Mammary Diseases in a Captive Reared Agouti (Dasyprocta leporina) in Trinidad
Abstract
1. Introduction
2. Materials and Method
2.1. Ethical Approval
2.2. Housing and Environment of the Agouti
2.3. History and Physical Examination
2.4. Haematological and Serum Biochemical Test
2.5. Milk Sample Test
2.6. Tissue Biopsy and Histology
3. Results
3.1. Gross Findings
3.2. Blood and Milk Samples Findings
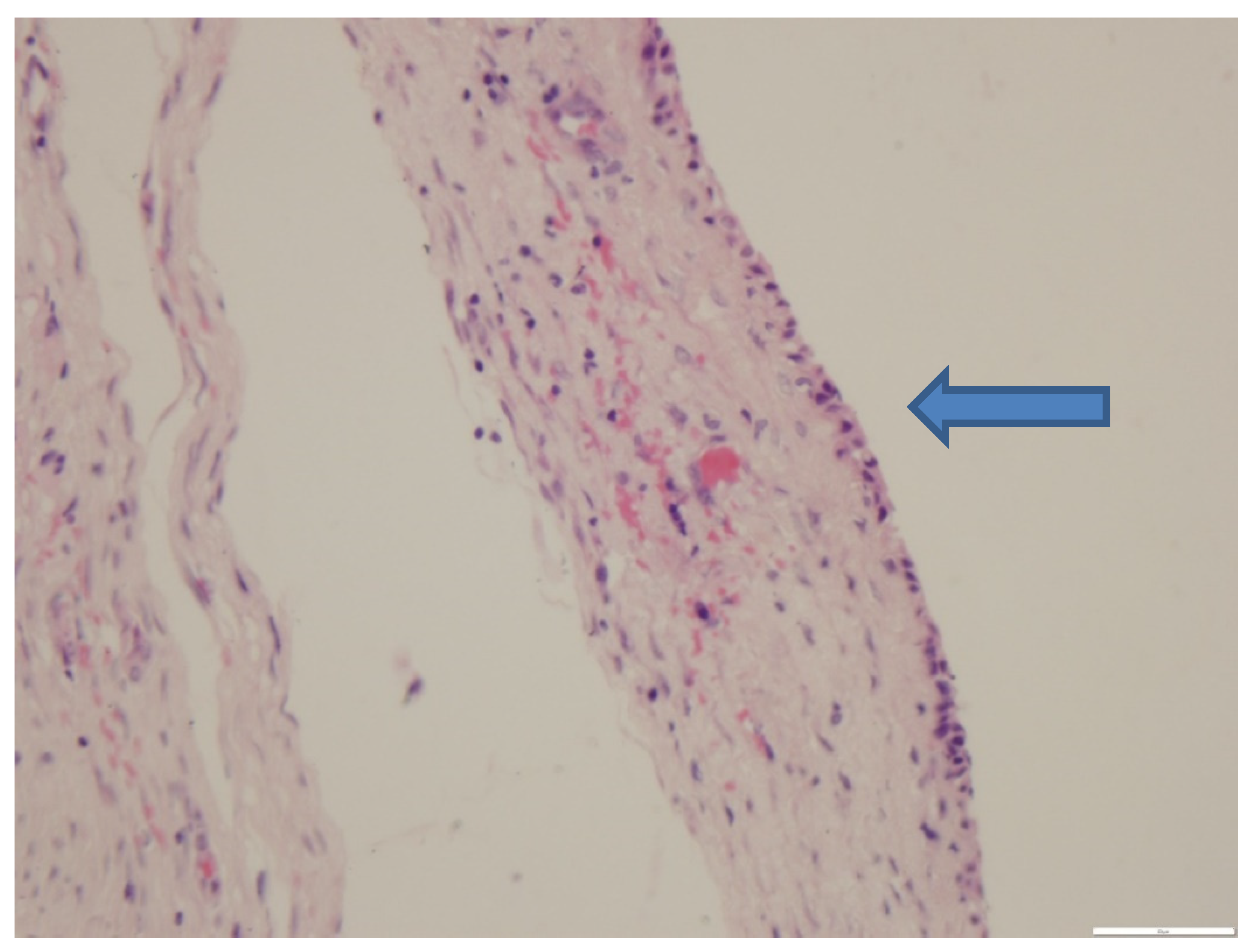
3.3. Histopathological Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Emmons, L.; Feer, F. Neo-Tropical Rainforest Mammals: A Field Guide; London University of Chicago Press: Chicago, IL, USA, 1997. [Google Scholar]
- Nunes, A.V.; Guariento, R.G.; Santos, B.A.; Fisher, E. Wild meat sharing among indigenous people in the southwestern Amazon. Behav. Ecol. Sociobiol. 2019, 73, 26. [Google Scholar] [CrossRef]
- Brown-Uddenberg, R.; Garcia, W.G.; Baptiste, Q.S.; Counand, T.; Adogwa, A.; Sampson, T. The Agouti (Dasyprocta leporina, D. agouti) Booklet and Production Manual; GWG Publications: St. Augustine, Trinidad, 2004. [Google Scholar]
- Hardouin, J.; Thys, E.; Joiris, V.; Fielding, D. Minilivestock breeding with indigenous species in the tropics. Livest. Res. Rural Dev. 2003, 15, 4. [Google Scholar]
- Lainson, R.; Carneiro, L.; Silveira, F.T. Observations on the Eineria species of the Dasyprocta leporina (Linnaeus, 1758) (Rodentia: Dasyproctidae) for the state of Para, North Brazil. Mem. Inst. Oswaldo Cruz 2007, 102, 183–189. [Google Scholar] [CrossRef] [PubMed]
- da Silva, M.K.; da Silva, A.S.; Olivieria, C.M.; Montiero, S.G. Gastrointestinal parasites of the agouti (Dasyprocta leporina). Cienc. Anim. Bras. 2008, 9, 128–131. [Google Scholar]
- Zimmerman, D.M.; Douglass, M.; Reavill, D.R.; Greiner, E.C. Echinococcus oligarthrus cystic hydatidosis in Brazillian agouti (Dasyprocta leporina). J. Zoo Wildl. Med. 2009, 40, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Suepaul, R.; Charles, R.; Dziva, F. Aerobic microflora and endoparasites of freshly shot wild Agouti (Dasyprocta leporina) in Trindad, West Indies. J. Zoo Wildl. Med. 2016, 47, 1044–1048. [Google Scholar] [CrossRef]
- Jones, K.R.; Garcia, G.W. A survey of the gastrointestinal parasites present in the Agouti (Dasyprocta leporina) reared intensively in Trinidad. Livest. Res. Rural Dev. 2017, 29, 1–7. [Google Scholar]
- Jones, K.R.; Garcia, G.W. Observations on endoparasitic load in captive reared agoutis (Dasyprocta leporina) without anthelmintic exposure in Trinidad, Republic of Trinidad and Tobago. Livest. Res. Rural Dev. 2018, 30, 1–7. [Google Scholar]
- Jones, K.R.; Lall, K.R.; Garcia, G.W. Endoparasites of non-domesticated mammals from the Neo-tropics (New World Tropics). Vet. Sci. 2019, 4, 87. [Google Scholar] [CrossRef]
- Ayala, S.C.; D’ Alessandro, A.; Mackenzie, R.; Angel, D. Hemoparasites in 830 wild animals from Eastern Llanos of Colombia. J. Parasitol. 1973, 59, 52–59. [Google Scholar] [CrossRef]
