Topical Use of Sucralfate in Cutaneous Wound Management: A Narrative Review with a Veterinary Perspective
Simple Summary
Abstract
1. Introduction
2. Sucralfate’s Mechanism of Action
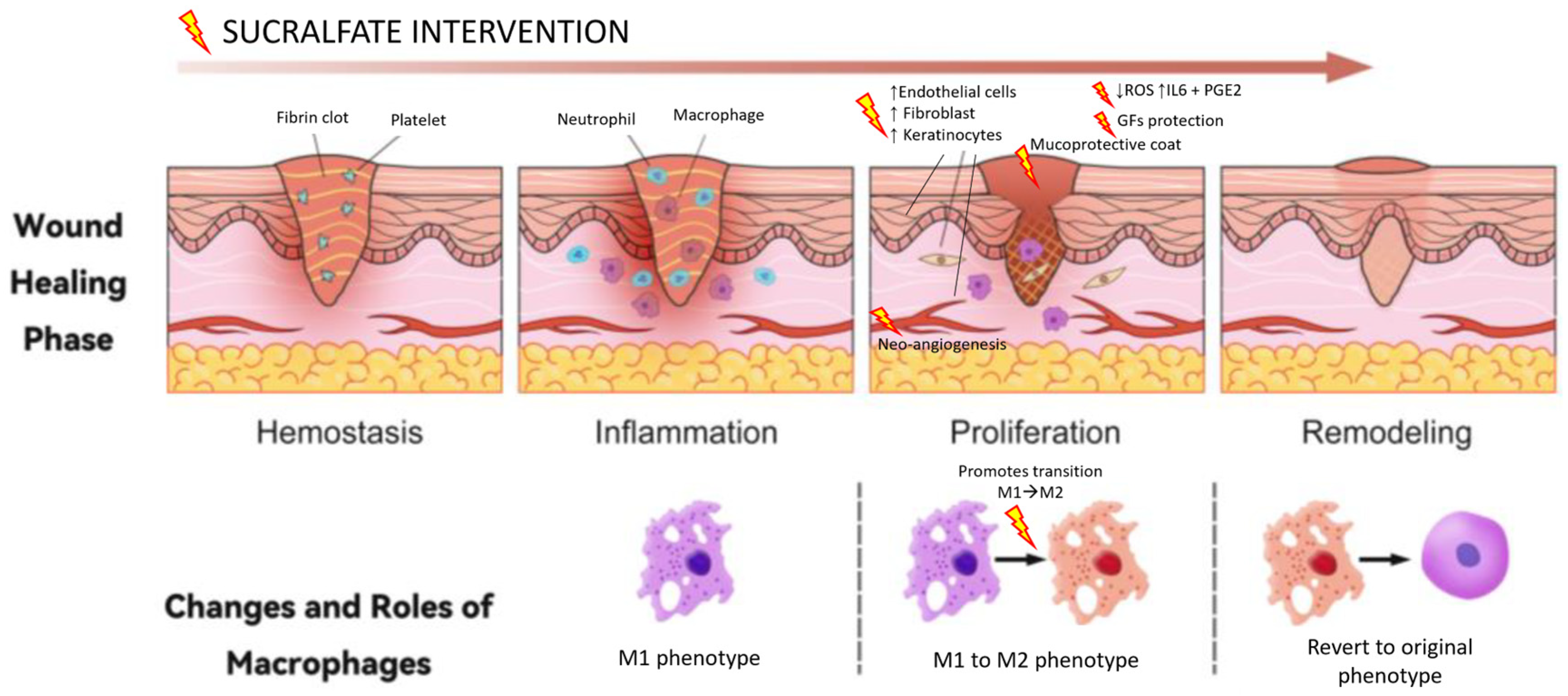
3. Sucralfate’s Topical Use in Human Medicine
4. Topical Sucralfate’s Experimental Use in Veterinary Medicine
5. Discussion: Challenges and the Potential Role of Topical Sucralfate
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Harris-Tryon, T.A.; Grice, E. Microbiota and maintenance of skin barrier function. Science 2022, 376, 940–945. [Google Scholar] [CrossRef] [PubMed]
- Borena, B.M.; Martens, A.; Broeckx, S.Y.; Meyer, E.; Chiers, K.; Duchateau, L.; Spaas, J.H. Regenerative skin wound healing in mammals: State-of-the-art on growth factor and stem cell-based treatments. Cell. Physiol. Biochem. 2015, 36, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Sorg, H.; Tilkorn, D.J.; Hager, S.; Hauser, J.; Mirastschijski, U. Skin wound healing: An update on the current knowledge and concepts. Eur. Surg. Res. 2017, 58, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Tambella, A.M.; Attili, A.R.; Dupre, G.; Cantalamessa, A.; Martin, S.; Cuteri, V.; Marcazzan, S.; Del Fabbro, M. Platelet-rich plasma to treat experimentally-induced skin wounds in animals: A systematic review and meta-analysis. PLoS ONE 2018, 13, e0191093. [Google Scholar] [CrossRef]
- Lux, C.N. Wound healing in animals: A review of physiology and clinical evaluation. Vet. Dermatol. 2022, 33, 91-e27. [Google Scholar] [CrossRef]
- Le, Z.; Ramos, M.C.; Shou, Y.; Li, R.R.; Cheng, H.S.; Jang, C.J.; Liu, L.; Xue, C.; Li, X.; Liu, H.; et al. Bioactive sucralfate-based microneedles promote wound healing through reprogramming macrophages and protecting endogenous growth factors. Biomaterials 2024, 311, 122700. [Google Scholar] [CrossRef]
- Harman, R.M.; Theoret, C.L.; Van de Walle, G.R. The horse as a model for the study of cutaneous wound healing. Adv. Wound Care 2021, 10, 381–399. [Google Scholar] [CrossRef]
- Hill, P.B.; Lo, A.; Eden, C.A.N.; Huntley, S.; Morey, V.; Ramsey, S.; Richardson, C.; Smith, D.J.; Sutton, C.; Taylor, M.D.; et al. Survey of the prevalence, diagnosis and treatment of dermatological conditions in small animals in general practice. Vet. Rec. 2006, 158, 533–539. [Google Scholar] [CrossRef]
- Hill, P. Small Animal Dermatology: A Practical Guide to the Diagnosis and Management of Skin Diseases in Dogs and Cats; Butterworth-Heinemann: Oxford, UK, 2002; pp. 143–147. [Google Scholar]
- Marsella, R.; Olivry, T.; Carlotti, D.N. Current evidence of skin barrier dysfunction in human and canine atopic dermatitis. Vet. Dermatol. 2011, 22, 239–248. [Google Scholar] [CrossRef]
- Woldemeskel, M. Nutraceuticals in Dermatological Disorders. In Nutraceuticals in Veterinary Medicine; Springer: Cham, Switzerland, 2019; pp. 563–568. [Google Scholar]
- Aisa, J.; Parlier, M. Local wound management: A review of modern techniques and products. Vet. Dermatol. 2022, 33, 463–478. [Google Scholar] [CrossRef]
- Rudiman, R.; Hanafi, R.V.; Evan, C.; Halim, F. The efficacy of topical sucralfate in improving pain and wound healing after haemorrhoidectomy procedure: A systematic review, meta-analysis, and meta-regression of randomised clinical trials. Int. Wound J. 2023, 20, 543–553. [Google Scholar] [CrossRef]
