Effects of Intra-uterine Ceftiofur on the Equine Uterine Microbiome
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Work
2.1.1. Sample Collection
2.1.2. Artificial Insemination
2.2. Laboratory Analysis
2.2.1. Endometrial Culture and Cytology
2.2.2. DNA Extraction, Sequencing, and Metagenomic Analyses
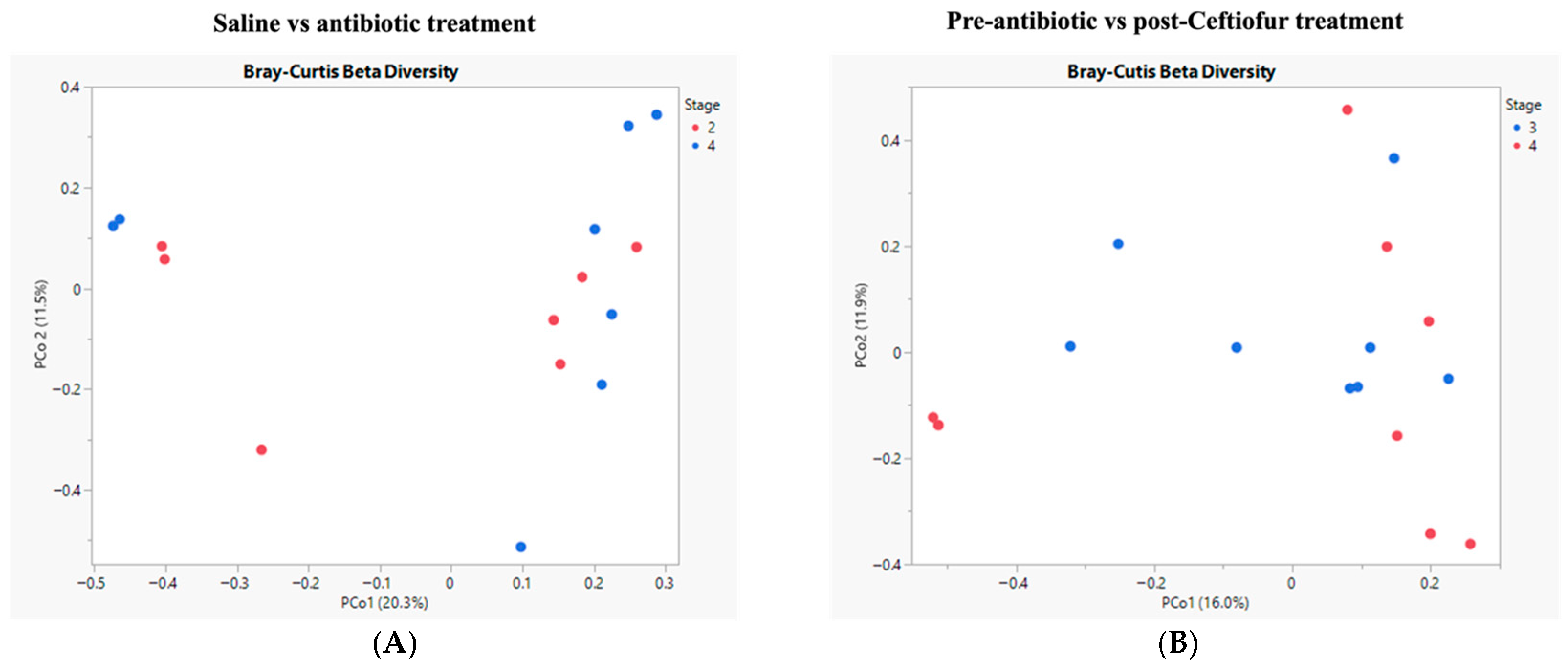
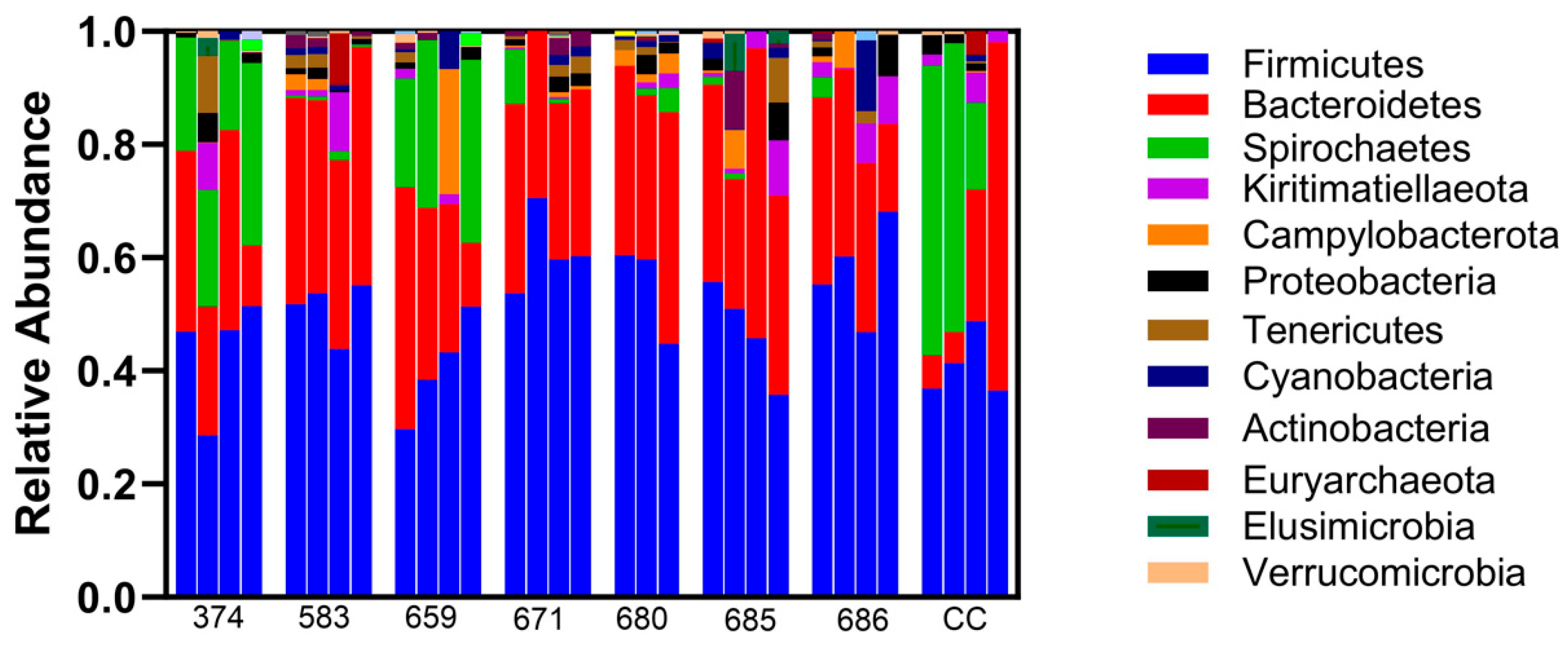
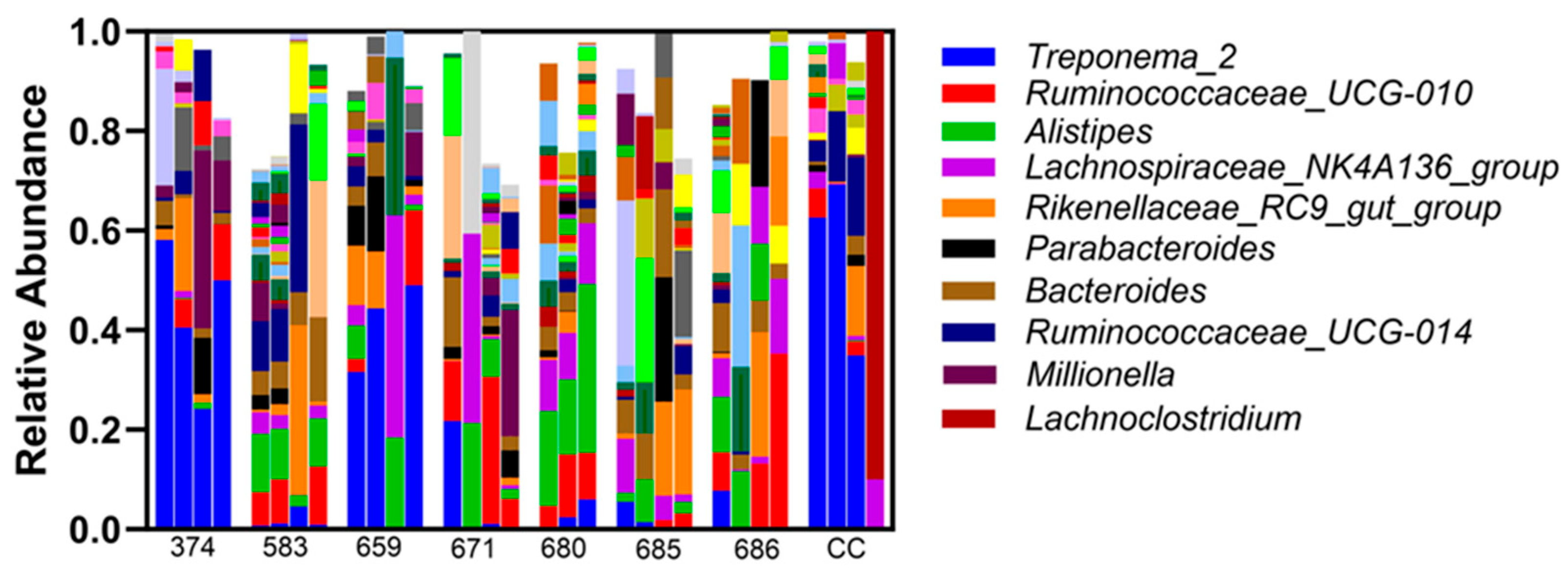
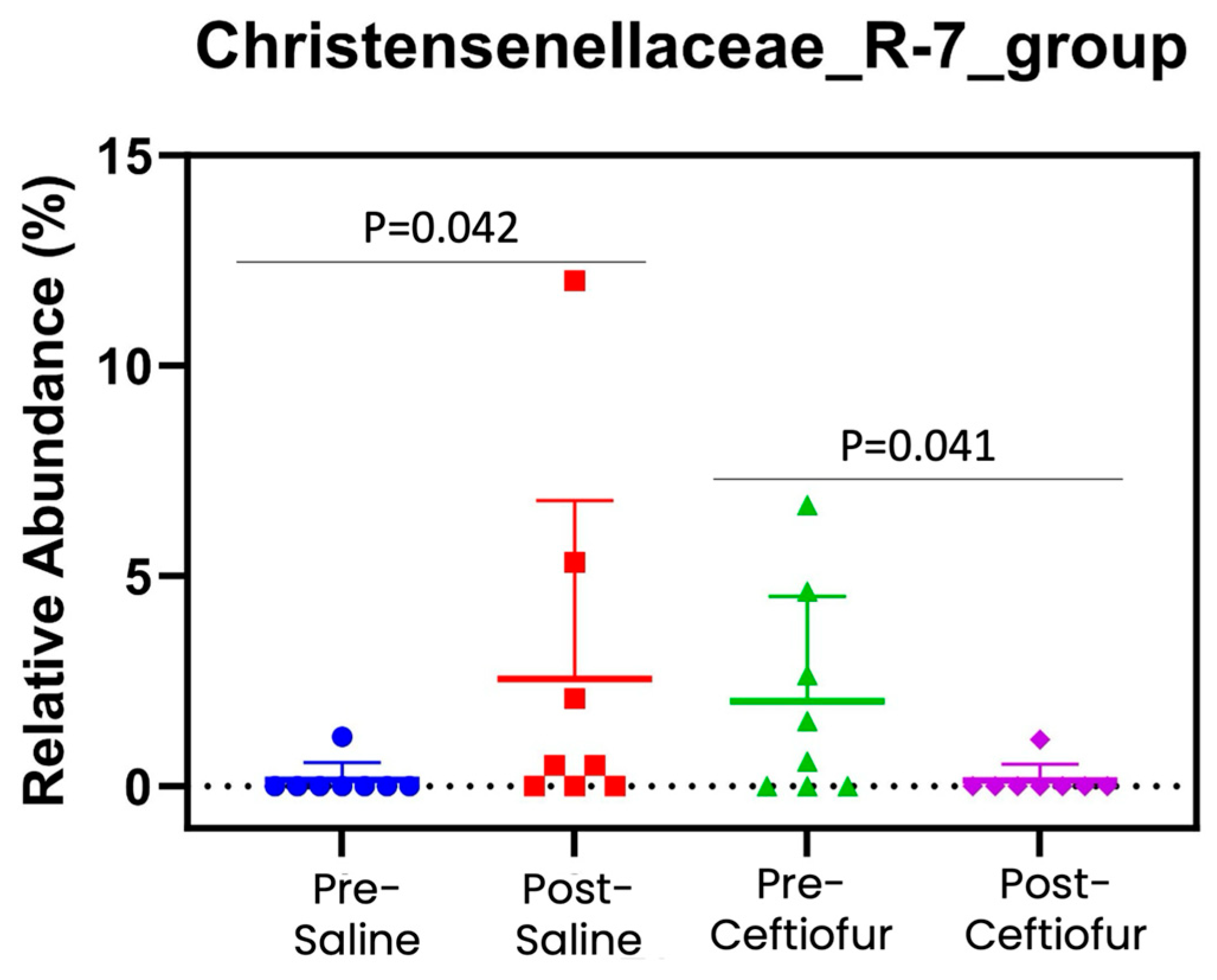
3. Results
Microbiome
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gil-Miranda, A.; Macnicol, J.; Orellana-Guerrero, D.; Samper, J.C.; Gomez, D.E. Reproductive Tract Microbiota of Mares. Vet. Sci. 2024, 11, 324. [Google Scholar] [CrossRef]
- Holyoak, G.R.; Lyman, C.C. The equine endometrial microbiome: A brief review. Am. J. Biomed. Sci. Res. 2021, 11, 532–534. [Google Scholar] [CrossRef]
- Heil, B.A.; van Heule, M.; Thompson, S.K.; Kearns, T.A.; Oberhaus, E.L.; King, G.; Daels, P.; Dini, P.; Sones, J.L. Effect of Sampling Method on Detection of the Equine Uterine Microbiome during Estrus. Vet. Sci. 2023, 10, 644. [Google Scholar] [CrossRef] [PubMed]
- Virendra, A.; Gulavane, S.U.; Ahmed, Z.A.; Reddy, R.; Chaudhari, R.J.; Gaikwad, S.M.; Shelar, R.R.; Ingole, S.D.; Thorat, V.D.; Khanam, A.; et al. Metagenomic analysis unravels novel taxonomic differences in the uterine microbiome between healthy mares and mares with endometritis. Vet. Med. Sci. 2024, 10, e1369. [Google Scholar] [CrossRef]
- Heil, B.A.; van Heule, M.; Thompson, S.K.; Kearns, T.A.; Oberhaus, E.L.; King, G.; Daels, P.; Dini, P.; Sones, J.L. Metagenomic characterization of the equine endometrial microbiome during anestrus. J. Equine Vet. Sci. 2024, 140, 105134. [Google Scholar] [CrossRef]
