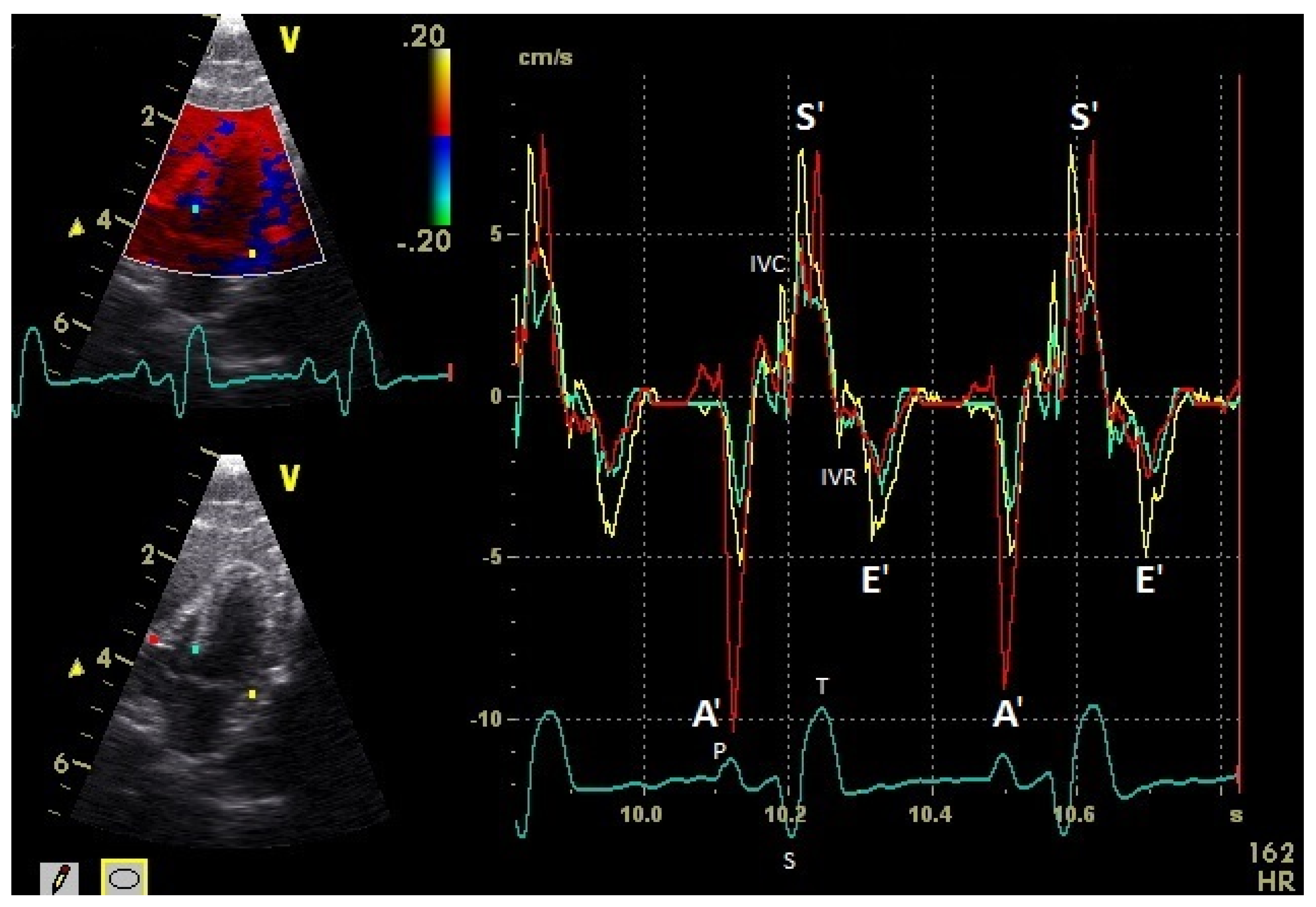
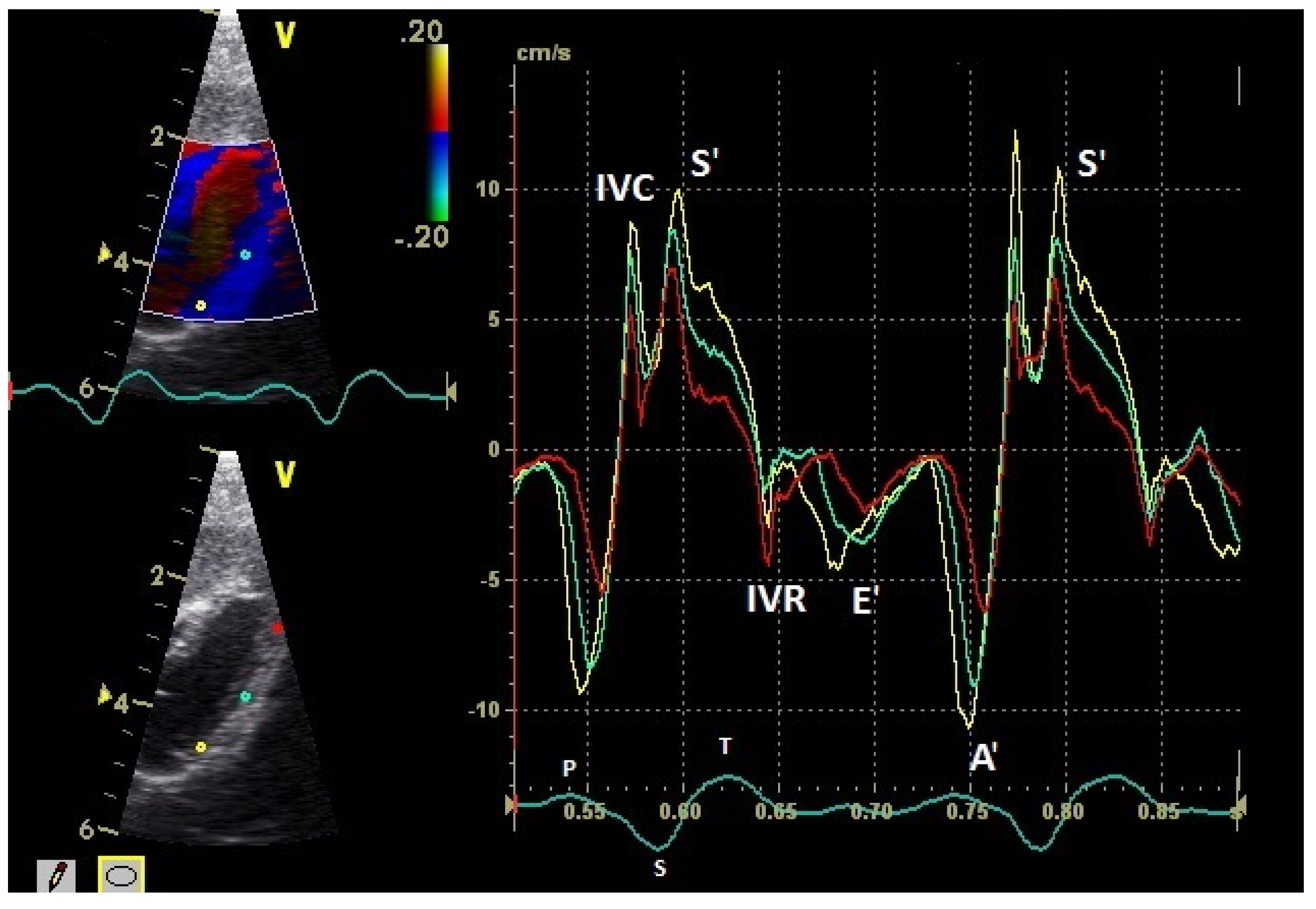
| Myocardium | TDI | Basal (cm/s) | Middle (cm/s) | Apical (cm/s) | Heart Rate (bpm) | Sonographic Angle |
|---|---|---|---|---|---|---|
| Septum 1 | S’ wave | 5.4 ± 1.7; 4.7; 3.1–12.4 | 4.5 ± 1.3; 4.4; 2.7–8.0 | 3.5 ± 1.4; 3.3; 1.2–7.1 | 206.3 ± 40.8 | 18.6 ± 11.6 |
| E’ wave | −2.4 ± 0.6; −2.3; −1.1–−3.7 | −1.9 ± 0.5; −1.8; −0.9–−3.4 | −1.4 ± 0.5; −1.4; −0.4–−2.5 | |||
| A’ wave | −3.9 ± 1.4; −3.7; −2.0–−7.4 | −3.6 ± 1.4; −3.3; −1.7–−6.7 | −2.7 ± 1.2; −2.5; −1.2–−5.2 | |||
| Left free wall 1 | S’ wave | 8.7 ± 1.9; 8.7; 3.7–12.3 | 7.4 ± 1.9; 7.2; 3.4–11.8 | 5.4 ± 1.9; 5.2; 1.8–9.9 | 210.5 ± 33.9 | 26.3 ± 7.5 |
| E’ wave | −4.4 ± 1.3; −4.3; −1.7–−7.0 | −3.4 ± 1.0; −3.3; −1.4–−6.1 | −2.3 ± 0.7; −2.3; −1.1–−4.0 | |||
| A’ wave | –6.7 ± 3.0; –8.7; −2.3–−16.3 | −5.9 ± 3.2; −5.1; −2.2–−18.5 | −3.8 ± 2.5; −3.1; −0.6–−13.4 | |||
| Right free wall 2 | S’ wave | 8.9 ± 1.9; 8.7; 5.4–12.6 | 7.2 ± 1.3; 6.9; 4.2–9.6 | 4.8 ± 1.2; 5.0; 1.8–7.2 | 200.1 ± 36.7 | 38.9 ± 7.9 |
| E’ wave | −4.5 ± 1.9; −4.3; −2.3–−14.5 | −3.7 ± 1.7; −3.6; −1.2–−11.5 | −2.8 ± 0.8; −2.7; −1.2–−5.0 | |||
| A’ wave | −8.8 ± 2.6; −8.7; −4.4–−12.7 | −7.2 ± 2.5; −7.2; −3.7–−11.3 | −4.4 ± 1.8; −4.4; −0.9–−9.6 | |||
