Identification of Candidate Genes for Min Pig Villi Hair Traits by Genome-Wide Association of Copy Number Variation
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals and Phenotype Recording
2.3. Genotyping and Quality Control
2.4. Genome-Wide Association Study
2.5. Gene Enrichment Analysis and QTL Co-Location
2.6. Validation of CNVs by Real-Time Quantitative PCR (qRT-PCR)
3. Results
3.1. Phenotype Description
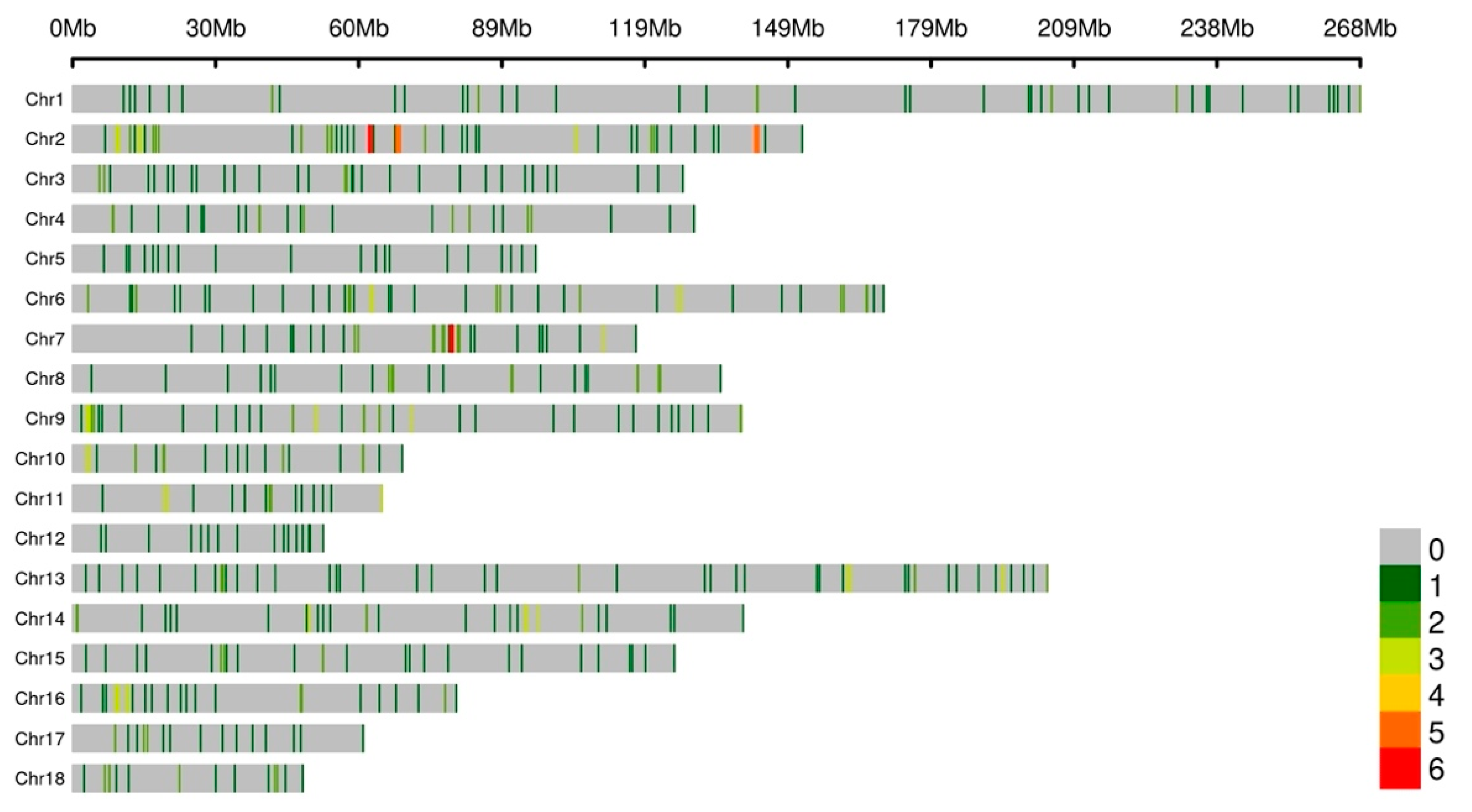
3.2. Copy Number Variation Genotyping
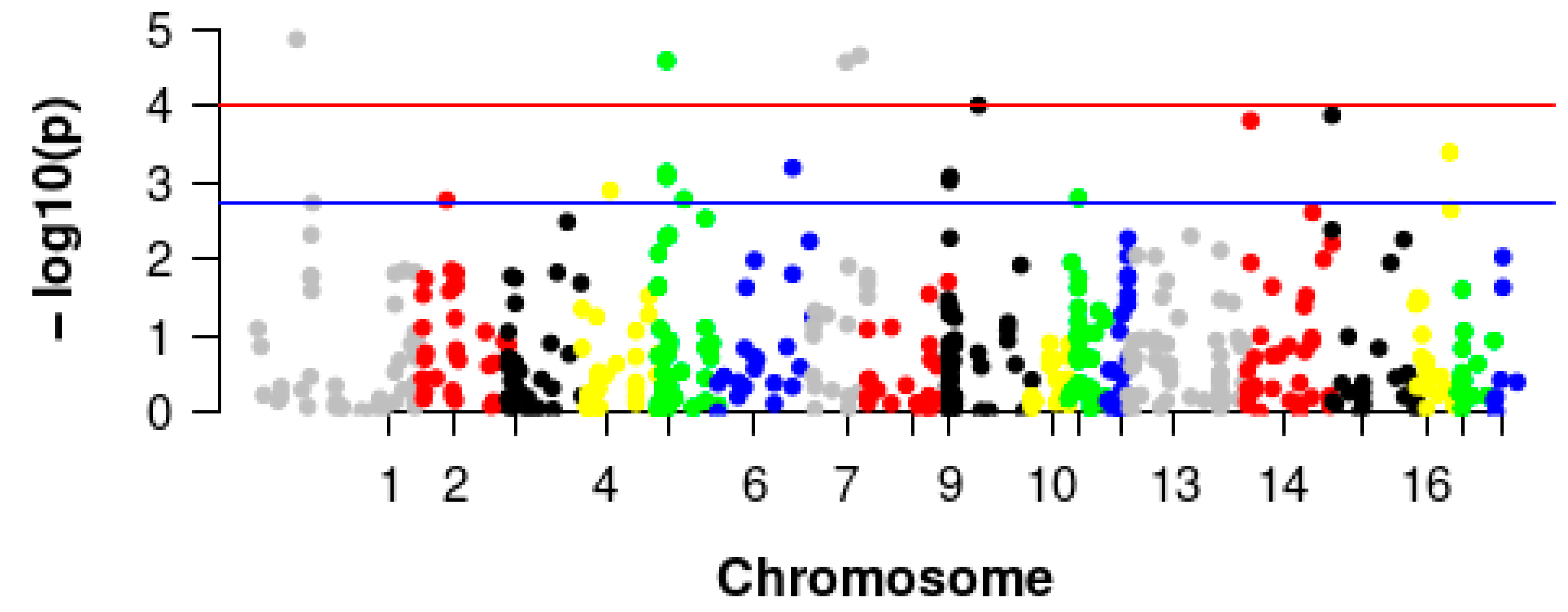
3.3. GWAS of Pig Villi Hair
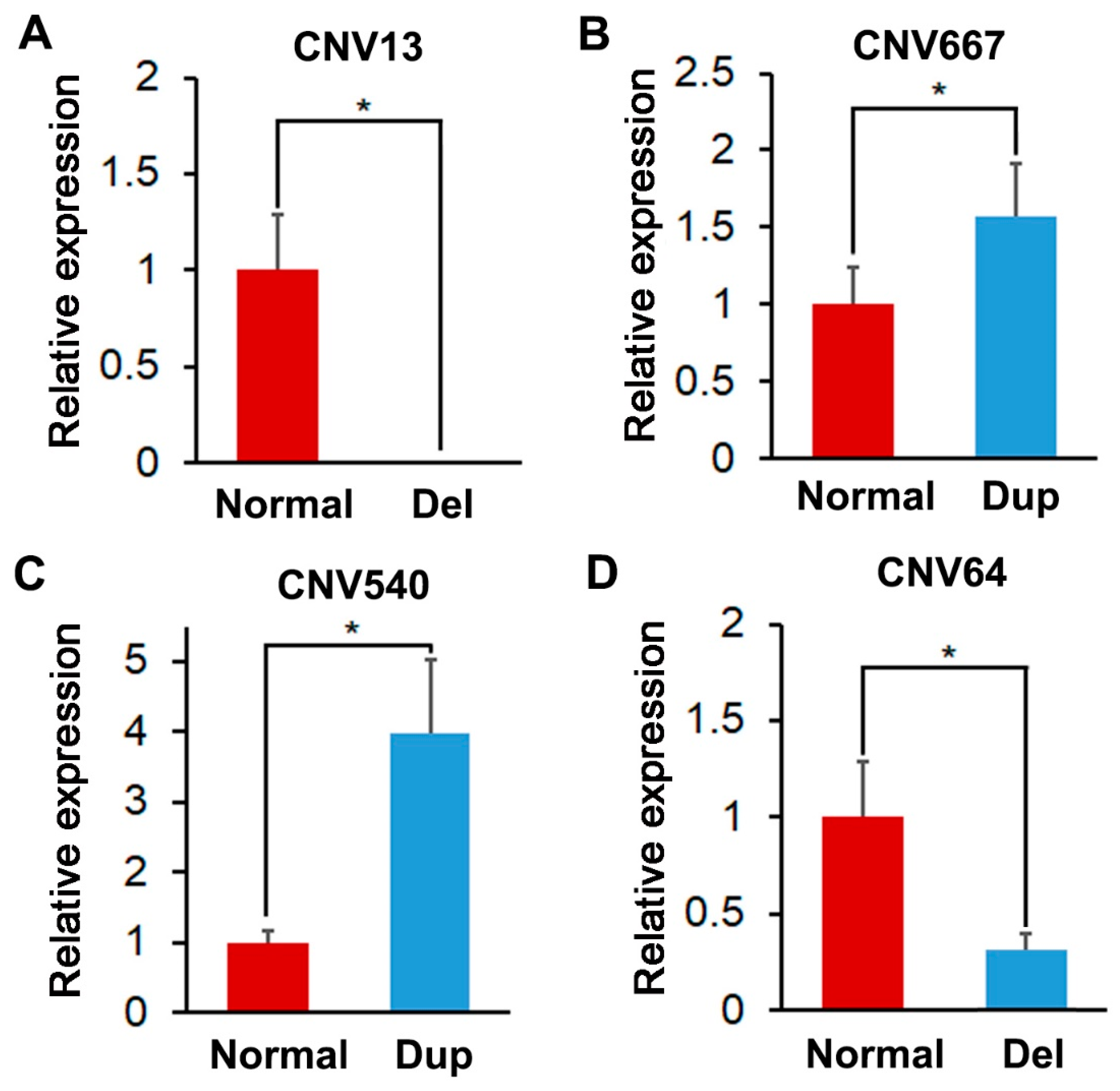
3.4. Identification and Validation of the Selected CNVRs
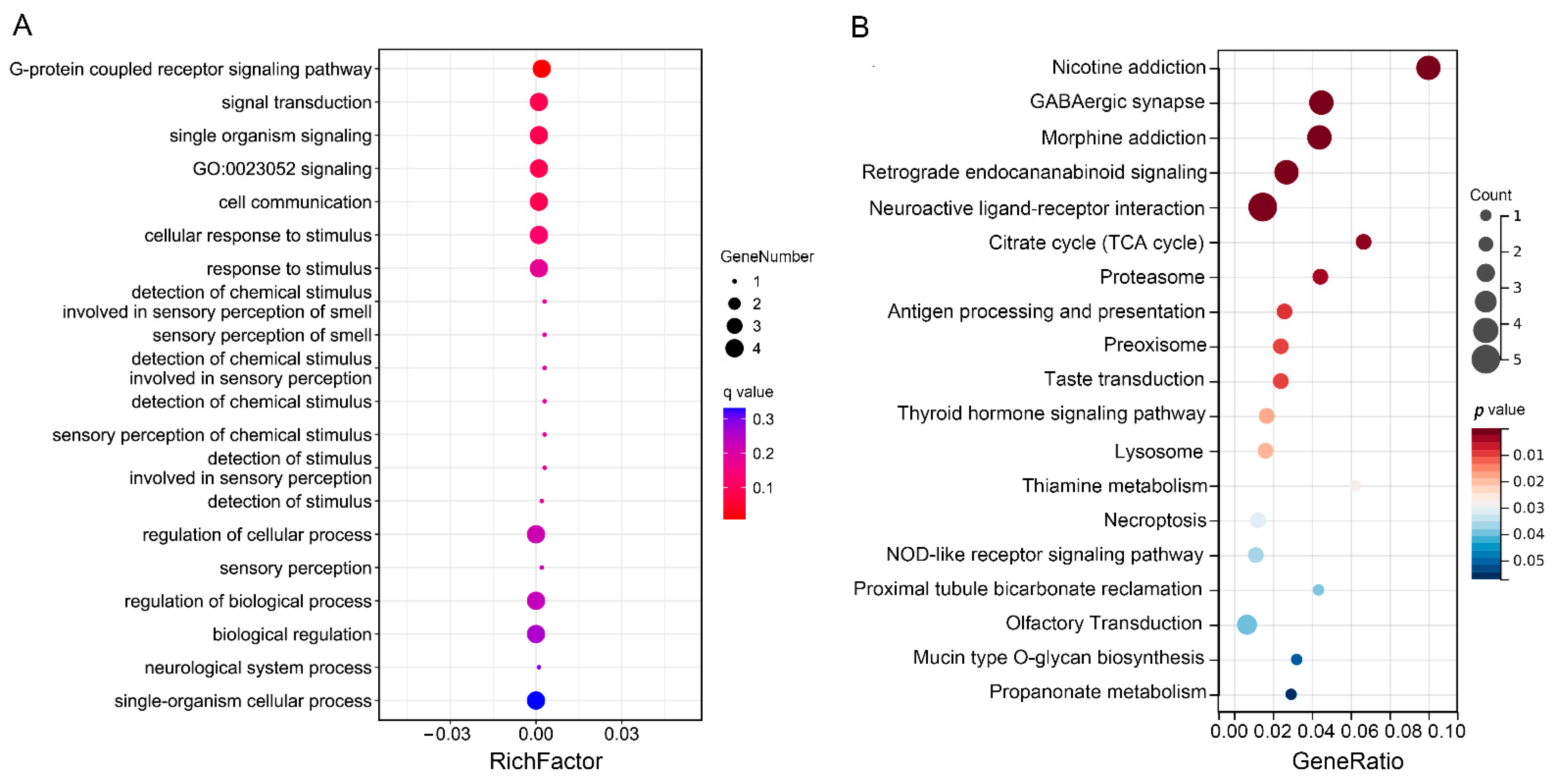
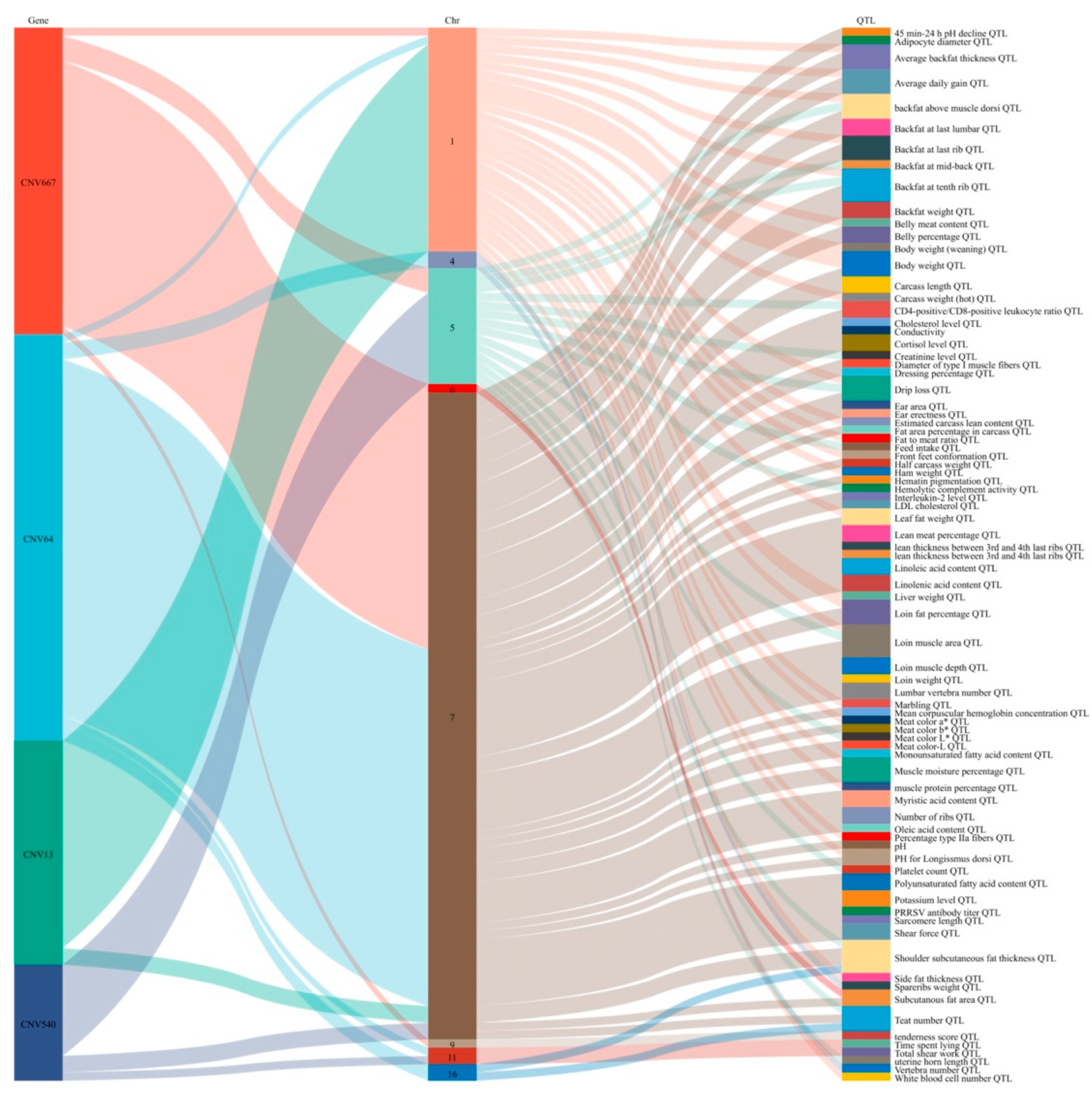
3.5. Functional Enrichment and Annotation Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fokkema, W.; van der Jeugd, H.P.; Lameris, T.K.; Dokter, A.M.; Ebbinge, B.S.; de Roos, A.M.; Nolet, B.A.; Piersma, T.; Olff, H. Ontogenetic niche shifts as a driver of seasonal migration. Oecologia 2020, 193, 285–297. [Google Scholar] [CrossRef]
- Xu, J.; Fu, Y.; Hu, Y.; Yin, L.; Tang, Z.; Yin, D.; Zhu, M.; Yu, M.; Li, X.; Zhou, Y.; et al. Whole genome variants across 57 pig breeds enable comprehensive identification of genetic signatures that underlie breed features. J. Anim. Sci. Biotechnol. 2020, 11, 115. [Google Scholar] [CrossRef]