- Troedsson, M.H. Uterine clearance and resistance to persistent endometritis in the mare. Theriogenology 1999, 52, 461–471. [Google Scholar] [CrossRef]
- Katila, T. Post-mating inflammatory responses of the uterus. Reprod. Domest. Anim. 2012, 47 (Suppl. 5), 31–41. [Google Scholar] [CrossRef]
- Zent, W.W.; Troedsson, M.H.T.; Xue, J.L. Postbreeding uterine fluid accumulation in a normal population of Thoroughbred mares: A field study. In Proceedings of the Annual Convention of the American Association of Equine Practitioners, Baltilmore, MD, USA, 9 December 1998; pp. 64–65. [Google Scholar]
- LeBlanc, M.M.; Causey, R.C. Clinical and subclinical endometritis in the mare: Both threats to fertility. Reprod. Domest. Anim. 2009, 44 (Suppl. 3), 10–22. [Google Scholar] [CrossRef]
- Ball, B.A. Embryonic loss in mares. Incidence, possible causes, and diagnostic considerations. Vet. Clin. North Am. Equine Pract. 1988, 4, 263–290. [Google Scholar] [CrossRef] [PubMed]
- Canisso, I.F.; Segabinazzi, L.G.T.M.; Fedorka, C.E. Persistent Breeding-Induced Endometritis in Mares—A Multifaceted Challenge: From Clinical Aspects to Immunopathogenesis and Pathobiology. Int. J. Mol. Sci. 2020, 21, 1432. [Google Scholar] [CrossRef]
- Nielsen, J.M. Endometritis in the mare: A diagnostic study comparing cultures from swab and biopsy. Theriogenology 2005, 64, 510–518. [Google Scholar] [CrossRef]
- Holyoak, G.R.; Premathilake, H.U.; Lyman, C.C.; Sones, J.L.; Gunn, A.; Wieneke, X.; DeSilva, U. The healthy equine uterus harbors a distinct core microbiome plus a rich and diverse microbiome that varies with geographical location. Sci. Rep. 2022, 12, 14790. [Google Scholar] [CrossRef]
