Review of Diagnostic and Therapeutic Approach to Canine Myxomatous Mitral Valve Disease
Abstract
:1. Introduction
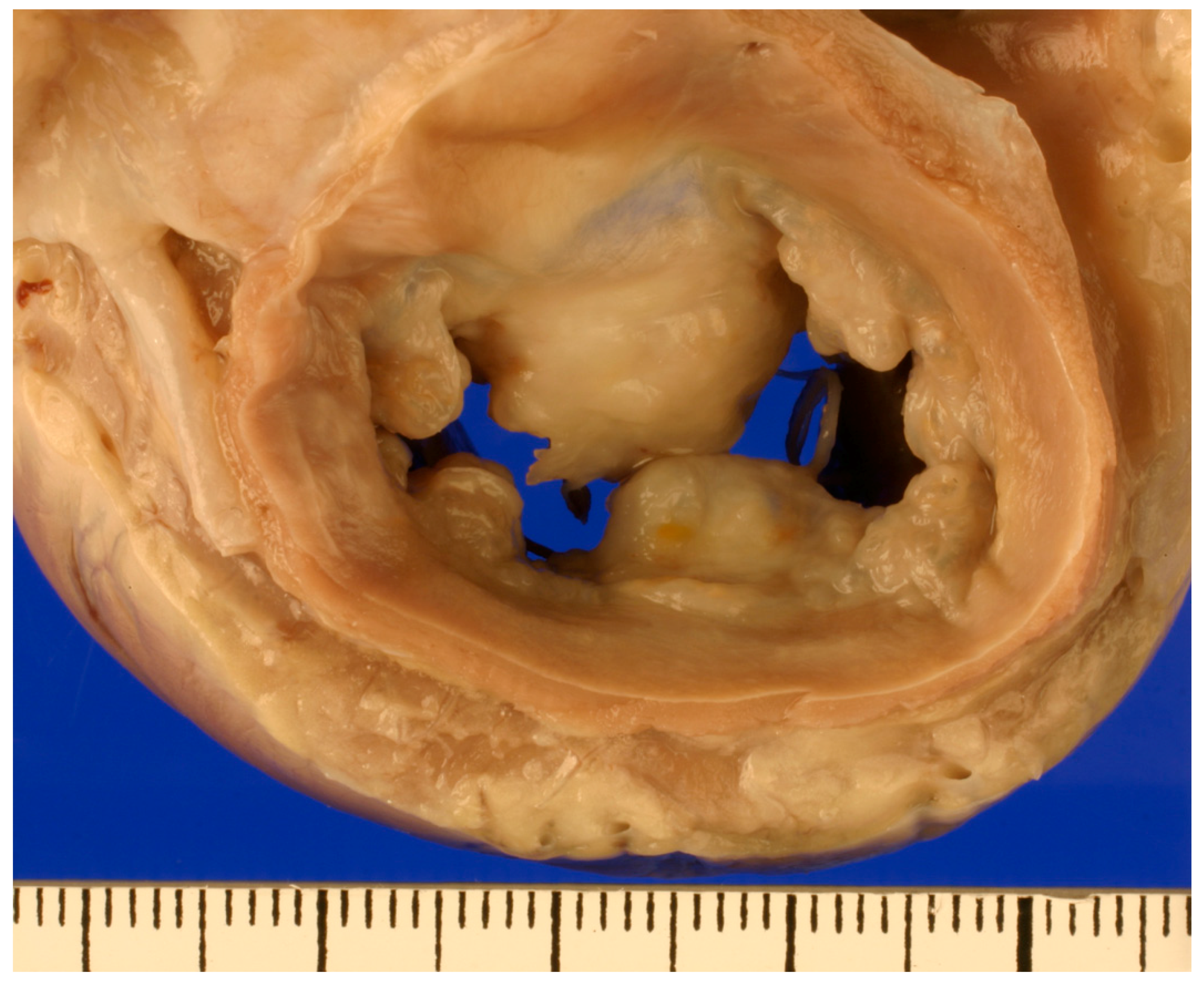
2. Diagnosis
3. Assessing Disease Severity
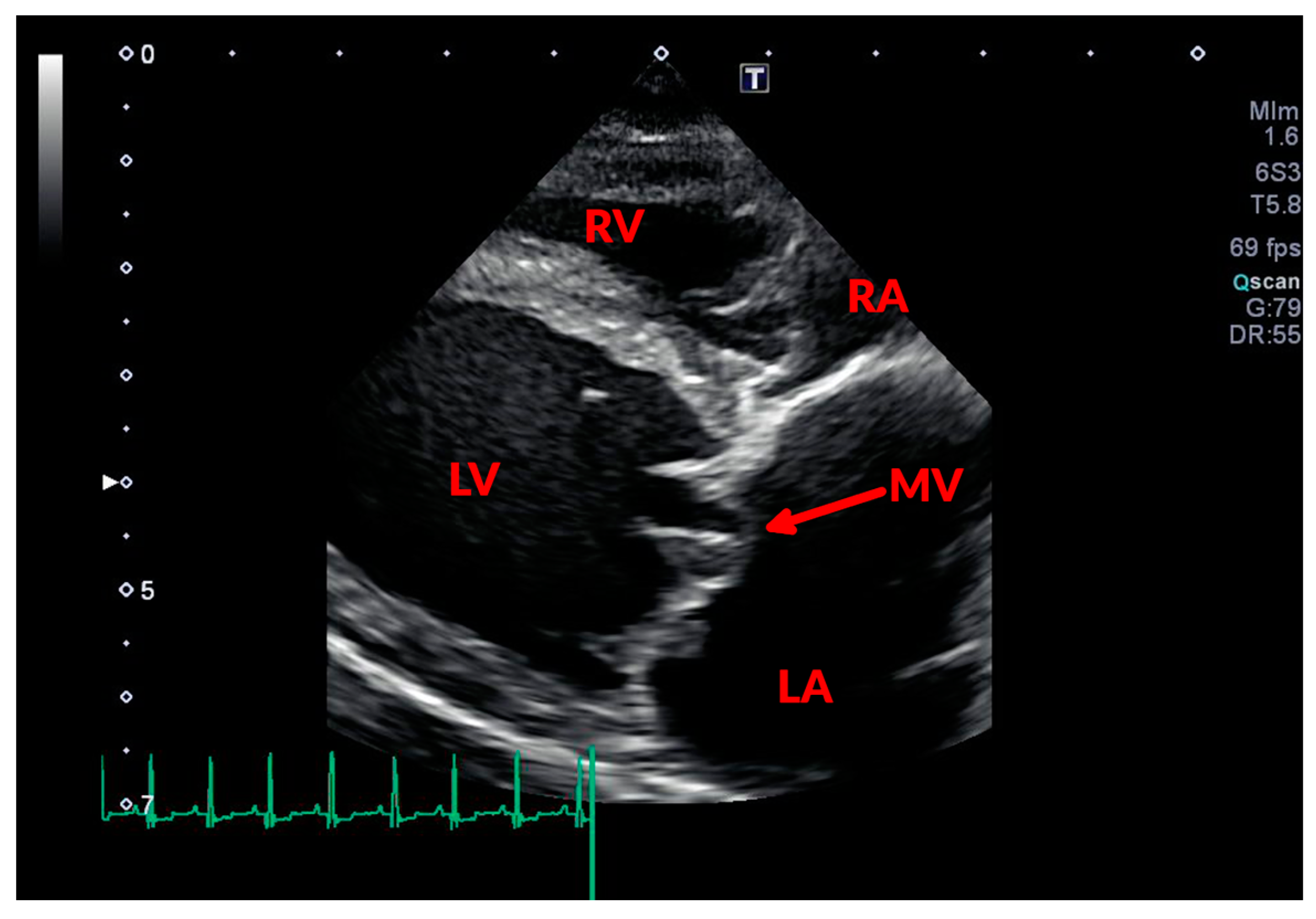
3.1. Conventional Echocardiography
3.1.1. Assessing MR Severity
3.1.2. Assessing Cardiac Remodeling
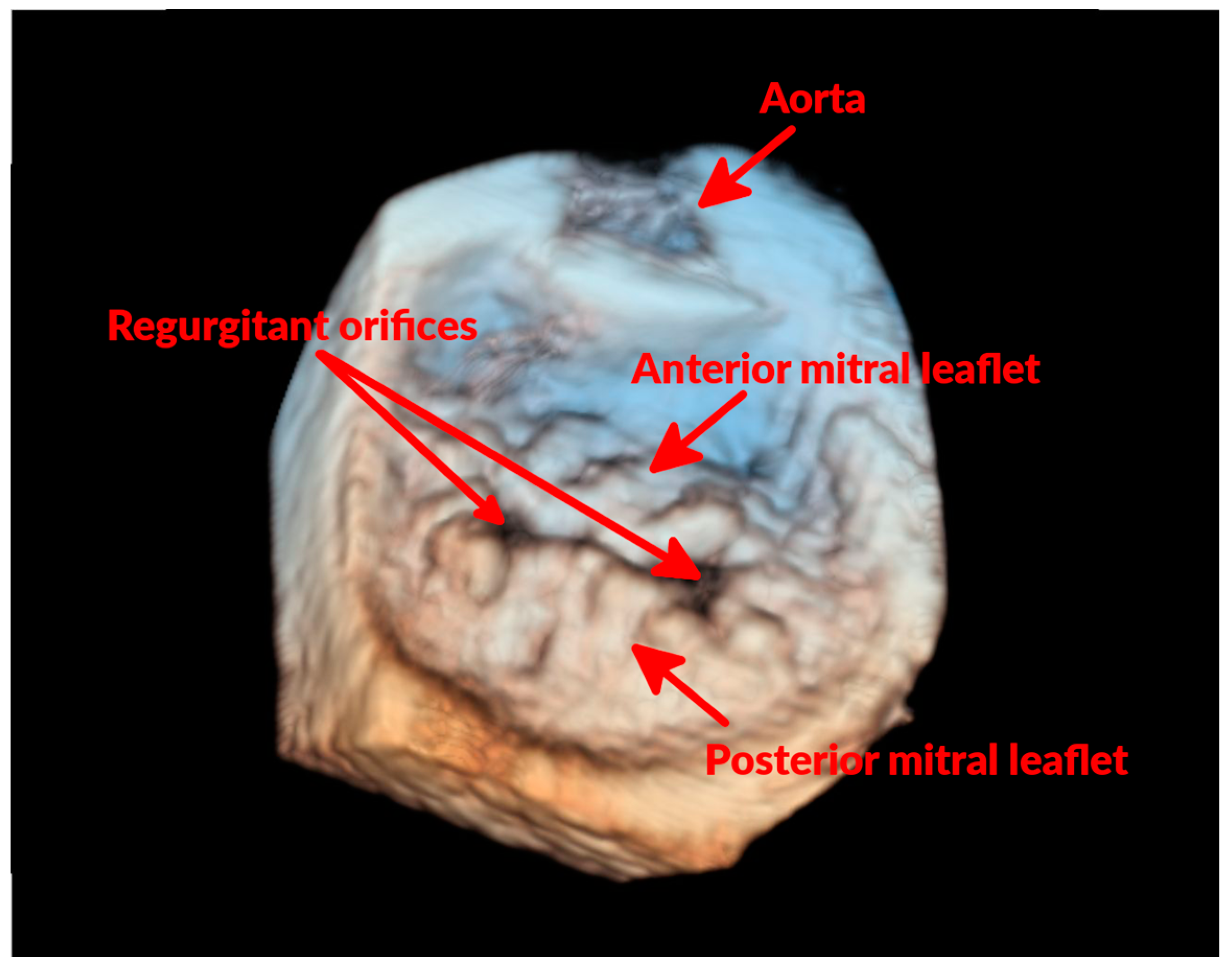
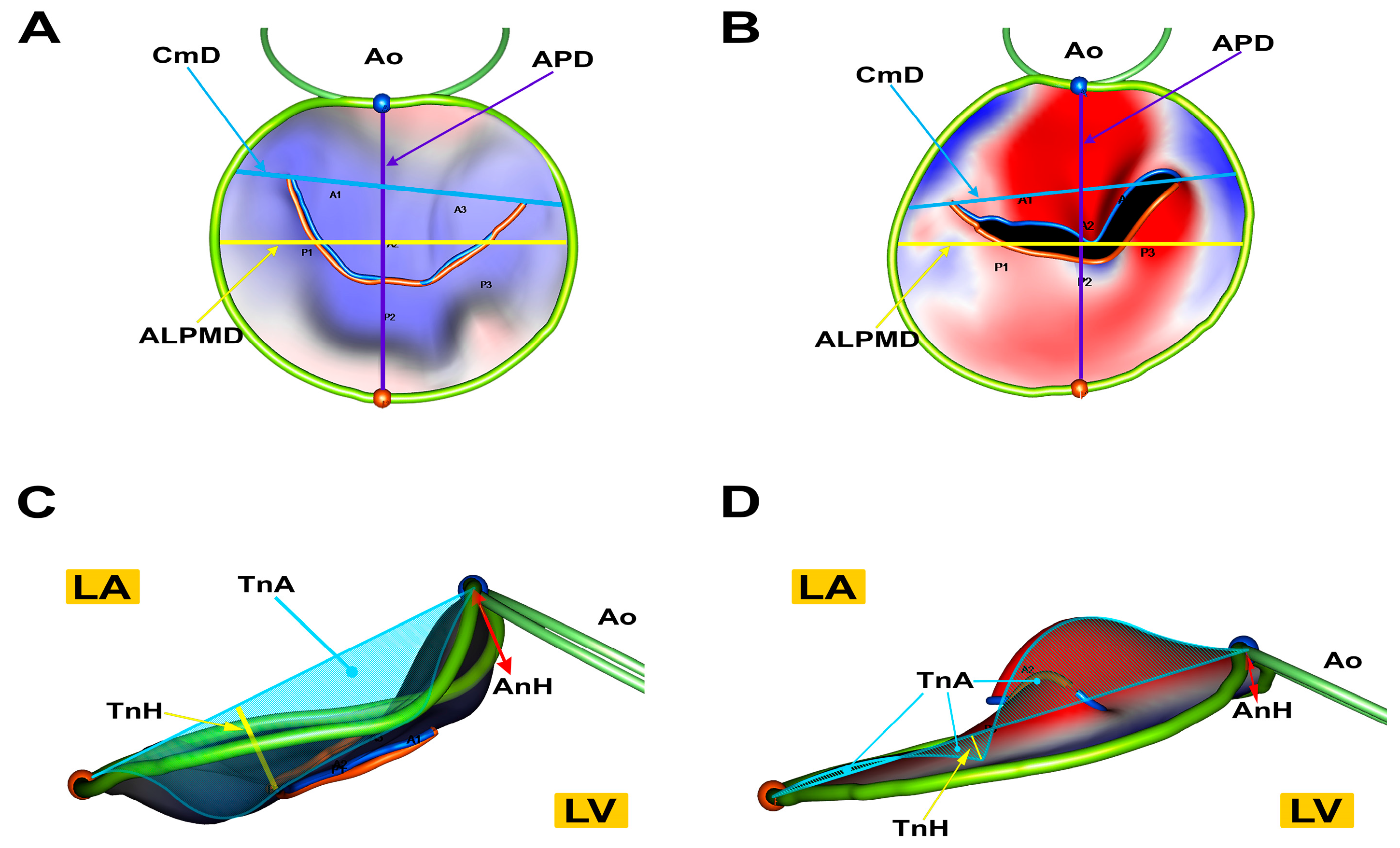
3.2. Advanced Echocardiographic Techniques
4. Therapy
4.1. Medical Therapy
4.2. Surgical Therapy
4.2.1. Mitral Valve Replacement
4.2.2. Minimally-Invasive Approaches
Conflicts of Interest
References
- Häggström, J.; Hansson, K.; Kvart, C.; Swenson, L. Chronic valvular disease in the cavalier King Charles spaniel in Sweden. Vet. Rec. 1992, 131, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Detweiler, D.K.; Patterson, D.F. The prevalence and types of cardiovascular disease in dogs. Ann. N. Y. Acad. Sci. 1965, 127, 481–516. [Google Scholar] [CrossRef] [PubMed]
- Borgarelli, M.; Zini, E.; D’Agnolo, G.; Tarducci, A.; Santilli, R.A.; Chiavegato, D.; Tursi, M.; Prunotto, M.; Häggström, J. Comparison of primary mitral valve disease in German Shepherd dogs and in small breeds. J. Vet. Cardiol. 2004, 6, 27–34. [Google Scholar] [CrossRef]
- Borgarelli, M.; Tarducci, A.; Zanatta, R.; Haggstrom, J. Decreased systolic function and inadequate hypertrophy in large and small breed dogs with chronic mitral valve insufficiency. J. Vet. Intern. Med. 2007, 21, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Swenson, L.; Häggström, J.; Kvart, C.; Juneja, R.K. Relationship between parental cardiac status in Cavalier King Charles spaniels and prevalence and severity of chronic valvular disease in offspring. J. Am. Vet. Med. Assoc. 1996, 208, 2009–2012. [Google Scholar] [PubMed]
- Olsen, L.H.; Fredholm, M.; Pedersen, H.D. Epidemiology and inheritance of mitral valve prolapse in Dachshunds. J. Vet. Intern. Med. 1999, 13, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Lewis, T.; Swift, S.; Woolliams, J.A.; Blott, S. Heritability of premature mitral valve disease in Cavalier King Charles spaniels. Vet. J. 2011, 188, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Oyama, M.A.; Levy, R.J. Insights into serotonin signaling mechanisms associated with canine degenerative mitral valve disease. J. Vet. Intern. Med. 2010, 24, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Cremer, S.E.; Zois, N.E.; Moesgaard, S.G.; Ravn, N.; Cirera, S.; Honge, J.L.; Smerup, M.H.; Hasenkam, J.M.; Sloth, E.; Leifsson, P.S.; et al. Serotonin markers show altered transcription levels in an experimental pig model of mitral regurgitation. Vet. J. 2015, 203, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Cremer, S.E.; Singletary, G.E.; Olsen, L.H.; Wallace, K.; Häggström, J.; Ljungvall, I.; Höglund, K.; Reynolds, C.A.; Pizzinat, N.; Oyama, M.A. Serotonin concentrations in platelets, plasma, mitral valve leaflet, and left ventricular myocardial tissue in dogs with myxomatous mitral valve disease. J. Vet. Intern. Med. 2014, 28, 1534–1540. [Google Scholar] [CrossRef] [PubMed]
- Ljungvall, I.; Höglund, K.; Lilliehöök, I.; Oyama, M.A.; Tidholm, A.; Tvedten, H.; Häggström, J. Serum serotonin concentration is associated with severity of myxomatous mitral valve disease in dogs. J. Vet. Intern. Med. 2013, 27, 1105–1112. [Google Scholar] [CrossRef] [PubMed]
- Orton, E.C.; Lacerda, C.M.R.; MacLea, H.B. Signaling pathways in mitral valve degeneration. J. Vet. Cardiol. 2012, 14, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Lacerda, C.M.R.; Kisiday, J.; Johnson, B.; Orton, E.C. Local serotonin mediates cyclic strain-induced phenotype transformation, matrix degradation, and glycosaminoglycan synthesis in cultured sheep mitral valves. Am. J. Physiol. 2012, 302, H1983–H1990. [Google Scholar] [CrossRef] [PubMed]