- Gong, G.; Fan, Y.; Li, W.; Yan, X.; Yan, X.; Zhang, L.; Wang, N.; Chen, O.; Zhang, Y.; Wang, R.; et al. Identification of the Key Genes Associated with Different Hair Types in the Inner Mongolia Cashmere Goat. Animals 2022, 12, 1456. [Google Scholar] [CrossRef]
- Zheng, Q.; Ye, N.; Bao, P.; Zhang, X.; Wang, F.; Ma, L.; Chu, M.; Guo, X.; Liang, C.; Pan, H.; et al. Construction of transcriptome atlas of white yak hair follicle during anagen and catagen using single-cell RNA sequencing. BMC Genom. 2022, 23, 813. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Qiu, X.; Piao, J.; Zhang, L.; Piao, J.; Zhao, F. Study on the roles of melatonin in regulating dermal fibroblast growth in Liaoning cashmere goats by transcriptome sequencing. Anim. Biotechnol. 2022, 33, 1255–1267. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; He, X.; Wang, W.; Feng, Y.; Zhang, D.; Li, Z.; Liu, D. Transcriptome Analysis Reveals Genes Contributed to Min Pig Villi Hair Follicle in Different Seasons. Vet. Sci. 2022, 9, 639. [Google Scholar] [CrossRef]
- Shang, F.; Wang, Y.; Ma, R.; Rong, Y.; Wang, M.; Wu, Z.; Hai, E.; Pan, J.; Liang, L.; Wang, Z.; et al. Screening of microRNA and mRNA related to secondary hair follicle morphogenesis and development and functional analysis in cashmere goats. Funct. Integr. Genom. 2022, 22, 835–848. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Li, X.; Wang, L.; Wang, H.; Yang, K.; Wang, Z.; Wang, R.; Su, R.; Liu, Z.; Zhao, Y.; et al. Expression of fox-related genes in the skin follicles of Inner Mongolia cashmere goat. Asian-Australas. J. Anim. Sci. 2018, 31, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Manolio, T.A.; Collins, F.S.; Cox, N.J.; Goldstein, D.B.; Hindorff, L.A.; Hunter, D.J.; McCarthy, M.I.; Ramos, E.M.; Cardon, L.R.; Chakravarti, A.; et al. Finding the missing heritability of complex diseases. Nature 2009, 461, 747–753. [Google Scholar] [CrossRef]
- Wei, X.; Shu, Z.; Wang, L.; Zhang, T.; Zhang, L.; Hou, X.; Yan, H.; Wang, L. Copy Number Variations Contribute to Intramuscular Fat Content Differences by Affecting the Expression of PELP1 Alternative Splices in Pigs. Animals 2022, 12, 1382. [Google Scholar] [CrossRef] [PubMed]
- Curtin, F.; Schulz, P. Multiple correlations and bonferroni’s correction. Biol. Psychiatry 1998, 44, 775–777. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.L.; Park, C.A.; Reecy, J.M. Building a livestock genetic and genomic information knowledgebase through integrative devel-opments of Animal QTLdb and CorrDB. Nucleic Acids Res. 2019, 47, D701–D710. [Google Scholar] [CrossRef] [PubMed]
- Quinlan, A.R. BEDTools: The Swiss-Army Tool for Genome Feature Analysis. Curr. Protoc. Bioinform. 2014, 47, 11–12. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Liu, X.; Huang, X.; Li, N.; Zhang, L.; Yan, H.; Hou, X.; Wang, L.; Wang, L. Integrated Proteotranscriptomics Reveals Differences in Molecular Immunity between Min and Large White Pig Breeds. Biology 2022, 11, 1708. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, X.; Jing, X.; He, X.; Wang, L.; Liu, Y.; Liu, D. Transcriptomics Analysis on Excellent Meat Quality Traits of Skeletal Muscles of the Chinese Indigenous Min Pig Compared with the Large White Breed. Int. J. Mol. Sci. 2017, 19, 21. [Google Scholar] [CrossRef]