- Abtahi-Naeini, B.; Saffaei, A.; Sabzghabaee, A.M.; Amiri, R.; Hosseini, N.S.; Niknami, E.; Dehghani, S. Topical sucralfate for treatment of mucocutaneous conditions: A systematic review on clinical evidences. Dermatol. Ther. 2022, 35, e15334. [Google Scholar] [CrossRef] [PubMed]
- Masuelli, L.; Tumino, G.; Turriziani, M.; Modesti, A.; Bei, R. Topical use of sucralfate in epithelial wound healing: Clinical evidence and molecular mechanisms of action. Recent Pat. Inflamm. Allergy Drug Discov. 2010, 4, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Tolbert, K.; Stubbs, E. Rational use of gastroprotectants in cats: An evidence-based approach. J. Feline Med. Surg. 2024, 26. [Google Scholar] [CrossRef] [PubMed]
- Vokes, J.; Lovett, A.; Sykes, B. Equine gastric ulcer syndrome: An update on current knowledge. Animals 2023, 13, 1261. [Google Scholar] [CrossRef]
- Yildizhan, E.; Ulger, B.V.; Akkus, M.; Akinci, D.; Basol, O. Comparison of topical sucralfate with dexpanthenol in rat wound model. Int. J. Exp. Pathol. 2022, 103, 164–170. [Google Scholar] [CrossRef]
- Yuniati, R.; Innelya, I.; Rachmawati, A.; Charlex, H.J.M.; Rahmatika, A.; Khrisna, M.B.; Mundhofir, F.E.P.; Seno, T.N.; Kristina, T.N. Application of topical sucralfate and topical platelet-rich plasma improves wound healing in diabetic ulcer rats wound model. J. Exp. Pharmacol. 2021, 13, 797–806. [Google Scholar] [CrossRef]
- Abdel Sattar, A.M.; Al Batanony, A.A.; Al Khateeb, Y.M. Evaluation of the role of sucralfate cream in decreasing pain intensity and improving healing following open hemorrhoidectomy: A randomized controlled study. Menoufia Med. J. 2019, 32, 506–510. [Google Scholar] [CrossRef]
- Gutta, S.H.; Kotennavar, M.S.; Patil, A.; Benakatti, R.; Jaju, P.; Savanth, M. Topical Solutions for Chronic Lower Limb Ulcers: A Comparative Study of Sucralfate and 5% Povidone-Iodine. Int. J. Sci. Study 2023, 11, 45–49. [Google Scholar]
- Yaşar, Ş.; Yaşar, B.; Yörüsün, A.; Güneş, P.; Kayadibi, H.; Ercin, Z.; Aytekin, S. A Controlled Study to Examine the Effect of Topical Sucralfate on Radiofrequency-induced Burn Wounds in Rats. Wound Manag. Prev. 2020, 66, 34–42. [Google Scholar] [CrossRef]
- Bryan, N.; Ahswin, H.; Smart, N.; Bayon, Y.; Wohlert, S.; Hunt, J.A. Reactive oxygen species (ROS)—A family of fate deciding molecules pivotal in constructive inflammation and wound healing. Eur. Cells Mater. 2012, 24, e65. [Google Scholar] [CrossRef] [PubMed]
- Ala, S.; Saeedi, M.; Gholipour, A.; Ahmadi, M.; Asoodeh, A.; Shiva, A. Effectiveness of topical sucralfate in the management of pressure ulcer in hospitalized patients: A prospective, randomized, placebo-controlled trial. Am. J. Ther. 2019, 26, e5–e11. [Google Scholar] [CrossRef] [PubMed]
- Freeman, S.B.; Markwell, J.K. Sucralfate in alleviating post-tonsillectomy pain. Laryngoscope 1992, 102, 1242–1246. [Google Scholar] [CrossRef] [PubMed]
- Özcan, M.; Altuntasl, A.; Ünal, A.; Nalcla, Y.; Aslan, A. Sucralfate for posttonsillectomy analgesia. Otolaryngol.–Head Neck Surg. 1998, 119, 700–704. [Google Scholar] [CrossRef]