- Lacerda, C.M.R.; MacLea, H.B.; Kisiday, J.D.; Orton, E.C. Static and cyclic tensile strain induce myxomatous effector proteins and serotonin in canine mitral valves. J. Vet. Cardiol. 2012, 14, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Aupperle, H.; Disatian, S. Pathology, protein expression and signaling in myxomatous mitral valve degeneration: Comparison of dogs and humans. J. Vet. Cardiol. 2012, 14, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Disatian, S.; Ehrhart, E.J.; Zimmerman, S.; Orton, E.C. Interstitial cells from dogs with naturally occurring myxomatous mitral valve disease undergo phenotype transformation. J. Heart Valve Dis. 2008, 17, 402–411; discussion 412. [Google Scholar] [PubMed]
- Richards, J.M.; Farrar, E.J.; Kornreich, B.G.; Moїse, N.S.; Butcher, J.T. The mechanobiology of mitral valve function, degeneration, and repair. J. Vet. Cardiol. 2012, 14, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Pomerance, A.; Whitney, J.C. Heart valve changes common to man and dog: A comparative study. Cardiovasc. Res. 1970, 4, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Côté, E.; Edwards, N.J.; Ettinger, S.J.; Fuentes, V.L.; MacDonald, K.A.; Scansen, B.A.; Sisson, D.D.; Abbott, J.A. Management of incidentally detected heart murmurs in dogs and cats. J. Am. Vet. Med. Assoc. 2015, 246, 1076–1088. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, H.D.; Häggström, J.; Falk, T.; Mow, T.; Olsen, L.H.; Iversen, L.; Jensen, A.L. Auscultation in mild mitral regurgitation in dogs: Observer variation, effects of physical maneuvers, and agreement with color doppler echocardiography and phonocardiography. J. Vet. Intern. Med. 1999, 13, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Terzo, E.; Di Marcello, M.; McAllister, H.; Glazier, B.; Lo Coco, D.; Locatelli, C.; Palermo, V.; Brambilla, P.G. Echocardiographic assessment of 537 dogs with mitral valve prolapse and leaflet involvement. Vet. Radiol. Ultrasound 2009, 50, 416–422. [Google Scholar] [CrossRef] [PubMed]
- López-Alvarez, J.; Elliott, J.; Pfeiffer, D.; Chang, Y.-M.; Mattin, M.; Moonarmart, W.; Hezzell, M.J.; Boswood, A. Clinical severity score system in dogs with degenerative mitral valve disease. J. Vet. Intern. Med. 2015, 29, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Borgarelli, M.; Savarino, P.; Crosara, S.; Santilli, R.A.; Chiavegato, D.; Poggi, M.; Bellino, C.; La Rosa, G.; Zanatta, R.; Haggstrom, J.; et al. Survival characteristics and prognostic variables of dogs with mitral regurgitation attributable to myxomatous valve disease. J. Vet. Intern. Med. 2008, 22, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Borgarelli, M.; Crosara, S.; Lamb, K.; Savarino, P.; La Rosa, G.; Tarducci, A.; Haggstrom, J.; Santilli, R.A.; Chiavegato, D.; Poggi, M.; et al. Survival characteristics and prognostic variables of dogs with preclinical chronic degenerative mitral valve disease attributable to myxomatous degeneration. J. Vet. Intern. Med. 2012, 26, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, J.W.; Bücheler, J. Vertebral scale system to measure canine heart size in radiographs. J. Am. Vet. Med. Assoc. 1995, 206, 194–199. [Google Scholar] [PubMed]
- Atkins, C.; Bonagura, J.; Ettinger, S.; Fox, P.; Gordon, S.; Haggstrom, J.; Hamlin, R.; Keene, B.; Luis-Fuentes, V.; Stepien, R. Guidelines for the diagnosis and treatment of canine chronic valvular heart disease. J. Vet. Intern. Med. 2009, 23, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Diana, A.; Guglielmini, C.; Pivetta, M.; Sanacore, A.; di Tommaso, M.; Lord, P.F.; Cipone, M. Radiographic features of cardiogenic pulmonary edema in dogs with mitral regurgitation: 61 Cases (1998–2007). J. Am. Vet. Med. Assoc. 2009, 235, 1058–1063. [Google Scholar] [CrossRef] [PubMed]
- Chetboul, V.; Tissier, R. Echocardiographic assessment of canine degenerative mitral valve disease. J. Vet. Cardiol. 2012, 14, 127–148. [Google Scholar] [CrossRef] [PubMed]