- An, S.; Cutler, G.; Zhao, J.J.; Huang, S.G.; Tian, H.; Li, W.; Liang, L.; Rich, M.; Bakleh, A.; Du, J.; et al. Identification and characterization of a melanin-concentrating hormone receptor. Proc. Natl. Acad. Sci. USA 2001, 98, 7576–7581. [Google Scholar] [CrossRef]
- Sailer, A.W.; Sano, H.; Zeng, Z.; McDonald, T.P.; Pan, J.; Pong, S.-S.; Feighner, S.D.; Tan, C.P.; Fukami, T.; Iwaasa, H.; et al. Identification and characterization of a second melanin-concentrating hormone receptor, MCH-2R. Proc. Natl. Acad. Sci. USA 2001, 98, 7564–7569. [Google Scholar] [CrossRef]
- Gao, X.-B. Electrophysiological effects of MCH on neurons in the hypothalamus. Peptides 2009, 30, 2025–2030. [Google Scholar] [CrossRef]
- Stuber, G.D.; Wise, R.A. Lateral hypothalamic circuits for feeding and reward. Nat. Neurosci. 2016, 19, 198–205. [Google Scholar] [CrossRef]
- Fischer, J.; Degenhardt, F.; Hofmann, A.; Redler, S.; Basmanav, F.B.; Heilmann-Heimbach, S.; Hanneken, S.; Giehl, K.A.; Wolff, H.; Moebus, S.; et al. Genome wide analysis of copy number variants in alopecia areata in a Central European cohort reveals association with MCHR2. Exp. Dermatol. 2017, 26, 536–541. [Google Scholar] [CrossRef]
