Spermatogonial Stem Cells in Domestic Animals: Current Insights and Future Directions with a Focus on Dogs
Simple Summary
Abstract
1. Introduction
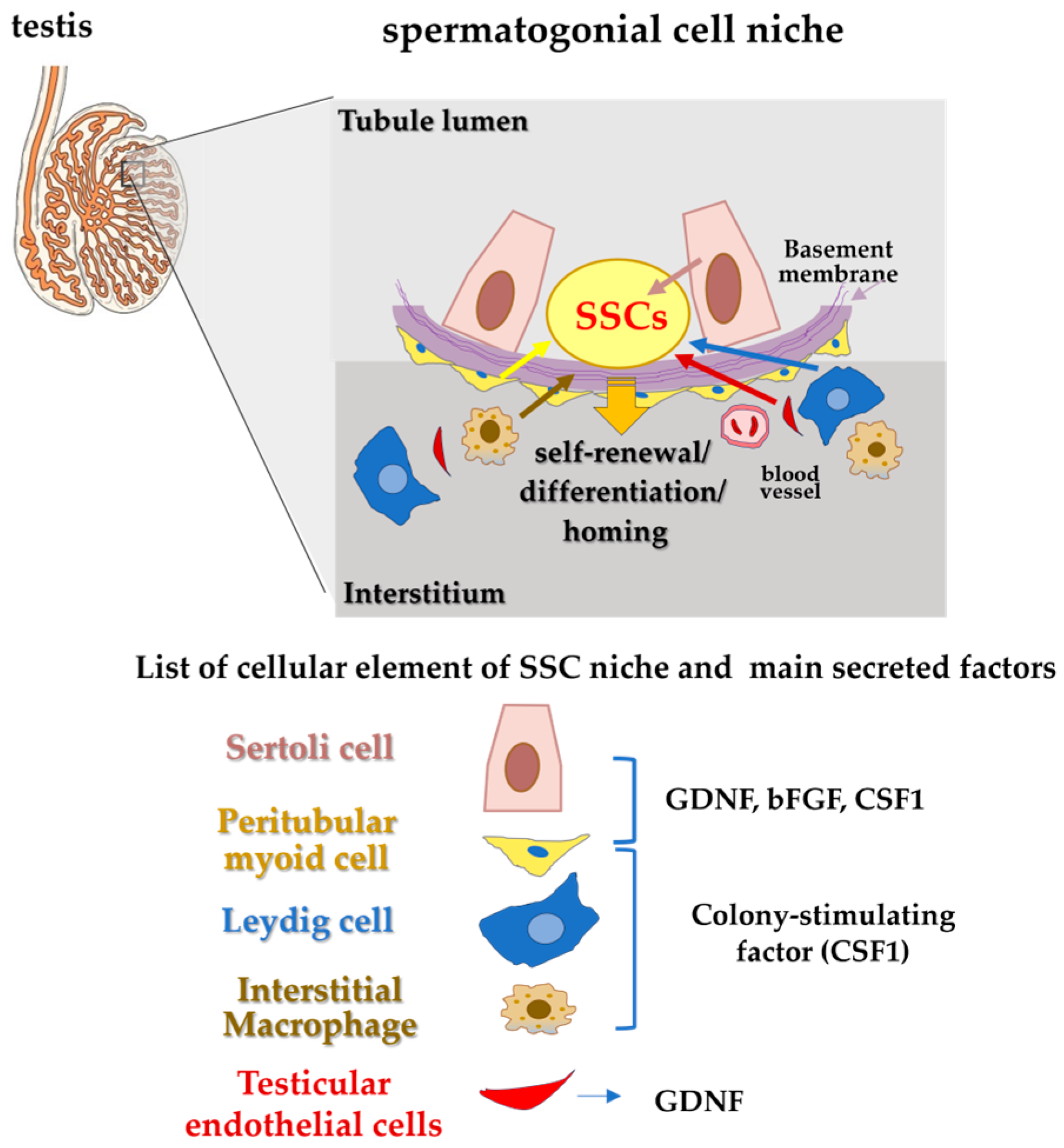
2. Spermatogonial Stem Cells (SSCs): Characteristics and Main Roles
| Factor | Role at Level of SSC | Mechanism Involved | Species | Reference |
|---|---|---|---|---|
| GDNF | self-renewal | Nanos2, ETV5, Lhx1, T, BCL6b, Id1, and CXCR4 | mouse | [26,35,41] |
| self-renewal and proliferation | n.d. | swine | [34] | |
| IGF1, IGFBP7, NKCC1, and protein-tyrosine phosphatase | self-renewal and proliferation | CCL24, IGFBP7, and TEK | mouse | [37] |
| retinoic acid | differentiation | downregulation of GDNF expression activation of differentiation factors (BMP and SCF), SOHLH1, SOHL2 | mouse, rat | [42,43,44,45] |
| PLZF transcription factor | self-renewal | SALL4 protein | mouse | [46] |
| FOXO1 transcription factor | self-renewal | PI3K-Akt signaling | mouse | [47] |
| miR-30 | self-renewal and proliferation | n.d. | mouse | [48] |
| miR-34c | differentiation | Inhibition of the function of NANOS2 gene | mouse | [49] |
| miR-202 | self-renewal | Influence of regulators such as STRA8 and DMRT6 | mouse | [50] |
| miR-17-92 and miR-202 | spermatogenesis | Involvement of Bcl2l11, Kit, SOCS3, and Stat3 | mouse | [50,51,52] |
| miR-486-5p | differentiation | up regulating the expression of STRA8 and SYCP3 | mouse | [53] |
| miR-204 | self-renewal and differentiation | SIRT1 | goat | [54] |
| bta-miR-146b | inhibit proliferation and promote apoptosis | n.d. | bovine | [55] |
3. Spermatogonial Stem Cells from Domestic Animal Species: Isolation and In Vitro Expansion Techniques
| Animal | Optimal (Time) for Testis Collection (Days) | Isolation (IM) and Enrichment Method (EM) | Factors Added to the Culture Medium | Evaluation Time of SSC Proliferation (Days) | Reference |
|---|---|---|---|---|---|
| cat | IM = two-step enzymatic digestion EM = gelatin-coated method | GDNF | 43 days | [80] | |
| dog | 90–150 (pre-pubertal stage) | IM = CLS digestion | GDNF, FGF2, EGF, soluble GRFA1, LIF, and a laminin substratum | Note: the enriched cells can survive for several weeks | [77] |
| buffalo | n.d. | IM = two-step enzymatic digestion | FBS (2.5%), GDNF | [83] | |
| calf | 150–210 | IM = three-step enzymatic digestion 1° (CLS IV), 2° (CLS IV+ HYAL), 3° (trypsin and DNase I) EM = poly-L-lysine-coated method | KSR (15%) | >60 | [79] |
| chicken | 21 | IM = two-step enzymatic digestion EM = differential plating | FBS (2%), GDNF, bFGF or LIF | 7 | [84] |
| goat | 120 | IM = two-step enzymatic digestion EM = Percoll gradient (32%) | LIF, EGF, bFGF, GDNF | 15 | [85] |
| horse | n.d. | IM = two-step enzymatic digestion EM = Percoll gradient (40%) | FBS (10%) | Note: isolated SSCs thawed after cryopreservation demonstrated as much metabolic activity as the fresh cells | [86] |
| pig | 30 | IS = two-step enzymatic digestion EM = differential plating (laminin and PLL) in gelatin-coated plates | GDNF, FGF2, IGF1, LIF, EGF | 25 | [87] |
| 7–15 | IM = two-step enzymatic treatment (CLS, HYAL II, DNase I and trypsin-EDTA) EM = SSC plating in presence of Sertoli cell feeder layer | FGF, GDNF, KSR | >30 | [78] | |
| rabbit | 90–120 | IM = CLS digestion EM = Percoll gradient (32%) | GDNF, FGF2, GRFA1 | 15 | [88] |
| sheep | n.d. | IM = two-step enzymatic digestion EM = Ficoll gradient (12%) and plating (laminin 20 μg/mL in combination with BSA) | GDNF, EGF, IGF1 | 30 | [33] |
4. Spermatogonial Stem Cells from Domestic Animal Species: Current Insights into Cryopreservation and Transplantation Techniques
5. Potential Effects of Xenobiotic and External Factors on the Biology of Spermatogonial Stem Cells
6. Canine Spermatogonial Stem Cells: Characteristics, Pathophysiological Conditions Affecting Fertility and Methods for Their Transplantation
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| As | spermatogonia type A |
| AT-MSCs | adipose-derived mesenchymal stem cells |
| BAX-BCL2 | B-cell lymphoma 2 (BCL2) -associated X protein |
| BPF | 4,4′-dihydroxydiphenylmethane |
| BMP | Bone Morphogenetic Protein |
| BPS | 4,4′-sulfonyldiphenol |
| BSA | bovine serum albumin |
| CDH1 | cadherin 1 |
| CCL24 | C-C motif chemokine ligand 24 |
| CLS | collagenase |
| CSF1 | and colony stimulating factor 1 |
| CXCR4 | C-X-C motif chemokine receptor 4 |
| CRISPR | clustered regularly interspaced short palindromic repeats |
| DAZL | deleted in azoospermia-like |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DMSO | dimethyl sulfoxide |
| DMRT6 | Doublesex and Mab-3-Related Transcription Factor 6 |
| eCG | equine chorionic gonadotropin hormone |
| ECM | extracellular matrix |
| EDCs | endocrine-disrupting chemicals |
| EGF | epidermal growth factor |
| ESCs | embryonic stem cells |
| ETV5 | ETS variant transcription factor 5 |
| FBS | fetal bovine serum |
| FGF2 | fibroblast growth factor 2 |
| FOX | forkhead box |
| GATA4 | GATA Binding Protein 4 |
| GDNF | glial cell-derived neurotrophic factor |
| GFP | green fluorescent protein |
| GRFA1 | glial cell line-derived neurotrophic factor family receptor alpha 1 |
| H3K9me3 | histone 3 lysine 9 trimethylation |
| HYAL | hyaluronidase |
| Id1 | DNA-binding protein inhibitor ID-1 |
| IGF1 | insulin-like growth factor 1 |
| IGFBP | Insulin growth factor-binding protein |
| JAK/STAT | Janus Kinase/Signal Transducer and Activator of Transcription |
| Kit | c-kit proto-oncogene |
| KSR | knockout serum replacement |
| Lhx1 | LIM homeobox 1 |
| LIF | leukemia inhibitory factor |
| MAPK | mitogen-activated protein kinase |
| MCS | methylcellulose culture system |
| MEHP | mono(2-ethylhexyl) phthalate |
| MRT6 | mab-3-related transcription factor B1 |
| MSC | mesenchymal stromal cells |
| MT1/MT2 | melatonin receptor 1A/melatonin receptor 1B |
| Nanos2 | nanos C2HC-type zinc finger 2 |
| NKCC1 | Na+-K+-Cl transporter isoform 1 |
| OCT4 | octamer-binding transcription factor 4 |
| PB-MSCs | peripheral blood-derived mesenchymal stem cells |
| PGP9.5 | protein gene product 9.5 |
| PI3K-Akt | phosphatidylinositol 3-kinase and protein kinase B |