- Gupta, P.J.; Heda, P.S.; Kalaskar, S.; Tamaskar, V.P. Topical sucralfate decreases pain after hemorrhoidectomy and improves healing: A randomized, blinded, controlled study. Dis. Colon Rectum 2008, 51, 231–234. [Google Scholar] [CrossRef]
- Tryba, M.; Mantey-Stiers, F. Antibacterial activity of sucralfate in human gastric juice. Am. J. Med. 1987, 83, 125–127. [Google Scholar] [CrossRef]
- Yen, T.; Boord, M.J.; Ghubash, R.; Blondeau, J.M. A pilot study investigating the in vitro efficacy of sucralfate against common veterinary cutaneous pathogens. J. Small Anim. Pract. 2018, 59, 691–694. [Google Scholar] [CrossRef]
- Li, J.; Xie, J.; Wang, Y.; Li, X.; Yang, L.; Zhao, M.; Chen, C. Development of Biomaterials to Modulate the Function of Macrophages in Wound Healing. Bioengineering 2024, 11, 1017. [Google Scholar] [CrossRef]
- Linata, A.M.; Fawzy, A. Does The Scientific Evidence Support the Idea of Promoting Sucralfate as The First Aid Topical Management for Burn Wound? A Literature Review. Int. J. Med. Sci. Clin. Res. Stud. 2024, 4, 1226–1230. [Google Scholar] [CrossRef]
- Godhi, A.S.; Ram, P.; Powar, R. Efficacy of topical sucralfate versus silver sulfadiazine in the management of burns: A 1-year randomized controlled trial. J. West Afr. Coll. Surg. 2017, 7, 57. [Google Scholar]
- Koshariya, M.; Shitole, A.; Agarwal, V.; Dave, S. Role of topical Sucralfate in healing of burn wounds. Int. Surg. J. 2018, 5, 2995–3001. [Google Scholar] [CrossRef]
- Saei, S.; Sahebnasagh, A.; Ghasemi, A.; Akbari, J.; Alipour, A.; Lashkardoost, H.; Joybari, A.Y.; Dadgar, N.F.; Ala, S.; Salehifar, E. Efficacy of sucralfate ointment in the prevention of acute proctitis in cancer patients: A randomized controlled clinical trial. Casp. J. Intern. Med. 2020, 11, 410. [Google Scholar] [CrossRef]
- Shum, N.F.; Choi, H.K.; Wei, R.; Dominic, C.C.F. Evaluating self-administered sucralfate enemas for reducing rectal bleeding in patients with radiation proctitis. Cancer Nurs. Pract. 2024, 23. [Google Scholar] [CrossRef]
- Kouloulias, V.; Asimakopoulos, C.; Tolia, M.; Filippou, G.; Platoni, K.; Dilvoi, M.; Beli, I.; Georgakopoulos, J.; Patatoukas, G.; Kelekis, N. Sucralfate gel as a radioprotector against radiation induced dermatitis in a hypo-fractionated schedule: A non-randomized study. Hippokratia 2013, 17, 126. [Google Scholar] [PubMed]
- Marik, A.R.; Miklós, I.; Csukly, G.; Hársfalvi, P.; Novák, A. Effectiveness and tolerability of rectal ointment and suppositories containing sucralfate for hemorrhoidal symptoms: A prospective, observational study. Int. J. Color. Dis. 2024, 39, 72. [Google Scholar] [CrossRef] [PubMed]
- Lamb, S.; Coulter, L.; Hudson, J. Is topical sucralfate an effective therapy for noncandidal diaper rash? Evid.-Based Pract. 2016, 19, 7–8. [Google Scholar] [CrossRef]
- Markham, T.; Kennedy, F.; Collins, P. Topical sucralfate for erosive irritant diaper dermatitis. Arch. Dermatol. 2000, 136, 1199–1200. [Google Scholar] [CrossRef][Green Version]
- Tumino, G.; Masuelli, L.; Bei, R.; Simonelli, L.; Santoro, A.; Francipane, S. Topical treatment of chronic venous ulcers with sucralfate: A placebo controlled randomized study. Int. J. Mol. Med. 2008, 22, 17–23. [Google Scholar] [CrossRef]