- de Thoisy, B.; Michel, J.-C.; Vogel, I.; Vie, J.-C. A survey of hemoparasite infection in free ranging mammals and reptiles in French Guina. J. Parasitol. 2000, 86, 1035–1040. [Google Scholar] [CrossRef]
- Jones, K.R.; Lall, K.R.; Garcia, G.W. Haematological and Serum biochemical values of the agouti (Dasyprocta leporina) reared intensively in Trinidad, Republic of Trinidad and Tobago. Livest. Res. Rural Dev. 2019, 31, 1–8. [Google Scholar]
- Lall, K.R.; Jones, K.R.; Garcia, G.W. Infectious Diseases of Six Non-Domesticated Neo-Tropical Animals in Trinidad and Tobago. Int. J. Trop. Vet. Biomed. Res. 2018, 3, 1–31. [Google Scholar] [CrossRef]
- Kenny, D.; Cambre, R.C.; Lewandowski, A.; Pelto, J.A.; Iribeck, N.A.; Wilson, H.; Mierau, G.W.; Sill, M.G.; Garcia, A.P. Suspected vitamin D3 toxicity in pacas (Cuniculus paca) and agoutis (Dasyprocta aguti). J. Zoo Wildl. Med. 1993, 24, 129–139. [Google Scholar]
- Anderson, K.M.; Lewandowski, A.; Dennis, P.M. Suspected hypervitaminosis in red-rumped agouti (Dasyprocta leporina) receiving a commercial rodent diet. J. Zoo Wildl. Med. 2018, 49, 96–200. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.R.; Lall, K.R.; Garcia, G.W. Dystocia in a captive reared Agouti (Dasyprocta leporina) in Trinidad and Tobago, West Indies. Vet. Sci. 2020, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Pang, V.F.; Chang, P.H.; Wang, F.I.; Chin, S.C.; Jeng, C.R.; Liu, C.H.; Chu, P.Y.; Zhuo, Y.X. Spontaneous neoplasms in zoo mammals, birds, and reptiles in Taiwan–a 10-year survey. Anim. Biol. 2012, 62, 95–110. [Google Scholar] [CrossRef]
- Batista, J.S.; Freitas, C.J.A.; Brilhante, F.S.; Viana, G.A.; Olinda, R.G.; Cavalcante, T.V.; de Paiva, K.A.R.; de Oliveira, M.F. Pathological changes of the genital system of agoutis (Dasyprocta aguti Linnaeus, 1758) females bred in captivity. Braz. Vet. J. 2016, 36, 634–641. [Google Scholar]
- Batista, J.S.; Olinda, R.G.; Silva, T.M.F.; Rodriguez, C.M.F.; Oliveira, A.F.; Queiroz, S.A.C.; Morais, S.R.L.; Oliveira, M.F. Diseases of agouti (Dasyprocta aguti) raised in captivity diagnosed by pathological examination. Braz. Vet. J. 2010, 30, 497–502. [Google Scholar]
- Müller, D.W.; Szentiks, C.A.; Wibbelt, G. Polycystic kidney disease in adult Brazilian agoutis (Dasyprocta leporina). Vet. Pathol. 2009, 46, 656–661. [Google Scholar] [CrossRef]
- Raus, J.; Love, D.N. Characterization of Coagulase-Positive Staphyloccocus intermedius and Staphyloccus aureus Isolated from Veterinary Clinical Specimens. J. Clin. Microbiol. 1983, 18, 789–792. [Google Scholar] [CrossRef] [PubMed]
- Lainhart, W.; Yarbrough, L.; Burnham, C.-A. The Brief Case: Staphyloccus intermedius Group-Look What the Dog Dragged in. J. Clin. Microbiol. 2018, 56. [Google Scholar] [CrossRef]
- Baas, E.J.; Potkay, S.; Bacher, J. The agouti (Dasyprocta sp) in biomedical research and captivity. Lab. Anim. Sci. 1976, 26, 788–796. [Google Scholar] [PubMed]