| PLZF | promyelocytic leukemia zinc finger |
| PTM | peritubular myoid |
| PTPN11 | protein tyrosine phosphatase and non-receptor type 11 |
| RA | retinoic acid |
| SALL4 | spalt-like transcription factor 4 |
| SCF | stem cell factor |
| SIRT1 | sirtuin 1 |
| SOCS3 | suppressor of cytokine signaling 3 |
| SOHLH1 | spermatogenesis- and oogenesis-specific basic helix–loop–helix 1 |
| SSCs | spermatogonial stem cells |
| STAT3 | signal transducer and activator of transcription 3 |
| Stra8 | stimulated by retinoic acid 8 |
| SYCP3 | synaptonemal complex protein 3 |
| TEK | receptor tyrosine kinase |
| ZEA | zearalenone |
References
- Yoshida, S.; Sukeno, M.; Nabeshima, Y. A vasculature-associated niche for undifferentiated spermatogonia in the mouse testis. Science 2007, 317, 1722–1726. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.L.; Wagers, A.J. No place like home: Anatomy and function of the stem cell niche. Nat. Rev. Mol. Cell Biol. 2008, 9, 11–21. [Google Scholar] [CrossRef]
- La, H.M.; Hobbs, R.M. Mechanisms regulating mammalian spermatogenesis and fertility recovery following germ cell depletion. Cell. Mol. Life Sci. 2019, 76, 4071–4102. [Google Scholar] [CrossRef]
- Ghasemi, N.; Azizi, H.; Qorbanee, A.; Skutella, T. From unipotency to pluripotency: Deciphering protein networks and signaling pathways in the generation of embryonic stem-like cells from murine spermatogonial stem cells. BMC Genom. 2025, 26, 426. [Google Scholar] [CrossRef]
- Tsai, K.L.; Clark, L.A.; Murphy, K.E. Understanding hereditary diseases using the dog and human as companion model systems. Mamm. Genome 2007, 18, 444–451. [Google Scholar] [CrossRef]
- Mukherjee, P.; Roy, S.; Ghosh, D.; Nandi, S.K. Role of animal models in biomedical research: A review. Lab. Anim. Res. 2022, 38, 18. [Google Scholar] [CrossRef]
- Lindblad-Toh, K.; Wade, C.M.; Mikkelsen, T.S.; Karlsson, E.K.; Jaffe, D.B.; Kamal, M.; Clamp, M.; Chang, J.L.; Kulbokas, E.J.; Zody, M.C.; et al. Genome sequence, comparative analysis and haplotype structure of the domestic dog. Nature 2005, 438, 803–819. [Google Scholar] [CrossRef]
- Pallotti, S.; Piras, I.S.; Marchegiani, A.; Cerquetella, M.; Napolioni, V. Dog-human translational genomics: State of the art and genomic resources. J. Appl. Genet. 2022, 63, 703–716. [Google Scholar] [CrossRef]
- Mazzatenta, A.; Carluccio, A.; Robbe, D.; Di Giulio, C.; Cellerino, A. The companion dog as a unique translational model for aging. Semin. Cell Dev. Biol. 2017, 70, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Schiffman, J.D.; Breen, M. Comparative oncology: What dogs and other species can teach us about humans with cancer. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140231. [Google Scholar] [CrossRef] [PubMed]
- Wright, S.J. Spotlight on Reproduction in Domestic Dogs as a Model for Human Reproduction. In Animal Models and Human Reproduction; John Wiley & Sons, Inc.: New York, NY, USA, 2017; pp. 247–358. [Google Scholar]
- Sumner, R.N.; Harris, I.T.; Van der Mescht, M.; Byers, A.; England, G.C.W.; Lea, R.G. The dog as a sentinel species for environmental effects on human fertility. Reproduction 2020, 159, R265–R276. [Google Scholar] [CrossRef] [PubMed]
- Ibtisham, F.; Awang-Junaidi, A.H.; Honaramooz, A. The study and manipulation of spermatogonial stem cells using animal models. Cell Tissue Res. 2020, 380, 393–414. [Google Scholar] [CrossRef]
- Potter, S.J.; De Falco, T. Role of the testis interstitial compartment in spermatogonial stem cell function. Reproduction 2017, 153, R151–R162. [Google Scholar] [CrossRef] [PubMed]
- Oatley, J.M.; Brinster, R.L. The germline stem cell niche unit in mammalian testes. Physiol. Rev. 2012, 92, 577–595. [Google Scholar] [CrossRef]
- Guo, J.; Grow, E.J.; Mlcochova, H.; Maher, G.J.; Lindskog, C.; Nie, X.; Guo, Y.; Takei, Y.; Yun, J.; Cai, L.; et al. The adult human testis transcriptional cell atlas. Cell Res. 2018, 28, 1141–1157. [Google Scholar] [CrossRef]
- Bhang, D.H.; Kim, B.J.; Kim, B.G.; Schadler, K.; Baek, K.H.; Kim, Y.H.; Hsiao, W.; Ding, B.S.; Rafii, S.; Weiss, M.J.; et al. Testicular endothelial cells are a critical population in the germline stem cell niche. Nat. Commun. 2018, 9, 4379. [Google Scholar] [CrossRef] [PubMed]
- Gaskell, T.L.; Esnal, A.; Robinson, L.L.L.; Anderson, R.A.; Saunders, P.T.K. Immunohistochemical Profiling of Germ Cells Within the Human Fetal Testis: Identification of Three Subpopulations. Biol. Reprod. 2004, 71, 2012–2021. [Google Scholar] [CrossRef]
- Law, N.C.; Oatley, J.M. Developmental underpinnings of spermatogonial stem cell establishment. Andrology 2020, 8, 852–861. [Google Scholar] [CrossRef]
- Du, G.; Oatley, M.J.; Law, N.C.; Robbins, C.; Wu, X.; Oatley, J.M. Proper timing of a quiescence period in precursor prospermatogonia is required for stem cell pool establishment in the male germline. Development 2021, 148, 194571. [Google Scholar] [CrossRef]
- Brinster, R.L.; Avarbock, M.R. Germline transmission of donor haplotype following spermatogonial transplantation. Proc. Natl. Acad. Sci. USA 1994, 91, 11303–11307. [Google Scholar] [CrossRef]
- Nakagawa, T.; Sharma, M.; Nabeshima, Y.; Braun, R.E.; Yoshida, S. Functional hierarchy and reversibility within the murine spermatogenic stem cell compartment. Science 2010, 328, 62–67. [Google Scholar] [CrossRef]
- Hara, K.; Nakagawa, T.; Enomoto, H.; Suzuki, M.; Yamamoto, M.; Simons, B.D.; Yoshida, S. Mouse spermatogenic stem cells continually interconvert between equipotent singly isolated and syncytial states. Cell Stem Cell 2014, 14, 658–672. [Google Scholar] [CrossRef]
- Meng, X.; Lindahl, M.; Hyvonen, M.E.; Parvinen, M.; de Rooij, D.G.; Hess, M.W.; Raatikainen-Ahokas, A.; Sainio, K.; Rauvala, H.; Lakso, M.; et al. Regulation of cell fate decision of undifferentiated spermatogonia by GDNF. Science 2000, 287, 1489–1493. [Google Scholar] [CrossRef]
- Hofmann, M.C.; Braydich-Stolle, L.; Dym, M. Isolation of male germ-line stem cells; influence of GDNF. Dev. Biol. 2005, 279, 114–124. [Google Scholar] [CrossRef]