- Bhatmule, A.; Janugade, H.B.; Nangare, N. A competitive study of topical sucralfate and ordinary saline for diabetic ulcer dressing. J. Pharm. Negat. Results 2022, 13, 577–580. [Google Scholar] [CrossRef]
- Chatterjee, S.; Sen, S.; Hazra, A.; Das, A.K. Randomized controlled trial of topical mupirocin versus mupirocin with sucralfate combination in chronic skin ulcers. Indian J. Pharmacol. 2019, 51, 316–322. [Google Scholar] [CrossRef]
- Pourandish, Y.; Mehrabi, F.; Veldani, N.A.; Tabar, R.M. Pressure ulcer healing by daily topical sucralfate and silver sulfadiazine: A case report study. J. Nurs. Midwifery Sci. 2021, 8, 219. [Google Scholar] [CrossRef]
- Chatterjee, N.; Ekka, N.M.; Mahajan, M.; Kumar, B.; Kumar, N.; Zia, A.; Devarajan, A.; Kujur, A.D.; Sinha, D.K. Effectiveness of Topical Sucralfate in the Management of Diabetic Foot Ulcers: An Open-Labeled Randomized Study. Cureus 2023, 15, e37570. [Google Scholar] [CrossRef]
- Vejdan, A.K.; Khosravi, M.; Amirian, Z.; Daneshmand, M.; Babak, B.; Samira, K.; Azin, S.; Kosar, S.; Razie, K. Evaluation of the efficacy of topical sucralfate on healing haemorrhoidectomy incision wounds and reducing pain severity: A randomised clinical trial. Int. Wound J. 2020, 17, 1047–1051. [Google Scholar] [CrossRef] [PubMed]
- Siupsinskiene, N.; Žekonienė, J.; Padervinskis, E.; Žekonis, G.; Vaitkus, S. Efficacy of sucralfate for the treatment of post-tonsillectomy symptoms. Eur. Arch. Oto-Rhino-Laryngol. 2015, 272, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Suparakchinda, C.; Rawangban, W. Effectiveness of Sucralfate comparing to normal saline as an oral rinse in pain reduction and wound healing promotion in oral surgery. Laryngoscope Investig. Otolaryngol. 2023, 8, 1226–1232. [Google Scholar] [CrossRef] [PubMed]
- Giua, C.; Minerba, L.; Piras, A.; Floris, N.; Romano, F. The effect of sucralfate-containing ointment on quality of life in people with symptoms associated with haemorrhoidal disease and its complications: The results of the EMOCARE survey. Acta Biomed. Atenei Parm. 2021, 92, e2021029. [Google Scholar] [CrossRef]
- Beheshti, A.; Shafigh, Y.; Zangivand, A.A.; Samiee-Rad, F.; Hassanzadeh, G.; Shafigh, N. Comparison of topical sucralfate and silver sulfadiazine cream in second degree burns in rats. Adv. Clin. Exp. Med. 2013, 22, 481–487. [Google Scholar] [PubMed]
- Ribeiro, G.; Carvalho, L.; Borges, J.; Prazeres, J. The Best Protocol to Treat Equine Skin Wounds by Second Intention Healing: A Scoping Review of the Literature. Animals 2024, 14, 1500. [Google Scholar] [CrossRef]
- Eggleston, R.B. Wound management: Wounds with special challenges. Vet. Clin. Equine Pract. 2018, 34, 511–538. [Google Scholar] [CrossRef]
- Hamed, M.A.; Abouelnasr, K.S.; El-Adl, M.; Elfadl, E.A.A.; Farag, A.; Lashen, S. Effectiveness of allogeneic platelet-rich fibrin on second-intention wound healing of experimental skin defect in distal limb in donkeys (Equus asinus). J. Equine Vet. Sci. 2019, 73, 131–138. [Google Scholar] [CrossRef]
- Marchant, K.; Hendrickson, D.A.; Pezzanite, L.M. Review of the role of biofilms in equine wounds: Clinical indications and treatment strategies. Equine Vet. Educ. 2024, 36, 152–168. [Google Scholar] [CrossRef]