- Reimann, M.J.; Møller, J.E.; Häggström, J.; Martinussen, T.; Zatrazemi, S.S.C.; Svanholm, L.; Nielsen, L.B.M.; Pedersen, H.D.; Olsen, L.H. Mitral regurgitation severity and left ventricular systolic dimension predict survival in young cavalier king charles spaniels. J. Vet. Intern. Med. 2017, 31, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Sargent, J.; Muzzi, R.; Mukherjee, R.; Somarathne, S.; Schranz, K.; Stephenson, H.; Connolly, D.; Brodbelt, D.; Fuentes, V.L. Echocardiographic predictors of survival in dogs with myxomatous mitral valve disease. J. Vet. Cardiol. 2015, 17, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zoghbi, W.A.; Enriquez-Sarano, M.; Foster, E.; Grayburn, P.A.; Kraft, C.D.; Levine, R.A.; Nihoyannopoulos, P.; Otto, C.M.; Quinones, M.A.; Rakowski, H.; et al. Recommendations for evaluation of the severity of native valvular regurgitation with two-dimensional and Doppler echocardiography. J. Am. Soc. Echocardiogr. 2003, 16, 777–802. [Google Scholar] [CrossRef]
- Serres, F.; Chetboul, V.; Tissier, R.; Sampedrano, C.C.; Gouni, V.; Nicolle, A.P.; Pouchelon, J.-L. Chordae tendineae rupture in dogs with degenerative mitral valve disease: Prevalence, survival, and prognostic factors (114 cases, 2001–2006). J. Vet. Intern. Med. 2007, 21, 258–264. [Google Scholar] [CrossRef]
- Muzzi, R.A.L.; de Araújo, R.B.; Muzzi, L.A.L.; Pena, J.L.B.; Silva, E.F. Regurgitant jet area by Doppler color flow mapping: Quantitative assessment of mitral regurgitation severity in dogs. J. Vet. Cardiol. 2003, 5, 33–38. [Google Scholar] [CrossRef]
- Gouni, V.; Serres, F.J.; Pouchelon, J.-L.; Tissier, R.; Lefebvre, H.P.; Nicolle, A.P.; Sampedrano, C.C.; Chetboul, V. Quantification of mitral valve regurgitation in dogs with degenerative mitral valve disease by use of the proximal isovelocity surface area method. J. Am. Vet. Med. Assoc. 2007, 231, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Biner, S.; Rafique, A.; Rafii, F.; Tolstrup, K.; Noorani, O.; Shiota, T.; Gurudevan, S.; Siegel, R.J. Reproducibility of proximal isovelocity surface area, vena contracta, and regurgitant jet area for assessment of mitral regurgitation severity. JACC Cardiovasc. Imaging 2010, 3, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Zoghbi, W.A.; Adams, D.; Bonow, R.O.; Enriquez-Sarano, M.; Foster, E.; Grayburn, P.A.; Hahn, R.T.; Han, Y.; Hung, J.; Lang, R.M.; et al. Recommendations for noninvasive evaluation of native valvular regurgitation: A report from the american society of echocardiography developed in collaboration with the society for cardiovascular magnetic resonance. J. Am. Soc. Echocardiogr. 2017, 30, 303–371. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.F.; Eidenvall, L.; Ask, P.; Loyd, D.; Wranne, B. Understanding continuous-wave Doppler signal intensity as a measure of regurgitant severity. J. Am. Soc. Echocardiogr. 1997, 10, 613–622. [Google Scholar] [CrossRef]
- Di Marcello, M.; Terzo, E.; Locatelli, C.; Palermo, V.; Sala, E.; Dall’Aglio, E.; Bussadori, C.M.; Spalla, I.; Brambilla, P.G. Assessment of mitral regurgitation severity by Doppler color flow mapping of the vena contracta in dogs. J. Vet. Intern. Med. 2014, 28, 1206–1213. [Google Scholar] [CrossRef] [PubMed]
- Dillon, A.R.; Dell’Italia, L.J.; Tillson, M.; Killingsworth, C.; Denney, T.; Hathcock, J.; Botzman, L. Left ventricular remodeling in preclinical experimental mitral regurgitation of dogs. J. Vet. Cardiol. 2012, 14, 73–92. [Google Scholar] [CrossRef] [PubMed]
- Lord, P.; Hansson, K.; Kvart, C.; Häggström, J. Rate of change of heart size before congestive heart failure in dogs with mitral regurgitation. J. Small Anim. Pract. 2010, 51, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, C.A.; Brown, D.C.; Rush, J.E.; Fox, P.R.; Nguyenba, T.P.; Lehmkuhl, L.B.; Gordon, S.G.; Kellihan, H.B.; Stepien, R.L.; Lefbom, B.K.; et al. Prediction of first onset of congestive heart failure in dogs with degenerative mitral valve disease: The PREDICT cohort study. J. Vet. Cardiol. 2012, 14, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Rishniw, M.; Erb, H.N. Evaluation of four 2-dimensional echocardiographic methods of assessing left atrial size in dogs. J. Vet. Intern. Med. 2000, 14, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Hansson, K.; Häggström, J.; Kvart, C.; Lord, P. Left atrial to aortic root indices using two-dimensional and M-mode echocardiography in cavalier King Charles spaniels with and without left atrial enlargement. Vet. Radiol. Ultrasound 2002, 43, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Wesselowski, S.; Borgarelli, M.; Bello, N.M.; Abbott, J. Discrepancies in identification of left atrial enlargement using left atrial volume versus left atrial-to-aortic root ratio in dogs. J. Vet. Intern. Med. 2014, 28, 1527–1533. [Google Scholar] [CrossRef] [PubMed]