- Ryan, C.A.; McNeal, C.D.; Credille, B.C. Ceftiofur use and antimicrobial stewardship in the horse. Equine Vet. J. 2023, 55, 944–961. [Google Scholar] [CrossRef]
- Scofield, D.; Black, J.; Wittenburg, L.; Gustafson, D.; Ferris, R.A.; Hatzel, J.N.; Traub-Dargatz, J.; McCue, P. Endometrial Tissue and Blood Plasma Concentration of Ceftiofur and Metabolites After Intramuscular Administration of Ceftiofur Crystalline-Free Acid to Mares. Am. Assoc. Equine Pract. 2012, 58, 519–520. [Google Scholar]
- Fleury, M.A.; Mourand, G.; Jouy, E.; Touzain, F.; Le Devendec, L.; de Boisseson, C.; Eono, F.; Cariolet, R.; Guérin, A.; Le Goff, O.; et al. Impact of ceftiofur injection on gut microbiota and Escherichia coli resistance in pigs. Antimicrob. Agents Chemother. 2015, 59, 5171–5180. [Google Scholar] [CrossRef] [PubMed]
- Vasco, K.A.; Carbonell, S.; Sloup, R.E.; Bowcutt, B.; Colwell, R.R.; Graubics, K.; Erskine, R.; Norby, B.; Ruegg, P.L.; Zhang, L.; et al. Persistent effects of intramammary ceftiofur treatment on the gut microbiome and antibiotic resistance in dairy cattle. Anim. Microbiome 2023, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Fuertes, M.; Sibila, M.; Franzo, G.; Obregon-Gutierrez, P.; Illas, F.; Correa-Fiz, F.; Aragón, V. Ceftiofur treatment of sows results in long-term alterations in the nasal microbiota of the offspring that can be ameliorated by inoculation of nasal colonizers. Anim. Microbiome 2023, 5, 53. [Google Scholar] [CrossRef]