- Kruse, C.R.; Singh, M.; Targosinski, S.; Sinha, I.; Sørensen, J.A.; Eriksson, E.; Nuutila, K. The effect of pH on cell viability, cell migration, cell proliferation, wound closure, and wound reepithelialization: In vitro and in vivo study. Wound Repair Regen. 2017, 25, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Bohling, M.W.; Henderson, R.A.; Swaim, S.F.; Kincaid, S.A.; Wright, J.C. Cutaneous wound healing in the cat: A macroscopic description and comparison with cutaneous wound healing in the dog. Vet. Surg. 2004, 33, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Tashkandi, H. Honey in wound healing: An updated review. Open Life Sci. 2021, 16, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Zainuddin, A.N.Z.; Mustakim, N.N.; Rosemanzailani, F.A.; Fadilah, N.I.M.; Maarof, M.; Fauzi, M.B. A Comprehensive Review of Honey-Containing Hydrogel for Wound Healing Applications. Gels 2025, 11, 194. [Google Scholar] [CrossRef]
- Rybka, M.; Mazurek, L.; Konop, M. Beneficial effect of wound dressings containing silver and silver nanoparticles in wound healing—From experimental studies to clinical practice. Life 2023, 13, 69. [Google Scholar] [CrossRef]
- Jangid, H.; Singh, S.; Kashyap, P.; Singh, A.; Kumar, G. Advancing biomedical applications: An in-depth analysis of silver nanoparticles in antimicrobial, anticancer, and wound healing roles. Front. Pharmacol. 2024, 15, 1438227. [Google Scholar] [CrossRef]
- Popescu, I.; Constantin, M.; Solcan, G.; Ichim, D.L.; Rata, D.M.; Horodincu, L.; Solcan, C. Composite Hydrogels with Embedded Silver Nanoparticles and Ibuprofen as Wound Dressing. Gels 2023, 9, 654. [Google Scholar] [CrossRef]
- Farabi, B.; Roster, K.; Hirani, R.; Tepper, K.; Atak, M.F.; Safai, B. The Efficacy of Stem Cells in Wound Healing: A Systematic Review. Int. J. Mol. Sci. 2024, 25, 3006. [Google Scholar] [CrossRef]
- Gao, M.; Guo, H.; Dong, X.; Wang, Z.; Yang, Z.; Shang, Q.; Wang, Q. Regulation of inflammation during wound healing: The function of mesenchymal stem cells and strategies for therapeutic enhancement. Front. Pharmacol. 2024, 15, 1345779. [Google Scholar] [CrossRef]
- Scapagnini, G.; Marchegiani, A.; Rossi, G.; Zago, M.; Jowarska, J.; Wael, M.; Campbell, S.E.; Schiffman, Z.; Buonamici, E.; Garvao, R.; et al. Management of all three phases of wound healing through the induction of fluorescence biomodulation using fluorescence light energy. In Proceedings of the Photonic Diagnosis Treatment Infections Inflammatory Diseases II, San Francisco, CA, USA, 4–5 February 2019; pp. 108630W1–108630W17. [Google Scholar] [CrossRef]
- Nikolis, A.; Grimard, D.; Pesant, Y.; Scapagnini, G.; Vezina, D.A. A prospective case series evaluating the safety and efficacy of the Klox BioPhotonic System in venous leg ulcers. Chronic Wound Care Manag. Res. 2016, 3, 101–111. [Google Scholar] [CrossRef]

| Study (Author, Year, Type) | Condition | Protocol | Main Outcomes |
|---|---|---|---|
| Godhi et al., 2017, Randomized controlled trial [32] | Second-degree burn wounds | Topical sucralfate dressing vs. 1% silver sulfaziadine dressing | Faster granulation tissue formation compared to silver sulfadiazine. Comparable antimicrobial effectiveness. |