- Kubota, H.; Avarbock, M.R.; Brinster, R.L. Growth factors essential for selfrenewal and expansion of mouse spermatogonial stem cells. Proc. Natl. Acad. Sci. USA 2004, 101, 16489–16494. [Google Scholar] [CrossRef]
- Martin Laura, A.; Assif, N.; Gilbert, M.; Wijewarnasuriya, D.; Seandel, M. Enhanced fitness of adult spermatogonial stem cells bearing a paternal age-associated FGFR2 mutation. Stem Cell Rep. 2014, 3, 219–226. [Google Scholar] [CrossRef]
- Chen, S.R.; Liu, Y.X. Regulation of spermatogonial stem cell self-renewal and spermatocyte meiosis by Sertoli cell signaling. Reproduction 2015, 149, R159–R167. [Google Scholar] [CrossRef] [PubMed]
- De Falco, T.; Potter, S.J.; Williams, A.V.; Waller, B.; Kan, M.J.; Capel, B. Macrophages contribute to the spermatogonial niche in the adult testis. Cell Rep. 2015, 12, 1107–1119. [Google Scholar] [CrossRef] [PubMed]
- Sawaied, A.; Arazi, E.; AbuElhija, A.; Lunenfeld, E.; Huleihel, M. The presence of colony-stimulating factor-1 and its receptor in different cells of the testis; it involved in the development of spermatogenesis in vitro. Int. J. Mol. Sci. 2021, 22, 2325. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Du, L.; Li, J.; He, Y.; Tang, M. Microenvironment of spermatogonial stem cells: A key factor in the regulation of spermatogenesis. Stem Cell Res. Ther. 2024, 15, 294. [Google Scholar] [CrossRef]
- Masaki, K.; Sakai, M.; Kuroki, S.; Jo, J.I.; Hoshina, K.; Fujimori, Y.; Oka, K.; Amano, T.; Yamanaka, T.; Tachibana, M.; et al. FGF2 has distinct molecular functions from GDNF in the mouse germline niche. Stem Cell Rep. 2018, 10, 1782–1792. [Google Scholar] [CrossRef] [PubMed]
- Binsila, B.K.; Selvaraju, S.; Ghosh, S.K.; Ramya, L.; Arangasamy, A.; Ranjithkumaran, R.; Bhatta, R. EGF, GDNF, and IGF-1 influence the proliferation and stemness of ovine spermatogonial stem cells in vitro. J. Assist. Reprod. Genet. 2020, 37, 2615–2630. [Google Scholar] [CrossRef]
- Zhao, H.; Li, T.; Yang, H.; Mehmood, M.U.; Lu, Y.; Liang, X.; Yang, X.; Xu, H.; Lu, K.; Lu, S. The effects of growth factors on proliferation of spermatogonial stem cells from Guangxi Bama mini-pig. Reprod. Dom. Anim. 2019, 54, 1574–1582. [Google Scholar] [CrossRef]
- Oatley, J.M.; Avarbock, M.R.; Brinster, R.L. Glial cell line-derived neurotrophic factor regulation of genes essential for self-renewal of mouse spermatogonial stem cells is dependent on Src family kinase signaling. J. Biol. Chem. 2007, 282, 25842–25851. [Google Scholar] [CrossRef] [PubMed]
- Parker, N.; Laychur, A.; Sukwani, M.; Orwig, K.E.; Oatley, J.M.; Zhang, C.; Rutaganira, F.U.; Shokat, K.; Wright, W.W. Spermatogonial stem cell numbers are reduced by transient inhibition of GDNF signaling but restored by self-renewing replication when signaling resumes. Stem Cell Rep. 2021, 16, 597–609. [Google Scholar] [CrossRef]
- Jin, C.; Wang, Z.; Li, P.; Tang, J.; Jiao, T.; Li, Y.; Ou, J.; Zou, D.; Li, M.; Mang, X.; et al. Decoding the spermatogonial stem cell niche under physiological and recovery conditions in adult mice and humans. Sci. Adv. 2023, 9, eabq3173. [Google Scholar] [CrossRef]
- Aponte, P.M.; Soda, T.; Teerds, K.J.; Canan Mizrak, S.; van de Kant, H.J.G.; de Rooij, D.G. Propagation of bovine spermatogonial stem cells in vitro. Reproduction 2008, 136, 543–557. [Google Scholar] [CrossRef]
- Kadam, P.H.; Kala, S.; Agrawal, H.; Singh, K.P.; Singh, M.K.; Chauhan, M.S.; Palta, P.; Singla, S.K.; Manik, R.S. Effects of glial cell line-derived neurotrophic factor, fibroblast growth factor 2 and epidermal growth factor on proliferation and the expression of some genes in buffalo (Bubalus bubalis) spermatogonial cells. Reprod. Fertil. Dev. 2013, 25, 1149–1157. [Google Scholar] [CrossRef]
- Kanatsu-Shinohara, M.; Ogonuki, N.; Inoue, K.; Miki, H.; Ogura, A.; Toyokuni, S.; Shinohara, T. Long-term proliferation in culture and germline transmission of mouse male germline stem cells. Biol. Reprod. 2003, 69, 612–616. [Google Scholar] [CrossRef]
- Yang, Q.E.; Kim, D.; Kaucher, A.; Oatley, M.J.; Oatley, J.M. CXCL12-CXCR4 signaling is required for the maintenance of mouse spermatogonial stem cells. J. Cell Sci. 2013, 126, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, M.; Filipponi, D.; Gori, M.; Barrios, F.; Lolicato, F.; Grimaldi, P.; Rossi, P.; Jannini, E.A.; Geremia, R.; Dolci, S. ATRA and KL promote differentiation toward the meiotic program of male germ cells. Cell Cycle 2008, 7, 3878–3888. [Google Scholar] [CrossRef]
- Carlomagno, G.; van Bragt, M.P.; Korver, C.M.; Repping, S.; de Rooij, D.G.; van Pelt, A.M. BMP4-induced differentiation of a rat spermatogonial stem cell line causes changes in its cell adhesion properties. Biol. Reprod. 2010, 83, 742–749. [Google Scholar] [CrossRef]
- Barrios, F.; Filipponi, D.; Campolo, F.; Gori, M.; Bramucci, F.; Pellegrini, M.; Ottolenghi, S.; Rossi, P.; Jannini, E.A.; Dolci, S. SOHLH1 and SOHLH2 control kit expression during postnatal male germ cell development. J. Cell. Sci. 2012, 125, 1455–1464. [Google Scholar]
- Yang, Q.E.; Racicot, K.E.; Kaucher, A.V.; Oatley, M.J.; Oatley, J.M. MicroRNAs 221 and 222 regulate the undifferentiated state in mammalian male germ cells. Development 2013, 140, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, R.M.; Fagoonee, S.; Papa, A.; Webster, K.; Altruda, F.; Nishinakamura, R.; Chai, L.; Pandolfi, P.P. Functional antagonism between Sall4 and Plzf defines germline progenitors. Cell Stem Cell 2012, 10, 284–298. [Google Scholar] [CrossRef]
- Goertz, M.J.; Wu, Z.; Gallardo, T.D.; Hamra, F.K.; Castrillon, D.H. Foxo1 is required in mouse spermatogonial stem cells for their maintenance and the initiation of spermatogenesis. J. Clin. Investig. 2011, 121, 3456–3466. [Google Scholar] [CrossRef]
- Khanehzad, M.; Abolhasani, F.; Hassanzadeh, G.; Nourashrafeddin, S.M.; Hedayatpour, A. Determination of the excitatory effects of MicroRNA-30 in the self-renewal and differentiation process of neonatal mouse spermatogonial stem cells. Galen Med. J. 2020, 9, e1829. [Google Scholar] [CrossRef]
- Yu, M.; Mu, H.; Niu, Z.; Chu, Z.; Zhu, H.; Hua, J. miR-34c enhances mouse spermatogonial stem cells differentiation by targeting Nanos2. J. Cell. Biochem. 2014, 115, 232–242. [Google Scholar] [CrossRef]