- Chiang, M.S.; Yang, J.R.; Liao, S.C.; Hsu, C.C.; Hsu, C.W.; Yuan, K. Latent transforming growth factor-β binding proteins (LTBP-1 and LTBP-2) and gingiva keratinization. Oral Dis. 2015, 21, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Wollina, U.; Lange, D.; Funa, K.; Paus, R. Expression of transforming growth factor beta isoforms and their receptors during hair growth phases in mice. Histol. Histopathol. 1996, 11, 431–436. [Google Scholar] [PubMed]
- Niu, N.; Liu, Q.; Hou, X.; Liu, X.; Wang, L.; Zhao, F.; Gao, H.; Shi, L.; Wang, L.; Zhang, L. Genome-wide association study revealed ABCD4 on SSC7 and GREB1L and MIB1 on SSC6 as crucial candidate genes for rib number in Beijing Black pigs. Anim. Genet. 2022, 53, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Takeichi, T.; Hsu, C.K.; Yang, H.S.; Chen, H.Y.; Wong, T.W.; Tsai, W.L.; Chao, S.C.; Lee, J.Y.; Akiyama, M.; Simpson, M.A.; et al. Pro-gressive hyperpigmentation in a Taiwanese child due to an inborn error of vitamin B12 metabolism (cblJ). Br. J. Dermatol. 2015, 172, 1111–1115. [Google Scholar] [CrossRef] [PubMed]
- Sandmark, J.; Dahl, G.; Öster, L.; Xu, B.; Johansson, P.; Akerud, T.; Aagaard, A.; Davidsson, P.; Bigalke, J.M.; Winzell, M.S.; et al. Structure and biophysical characterization of the human full-length neurturin–GFRa2 complex: A role for heparan sulfate in signaling. J. Biol. Chem. 2018, 293, 5492–5508. [Google Scholar] [CrossRef]
- Samos, A.; McGaughey, V.; Rieger, S.; Lisse, T.S. Reawakening GDNF’s regenerative past in mice and humans. Regen Ther. 2022, 20, 78–85. [Google Scholar] [CrossRef]