| Right free wall 1 | S’ wave | 6.1 ± 2.11; 5.8; 2.7–12.6 | 5.0 ± 1.8; 4.6; 2.0–9.4 | 3.7 ± 1.6; 1.3; 1.3–8.2 | 206.9 ± 37.4 | 48.0 ± 11.0 |
| E’ wave | −2.6 ± 0.7; −2.6; −0.7–−3.8 | −2.2 ± 0.8; −2.2; −0.7–−3.8 | −1.7 ± 0.7; −1.7; −0.5–−3.0 | |||
| A’ wave | −7.4 ± 2.9; –6.6; −3.2–−15.8 | −5.7 ± 2.6; −4.8; −2.4–−11.8 | −3.2 ± 2.2; −2.3; −0.7–−9.8 |
| Measuring Point | Early (E Wave) Diastolic Velocities | Late (A Wave) Diastolic Velocities | Systolic (S Wave) Velocities |
|---|---|---|---|
| Left atrioventricular valve 1 | 0.37 ± 0.06 (0.37; 0.25–0.53) | 0.57 ± 0.20 (0.57; 0.24–0.96) | |
| Aorta 2 | 1.19 ± 0.16 (1.19; 0.79–1.55) | ||
| Right atrioventricular valve 1 | 0.22 ± 0.06 (0.21; 0.12–0.42) | 0.53 ± 0.09 (0.52; 0.31–0.70) | |
| Pulmonary artery 1 | 1.08 ± 0.2 (1.06; 0.7–1.59) |
| Myocardium | TDI 1 | CV (%) |
|---|---|---|
| Septum | S’ wave | 9.5 ± 4.7 (3–28) |
| E’ wave | 14.0 ± 9.6 (5–53) | |
| A’ wave | 16.3 ± 12.0 (6–75) | |
| Left free wall | S’ wave | 7.9 ± 4.6 (2–24) |
| E’ wave | 14.3 ± 9.3 (4–40) | |
| A’ wave | 13.6 ± 5.6 (3–29) | |
| Right free wall | S’ wave | 12.1 ± 5.9 (3–33) |
| E’ wave | 16.9 ± 8.5 (6–36) | |
| A’ wave | 7.5 ± 3.0 (3–16) |
| Myocardium | TDI 1 | CV (%) |
|---|---|---|
| Septum | S’ wave | 19.2 ± 3.5 (15–24) |