- Chen, J.; Gao, C.; Lin, X.; Ning, Y.; He, W.; Zheng, C.; Zhang, D.; Yan, L.; Jiang, B.; Zhao, Y.; et al. The microRNA miR-202 prevents precocious spermatogonial differentiation and meiotic initiation during mouse spermatogenesis. Development 2021, 148, 199799. [Google Scholar] [CrossRef] [PubMed]
- Tong, M.H.; Mitchell, D.A.; McGowan, S.D.; Evanoff, R.; Griswold, M.D. Two miRNA clusters, Mir-17-92 (Mirc1) and Mir-106b-25 (Mirc3), are involved in the regulation of spermatogonial differentiation in mice. Biol. Reprod. 2012, 86, 72. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Lin, X.; Du, T.; Xu, K.; Shen, H.; Wei, F.; Hao, W.; Lin, T.; Lin, X.; Qin, Y.; et al. Targeted disruption of miR-17-92 impairs mouse spermatogenesis by activating mTOR signaling pathway. Medicine 2016, 95, e2713. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Li, H.; Liang, J.; Mei, J.; Cao, Z.; Zhang, L.; Luo, J.; Tang, Y.; Huang, R.; Xia, H.; et al. Sertoli cell-derived exosomal microRNA-486-5p regulates differentiation of spermatogonial stem cell through PTEN in mice. J. Cell. Mol. Med. 2021, 25, 3950–3962. [Google Scholar] [CrossRef] [PubMed]
- Niu, B.; Wu, J.; Mu, H.; Li, B.; Wu, C.; He, X.; Bai, C.; Li, G.; Hua, J. miR-204 regulates the proliferation of dairy goat spermatogonial stem cells via targeting to Sirt1. Rejuvenation Res. 2016, 19, 120–130. [Google Scholar] [CrossRef]
- Gao, Y.; Wu, F.; Ren, Y.; Zhou, Z.; Chen, N.; Huang, Y.; Lei, C.; Chen, H.; Dang, R. MiRNAs expression profiling of bovine (Bos taurus) testes and effect of bta-miR-146b on proliferation and apoptosis in bovine male germline stem cells. Int. J. Mol. Sci. 2020, 21, 3846. [Google Scholar] [CrossRef]
- Kanatsu-Shinohara, M.; Toyokuni, S.; Shinohara, T. CD9 is a surface marker on mouse and rat male germline stem cells. Biol. Reprod. 2004, 70, 70–75. [Google Scholar] [CrossRef]
- Kubota, H.; Avarbock, M.R.; Brinster, R.L. Spermatogonial stem cells share some, but not all, phenotypic and functional characteristics with other stem cells. Proc. Natl. Acad. Sci. USA 2003, 100, 6487–6492. [Google Scholar] [CrossRef]
- Shinohara, T.; Avarbock, M.R.; Brinster, R.L. beta1- and alpha6-integrin are surface markers on mouse spermatogonial stem cells. Proc. Natl. Acad. Sci. USA 1999, 96, 5504–5509. [Google Scholar] [CrossRef]
- Zi, H.; Kokkinaki, M.; Jiang, J.; Dobrinski, I.; Dym, M. Isolation, characterization, and culture of human spermatogonia. Biol. Reprod. 2010, 82, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Schmidt, J.A.; Avarbock, M.R.; Tobias, Y.W.; Carlson, C.A.; Kolon, T.F.; Ginsberg, J.P.; Brinster, R.L. Prepubertal human spermatogonia and mouse gonocytes share conserved gene expressionof germline stem cell regulatory molecules. Proc. Natl. Acad. Sci. USA 2009, 106, 21672–21677. [Google Scholar] [CrossRef]
- Dym, M.; Kokkinaki, M.; He, Z. Spermatogonial stem cells: Mouse and human comparisons. Birth Defects Res. Part C Embryo Today Rev. 2009, 87, 27–34. [Google Scholar] [CrossRef]
- Oatley, J.M.; Avarbock, M.R.; Telaranta, A.I.; Fearon, D.T.; Brinster, R.L. Identifying genes important for spermatogonial stem cell selfrenewal and survival. Proc. Natl. Acad. Sci. USA 2006, 103, 9524–9529. [Google Scholar] [CrossRef] [PubMed]
- Oatley, J.M.; Brinster, R.L. Regulation of spermatogonial stem cell self-renewal in mammals. Ann. Rev. Cell. Dev. Biol. 2008, 24, 263–286. [Google Scholar] [CrossRef] [PubMed]
- Kanatsu-Shinohara, M.; Chen, G.; Morimoto, H.; Shinohara, T. CD2 is a surface marker for mouse and rat spermatogonial stem cells. J. Reprod. Develop. 2020, 66, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jin, C.; Li, P.; Li, Y.; Tang, J.; Yu, Z.; Jiao, T.; Ou, J.; Wang, H.; Zou, D.; et al. FOXC2 marks and maintains the primitive spermatogonial stem cells subpopulation in the adult testis. eLife 2023, 12, RP85380. [Google Scholar] [CrossRef]
- Wu, J.; Liao, M.; Zhu, H.; Kang, K.; Mu, H.; Song, W.; Niu, Z.; He, X.; Bai, C.; Li, G.; et al. CD49f-positive testicular cells in Saanen dairy goat were identified as spermatogonia-like cells by miRNA profiling analysis. J. Cell. Biochem. 2014, 115, 1712–1723. [Google Scholar] [CrossRef]
- Lu, X.; Yin, P.; Li, H.; Gao, W.; Jia, H.; Ma, W. Transcriptome analysis of key genes involved in the initiation of spermatogonial stem cell differentiation. Genes 2024, 15, 141. [Google Scholar] [CrossRef]
- Zhuo, L.; Zhou, Y.; Tian, J.; Li, Y.; Xie, Z.; Pei, C.; Yan, B.; Ma, L. The role of miR-199a-3p in inhibiting the proliferation of spermatogonial stem cells under heat stress. Theriogenology 2023, 211, 56–64. [Google Scholar] [CrossRef]
- Geng, Q.; Hu, C.; Zhao, Z.; Wang, Z.; Cheng, F.; Chen, J.; Zuo, Q.; Zhang, Y. miR-1458 is inhibited by low concentrations of Vitamin B6 and targets TBX6 to promote the formation of spermatogonial stem cells in Rugao Yellow Chicken. Poult. Sci. 2025, 104, 104583. [Google Scholar] [CrossRef]
- Yeh, J.R.; Zhang, X.; Nagano, M.C. Establishment of a short-term in vitro assay for mouse spermatogonial stem cells. Biol. Reprod. 2007, 77, 897–904. [Google Scholar] [CrossRef][Green Version]
- Di Persio, S.; Neuhaus, N. Human spermatogonial stem cells and their niche in male (in)fertility: Novel concepts from single-cell RNA-sequencing. Hum. Reprod. 2023, 38, 1–13. [Google Scholar] [CrossRef]
- Izadyar, F.; Spierenberg, G.T.; Creemers, L.B.; den Ouden, K.; de Rooij, D.G. Isolation and purification of type A spermatogonia from the bovine testis. Reproduction 2002, 124, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Damyanova, K.B.; Nixon, B.; Johnston, S.D.; Gambini, A.; Benitez, P.P.; Lord, T. Spermatogonial stem cell technologies: Applications from human medicine to wildlife conservation. Biol. Reprod. 2024, 111, 757–779. [Google Scholar] [CrossRef]
- Xi, H.M.; Ren, Y.J.; Ren, F.; Li, Y.; Feng, T.Y.; Wang, Z.; Du, Y.Q.; Zhang, L.K.; Hu, J.H. Recent advances in isolation, identification, and culture of mammalian spermatogonial stem cells. Asian J. Androl. 2022, 24, 5–14. [Google Scholar] [CrossRef]