- Häggström, J.; Boswood, A.; O’Grady, M.; Jöns, O.; Smith, S.; Swift, S.; Borgarelli, M.; Gavaghan, B.; Kresken, J.-G.G.; Patteson, M.; et al. Effect of pimobendan or benazepril hydrochloride on survival times in dogs with congestive heart failure caused by naturally occurring myxomatous mitral valve disease: The QUEST study. J. Vet. Intern. Med. 2008, 22, 1124–1135. [Google Scholar] [CrossRef] [PubMed]
- Höllmer, M.; Willesen, J.L.; Tolver, A.; Koch, J. Left atrial volume and function in dogs with naturally occurring myxomatous mitral valve disease. J. Vet. Cardiol. 2017, 19, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Teichholz, L.E.; Kreulen, T.; Herman, M.V.; Gorlin, R. Problems in echocardiographic volume determinations: Echocardiographic-angiographic correlations in the presence or absence of asynergy. Am. J. Cardiol. 1976, 37, 7–11. [Google Scholar] [CrossRef]
- Boon, J.; Wingfield, W.E.; Miller, C.W. Echocardiographic indices in the normal dog. Vet. Radiol. 1983, 24, 214–221. [Google Scholar] [CrossRef]
- Goncalves, A.C.; Orton, E.C.; Boon, J.A.; Salman, M.D. Linear, logarithmic, and polynomial models of M-mode echocardiographic measurements in dogs. Am. J. Vet. Res. 2002, 63, 994–999. [Google Scholar] [CrossRef] [PubMed]
- Cornell, C.C.; Kittleson, M.D.; Della Torre, P.; Häggström, J.; Lombard, C.W.; Pedersen, H.D.; Vollmar, A.; Wey, A. Allometric scaling of M-mode cardiac measurements in normal adult dogs. J. Vet. Intern. Med. 2004, 18, 311–321. [Google Scholar] [CrossRef] [PubMed]
- McGinley, J.C.; Berretta, R.M.; Chaudhary, K.; Rossman, E.; Bratinov, G.D.; Gaughan, J.P.; Houser, S.; Margulies, K.B. Impaired contractile reserve in severe mitral valve regurgitation with a preserved ejection fraction. Eur. J. Heart Fail. 2007, 9, 857–864. [Google Scholar] [CrossRef] [PubMed]
- Tidholm, A.; Nicolle, A.P.; Carlos, C.; Gouni, V.; Caruso, J.L.; Pouchelon, J.L.; Chetboul, V. Tissue Doppler imaging and echo-Doppler findings associated with a mitral valve stenosis with an immobile posterior valve leaflet in a bull terrier. Transbound. Emerg. Dis. 2004, 51, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Bonagura, J.D.; Schober, K.E. Can ventricular function be assessed by echocardiography in chronic canine mitral valve disease? J. Small Anim. Pract. 2009, 50, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Park, H.-M. Usefulness of conventional and tissue doppler echocardiography to predict congestive heart failure in dogs with myxomatous mitral valve disease. J. Vet. Intern. Med. 2015, 29, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Baron Toaldo, M.; Poser, H.; Menciotti, G.; Battaia, S.; Contiero, B.; Cipone, M.; Diana, A.; Mazzotta, E.; Guglielmini, C. Utility of tissue doppler imaging in the echocardiographic evaluation of left and right ventricular function in dogs with myxomatous mitral valve disease with or without pulmonary hypertension. J. Vet. Intern. Med. 2016, 30, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Lisauskas, J.; Singh, J.; Courtois, M.; Kovács, S.J. The relation of the peak Doppler E-wave to peak mitral annulus velocity ratio to diastolic function. Ultrasound Med. Biol. 2001, 27, 499–507. [Google Scholar] [CrossRef]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F.; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P.; et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: An update from the American society of echocardiography and the European association of cardiovascular imaging. J. Am. Soc. Echocardiogr. 2016, 29, 277–314. [Google Scholar] [CrossRef] [PubMed]