- Rutjens, S.; Vereecke, N.; De Spiegelaere, W.; Croubels, S.; Devreese, M. Intestinal Exposure to Ceftiofur and Cefquinome after Intramuscular Treatment and the Impact of Ceftiofur on the Pig Fecal Microbiome and Resistome. Antibiotics 2022, 11, 342. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.J.; Cunha, F.; Daetz, R.; Bicalho, R.C.; Lima, S.; Galvão, K.N. Ceftiofur reduced Fusobacterium leading to uterine microbiota alteration in dairy cows with metritis. Anim. Microbiome 2021, 3, 15. [Google Scholar] [CrossRef]
- Jeon, S.J.; Lima, F.S.; Vieira-Neto, A.; Machado, V.S.; Lima, S.F.; Bicalho, R.C.; Santos, J.E.P.; Galvão, K.N. Shift of uterine microbiota associated with antibiotic treatment and cure of metritis in dairy cows. Vet. Microbiol. 2018, 214, 132–139. [Google Scholar] [CrossRef]
- Galvão, K.N.; Greco, L.F.; Vilela, J.M.; Filho, M.F.S.; Santos, J.E.P. Effect of intrauterine infusion of ceftiofur on uterine health and fertility in dairy cows. J. Dairy Sci. 2009, 92, 1532–1542. [Google Scholar] [CrossRef]
- Scott, H.M.; Schouten, M.J.; Gaiser, J.C.; Belschner, A.P.; Jordan, E.R. Effect of intrauterine administration of ceftiofur on fertility and risk of culling in postparturient cows with retained fetal membranes, twins, or both. J. Am. Vet. Med. Assoc. 2005, 226, 2044–2052. [Google Scholar] [CrossRef]
- Ricketts, S.W. Treatment of equine endometritis with intrauterine irrigation of ceftiofur sodium. Pferdeheilkunde 1997, 5, 486–489. [Google Scholar] [CrossRef]
- Troedsson, M.H.; Liu, I.K.; Crabo, B.G. Sperm transport and survival in the mare. Theriogenology 1998, 49, 905–915. [Google Scholar] [CrossRef]
- Sieme, H.; Bonk, A.; Hamann, H.; Klug, E.; Katila, T. Effects of different artificial insemination techniques and sperm doses on fertility of normal mares and mares with abnormal reproductive history. Theriogenology 2004, 62, 915–928. [Google Scholar] [CrossRef] [PubMed]