- Adly, M.A.; Assaf, H.A.; Pertile, P.; Hussein, M.R.; Paus, R. Expression patterns of the glial cell line–derived neurotrophic factor, neurturin, their cognate receptors GFRα-1, GFRα-2, and a common signal transduction element c-Ret in the human skin hair follicles. J. Am. Acad. Dermatol. 2008, 58, 238–250. [Google Scholar] [CrossRef]
- Bhat, B.; Yaseen, M.; Singh, A.; Ahmad, S.M.; Ganai, N.A. Identification of potential key genes and pathways associated with the Pashmina fiber initiation using RNA-Seq and integrated bioinformatics analysis. Sci. Rep. 2021, 11, 1766. [Google Scholar] [CrossRef]
- Moyer, A.M. NUDT15: A bench to bedside success story. Clin. Biochem. 2021, 92, 1–8. [Google Scholar] [CrossRef]
- Kakuta, Y.; Naito, T.; Onodera, M.; Kuroha, M.; Kimura, T.; Shiga, H.; Endo, K.; Negoro, K.; Kinouchi, Y.; Shimosegawa, T. NUDT15 R139C causes thiopurine-induced early severe hair loss and leukopenia in Japanese patients with IBD. Pharm. J. 2016, 16, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.; Choe, B.-H.; Kang, B. Prevention of thiopurine-induced early leukopenia in a Korean pediatric patient with Crohn’s disease who turned out to possess homozygous mutations in NUDT15 R139C. Yeungnam Univ. J. Med. 2020, 37, 332–336. [Google Scholar] [CrossRef]
- Wang, R.; Liu, B.; Li, J.; Xu, J.; Wang, X.; Zhao, Z.; Zhao, L. Association between the c.415C > T, c.52G > A, and 36_37insGGAGTC polymor-phisms of NUDT 15 and thiopurine-induced leukopenia, thiopurine intolerance, and severe hair loss: An updated me-ta-analysis. Drug Des. Dev. Ther. 2019, 13, 2729–2744. [Google Scholar] [CrossRef] [PubMed]