| E’ wave | 20.0 ± 3.0 (17–25) | |
| A’ wave | 26.8 ± 9.7 (15–41) | |
| Left free wall | S’ wave | 17.2 ± 6.1 (6–23) |
| E’ wave | 20.8 ± 4.7 (17–30) | |
| A’ wave | 25.2 ± 6.0 (3–29) | |
| Right free wall | S’ wave | 21.8 ± 7.0 (10–30) |
| E’ wave | 25.5 ± 9.9 (17–45) | |
| A’ wave | 21.7 ± 8.7 (12–37) |
| Myocardium | TDI 1 | Pigeon | Day | Observer |
|---|---|---|---|---|
| Septum | S’ wave | 40.4% | 45.6% | 14.0% |
| E’ wave | 15.1% | 51.7% | 33.2% | |
| A’ wave | 19.4% | 47.6% | 33.0% | |
| Left free wall | S’ wave | 24.3% | 61.1% | 14.6% |
| E’ wave | 6.7% | 62.1% | 31.2% | |
| A’ wave | 47.9% | 41.6% | 10.5% | |
| Right free wall | S’ wave | 18.6% | 61.0% | 20.4% |
| E’ wave | 28.4% | 47.3% | 24.3% | |
| A’ wave | 35.6% | 54.3% | 10.1% |
| Location | Sonographic Angle | S’ Wave, cm/s | E’ Wave, cm/s | A’ Wave, cm/s | Heart Rate, bpm |
|---|---|---|---|---|---|
| Septum 1 | 19.8 ± 6.4 | 4.8 ± 1.3 | −2.5 ± 0.5 | −3.8 ± 1.1 | 216 ± 50.8 |
| Septum 2 | 44.5 ± 6.3 | 4.1 ± 0.7 | −2.4 ± 0.4 | −4.5 ± 1.7 | 243 ± 43.7 |
| Left free wall 1 | 29.0 ± 3.8 | 8.6 ± 1.2 | −4.5 ± 1.2 | −6.5 ± 2.0 | 228 ± 46.5 |
| Left free wall 2 | 39.8 ± 2.5 | 6.9 ± 1.7 | −3.7 ± 1.4 | −4.6 ± 1.2 | 223 ± 52.5 |
| Right free wall 1 | 45.7 ± 6.2 | 6.3 ± 1.2 | −2.5 ± 0.9 | −9.7 ± 2.6 | 210 ± 47.4 |