- Jones, K.R.; Garcia, G.W. Haematology and Serum Biochemistry in the Agouti (Dasyprocta spp.): A Neo-Tropical Rodent with the Potential for Domestication. Concepts Dairy Vet. Sci. 2019, 3, 1. [Google Scholar]
- Jones, K.R.; Garcia, G.W. Understanding of the Blood and Serum values of the Agouti (Dasyprocta spp.): A Rodent of the Neo-Tropics with the potential to be domesticated. Trop. Agric. 2019, 96, 117–126. [Google Scholar]
- Singh, M.D.; Singh, S.; Garcia, G.W. Understanding mammary activity in red-rumped agouti and implications for management and conservation of this Neotropical game species. Braz. J. Biol. 2018, 78, 540–547. [Google Scholar] [CrossRef]
- Kinkler Jr, R.J.; Wagner, J.E.; Doyle, R.E.; Owens, D.R. Bacterial mastitis in guinea pigs. Lab. Anim. Sci. 1976, 26, 214–217. [Google Scholar]
- Hong, C.C.; Ediger, R.D. Chronic necrotizing mastitis in rats caused by Pasteurella pneumotropica. Lab. Anim. Sci. 1978, 28, 317–320. [Google Scholar]
- Gupta, B.N.; Langham, R.F.; Conner, G.H. Mastitis in Guinea Pigs. Am. J. Vet. Res. 1970, 31, 1703–1707. [Google Scholar]
- Hermans, K.; De Herdt, P.; Devriese, L.A.; Hendrickx, W.; Godard, C.; Haesebrouck, F. Colonization of rabbits with Staphylococcus aureus in flocks with and without chronic staphylococcosis. Vet. Microbiol. 1999, 67, 37–46. [Google Scholar] [CrossRef]
- Viana, D.; Selva, L.; Callanan, J.J.; Ferrian, S.; Corpa, J.M. Strains of Staphylococcus aureus and pathology associated with chronic suppurative mastitis in rabbits. Vet. J. 2013, 190, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, I.; Ferrian, S.; Panades, M.; Garcia-Quiros, A.; Pascual, J.J.; Selva, L.; Viana, D.; Copra, J.M. Host responses associated with chronic staphylococcal mastitis in rabbits. Vet. J. 2015, 204, 338–344. [Google Scholar] [CrossRef] [PubMed]
| Morphological Characteristics/Tests | Results |
|---|---|
| Gram Stain | Gram (+) cocci |
| Catalase Test | Catalase (+) |
| Appearance on Blood Agar | Cream pinpoint colonies with zones of hemolysis |
| Coagulase test | Coagulase (+) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, K.R.; Lall, K.R.; Suepaul, R.; Garcia, G.W. Mammary Diseases in a Captive Reared Agouti (Dasyprocta leporina) in Trinidad. Vet. Sci. 2020, 7, 137. https://doi.org/10.3390/vetsci7030137
Jones KR, Lall KR, Suepaul R, Garcia GW. Mammary Diseases in a Captive Reared Agouti (Dasyprocta leporina) in Trinidad. Veterinary Sciences. 2020; 7(3):137. https://doi.org/10.3390/vetsci7030137
Chicago/Turabian StyleJones, Kegan Romelle, Kavita Ranjeeta Lall, Rod Suepaul, and Gary Wayne Garcia. 2020. "Mammary Diseases in a Captive Reared Agouti (Dasyprocta leporina) in Trinidad" Veterinary Sciences 7, no. 3: 137. https://doi.org/10.3390/vetsci7030137
APA StyleJones, K. R., Lall, K. R., Suepaul, R., & Garcia, G. W. (2020). Mammary Diseases in a Captive Reared Agouti (Dasyprocta leporina) in Trinidad. Veterinary Sciences, 7(3), 137. https://doi.org/10.3390/vetsci7030137