- Köhne, M.; Hegger, A.; Görgens, A.; Martinsson, G.; Sieme, H. Success of different therapy concepts for bacterial endometritis in stud farm practice. J. Equine Vet. Sci. 2023, 125, 104723. [Google Scholar] [CrossRef]
- Shanahan, F.; Ghosh, T.S.; O’Toole, P.W. Human microbiome variance is underestimated. Curr. Opin. Microbiol. 2023, 73, 102288. [Google Scholar] [CrossRef] [PubMed]
- Healey, G.R.; Murphy, R.; Brough, L.; A Butts, C.; Coad, J. Interindividual variability in gut microbiota and host response to dietary interventions. Nutr. Rev. 2017, 75, 1059–1080. [Google Scholar] [CrossRef] [PubMed]
- Favari, C.; de Alvarenga, J.F.R.; Sánchez-Martínez, L.; Tosi, N.; Mignogna, C.; Cremonini, E.; Manach, C.; Bresciani, L.; Del Rio, D.; Mena, P. Factors driving the inter-individual variability in the metabolism and bioavailability of (poly)phenolic metabolites: A systematic review of human studies. Redox Biol. 2024, 71, 103095. [Google Scholar] [CrossRef]
- Wei, Z.; Li, F.; Pi, G. Association Between Gut Microbiota and Osteoarthritis: A Review of Evidence for Potential Mechanisms and Therapeutics. Front. Cell. Infect. Microbiol. 2022, 12, 812596. [Google Scholar] [CrossRef]
- Elkafas, H.; Walls, M.; Al-Hendy, A.; Ismail, N. Gut and genital tract microbiomes: Dysbiosis and link to gynecological disorders. Front. Cell. Infect. Microbiol. 2022, 12, 1059825. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Zeng, W.; Hou, T.; Yang, H.; Wu, B.; Pan, R.; Huang, L. Gut microbiome and reproductive endocrine diseases: A Mendelian randomization study. Front. Endocrinol. 2023, 14, 1164186. [Google Scholar] [CrossRef]
