Dynamics of Macrophages and Polymorphonuclear Leukocytes Milk-Secreted by Buffaloes with Udders Characterized by Different Clinical Status
Abstract
1. Introduction
2. Materials and Methods
2.1. General and Ethical Animal Care
2.2. Farm and Study Population
2.3. Clinical Procedures and Udder Health Status
2.4. QSCC and Bacteriological Investigations
2.5. DSCC
2.6. Statistical Analysis
3. Results
3.1. Clinical Procedures and Udder Health Status
3.2. QSCC and Bacteriological Investigations
3.3. DSCC
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BMC | Bacteriological milk culture |
| CI | Confidence Interval |
| CM | Clinical mastitis |
| DIM | Days in milk |
| DSCC | Differential somatic cell count |
| IMI | Intramammary Infection |
| MBs | Mediterranean Buffaloes |
| NAS | Non-aureus Staphylococci |
| PMNL | Polymorphonuclear leukocytes |
| QSCC | Quantitative somatic cell count |
| SCM | Subclinical Mastitis |
| SE | Standard Error |
| SD | Standard Deviation |
| CI | Confidence Interval |
References
- Guccione, J.; Cosandey, A.; Pesce, A.; Di Loria, A.; Pascale, M.; Piantedosi, D.; Steiner, A.; Graber, H.U.; Ciaramella, P. Clinical outcomes and molecular genotyping of Staphylococcus aureus isolated from milk samples of dairy primiparous Mediterranean buffaloes (Bubalus bubalis). J. Dairy Sci. 2014, 97, 7606–7613. [Google Scholar] [CrossRef] [PubMed]
- Guccione, J.; D’Andrea, L.; Pesce, A.; Toni, F.; Borriello, G.; Salzano, C.; Diuccio, F.; Pascale, M.; Ciaramella, P. Antibiotic dry buffalo therapy: Effect of intramammary administration of benzathine cloxacillin against Staphylococcus aureus mastitis in dairy water buffalo. BMC Veter.-Res. 2020, 16, 191. [Google Scholar] [CrossRef] [PubMed]
- SVIMEZ. Associazione per lo Sviluppo Dell’industria del Mezzogiorno. Available online: http://lnx.svimez.info/svimez/wp-content/uploads/2019/06/2019_06_20_filiera_bufalina_slides.pdf (accessed on 16 December 2020).
- Puggioni, G.M.G.; Tedde, V.; Uzzau, S.; Guccione, J.; Ciaramella, P.; Pollera, C.; Moroni, P.; Bronzo, V.; Addis, M.F. Evaluation of a bovine cathelicidin ELISA for detecting mastitis in the dairy buffalo: Comparison with milk somatic cell count and bacteriological culture. Res. Veter.-Sci. 2019, 128, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Pisanu, S.; Cacciotto, C.; Pagnozzi, D.; Puggioni, G.M.G.; Uzzau, S.; Ciaramella, P.; Guccione, J.; Penati, M.; Pollera, C.; Moroni, P.; et al. Proteomic changes in the milk of water buffaloes (Bubalus bubalis) with subclinical mastitis due to intramammary infection by Staphylococcus aureus and by non-aureus staphylococci. Sci. Rep. 2019, 9, 15850. [Google Scholar] [CrossRef] [PubMed]
- Martini, V.; Bernardi, S.; Russo, V.; Guccione, J.; Comazzi, S.; Roperto, S. Blood lymphocyte subpopulations in healthy water buffaloes (Bubalus bubalis, Mediterranean lineage): Reference intervals and influence of age and reproductive history. Veter.-Immunol. Immunopathol. 2019, 211, 58–63. [Google Scholar] [CrossRef]
- Fink, S.L.; Campbell, S. Infection and host response. In Essential Concepts in Molecular Pathology, 2nd ed.; Coleman, V.B., Tsongalis, G.J., Eds.; Academic Press: London, UK, 2010; Volume 3, pp. 33–54. [Google Scholar]
- Piccinini, R.; Borromeo, V.; Zecconi, A. Relationship between S. aureus gene pattern and dairy herd mastitis prevalence. Veter.-Microbiol. 2010, 145, 100–105. [Google Scholar] [CrossRef]
- Damm, M.; Holm, C.; Blaabjerg, M.; Bro, M.N.; Schwarz, D. Differential somatic cell count—A novel method for routine mastitis screening in the frame of Dairy Herd Improvement testing programs. J. Dairy Sci. 2017, 100, 4926–4940. [Google Scholar] [CrossRef]