- Schober, K.E.; Bonagura, J.D.; Scansen, B.A.; Stern, J.A.; Ponzio, N.M. Estimation of left ventricular filling pressure by use of Doppler echocardiography in healthy anesthetized dogs subjected to acute volume loading. Am. J. Vet. Res. 2008, 69, 1034–1049. [Google Scholar] [CrossRef] [PubMed]
- Jacques, D.C.; Pinsky, M.R.; Severyn, D.; Gorcsan, J. Influence of alterations in loading on mitral annular velocity by tissue doppler echocardiography and its associated ability to predict filling pressures. Chest 2004, 126, 1910–1918. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.N.; Bonagura, J.D.; Culwell, N.M.; Schober, K.E. Left ventricular function quantified by myocardial strain imaging in small-breed dogs with chronic mitral regurgitation. J. Vet. Cardiol. 2012, 14, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Tidholm, A.; Ljungvall, I.; Höglund, K.; Westling, A.B.; Häggström, J. Tissue doppler and strain imaging in dogs with myxomatous mitral valve disease in different stages of congestive heart failure. J. Vet. Intern. Med. 2009, 23, 1197–1207. [Google Scholar] [CrossRef] [PubMed]
- Serres, F.; Chetboul, V.; Gouni, V.; Tissier, R.; Sampedrano, C.C.; Pouchelon, J.-L. Diagnostic value of echo-Doppler and tissue Doppler imaging in dogs with pulmonary arterial hypertension. J. Vet. Intern. Med. 2007, 21, 1280–1289. [Google Scholar] [CrossRef] [PubMed]
- Soydan, L.C.; Kellihan, H.B.; Bates, M.L.; Stepien, R.L.; Consigny, D.W.; Bellofiore, A.; Francois, C.J.; Chesler, N.C. Accuracy of Doppler echocardiographic estimates of pulmonary artery pressures in a canine model of pulmonary hypertension. J. Vet. Cardiol. 2015, 17, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Testani, J.M.; St. John Sutton, M.G.; Wiegers, S.E.; Khera, A.V.; Shannon, R.P.; Kirkpatrick, J.N. Accuracy of noninvasively determined pulmonary artery systolic pressure. Am. J. Cardiol. 2010, 105, 1192–1197. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Matsuo, H.; Kitabatake, A.; Inoue, M.; Hamanaka, Y.; Tamura, S.; Tanaka, K.; Abe, H. Three-dimensional echocardiograms and two-dimensional echocardiographic images at desired planes by a computerized system. Ultrasound Med. Biol. 1977, 3, 163–178. [Google Scholar] [CrossRef]
- Geiser, E.A.; Ariet, M.; Conetta, D.A.; Lupkiewicz, S.M.; Christie, L.G.; Conti, C.R. Dynamic three-dimensional echocardiographic reconstruction of the intact human left ventricle: Technique and initial observations in patients. Am. Heart J. 1982, 103, 1056–1065. [Google Scholar] [CrossRef]
- Ghosh, A.; Nanda, N.C.; Maurer, G. Three-dimensional reconstruction of echocardiographic images using the rotation method. Ultrasound Med. Biol. 1982, 8, 655–661. [Google Scholar] [CrossRef]
- Matsumoto, M.; Inoue, M.; Tamura, S.; Tanaka, K.; Abe, H. Three-dimensional echocardiography for spatial visualization and volume calculation of cardiac structures. J. Clin. Ultrasound 1981, 9, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.A.; Handschumacher, M.D.; Sanfilippo, A.J.; Hagege, A.A.; Harrigan, P.; Marshall, J.E.; Weyman, A.E. Three-dimensional echocardiographic reconstruction of the mitral valve, with implications for the diagnosis of mitral valve prolapse. Circulation 1989, 80, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Bax, J.J.; Delgado, V. Advanced imaging in valvular heart disease. Nat. Rev. Cardiol. 2017, 14, 209–223. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American society of echocardiography and the European association of cardiovascular imaging. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 233–271. [Google Scholar] [CrossRef] [PubMed]
- Eaton, L.W.; Maughan, W.L.; Shoukas, A.A.; Weiss, J.L. Accurate volume determination in the isolated ejecting canine left ventricle by two-dimensional echocardiography. Circulation 1979, 60, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Tidholm, A.; Westling, A.B.B.; Höglund, K.; Ljungvall, I.; Häggström, J. Comparisons of 3-, 2-Dimensional, and M-Mode echocardiographical methods for estimation of left chamber volumes in dogs with and without acquired heart disease. J. Vet. Intern. Med. 2010, 24, 1414–1420. [Google Scholar] [CrossRef] [PubMed]