| Koshariya et al., 2018 Observational study [33] | Burn wounds | Topical sucralfate vs. topical silver sulfadiazine | Faster reepithelialization and granulation tissue formation, lower infection incidence and pain relief compared to silver sulfadiazine. |
| Saei et al., 2020, Randomized, double-blind, placebo-controlled clinical trial [34] | Radiotherapy-induced proctitis | Sucralfate ointment vs. placebo rectally | Reduced rectal bleeding, diarrhea and rectal pain compared to placebo. |
| Shum et al., 2024, Non-randomized clinical trial [35] | Radiotherapy-induced proctitis | Sucralfate enemas | Reduced rectal bleeding, decreased hospitalization, reduced need for invasive treatments. |
| Kouloulias et al., 2013, Non-randomized clinical trial [36] | Radiation dermatitis | Topical sucralfate humid gel (Skincol®, Erba, Italy) | Protective effect against dermatitis, reduced radiation-induced skin toxicity. |
| Rudiman et al., 2023, Systematic review and meta-analysis of randomized controlled trials [13] | Post-hemorrhoidectomy wounds | Topical sucralfate formulation | Reduced pain and analgesic consumption, improved wound healing. |
| Marik et al., 2024, Prospective observational study [37] | Hemorrhoidal symptoms | Sucralfate-based rectal ointment or suppositories | Reduction in hemorrhoidal symptoms, improved hemorrhoid grades. |
| Abdel Sattar et al., 2019, Randomized controlled trial [20] | Post-hemorrhoidectomy pain | Topical sucralfate formulation vs. placebo | Pain reduction and fast healing rate compared to placebo. |
| Vejdan et al., 2020, Randomized controlled trial [45] | Post-hemorrhoidectomy wounds | Topical sucralfate ointment vs. placebo | Significant pain reduction and analgesic consumption, accelerated healing. |
| Tumino et al., 2008, Randomized, double-blind, placebo-controlled trial [40] | Venous ulcers | Topical sucralfate cream vs. placebo | 95.6% complete healing vs. 10.9% placebo, reduced pain and inflammation, faster ulcer reduction and granulation. Enhanced reepithelialization and angiogenesis. |
| Bhatmule et al., 2022, Comparative study [41] | Diabetic ulcers | Topical sucralfate formulation vs. saline dressing | Higher wound healing rate, reduction in ulcer size, reduced time to achieve complete healing. |
| Chatterjee et al., 2023, Open-label, randomized controlled trial [44] | Diabetic foot ulcers | Topical sucralfate + mupirocin ointment vs. mupirocin ointment | Healing time comparable to control group. |
| Ala et al., 2019, Prospective, double-blind, randomized, placebo-controlled trial [24] | Stage II pressure ulcers | Topical sucralfate gel vs. placebo | Healing time comparable to control group. |
| Pourandish et al., 2021, Case report [43] | Stage IV pressure ulcer | Topical sucralfate formulation + silver sulfadiazine | Ulcer improvement (stage II), reduction in ulcer size and tissue appearance. |
| Markham et al., 2000, Case series/clinical observation [39] | Infant irritant diaper dermatitis | Topical sucralfate ointment | Reduction in inflammation and fast healing rate. |