- Dehghan-Nayeri, N.; Eshghi, P.; Pour, K.G.; Rezaei-Tavirani, M.; Omrani, M.D.; Gharehbaghian, A. Differential expression pattern of protein markers for predicting chemosensitivity of dexamethasone-based chemotherapy of B cell acute lymphoblastic leukemia. Cancer Chemother. Pharmacol. 2017, 80, 177–185. [Google Scholar] [CrossRef]
- Wang, X.; Wu, F.; Deng, Y.; Chai, J.; Zhang, Y.; He, G.; Li, X. Increased expression of PSME2 is associated with clear cell renal cell carcinoma invasion by regulating BNIP3-mediated autophagy. Int. J. Oncol. 2021, 59, 106. [Google Scholar] [CrossRef]
- Sun, P.; Li, R.; Meng, Y.; Xi, S.; Wang, Q.; Yang, X.; Peng, X.; Cai, J. Introduction to DOK2 and its Potential Role in Cancer. Physiol. Res. 2021, 70, 671–685. [Google Scholar] [CrossRef] [PubMed]
- Takii, R.; Fujimoto, M.; Tan, K.; Takaki, E.; Hayashida, N.; Nakato, R.; Shirahige, K.; Nakai, A. ATF1 Modulates the Heat Shock Response by Regulating the Stress-Inducible Heat Shock Factor 1 Transcription Complex. Mol. Cell. Biol. 2015, 35, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Igoshin, A.; Yudin, N.; Aitnazarov, R.; Yurchenko, A.A.; Larkin, D.M. Whole-Genome Resequencing Points to Candidate DNA Loci Affecting Body Temperature under Cold Stress in Siberian Cattle Populations. Life 2021, 11, 959. [Google Scholar] [CrossRef]
- Saravanan, K.A.; Panigrahi, M.; Kumar, H.; Parida, S.; Bhushan, B.; Gaur, G.K.; Dutt, T.; Mishra, B.P.; Singh, R.K. Genomic scans for se-lection signatures revealed candidate genes for adaptation and production traits in a variety of cattle breeds. Genomics 2021, 113, 955–963. [Google Scholar] [CrossRef]