- Tidholm, A.; Bodegård-Westling, A.; Höglund, K.; Ljungvall, I.; Häggström, J. Comparisons of 2- and 3-dimensional echocardiographic methods for estimation of left atrial size in dogs with and without myxomatous mitral valve disease. J. Vet. Intern. Med. 2011, 25, 1320–1327. [Google Scholar] [CrossRef] [PubMed]
- Ljungvall, I.; Höglund, K.; Carnabuci, C.; Tidholm, A.; Häggström, J. Assessment of global and regional left ventricular volume and shape by real-time 3-dimensional echocardiography in dogs with myxomatous mitral valve disease. J. Vet. Intern. Med. 2011, 25, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Menciotti, G.; Borgarelli, M.; Aherne, M.; Häggström, J.; Ljungvall, I.; Lahmers, S.M.; Abbott, J.A. Assessment of mitral valve morphology using three-dimensional echocardiography. Feasibility and reference values. J. Vet. Cardiol. 2016, 18, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Menciotti, G.; Borgarelli, M.; Aherne, M.; Wesselowski, S.; Häggström, J.; Ljungvall, I.; Lahmers, S.M.; Abbott, J.A. Mitral valve morphology assessed by three-dimensional transthoracic echocardiography in healthy dogs and dogs with myxomatous mitral valve disease. J. Vet. Cardiol. 2017, 19, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Boswood, A.; Häggström, J.; Gordon, S.G.; Wess, G.; Stepien, R.L.; Oyama, M.A.; Keene, B.W.; Bonagura, J.; MacDonald, K.A.; Patteson, M.; et al. Effect of pimobendan in dogs with preclinical myxomatous mitral valve disease and cardiomegaly: The EPIC Study—A Randomized clinical trial. J. Vet. Intern. Med. 2016, 30, 1765–1779. [Google Scholar] [CrossRef] [PubMed]
- Kvart, C.; Häggström, J.; Pedersen, H.D.; Hansson, K.; Eriksson, A.; Järvinen, A.-K.; Tidholm, A.; Bsenko, K.; Ahlgren, E.; Lives, M.; et al. Efficacy of enalapril for prevention of congestive heart failure in dogs with myxomatous valve disease and asymptomatic mitral regurgitation. J. Vet. Intern. Med. 2002, 16, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Atkins, C.E.; Keene, B.W.; Brown, W.A.; Coats, J.R.; Crawford, M.A.; DeFrancesco, T.C.; Edwards, N.J.; Fox, P.R.; Lehmkuhl, L.B.; Luethy, M.W.; et al. Results of the veterinary enalapril trial to prove reduction in onset of heart failure in dogs chronically treated with enalapril alone for compensated, naturally occurring mitral valve insufficiency. J. Am. Vet. Med. Assoc. 2007, 231, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Bernay, F.; Bland, J.M.; Häggström, J.; Baduel, L.; Combes, B.; Lopez, A.; Kaltsatos, V.; Ha, J. Efficacy of spironolactone on survival in dogs with naturally occurring mitral regurgitation caused by myxomatous mitral valve disease. J. Vet. Intern. Med. 2010, 24, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Pouchelon, J.-L.; Jamet, N.; Gouni, V.; Tissier, R.; Serres, F.; Carlos Sampedrano, C.; Castaignet, M.; Lefebvre, H.P.; Chetboul, V. Effect of benazepril on survival and cardiac events in dogs with asymptomatic mitral valve disease: A retrospective study of 141 cases. J. Vet. Intern. Med. 2008, 22, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Lombard, C.W.; Jöns, O.; Bussadori, C.M. Clinical efficacy of pimobendan versus benazepril for the treatment of acquired atrioventricular valvular disease in dogs. J. Am. Anim. Hosp. Assoc. 2006, 42, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Häggström, J.; Boswood, A.; O’Grady, M.; Jöns, O.; Smith, S.; Swift, S.; Borgarelli, M.; Gavaghan, B.; Kresken, J.G.; Patteson, M.; et al. Longitudinal analysis of quality of life, clinical, radiographic, echocardiographic, and laboratory variables in dogs with myxomatous mitral valve disease receiving pimobendan or benazepril: The QUEST study. J. Vet. Intern. Med. 2013, 27, 1441–1451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferasin, L.; Crews, L.; Biller, D.S.; Lamb, K.E.; Borgarelli, M. Risk factors for coughing in dogs with naturally acquired myxomatous mitral valve disease. J. Vet. Intern. Med. 2013, 27, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Guglielmini, C.; Diana, A.; Pietra, M.; Di Tommaso, M.; Cipone, M. Use of the vertebral heart score in coughing dogs with chronic degenerative mitral valve disease. J. Vet. Med. Sci. 2009, 71, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Uechi, M.; Matsuoka, M.; Kuwajima, E.; Kaneko, T.; Yamashita, K.; Fukushima, U.; Ishikawa, Y. The effects of the loop diuretics furosemide and torasemide on diuresis in dogs and cats. J. Vet. Med. Sci. 2003, 65, 1057–1061. [Google Scholar] [CrossRef] [PubMed]