- Binsila, K.B.; Selvaraju, S.; Ghosh, S.K.; Parthipan, S.; Archana, S.S.; Arangasamy, A.; Prasad, J.K.; Bhatta, R.; Ravindra, J.P. Isolation and enrichment of putative spermatogonial stem cells from ram (Ovis aries) testis. Anim. Reprod. Sci. 2018, 196, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Nabulindo, N.W.; Nguhiu-Mwangi, J.; Kipyegon, A.N.; Ogugo, M.; Muteti, C.; Tiambo, C.; Oatley, M.J.; Oatley, J.M.; Kemp, S. Culture of kenyan goat (Capra hircus) undifferentiated spermatogonia in feeder-free conditions. Front. Vet. Sci. 2022, 9, 894075. [Google Scholar] [CrossRef]
- Harkey, M.A.; Asano, A.; Zoulas, M.E.; Torok-Storb, B.; Nagashima, J.; Travis, A. Isolation, genetic manipulation, and transplantation of canine spermatogonial stem cells: Progress toward transgenesis through the male germline. Reproduction 2013, 146, 75–90. [Google Scholar] [CrossRef]
- Das Dipak Bhuyan, A.; Lalmalsawma, T.; Pratim Das, P.; Koushik, S.; Chauhan, M.S.; Bhuyan, M. Propagation of porcine spermatogonial stem cells in serum-free culture conditions using knockout serum replacement. Reprod. Dom. Anim. 2023, 58, 219–229. [Google Scholar]
- Sahare, M.; Kim, S.M.; Otomo, A.; Komatsu, K.; Minami, N.; Yamada, M.; Imai, H. Factors supporting long-term culture of bovine male germ cells. Reprod. Fertil. Dev. 2015, 28, 2039–2050. [Google Scholar] [CrossRef]
- Bedford-Guaus, S.J.; Kim, S.; Mulero, L.; Vaquero, J.M.; Morera, C.; Adan-Milanès, R.; Veiga, A.; Raya, A. Molecular markers of putative spermatogonial stem cells in the domestic cat. Reprod. Dom. Anim. 2017, 52, 177–186. [Google Scholar] [CrossRef]
- Pramod, R.K.; Varughese, D.; Jameel, A.J.; Panda, B.N.; Goswami, S.; Mitra, A. Cryopreserved ovine spermatogonial stem cells maintain stemness and colony forming ability in vitro. Asian Pac. J. Reprod. 2023, 12, 273–280. [Google Scholar] [CrossRef]
- Parekh, P.A.; Garcia, T.X.; Hofmann, M.C. Regulation of GDNF expression in Sertoli cells. Reproduction 2019, 157, R95–R107. [Google Scholar] [CrossRef]
- Feng, W.; Chen, S.; Do, D.; Liu, Q.; Deng, Y.; Lei, X.; Luo, C.; Huang, B.; Shi, D. Isolation and identification of prepubertal buffalo (Bubalus bubalis) spermatogonial stem cells. Asian Australas. J. Anim. Sci. 2016, 29, 1407–1415. [Google Scholar] [CrossRef]
- Ibtisham, F.; Tang, S.; Song, Y.; Wanze, W.; Xiao, M.; Honaramooz, A.; An, L. Optimal isolation, culture, and in vitro propagation of spermatogonial stem cells in Huaixiang chicken. Reprod. Dom. Anim. 2024, 59, e14661. [Google Scholar] [CrossRef]
- Shirazi, M.S.; Heidari, B.; Naderi, M.M.; Behzadi, B.; Sarvari, A.; Borjian-Boroujeni, S.; Farab, M.; Shirazi, A. Transplantation of goat spermatogonial stem cells into the mouse rete testis. Int. J. Anim. Biol. 2015, 1, 61–68. [Google Scholar]
- Costa, G.M.J.; Avelar, G.F.; Lacerda, S.M.S.N.; Figueiredo, A.F.A.; Tavares, A.O.; Rezende-Neto, J.V.; Martins, F.G.P.; França, L.R. Horse spermatogonial stem cell cryopreservation: Feasible protocols and potential biotechnological applications. Cell Tissue Res. 2017, 370, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wan, W.; Li, B.; Zhang, X.; Zhang, M.; Wu, Z.; Yang, H. Isolation and in vitro expansion of porcine spermatogonial stem cells. Reprod. Dom. Anim. 2022, 57, 210–220. [Google Scholar] [CrossRef]
- Kubota, H.; Wu, X.; Goodyear, S.M.; Avarbock, M.R.; Brinster, R.L. Glial cell line-derived neurotrophic factor and endothelial cells promote self-renewal of rabbit germ cells with spermatogonial stem cell properties. FASEB J. 2011, 25, 2604–2614. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Indu, S.; Devi, A.N.; Sahadevan, M.; Sengottaiyan, J.; Basu, A.; Raj, K.S.; Kumar, P.G. Expression profiling of stemness markers in testicular germline stem cells from neonatal and adult Swiss albino mice during their transdifferentiation in vitro. Stem Cell Res. Ther. 2024, 15, 93. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, Y.; Ikeda, S.; Minami, N.; Yamada, M.; Imai, H. Long-term culture of undifferentiated spermatogonia isolated from immature and adult bovine testes. Mol. Reprod. Dev. 2018, 85, 236–249. [Google Scholar]
- Zhang, X.Y.; Li, T.T.; Liu, Y.R.; Geng, S.S.; Luo, A.L.; Jiang, M.S.; Liang, X.W.; Shang, J.H.; Lu, K.H.; Yang, X.G. Transcriptome analysis revealed differences in the microenvironment of spermatogonial stem cells in seminiferous tubules between prepubertal and adult buffaloes. Reprod. Dom. Anim. 2021, 56, 629–641. [Google Scholar] [CrossRef]
- Cai, H.; Jiang, Y.; Zhang, S.; Cai, N.N.; Zhu, W.K.; Yang, R.; Tang, B.; Li, Z.Y.; Zhang, X.M. Culture bovine prospermatogonia with 2i medium. Andrologia 2021, 53, e14056. [Google Scholar] [CrossRef]
- Yang, R.; Zhang, B.; Wang, Y.; Zhang, Y.; Zhao, Y.; Jiang, D.; Chen, L.; Tang, B.; Zhang, X. H3K9me3 Levels Affect the Proliferation of Bovine Spermatogonial Stem Cells. Int. J. Mol. Sci. 2024, 25, 9215. [Google Scholar] [CrossRef]
- Baarends, W.; Grootegoed, J. Molecular biology of male gametogenesis. In Molecular Biology in Reproductive Medicine; Parthenon Publishing: Nashville, TN, USA, 1999; pp. 271–295. [Google Scholar]
- Kashfi, A.; Sani, R.N.; Ahmadi-hamedani, M. The beneficial effect of equine chorionic gonadotropin hormone (eCG) on the in vitro co-culture of bovine spermatogonial stem cell with Sertoli cells. Comp. Clin. Pathol. 2019, 28, 701–704. [Google Scholar] [CrossRef]
- Navid, S.; Rastegar, T.; Baazm, M.; Alizadeh, R.; Talebi, A.; Gholami, K.; Khosravi-Farsani, S.; Koruji, M.; Abbasi, M. In vitro effects of melatonin on colonization of neonate mouse spermatogonial stem cells. Syst. Biol. Reprod. Med. 2017, 63, 370–381. [Google Scholar] [CrossRef]
- Feng, T.Y.; Li, Q.; Ren, F.; Xi, H.M.; Lv, D.L.; Yu Li, Y.; Hu, J.H. Melatonin protects goat spermatogonial stem cells against oxidative damage during cryopreservation by improving antioxidant capacity and inhibiting mitochondrial apoptosis pathway. Oxid. Med. Cell. Longev. 2020, 31, 5954635. [Google Scholar] [CrossRef]
- Salimi, H.; Feyli, P.R.; Yari, K.; Wong, A.; Moghaddam, A.A. Low testosterone concentration improves colonisation and viability in the co-cultured goat spermatogonial stem cell with Sertoli cells. Reprod. Domest. Anim. 2024, 59, e14729. [Google Scholar] [CrossRef] [PubMed]