| Right free wall 2 | 66.8 ± 4.2 | 5.0 ± 2.0 | −1.9 ± 0.4 | −8.9 ± 3.5 | 225 ± 43.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Legler, M.; Koy, L.; Kummerfeld, N.; Fehr, M. Diastolic and Systolic Longitudinal Myocardial Velocities of Healthy Racing Pigeons (Columba livia f. domestica) Measured by Tissue Doppler Imaging. Vet. Sci. 2021, 8, 23. https://doi.org/10.3390/vetsci8020023
Legler M, Koy L, Kummerfeld N, Fehr M. Diastolic and Systolic Longitudinal Myocardial Velocities of Healthy Racing Pigeons (Columba livia f. domestica) Measured by Tissue Doppler Imaging. Veterinary Sciences. 2021; 8(2):23. https://doi.org/10.3390/vetsci8020023
Chicago/Turabian StyleLegler, Marko, Lajos Koy, Norbert Kummerfeld, and Michael Fehr. 2021. "Diastolic and Systolic Longitudinal Myocardial Velocities of Healthy Racing Pigeons (Columba livia f. domestica) Measured by Tissue Doppler Imaging" Veterinary Sciences 8, no. 2: 23. https://doi.org/10.3390/vetsci8020023
APA StyleLegler, M., Koy, L., Kummerfeld, N., & Fehr, M. (2021). Diastolic and Systolic Longitudinal Myocardial Velocities of Healthy Racing Pigeons (Columba livia f. domestica) Measured by Tissue Doppler Imaging. Veterinary Sciences, 8(2), 23. https://doi.org/10.3390/vetsci8020023