- Oyama, M.A.; Peddle, G.D.; Reynolds, C.A.; Singletary, G.E. Use of the loop diuretic torsemide in three dogs with advanced heart failure. J. Vet. Cardiol. 2011, 13, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Hori, Y.; Takusagawa, F.; Ikadai, H.; Uechi, M.; Hoshi, F.; Higuchi, S. Effects of oral administration of furosemide and torsemide in healthy dogs. Am. J. Vet. Res. 2007, 68, 1058–1063. [Google Scholar] [CrossRef] [PubMed]
- Uchida, T.; Hayashi, K.; Suzuki, Y.; Matsumura, Y. Effects of torasemide on renal haemodynamics and function in anaesthetized dogs. Clin. Exp. Pharmacol. Physiol. 1991, 18, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Chiavegato, D.; Borgarelli, M.; D’Agnolo, G.; Santilli, R.A. Pulmonary hypertension in dogs with mitral regurgitation attributable to myxomatous valve disease. Vet. Radiol. Ultrasound 2009, 50, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Kellihan, H.B.; Stepien, R.L. Pulmonary hypertension in canine degenerative mitral valve disease. J. Vet. Cardiol. 2012, 14, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Borgarelli, M.; Abbott, J.; Braz-Ruivo, L.; Chiavegato, D.; Crosara, S.; Lamb, K.; Ljungvall, I.; Poggi, M.; Santilli, R.A.; Haggstrom, J. Prevalence and prognostic importance of pulmonary hypertension in dogs with myxomatous mitral valve disease. J. Vet. Intern. Med. 2015, 29, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Davison, E.; Sleeper, M.M. Clinical efficacy of sildenafil in treatment of pulmonary arterial hypertension in dogs. J. Vet. Intern. Med. 2010, 24, 850–854. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.F.; Rozanski, E.A.; MacGregor, J.; Betkowski, J.M.; Rush, J.E. Retrospective evaluation of sildenafil citrate as a therapy for pulmonary hypertension in dogs. J. Vet. Intern. Med. 2006, 20, 1132–1135. [Google Scholar] [CrossRef] [PubMed]
- Kellum, H.B.; Stepien, R.L. Sildenafil citrate therapy in 22 dogs with pulmonary hypertension. J. Vet. Intern. Med. 2007, 21, 1258–1264. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, R.A.; Otto, C.M.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P.; Guyton, R.A.; O’Gara, P.T.; Ruiz, C.E.; Skubas, N.J.; Sorajja, P.; et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease. J. Am. Coll. Cardiol. 2014, 63, e57–e185. [Google Scholar] [CrossRef] [PubMed]
- Uechi, M. Mitral valve repair in dogs. J. Vet. Cardiol. 2012, 14, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Uechi, M.; Mizukoshi, T.; Mizuno, T.; Mizuno, M.; Harada, K.; Ebisawa, T.; Takeuchi, J.; Sawada, T.; Uchida, S.; Shinoda, A.; et al. Mitral valve repair under cardiopulmonary bypass in small-breed dogs: 48 Cases (2006–2009). J. Am. Vet. Med. Assoc. 2012, 240, 1194–1201. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, T.; Mizukoshi, T.; Uechi, M. Long-term outcome in dogs undergoing mitral valve repair with suture annuloplasty and chordae tendinae replacement. J. Small Anim. Pract. 2013, 54, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Nishida, M.; Kagawa, Y.; Mizukoshi, T.; Mizuno, M.; Mizuno, T.; Harada, K.; Uechi, M. Post-mortem evaluation of expanded polytetrafluoroethylene (ePTFE) used in mitral valve repair in dogs. J. Vet. Cardiol. 2012, 14, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Klement, P.; Feindel, C.M.; Scully, H.E.; Mesher, E.; Klement, G.; Del Nido, P.; Wilson, G.J. Mitral valve replacement in dogs. Surgical technique and postoperative management. Vet. Surg. 1987, 16, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Orton, E.C.; Hackett, T.B.; Mama, K.; Boon, J.A. Technique and outcome of mitral valve replacement in dogs. J. Am. Vet. Med. Assoc. 2005, 226, 1508–1511. [Google Scholar] [CrossRef] [PubMed]