- Jafarnejad, A.; Aminafshar, M.; Zandi, M.; Sanjabi, M.R.; Kashan, N.E. Optimization of in vitro culture and transfection condition of bovine primary spermatogonial stem cells. S. Afr. J. Anim. Sci. 2018, 48, 108–116. [Google Scholar] [CrossRef]
- Kushki, D.; Azarnia, M.; Gholami, M.R. Antioxidant effects of selenium on seminiferous tubules of immature mice testis. Zahedan J. Res. Med. Sci. 2015, 17, 29–33. [Google Scholar] [CrossRef]
- Casao, A.; Peña-Delgado, V.; Pérez-Pe, R. From spermatogenesis to fertilisation: The role of melatonin on ram spermatozoa. Domest. Anim. Endocrinol. 2025, 91, 106916. [Google Scholar] [CrossRef]
- Zolfaghari, K.; Zandi, M.; Sanjabi, M.R.; Ghaedrahmati, A. In Vitro culture of ovine spermatogonial stem cells: Effects of grape seed extracts and vitamin C. Gene Cell Tissue 2024, 11, e135750. [Google Scholar] [CrossRef]
- Ogawa, T.; Dobrinski, I.; Avarbock, M.R.; Brinster, R.L. Transplantation of male germ line stem cells restores fertility in infertile mice. Nat. Med. 2000, 6, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Arregui, L.; Dobrinski, I. Xenografting of testicular tissue pieces: Twelve years of an in vivo spermatogenesis system. Reproduction 2014, 148, R71–R84. [Google Scholar] [CrossRef]
- Vurchio, V.; Colombo, M.; Pasquariello, R.; Luvoni, G.C. Cryopreservation and culture strategies for testicular tissue and cells in small and large animals. Front. Vet. Sci. 2025, 12, 1638248. [Google Scholar] [CrossRef]
- Han, Z.; Liu, X.; Wang, H.; Qazi, I.H.; Wang, L.; Du, R.; Dai, X.; Xu, C. Testicular tissue cryopreservation and transplantation as a strategy for feline conservation: A review of research advances. Front. Vet. Sci. 2025, 12, 1572150. [Google Scholar] [CrossRef]
- Tomcy, T.A.; Binsila, B.; Sadikh, M.; Krishnappa, B.; Ramachandran, N.; Arangasamy, A.; Aranganathan, V.; Selvaraju, S. Fetal bovine serum in cryomedia protects sheep spermatogonial stem cells and preserves stemness characteristics during cryopreservation. Vitr. Cell Dev. Biol. Anim. 2025, 21. [Google Scholar] [CrossRef]
- Yang, F.; Zhao, J.; Li, Y.; Niu, C.; Zheng, Y. Cryopreservation of spermatogonial stem cells: An overview of cryoprotectants. Reprod. Biomed. Online 2025, 51, 105023. [Google Scholar] [CrossRef]
- Fathi, S.; Nazari, H.; Arabi, M.; Afzali, A.; Ahmadi, E. The antioxidant capacity and protective ability of astaxanthin in cryopreservation of mouse spermatogonial stem cells. Cryobiology 2025, 120, 105261. [Google Scholar] [CrossRef]
- Tang, S.; Jones, C.; Davies, J.; Lane, S.; Coward, K. A comparative analysis of vitrification and two slow freezing methods for gonocyte-containing neonatal calf testicular tissue and subsequent in vitro culture. Vitr. Model 2025, 4, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chicaiza-Cabezas, N.; Garcia-Herreros, M.; Aponte, P.M. Germplasm cryopreservation in bulls: Effects of gonadal tissue type, cryoprotectant agent, and freezing-thawing rates on sperm quality parameters. Cryobiology 2023, 110, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Kaya, C.; Esin, B.; Akar, M.; Can, C.; Çevik, M. Investigation of the efficacy of different cryoprotectants in the freezing of testicular tissue and epididymal sperm: Spermatogonial parameters, tissue viability and PARP-1 gene expression. Cryobiology 2024, 117, 104982. [Google Scholar] [CrossRef]
- Ciccarelli, M.; Giassetti, M.I.; Miao, D.; Oatleya, M.J.; Robbins, C.; Lopez-Biladeau, B.; Waqas, M.S.; Tibarya, A.; Whitelaw, B.; Lillico, S.; et al. Donor-derived spermatogenesis following stem cell transplantation in sterile NANOS2 knockout males. Proc. Natl. Acad. Sci. USA 2020, 117, 24195–24204. [Google Scholar] [CrossRef] [PubMed]
- Blank, M.H.; Kawaoku, A.J.T.; Rui, B.R.; Carreira, A.C.O.; Hamilton, T.R.S.; Goissis, M.D.; Pereira, R.J.G. Successful xenotransplantation of testicular cells following fractionated chemotherapy of recipient birds. Sci. Rep. 2024, 14, 3085. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Ding, L.J.; Sun, G.B.; Sun, P.X.; He, X.H.; Ni, L.G.; Li, B.C. Transgenic sperm produced by electrotransfection and allogeneic transplantation of chicken fetal spermatogonial stem cells. Mol. Reprod. Dev. 2010, 77, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Tang, L.; Bondareva, A.; Honaramooz, A.; Tanco, V.; Dores, C.; Megee, S.; Modelski, M.; Rodriguez-Sosa, J.R.; Paczkowski, M.; et al. Viral transduction of male germline stem cells results in transgene transmission after germcell transplantation in pigs. Biol. Reprod. 2013, 88, 27. [Google Scholar] [CrossRef]
- Snedaker, A.K.; Honaramooz, A.; Dobrinski, I. A game of cat and mouse: Xenografting of testis tissue from domestic kittens results in complete cat spermatogenesis in a mouse host. J. Androl. 2004, 25, 926–930. [Google Scholar] [CrossRef]
- Segunda, M.N.; Cortez, J.; Díaz, C.; Arancibia, R.; Torres, C.G.; Parraguez, V.H.; De Los Reyes, M.; Peralta, O.A. Potential of mesenchymal stromal/stem cells and spermatogonial stem cells for survival and colonization in bull recipient testes after allogenic transplantation. Theriogenology 2024, 230, 192–202. [Google Scholar] [CrossRef]
- Ogawa, T.; Aréchaga, J.M.; Avarbock, M.R.; Brinster, R.L. Transplantation of testis germinal cells into mouse seminiferous tubules. Int. J. Dev. Biol. 1997, 41, 111–122. [Google Scholar]
- Nagano, M.; Avarbock, M.R.; Brinster, R.L. Pattern and kinetics of mouse donor spermatogonial stem cell colonization in recipient testes. Biol. Reprod. 1999, 60, 1429–1436. [Google Scholar] [CrossRef]
- Aponte, P.M. Spermatogonial stem cells: Current biotechnological advances in reproduction and regenerative medicine. World J. Stem Cells 2015, 7, 669–680. [Google Scholar] [CrossRef]
- Zhao, X.; Wan, W.; Zhang, X.; Wu, Z.; Yang, H. Spermatogonial Stem Cell Transplantation in Large Animals. Animals 2021, 11, 918. [Google Scholar] [CrossRef]
- Oback, B.; Cossey, D.A. Chimaeras, complementation, and controlling the male germline. Trends Biotechnol. 2023, 41, 1237–1247. [Google Scholar] [CrossRef]
