Stimulation with Concanavalin-A Induces IL-17 Production by Canine Peripheral T Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Mitogen-Stimulation Promotes IL-17 Production by Canine CD4 and CD8 T Cells
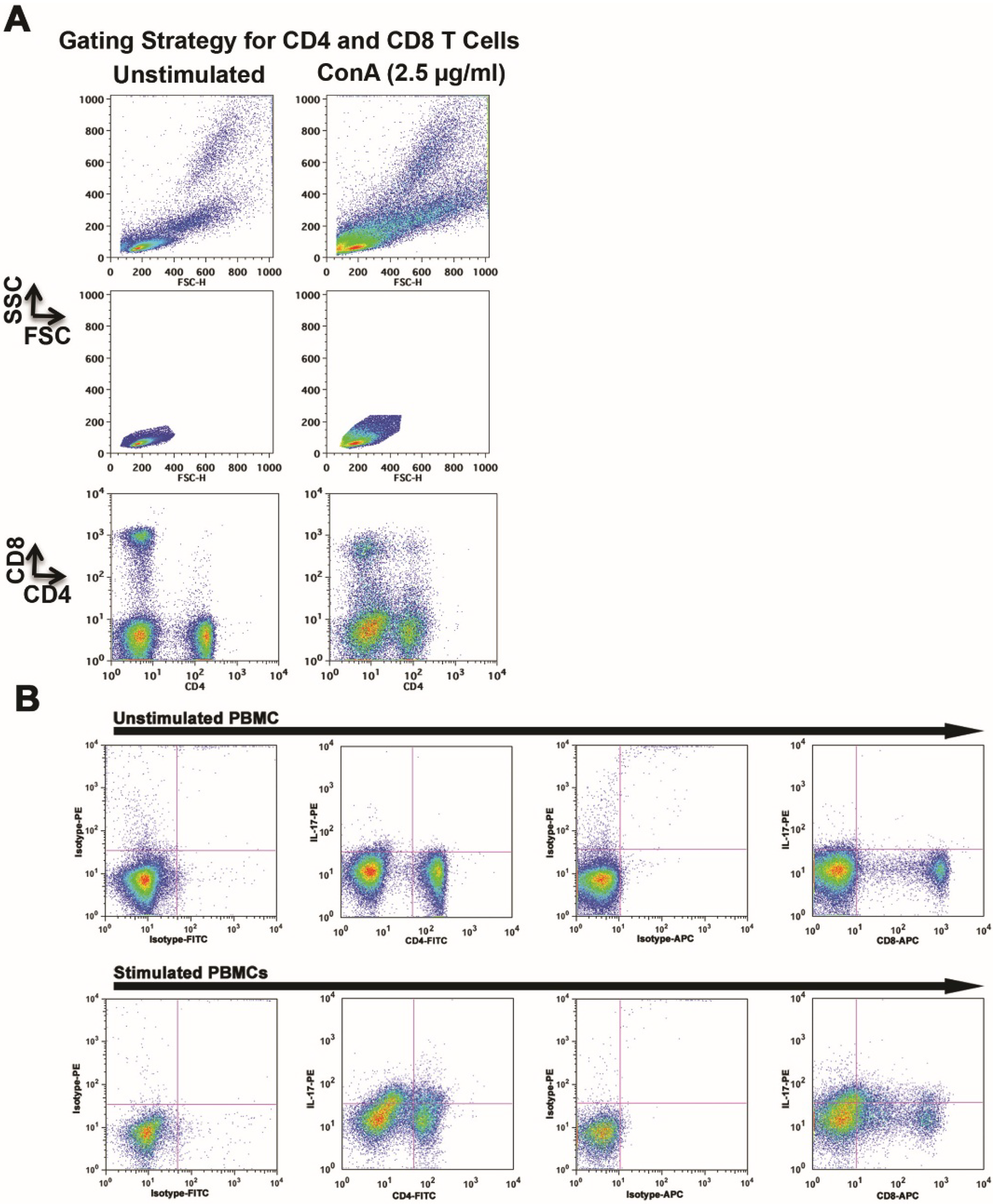
| %IL-17-Producing Cells in ConA Blasts (6 h) | ||
|---|---|---|
| Dog # | CD4 | CD8 |
| 1 | 12.4 | 17.6 |
| 2 | 4.5 | 4.8 |
| 3 | 11 | 5.2 |
| 4 | 1.0 | 8.8 |
2.2. Pro-Inflammatory Cytokines Promote Polarization of Canine T Helper Cells towards the Th17 Phenotype
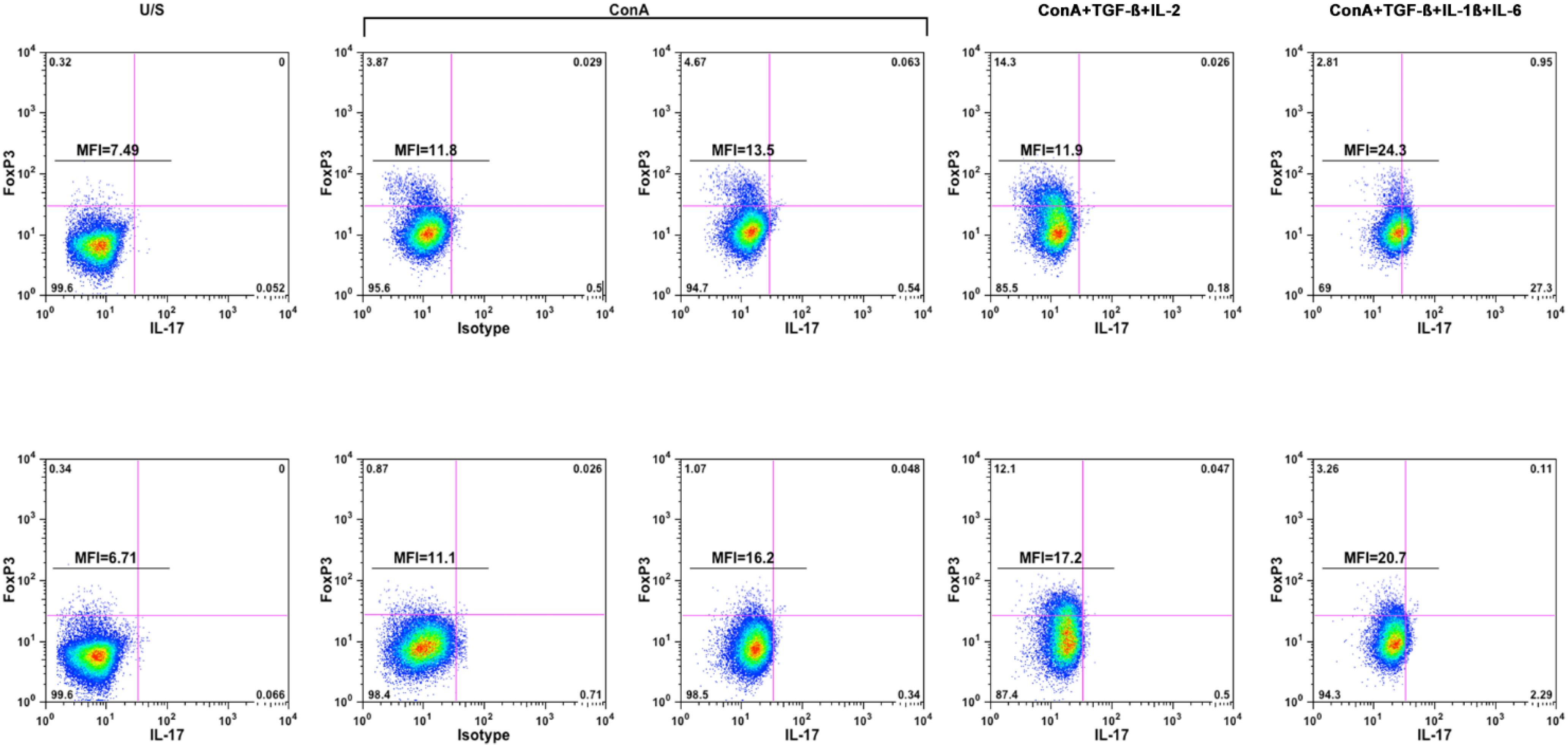
| Condition | Dog-1 | Dog-2 |
| IL-17 (±S. D.; ng/mL) | ||
| Unstimulated | 0 (0) | 4.5 (0.8) |
| ConA | 8.3 (0.3) | 8.4 (1.7) |
| ConA+TGF-ß+IL-2 | 8.4 (1.0) | 9.8 (0.4) |
| ConA+TGF-ß+IL-1ß+IL-6 | 9.5 (2.7) | 9.4 (0.3) |
3. Materials and Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Awasthi, A.; Kuchroo, V.K. Th17 cells: From precursors to players in inflammation and infection. Int. Immunol. 2009, 21, 489–498. [Google Scholar]
- Gutcher, I.; Donkor, M.K.; Ma, Q.; Rudensky, A.Y.; Flavell, R.A.; Li, M.O. Autocrine transforming growth factor-beta1 promotes in vivo th17 cell differentiation. Immunity 2011, 34, 396–408. [Google Scholar] [CrossRef] [PubMed]
- Bedoya, S.K.; Lam, B.; Lau, K.; Larkin, J., 3rd. Th17 cells in immunity and autoimmunity. Clin. Dev. Immunol. 2013. [Google Scholar] [CrossRef]
- Schmitz, S.; Garden, O.A.; Werling, D.; Allenspach, K. Gene expression of selected signature cytokines of T cell subsets in duodenal tissues of dogs with and without inflammatory bowel disease. Vet. Immunol. Immunopathol. 2012, 146, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Pan, H.F.; Ye, D.Q. Tc17 cells in immunity and systemic autoimmunity. Int. Rev. Immunol. 2014. [Google Scholar] [CrossRef]