- White, R.N.; Stepien, R.L.; Hammond, R.A.; Holden, D.J.; Torrington, A.M.; Milner, H.R.; Cobb, M.A.; Hellens, S.H. Mitral valve replacement for the treatment of congenital mitral dysplasia in a bull terrier. J. Small Anim. Pract. 1995, 36, 407–410. [Google Scholar] [CrossRef] [PubMed]
- White, R.N.; Boswood, A.; Garden, O.A.; Hammond, R.A. Surgical management of subvalvular aortic stenosis and mitral dysplasia in a golden retriever. J. Small Anim. Pract. 1997, 38, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Takashima, K.; Soda, A.; Tanaka, R.; Yamane, Y. Long-term clinical evaluation of mitral valve replacement with porcine bioprosthetic valves in dogs. J. Vet. Med. Sci. 2008, 70, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Feldman, T.; Wasserman, H.S.; Herrmann, H.C.; Gray, W.; Block, P.C.; Whitlow, P.; St. Goar, F.; Rodriguez, L.; Silvestry, F.; Schwartz, A.; et al. Percutaneous mitral valve repair using the edge-to-edge technique: Six-month results of the EVEREST phase I clinical trial. J. Am. Coll. Cardiol. 2005, 46, 2134–2140. [Google Scholar] [CrossRef] [PubMed]
- Feldman, T.; Kar, S.; Rinaldi, M.; Fail, P.; Hermiller, J.; Smalling, R.; Whitlow, P.L.; Gray, W.; Low, R.; Herrmann, H.C.; et al. Percutaneous mitral repair with the MitraClip system: Safety and midterm durability in the initial EVEREST (Endovascular Valve Edge-to-Edge REpair Study) cohort. J. Am. Coll. Cardiol. 2009, 54, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Whitlow, P.L.; Feldman, T.; Pedersen, W.R.; Lim, D.S.; Kipperman, R.; Smalling, R.; Bajwa, T.; Herrmann, H.C.; Lasala, J.; Maddux, J.T.; et al. Acute and 12-month results with catheter-based mitral valve leaflet repair: The EVEREST II (Endovascular Valve Edge-to-Edge Repair) High Risk Study. J. Am. Coll. Cardiol. 2012, 59, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Seeburger, J.; Rinaldi, M.; Nielsen, S.L.; Salizzoni, S.; Lange, R.; Schoenburg, M.; Alfieri, O.; Borger, M.A.; Mohr, F.W.; Aidietis, A. Off-pump transapical implantation of artificial neo-chordae to correct mitral regurgitation: The tact trial (transapical artificial chordae tendinae) proof of concept. J. Am. Coll. Cardiol. 2014, 63, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Gammie, J.S.; Wilson, P.; Bartus, K.; Gackowski, A.; Hung, J.; D’Ambra, M.N.; Kolsut, P.; Bittle, G.J.; Szymanski, P.; Sadowski, J.; et al. Transapical beating-heart mitral valve repair with an expanded polytetrafluoroethylene cordal implantation device. Circulation 2016, 134, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Borgarelli, M.; Lanz, O.; Pavlisko, N.; Abbott, J.A.; Menciotti, G.; Aherne, M.; Lahmers, S.M.; Lahmers, K.K.; Gammie, J.S. Mitral valve repair in dogs using an ePTFE chordal implantation device: A pilot study. J. Vet. Cardiol. 2017, 19, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Partida, R.A.; Elmariah, S. Transcatheter mitral valve interventions: Current therapies and future directions. Curr. Treat. Opt. Cardiovasc. Med. 2017, 19, 32. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menciotti, G.; Borgarelli, M. Review of Diagnostic and Therapeutic Approach to Canine Myxomatous Mitral Valve Disease. Vet. Sci. 2017, 4, 47. https://doi.org/10.3390/vetsci4040047
Menciotti G, Borgarelli M. Review of Diagnostic and Therapeutic Approach to Canine Myxomatous Mitral Valve Disease. Veterinary Sciences. 2017; 4(4):47. https://doi.org/10.3390/vetsci4040047
Chicago/Turabian StyleMenciotti, Giulio, and Michele Borgarelli. 2017. "Review of Diagnostic and Therapeutic Approach to Canine Myxomatous Mitral Valve Disease" Veterinary Sciences 4, no. 4: 47. https://doi.org/10.3390/vetsci4040047
APA StyleMenciotti, G., & Borgarelli, M. (2017). Review of Diagnostic and Therapeutic Approach to Canine Myxomatous Mitral Valve Disease. Veterinary Sciences, 4(4), 47. https://doi.org/10.3390/vetsci4040047