- Lara, N.M.L.; Goldsmith, T.; Rodriguez-Villamil, P.; Ongaratto, F.; Solin, S.; Webster, D.; Ganbaatar, U.; Hodgson, S.; Corbière, S.M.A.S.; Bondareva, A.; et al. DAZL Knockout Pigs as Recipients for Spermatogonial Stem Cell Transplantation. Cells 2023, 12, 2582. [Google Scholar] [CrossRef]
- Du, H.; Li, Y.; Ni, A.; Liu, S.; Chen, J.; Sun, Y. Research Progress in the Establishment of Sterile Hosts and Their Usage in Conservation of Poultry Genetic Resources. Agriculture 2024, 14, 1111. [Google Scholar] [CrossRef]
- Wang, J.; Xue, X.; Fan, K.; Liu, Q.; Zhang, S.; Peng, M.; Zhou, J.; Cao, Z. Moderate hypoxia modulates ABCG2 to promote the proliferation of mouse spermatogonial stem cells by maintaining mild ROS levels. Theriogenology 2020, 145, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Shakeel, M.; Choi, Y.; Yoon, M. Expression pattern of germ cell markers in cryptorchid stallion Testes. Reprod. Dom. Anim. 2024, 59, e14561. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gao, W.J.; Deng, S.L.; Liu, X.; Jia, H.; Ma, W.Z. High temperature suppressed SSC self-renewal through S phase cell cycle arrest but not apoptosis. Stem Cell Res. Ther. 2019, 10, 227. [Google Scholar] [CrossRef]
- Lymperi, S.; Giwercman, A. Endocrine disruptors and testicular function. Metabolism 2018, 86, 79–90. [Google Scholar] [CrossRef]
- Sharpe, R.M.; Skakkebaek, N.E. Are oestrogens involved in falling sperm counts and disorders of the male reproductive tract? Lancet 1993, 341, 1392–1396. [Google Scholar] [CrossRef] [PubMed]
- TranGuzman, A.; Moradian, R.; Cui, H.; Culty, M. In vitro impact of genistein and mono(2-ethylhexyl) phthalate (MEHP) on the eicosanoid pathway in spermatogonial stem cells. Reprod. Toxicol. 2022, 107, 150–165. [Google Scholar] [CrossRef]
- Kim, S.H.; Shin, S.H.; Kim, S.M.; Jung, S.E.; Shin, B.J.; Ahn, J.S.; Lim, K.T.; Kim, D.H.; Lee, K.; Ryu, B.J. Bisphenol analogs downregulate the self-renewal potential of spermatogonial stem cells. World J. Mens. Health 2025, 43, 154–165. [Google Scholar] [CrossRef]
- Stanojević, M.; Sollner Dolenc, M. Mechanisms of bisphenol A and its analogs as endocrine disruptors via nuclear receptors and related signaling pathways. Arch. Toxicol. 2025, 99, 2397–2417. [Google Scholar] [CrossRef]
- Ahn, J.S.; Won, J.H.; Kim, D.Y.; Jung, S.E.; Kim, B.J.; Kim, J.M.; Ryu, B.Y. Transcriptome alterations in spermatogonial stem cells exposed to bisphenol A. Anim. Cells Syst. 2022, 26, 70–83. [Google Scholar] [CrossRef]
- Cao, Y.; Wang, J.; Li, X.; Liu, B.; Li, C.; Sun, Y.; Zou, K. Gastrodin protects porcine Sertoli cells from zearalenone-induced abnormal secretion of glial cell line-derived neurotrophic factor through the NOTCH signaling pathway. Reprod. Biol. 2023, 23, 100781. [Google Scholar] [CrossRef] [PubMed]
- Duffin, K.; Mitchell, R.T.; Brougham, M.F.H.; Hamer, G.; van Pelt, A.M.M.; Mulder, C.L. Impacts of cancer therapy on male fertility: Past and present. Mol. Asp. Med. 2024, 100, 101308. [Google Scholar] [CrossRef] [PubMed]
- Lopes, F.; Tholeti, P.; Adiga, S.K.; Anderson, R.A.; Mitchell, R.T.; Spears, N. Chemotherapy induced damage to spermatogonial stem cells in prepubertal mouse in vitro impairs long-term spermatogenesis. Toxicol. Rep. 2021, 8, 114–123. [Google Scholar] [CrossRef]
- Tharmalingam, M.D.; Matilionyte, G.; Wallace, W.H.B.; Stukenborg, J.B.; Jahnukainen, K.; Oliver, E.; Goriely, A.; Lane, S.; Guo, J.; Cairns, B.; et al. Cisplatin and carboplatin result in similar gonadotoxicity in immature human testis with implications for fertility preservation in childhood cancer. BMC Med. 2020, 18, 374. [Google Scholar] [CrossRef]
- Soares, J.M.; Avelar, G.F.; De França, L.R. The seminiferous epithelium cycle and its duration in different breeds of dog (Canis familiaris). J. Anat. 2009, 215, 462–471. [Google Scholar] [CrossRef]
- Fayomi, A.P.; Orwig, K.E. Spermatogonial stem cells and spermatogenesis in mice, monkeys and men. Stem Cell Res. 2018, 29, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Pieri, N.C.G.; De Souza, A.F.; Mançanares, A.C.F.; Roballo, K.; Casals, J.; Martins, D.D.S.; Ambrósio, C.E. Immunolocalization of proteins in the spermatogenesis process of canine. Reprod. Domest. Anim. 2016, 52, 170–176. [Google Scholar] [CrossRef]
- Lee, K.H.; Lee, R.; Lee, W.Y.; Kim, D.H.; Chung, H.J.; Kim, J.H.; Kim, N.H.; Choi, S.H.; Kim, J.H.; Song, H. Identification and in vitro derivation of spermatogonia in beagle testis. PLoS ONE 2014, 9, e109963. [Google Scholar] [CrossRef]
- Lee, W.Y.; Lee, R.; Park, H.J.; Do, J.T.; Park, C.; Kim, J.H.; Jhunc, H.; Leed, J.H.; Hur, T.; Song, H. Analysis of putative biomarkers of undifferentiated spermatogonia in dog testis. Anim. Reprod. Sci. 2017, 185, 174–180. [Google Scholar] [CrossRef]
- Pieri, N.C.G.; Mançanares, A.C.F.; de Souza, A.F.; Fernandes, H.; Diaza, A.M.G.; Bressan, F.F.; Roballo, K.C.S.; Casals, J.B.; Binelli, M.; Ambrósio, C.E.; et al. Xenotransplantation of canine spermatogonial stem cells (cSSCs) regulated by FSH promotes spermatogenesis in infertile mice. Stem Cell Res. Ther. 2019, 10, 135. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Yan, G.J.; Ge, Q.Y.; Yu, F.; Zhao, X.; Diao, Z.Y.; Wang, Z.Q.; Yang, Z.Z.; Sun, H.X.; Hu, Y.L. FSH acts on the proliferation of type a spermatogonia via Nur77 that increases GDNF expression in the Sertoli cells. FEBS Lett. 2011, 15, 2437–2444. [Google Scholar] [CrossRef] [PubMed]
- Sluka, P.; O’Donnell, L.; Bartles, J.R.; Stanton, P.G. FSH regulates the formation of adherens junctions and ectoplasmic specialisations between rat Sertoli cells in vitro and in vivo. J. Endocrinol. 2006, 189, 381–395. [Google Scholar] [CrossRef] [PubMed]
- Orth, J.M.; McGuinness, M.P.; Qiu, J.; Jester, W.F.; Li, L.H. Use of in vitro systems to study male germ cell development in neonatal rats. Theriogenology 1998, 49, 431–439. [Google Scholar] [CrossRef]
- Kasimanickam, V.R.; Kasimanickam, R.K. Sertoli, Leydig, and spermatogonial cells’ specific gene and protein expressions as dog testes evolve from immature into mature states. Animals 2022, 12, 271. [Google Scholar] [CrossRef]