| Siupsinskiene et al., 2015, Randomized controlled trial [46] | Post-tonsillectomy or -adenotonsillectomy wounds | Topical sucralfate formulation vs. placebo | Reduced throat pain, odynophagia, otalgia. Reduced analgesic consumption and faster recovery. |
| Giua et al., 2021, Prospective, multicenter, observational study [48] | Hemorrhoidal disease | Topical rectal ointment with sucralfate and herbal extracts | Reduced pain, itching, bleeding and prolapse. |
| Study | Animal Model | Route/Formulation | Wound Type | Outcomes |
|---|---|---|---|---|
| Yildizhan et al., 2022 [18] | Rats | Topical application of 10% sucralfate cream | Full-thickness dorsal skin wounds, 2 cm diameter | Sucralfate significantly enhanced wound healing, with ↑ neovascularization, ↑ collagen organization, ↑ EGF expression and faster wound closure at days 7, 14 and 21 (p ≤ 0.05). |
| Le et al., 2023 [6] | Diabetic mice and pigs | Sucralfate-based microneedles delivering IL-4 | Full-thickness excisional wounds (model-dependent) | Dual-action system promoted M2 macrophage polarization and growth factor stabilization, leading to fastest wound closure, ↑ angiogenesis and ↑ collagen deposition vs. controls. |
| Yuniati et al., 2023 [19] | Diabetic rats | Topical sucralfate, PRP or combination | Chronic diabetic ulcers (dimensions not specified) | Sucralfate alone ↑ fibroblast activity, ↑ angiogenesis, ↑ local growth factors, ↑ tissue remodeling, leading to improved contraction and reepithelialization. |
| Yaşar et al., 2023 [22] | Rats | Topical application of 10% sucralfate cream | Radiofrequency-induced burns | Early ↑ vasodilation and leukocyte chemotaxis; from day 14, ↑ angiogenesis, ↑ fibroblasts, ↑ collagen synthesis and ↑ reepithelialization. Thick granulation tissue by week 4. |
| Beheshti et al., 2013 [49] | Rats | Topical sucralfate cream vs. silver sulfadiazine | Second-degree thermal burns | Sucralfate group showed 100% wound closure vs. 91% with SSD and 76% with control. Histology revealed more complete epidermal regeneration and skin appendage formation. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Accorroni, L.; Dini, F.; Pilati, N.; Marchegiani, A.; Bazzano, M.; Spaterna, A.; Laus, F. Topical Use of Sucralfate in Cutaneous Wound Management: A Narrative Review with a Veterinary Perspective. Vet. Sci. 2025, 12, 756. https://doi.org/10.3390/vetsci12080756
Accorroni L, Dini F, Pilati N, Marchegiani A, Bazzano M, Spaterna A, Laus F. Topical Use of Sucralfate in Cutaneous Wound Management: A Narrative Review with a Veterinary Perspective. Veterinary Sciences. 2025; 12(8):756. https://doi.org/10.3390/vetsci12080756
Chicago/Turabian StyleAccorroni, Lucrezia, Fabrizio Dini, Nicola Pilati, Andrea Marchegiani, Marilena Bazzano, Andrea Spaterna, and Fulvio Laus. 2025. "Topical Use of Sucralfate in Cutaneous Wound Management: A Narrative Review with a Veterinary Perspective" Veterinary Sciences 12, no. 8: 756. https://doi.org/10.3390/vetsci12080756
APA StyleAccorroni, L., Dini, F., Pilati, N., Marchegiani, A., Bazzano, M., Spaterna, A., & Laus, F. (2025). Topical Use of Sucralfate in Cutaneous Wound Management: A Narrative Review with a Veterinary Perspective. Veterinary Sciences, 12(8), 756. https://doi.org/10.3390/vetsci12080756