- Zhang, M.; Shi, Z.; Wu, C.; Yang, F.; Su, T.; Jing, X.; Shi, J.; Ren, H.; Jiang, L.; Jiang, Y.; et al. Cushing Syndrome Is Associated with Gut Microbial Dysbiosis and Cortisol-Degrading Bacteria. J. Clin. Endocrinol. Metab. 2024, 109, 1474–1484. [Google Scholar] [CrossRef] [PubMed]
- Beckers, K.F.; Gomes, V.C.L.; Crissman, K.R.; Liu, C.-C.; Schulz, C.J.; Childers, G.W.; Sones, J.L. Metagenetic Analysis of the Pregnant Microbiome in Horses. Animals 2023, 13, 1999. [Google Scholar] [CrossRef] [PubMed]
- Moraes, J.G.N.; Gull, T.; Ericsson, A.C.; Poock, S.E.; Caldeira, M.O.; Lucy, M.C. The microbiome of the pregnant uterus in Holstein dairy heifers and cows assessed by bacterial culture and 16S ribosomal RNA gene sequencing. Front. Microbiol. 2024, 15, 1385497. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beckers, K.F.; Liu, C.-C.; Gomes, V.C.L.; Schulz, C.J.; Childers, G.W.; Fedorka, C.E.; Sones, J.L. Effects of Intra-uterine Ceftiofur on the Equine Uterine Microbiome. Vet. Sci. 2025, 12, 837. https://doi.org/10.3390/vetsci12090837
Beckers KF, Liu C-C, Gomes VCL, Schulz CJ, Childers GW, Fedorka CE, Sones JL. Effects of Intra-uterine Ceftiofur on the Equine Uterine Microbiome. Veterinary Sciences. 2025; 12(9):837. https://doi.org/10.3390/vetsci12090837
Chicago/Turabian StyleBeckers, Kalie F., Chin-Chi Liu, Viviane C. L. Gomes, Christopher J. Schulz, Gary W. Childers, Carleigh E. Fedorka, and Jenny L. Sones. 2025. "Effects of Intra-uterine Ceftiofur on the Equine Uterine Microbiome" Veterinary Sciences 12, no. 9: 837. https://doi.org/10.3390/vetsci12090837
APA StyleBeckers, K. F., Liu, C.-C., Gomes, V. C. L., Schulz, C. J., Childers, G. W., Fedorka, C. E., & Sones, J. L. (2025). Effects of Intra-uterine Ceftiofur on the Equine Uterine Microbiome. Veterinary Sciences, 12(9), 837. https://doi.org/10.3390/vetsci12090837