- Reding, S.C.; Stepnoski, A.L.; Cloninger, L.W.; Oatley, J.M. THY1 is a conserved marker of undifferentiated spermatogonia in the pre-pubertal bull testis. Reproduction 2010, 139, 893–903. [Google Scholar] [CrossRef]
- Tarnawa, E.D.; Baker, M.D.; Aloisio, G.M.; Carr, B.R.; Castrillon, D.H. Gonadal expression of foxo1, but not foxo3, is conserved in diverse mammalian species. Biol. Reprod. 2013, 88, 103. [Google Scholar] [CrossRef]
- Yates, D.; Hayes, G.; Heffernan, M.; Beynon, R. Incidence of cryptorchidism in dogs and cats. Vet. Rec. 2003, 152, 502–504. [Google Scholar] [CrossRef]
- Lee, W.Y.; Lee, R.; Song, H.; Hur, T.Y.; Lee, S.; Ahn, J.; Jhun, H. Establishment of a surgically induced cryptorchidism canine recipient model for spermatogonial stem cell transplantation. Lab. Anim. Res. 2016, 32, 257–266. [Google Scholar] [CrossRef]
- Jhun, H.; Lee, W.Y.; Park, J.K.; Hwang, S.G.; Park, H.J. Transcriptomic analysis of testicular gene expression in a dog model of experimentally induced cryptorchidism. Cells 2022, 11, 2476. [Google Scholar] [CrossRef]
- Shiraishi, K.; Takihara, H.; Matsuyama, H. Elevated scrotal temperature, but not varicocele grade, reflects testicular oxidative stress-mediated apoptosis. World J. Urol. 2010, 28, 359–364. [Google Scholar] [CrossRef]
- Yin, Y.; Hawkins, K.L.; DeWolf, W.C.; Morgentaler, A. Heat stress causes testicular germ cell apoptosis in adult mice. J. Androl. 1997, 18, 159–165. [Google Scholar] [CrossRef]
- Agoulnik, A.I.; Huang, Z.; Ferguson, L. Spermatogenesis in cryptorchidism. Methods Mol. Biol. 2012, 825, 127–147. [Google Scholar] [PubMed]
- Reifarth, L.; Körber, H.; Packeiser, E.M.; Goericke-Pesch, S. Detection of spermatogonial stem cells in testicular tissue of dogs with chronic asymptomatic orchitis. Front. Vet. Sci. 2023, 10, 1205064. [Google Scholar] [CrossRef] [PubMed]
- Vorathavorn, V.I.; Sykes, J.E.; Feldman, D.G. Cryptococcosis as an emerging systemic mycosis in dogs. J. Vet. Emerg. Crit. Care 2013, 23, 489–497. [Google Scholar] [CrossRef]
- Malik, R.; Dill-Macky, E.; Martin, P.; Wigney, D.I.; Muir, D.B.; Love, D.N. Cryptococcosis in dogs: A retrospective study of 20 consecutive cases. J. Med. Vet. Mycol. 1995, 33, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Lester, S.J.; Kowalewich, N.J.; Bartlett, K.H.; Krockenberger, M.B.; Fairfax, T.M.; Malik, R. Clinicopathologic features of an unusual outbreak of cryptococcosis in dogs, cats, ferrets, and a bird: 38 cases (January to July 2003). J. Am. Vet. Med. Assoc. 2004, 225, 1716–1722. [Google Scholar] [CrossRef]
- Kuberka, M.; Przadka, P.; Dzimira, S. Concurrent Leydig and Sertoli Cell Tumors Associated with Testicular Mycosis in a Dog: A Case Report and Literature Review. Pathogens 2025, 14, 752. [Google Scholar] [CrossRef]
- Kim, Y.; Turner, D.; Nelson, J.; Dobrinski, I.; McEntee, M.; Travis, A.J. Production of donor-derived sperm after spermatogonial stem cell transplantation in the dog. Reproduction 2008, 136, 823–831. [Google Scholar] [CrossRef]
- Hur, T.J.; Lee, S.H.; Ock, S.A.; Song, H.; Park, H.J.; Lee, R.; Sung, S.H.; Jhun, H.; Lee, W.Y. Dose-dependent effects of busulfan on dog testes in preparation for spermatogonial stem cell transplantation. Lab. Anim. Res. 2017, 33, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Lee, W.Y.; Kim, D.H.; Lee, S.H.; Do, J.T.; Park, C.; Kim, J.H.; Choi, Y.S.; Song, H. Vitrified canine testicular cells allow the formation of spermatogonial stem cells and seminiferous tubules following their xenotransplantation into nude mice. Sci. Rep. 2016, 6, 21919. [Google Scholar] [CrossRef] [PubMed]
- Machado da Silva, L.D. Canine and Feline Testicular Preservation. Animals 2022, 12, 124. [Google Scholar] [CrossRef]
- Stukenborg, J.B. The use of testicular organoids in advancing future treatments for male factor infertility. Fertil. Steril. 2025, 124, 406–416. [Google Scholar] [CrossRef]
- Bourdon, G.; Cadoret, V.; Charpigny, G.; Couturier-Tarrade, A.; Dalbies-Tran, R.; Flores, M.-J.; Froment, P.; Raliou, M.; Reynaud, K.; Saint-Dizier, M.; et al. Progress and challenges in developing organoids in farm animal species for the study of reproduction and their applications to reproductive biotechnologies. Vet. Res. 2021, 52, 42. [Google Scholar] [CrossRef] [PubMed]
| Animal | Factor | Influence on Biological Aspects of SSCs | Reference |
|---|---|---|---|
| mouse | melatonin [100 µM] | cell viability improvement | [96] |
| goat | melatonin [1 μM] added to the freezing medium | cell viability improvement during cryopreservation | [97] |
| testosterone [60 μg/mL] | improvement in cell viability and colonization | [98] | |
| calf | eCG [5 IU/mL] | cell-colony formation | [95] |
| vitamin C [50 µg/mL] | improvement in cell viability and colonization | [99] | |
| α-tocopherol analog [5 µg/mL] | improvement in cell viability and colonization | [100] | |
| sheep | melatonin [10−7 M] | improvement in cell differentiation | [101] |
| vitamin C [50 µg/mL] | cell viability improvement | [102] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Squillacioti, C.; Mirabella, N.; Iasevoli, M.; Tafuri, S.; Iervolino, V.; Pelagalli, A. Spermatogonial Stem Cells in Domestic Animals: Current Insights and Future Directions with a Focus on Dogs. Vet. Sci. 2025, 12, 1047. https://doi.org/10.3390/vetsci12111047
Squillacioti C, Mirabella N, Iasevoli M, Tafuri S, Iervolino V, Pelagalli A. Spermatogonial Stem Cells in Domestic Animals: Current Insights and Future Directions with a Focus on Dogs. Veterinary Sciences. 2025; 12(11):1047. https://doi.org/10.3390/vetsci12111047
Chicago/Turabian StyleSquillacioti, Caterina, Nicola Mirabella, Mario Iasevoli, Simona Tafuri, Valeria Iervolino, and Alessandra Pelagalli. 2025. "Spermatogonial Stem Cells in Domestic Animals: Current Insights and Future Directions with a Focus on Dogs" Veterinary Sciences 12, no. 11: 1047. https://doi.org/10.3390/vetsci12111047
APA StyleSquillacioti, C., Mirabella, N., Iasevoli, M., Tafuri, S., Iervolino, V., & Pelagalli, A. (2025). Spermatogonial Stem Cells in Domestic Animals: Current Insights and Future Directions with a Focus on Dogs. Veterinary Sciences, 12(11), 1047. https://doi.org/10.3390/vetsci12111047