- Abbas, A.K.; Lichtman, A.H. Meccanismi effettori dell’immunità cellulo-mediata. In Immunologia Cellulare e Molecolare, 7th ed.; Abbas, A.K., Lichtman, A.H., Pillai, S., Eds.; Printer, Trento, S.r.l.: Trento, Italy, 2013; Volume 11, pp. 223–240. [Google Scholar]
- Oviedo-Boyso, J.; Valdez-Alarcón, J.J.; Cajero-Juárez, M.; Ochoa-Zarzosa, A.; López-Meza, J.E.; Bravo-Patiño, A.; Baizabal-Aguirre, V.M. Innate immune response of bovine mammary gland to pathogenic bacteria responsible for mastitis. J. Infect. 2007, 54, 399–409. [Google Scholar] [CrossRef]
- Guccione, J.; Pesce, A.; Pascale, M.; Tommasini, N.; Garofalo, F.; Di Loria, A.; Cortese, L.; Salzano, C.; Ciaramella, P. Effects of Systemic Dry Primiparous Mediterranean Buffalo (Bubalus bubalis) Treatment with Penethamate Hydriodide on Udder Health and Milk Yields. J. Dairy Sci. 2014, 97, 2219–2225. [Google Scholar] [CrossRef]
- Guccione, J.; Perreten, V.; Steiner, A.; Thomann, A.; Pesce, A.; Ciaramella, P.; Bodmer, M. Short communication: Role of Streptococcus pluranimalium in Mediterranean buffaloes (Bubalus bubalis) with different udder health statuses. J. Dairy Sci. 2016, 99, 2945–2949. [Google Scholar] [CrossRef]
- Guccione, J.; Pesce, A.; Pascale, M.; Salzano, C.; Tedeschi, G.; D’Andrea, L.; De Rosa, A.; Ciaramella, P. Efficacy of a polyvalent mastitis vaccine against Staphylococcus aureus on a dairy Mediterranean buffalo farm: Results of two clinical field trials. BMC Veter.-Res. 2016, 13, 1–9. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tripaldi, C.; Palocci, G.; Miarelli, M.; Catta, M.; Orlandini, S.; Amatiste, S.; Di Bernardini, R.; Catillo, G. Effect of Mastitis on Buffalo Milk Quality Asian-australas. J. Anim. Sci. 2010, 23, 1319–1324. [Google Scholar] [CrossRef]
- Guccione, J.; Rubino, V.; Pesce, A.; Piantedosi, D.; Palatucci, A.T.; Coppa, P.; Di Loria, A.; De Rosa, A.; Ciaramella, P. Diagnostic aspects of Mediterranean buffalo mastitis (Bubalus bubalis) through the use of flow cytometric differential cells counts. In Proceedings of the XIX ANEMBE International Congress, Oviedo, Spain, 26–28 June 2014. [Google Scholar]
- Schwarz, D.; Diesterbeck, U.; König, S.; Brügemann, K.; Schlez, K.; Zschöck, M.; Wolter, W.; Czerny, C.-P. Flow cytometric differential cell counts in milk for the evaluation of inflammatory reactions in clinically healthy and subclinically infected bovine mammary glands. J. Dairy Sci. 2011, 94, 5033–5044. [Google Scholar] [CrossRef]
- EMA, VICH GL9: Guideline on Good Clinical Practice. Available online: https://www.ema.europa.eu/en/vich-gl9-good-clinical-practices (accessed on 10 June 2021).
- Friedman, H. Simplified Determinations of Statistical Power, Magnitude of Effect and Research Sample Sizes. Educ. Psychol. Meas. 1982, 42, 521–526. [Google Scholar] [CrossRef]
- Guccione, J.; Borriello, G.; Ciaramella, P.; Di Loria, A. Clinical evaluation of poor milking procedures effects on dairy Mediterranean buffaloes udder health. Large Anim. Rev. 2017, 24, 155–157. [Google Scholar]
- Oliver, S.P. National Mastitis Council, Procedures for Collecting Milk Samples. In Microbiological Procedures for the Diagnosis of Bovine Udder Infection and Determination of Milk Quality, 4th ed.; Madison National Mastitis Council Inc.: Madison, WI, USA, 2004. [Google Scholar]
- National Mastitis Council, Diagnostic Procedures in Laboratory Handbook on Bovine Mastitis, 3rd ed.; National Mastitis Council Inc.: New Prague, MN, USA, 2017.