- Mou, Z.; Liu, D.; Okwor, I.; Jia, P.; Orihara, K.; Uzonna, J.E. Mhc class II restricted innate-like double negative T cells contribute to optimal primary and secondary immunity to leishmania major. PLoS Pathog. 2014, 10, e1004396. [Google Scholar] [CrossRef] [PubMed]
- Jergens, A.E.; Sonea, I.M.; O’Connor, A.M.; Kauffman, L.K.; Grozdanic, S.D.; Ackermann, M.R.; Evans, R.B. Intestinal cytokine mrna expression in canine inflammatory bowel disease: A meta-analysis with critical appraisal. Comp. Med. 2009, 59, 153–162. [Google Scholar] [PubMed]
- Ohta, H.; Takada, K.; Torisu, S.; Yuki, M.; Tamura, Y.; Yokoyama, N.; Osuga, T.; Lim, S.Y.; Murakami, M.; Sasaki, N.; et al. Expression of CD4+ T cell cytokine genes in the colorectal mucosa of inflammatory colorectal polyps in miniature dachshunds. Vet. Immunol. Immunopathol. 2013, 155, 259–263. [Google Scholar]
- Frantz, A.M.; Sarver, A.L.; Ito, D.; Phang, T.L.; Karimpour-Fard, A.; Scott, M.C.; Valli, V.E.; Lindblad-Toh, K.; Burgess, K.E.; Husbands, B.D.; et al. Molecular profiling reveals prognostically significant subtypes of canine lymphoma. Vet. Pathol. 2013, 50, 693–703. [Google Scholar]
- Lenarczyk, A.; Helsloot, J.; Farmer, K.; Peters, L.; Sturgess, A.; Kirkham, B. Antigen-induced IL-17 response in the peripheral blood mononuclear cells (PBMC) of healthy controls. Clin. Exp. Immunol. 2000, 122, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Aprile, J.; Gerhard-Miller, L.; Deeg, H.J. Cluster formation of canine dendritic cells and lymphocytes is calcium dependent and not inhibited by cyclosporine. Exp. Hematol. 1990, 18, 32–36. [Google Scholar] [PubMed]
- Korn, T.; Bettelli, E.; Oukka, M.; Kuchroo, V.K. IL-17 and Th17 cells. Annu. Rev. Immunol. 2009, 27, 485–517. [Google Scholar] [CrossRef] [PubMed]
- Lexberg, M.H.; Taubner, A.; Forster, A.; Albrecht, I.; Richter, A.; Kamradt, T.; Radbruch, A.; Chang, H.D. Th memory for interleukin-17 expression is stable in vivo. Eur. J. Immunol. 2008, 38, 2654–2664. [Google Scholar] [CrossRef] [PubMed]