- Schober, P.; Boer, C.; Schwarte, L.A. Correlation Coefficients: Appropriate Use and Interpretation. Anesth Analg. 2018, 126, 1763–1768. [Google Scholar] [CrossRef]
- Gianesella, M.; Fiore, E.; Arfuso, F.; Vecchio, D.; Curone, G.; Morgante, M.; Mazzotta, E.; Badon, T.; Rossi, P.; Bedin, S.; et al. Serum haptoglobin and protein electrophoretic fraction modifications in buffaloes (Bubalus bubalis) around calving and during early lactation. J. Dairy Res. 2019, 86, 291–295. [Google Scholar] [CrossRef]
- Fiore, E.; Arfuso, F.; Gianesella, M.; Vecchio, D.; Morgante, M.; Mazzotta, E.; Badon, T.; Rossi, P.; Bedin, S.; Piccione, G. Metabolic and hormonal adaptation in Bubalus bubalis around calving and early lactation. PLoS ONE 2018, 13, e0193803. [Google Scholar] [CrossRef]
- Halasa, T.; Kirkeby, C. Differential Somatic Cell Count: Value for Udder Health Management. Front. Veter.-Sci. 2020, 7, 609055. [Google Scholar] [CrossRef]
- Thomas, F.; Hehemann, J.; Rebuffet, E.; Czjzek, M.; Michel, G. Environmental and gut Bacteroidetes: The food connection. Front. Microbiol. 2011, 2, 93. [Google Scholar] [CrossRef]
- Michel, A.; Syring, C.; Steiner, A.; Graber, H.U. Intramammary infections with the contagious Staphylococcus aureus genotype B in Swiss dairy cows are associated with low prevalence of coagulase-negative staphylococci and Streptococcus spp. Veter.-J. 2011, 188, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Moroni, P.; Rossi, C.S.; Pisoni, G.; Bronzo, V.; Castiglioni, B.; Boettcher, P. Relationships between Somatic Cell Count and Intramammary Infection in Buffaloes. J. Dairy Sci. 2006, 89, 998–1003. [Google Scholar] [CrossRef]
- Nickerson, S. Immunological Aspects of Mammary Involution. J. Dairy Sci. 1989, 72, 1665–1678. [Google Scholar] [CrossRef]
- Pilla, R.; Malvisi, M.; Snel, G.; Schwarz, D.; König, S.; Czerny, C.-P.; Piccinini, R. Differential cell count as an alternative method to diagnose dairy cow mastitis. J. Dairy Sci. 2013, 96, 1653–1660. [Google Scholar] [CrossRef] [PubMed]
- Pilla, R.; Schwarz, D.; König, S.; Piccinini, R. Microscopic differential cell counting to identify inflammatory reactions in dairy cow quarter milk samples. J. Dairy Sci. 2012, 95, 4410–4420. [Google Scholar] [CrossRef] [PubMed]
- Zecconi, A.; Zanini, L.; Cipolla, M.; Stefanon, B. Factors Affecting the Patterns of Total Amount and Proportions of Leukocytes in Bovine Milk. Animals 2020, 10, 992. [Google Scholar] [CrossRef] [PubMed]
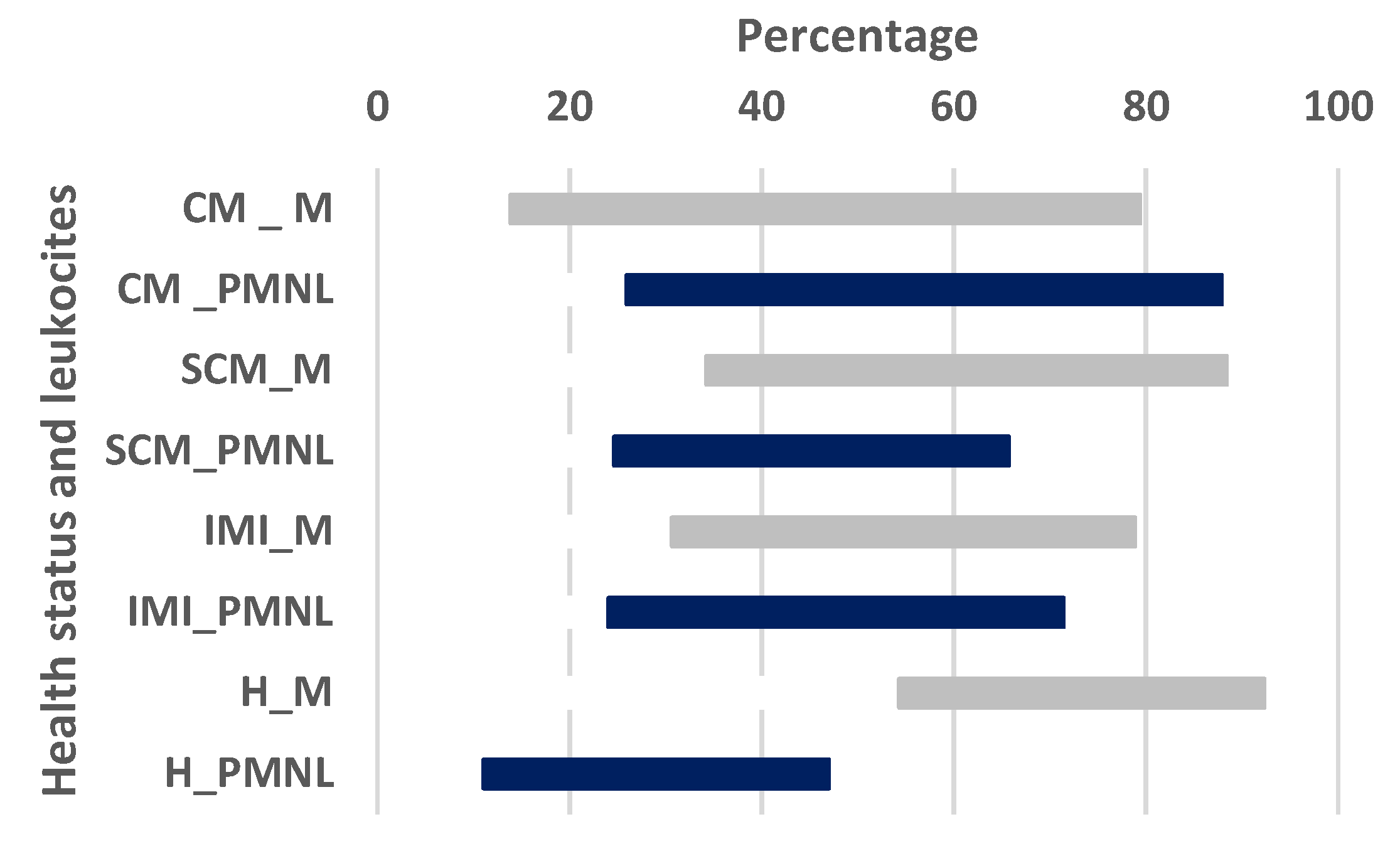

| QSCC (Log10 Cells/mL) | ||||||
| Ave. | S.E. | C.I. | ||||
| CM | 6.55 | 0.08 | 6.38–6.71 | |||
| SCM | 5.62 | 0.04 | 5.53–5.70 | |||
| IMI | 4.88 | 0.06 | 4.74–5.02 | |||
| H | 4.67 | 0.07 | 4.50–4.83 | |||
| DSCC (%) | ||||||
| Macrophages | Polymorphonuclear | |||||
| Ave. | S.E. | C.I. | Ave. | S.E. | C.I. | |
| CM | 49.08 a | 4.28 | 40.11–58.05 | 53.97 a | 4.11 | 45.36–62.57 |
| SCM | 56.42 c | 3.16 | 49.80–63.04 | 46.54 e | 3.37 | 39.46–53.61 |
| IMI | 57.79 | 3.15 | 51.18–64.39 | 42.17 e | 3.01 | 35.86–48.48 |
| H | 69.25 b,d | 2.28 | 64.77–74.33 | 29.88 b,f | 2.42 | 24.80–34.96 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alterisio, M.C.; Ciaramella, P.; Guccione, J. Dynamics of Macrophages and Polymorphonuclear Leukocytes Milk-Secreted by Buffaloes with Udders Characterized by Different Clinical Status. Vet. Sci. 2021, 8, 204. https://doi.org/10.3390/vetsci8100204
Alterisio MC, Ciaramella P, Guccione J. Dynamics of Macrophages and Polymorphonuclear Leukocytes Milk-Secreted by Buffaloes with Udders Characterized by Different Clinical Status. Veterinary Sciences. 2021; 8(10):204. https://doi.org/10.3390/vetsci8100204
Chicago/Turabian StyleAlterisio, Maria Chiara, Paolo Ciaramella, and Jacopo Guccione. 2021. "Dynamics of Macrophages and Polymorphonuclear Leukocytes Milk-Secreted by Buffaloes with Udders Characterized by Different Clinical Status" Veterinary Sciences 8, no. 10: 204. https://doi.org/10.3390/vetsci8100204
APA StyleAlterisio, M. C., Ciaramella, P., & Guccione, J. (2021). Dynamics of Macrophages and Polymorphonuclear Leukocytes Milk-Secreted by Buffaloes with Udders Characterized by Different Clinical Status. Veterinary Sciences, 8(10), 204. https://doi.org/10.3390/vetsci8100204