- Helfand, S.C.; Modiano, J.F.; Nowell, P.C. Immunophysiological studies of interleukin-2 and canine lymphocytes. Vet. Immunol. Immunopathol. 1992, 33, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ito, D.; O’Brien, T.D.; Modiano, J.F. Exclusion of cytoplasmic fragments in flow cytometric analysis of lymph node samples from dogs with lymphoma using membrane-permeable violet laser-excitable DNA-binding fluorescent dye (dyecycle violet). Vet. Clin. Pathol. 2010, 39, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.L.; Lin, G.G.; Zhang, K.; Wang, L.N.; Li, J.M. Application and effects of mouse foxp3 antibody and fixation/permeabilization buffer on the detection of CD4+ regulatory T cells in various mammal species. Genet. Mol. Res. 2013, 12, 6535–6545. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ritt, M.G.; Lindborg, B.A.; O'Brien, T.D.; Bisignano, J.; Modiano, J.F. Stimulation with Concanavalin-A Induces IL-17 Production by Canine Peripheral T Cells. Vet. Sci. 2015, 2, 43-51. https://doi.org/10.3390/vetsci2020043
Ritt MG, Lindborg BA, O'Brien TD, Bisignano J, Modiano JF. Stimulation with Concanavalin-A Induces IL-17 Production by Canine Peripheral T Cells. Veterinary Sciences. 2015; 2(2):43-51. https://doi.org/10.3390/vetsci2020043
Chicago/Turabian StyleRitt, Michelle G., Beth A. Lindborg, Timothy D. O'Brien, Joseph Bisignano, and Jaime F. Modiano. 2015. "Stimulation with Concanavalin-A Induces IL-17 Production by Canine Peripheral T Cells" Veterinary Sciences 2, no. 2: 43-51. https://doi.org/10.3390/vetsci2020043
APA StyleRitt, M. G., Lindborg, B. A., O'Brien, T. D., Bisignano, J., & Modiano, J. F. (2015). Stimulation with Concanavalin-A Induces IL-17 Production by Canine Peripheral T Cells. Veterinary Sciences, 2(2), 43-51. https://doi.org/10.3390/vetsci2020043