- Ai, H.; Huang, L.; Ren, J. Genetic Diversity, Linkage Disequilibrium and Selection Signatures in Chinese and Western Pigs Revealed by Genome-Wide SNP Markers. PLoS ONE 2013, 8, e56001. [Google Scholar] [CrossRef] [PubMed]
| Gene | Forward Primer | Reverse Primer |
|---|---|---|
| CNV13 | CCTGGCTTCCCTTCTCTCTT | GGAAGAATCGGGTGGGTATT |
| CNV667 | AGCTTCCTGCTGGACACAGT | GCTGCTTTGGGCTTATTCTG |
| CNV540 | CTTCAAAGCCAGCAATGTGA | GGATTATCCGAGTTGGCTCA |
| CNV64 | TCTGAAAGGGATGGATCTGG | GAACCTTTGCTGCCTGTCTC |
| GCG | GCAATATGGCTTTAGAATACACCTCTTA | GTCACTAATCAAGATCGTGTTCACAAC |
| CNVR ID | Location | Nearest Genes | CHISQ | Type | p-Value |
|---|---|---|---|---|---|
| CNV13 | 1:67141601-67155200 | ASCC3/MCHR2/SIM1 | 18.9 | Del | 1.38 × 10−5 |
| CNV667 | 7:97857601-97860000 | VSX2/ABCD4/VRTN/SYNDIG1L/ISCA2/LTBP2/NPC2/AREL1/FCF1/YLPM1/PROX2 | 17.97 | Dup | 2.24 × 10−5 |
| CNV540 | 5:16751601-16754400 | DIP2B/ATF1/TMPRSS12/SLC4A8/SCN8A | 17.66 | Dup | 2.64 × 10−5 |
| CNV64 | 7:75399601-75402400 | REC8/IRF9/FITM1/PSME1/RNF31/PSME2/EMC9/DCAF11/CARMIL3/CPNE6/PCK2/NRL/DHRS4/ZFHX2/JPH4/AP1G2/THTPA/MYH7/ENSSSCG00000002020 | 17.6 | Del | 2.73 × 10−5 |
| CNV734 | 9:51430801-51434000 | OR8D1/OR8D2/ENSSSCG00000015179/ENSSSCG00000061653/ENSSSCG00000053222/ENSSSCG00000015193/ENSSSCG00000053475/OR8B8/ENSSSCG00000056190/ENSSSCG00000055348/ENSSSCG00000061394/ENSSSCG00000015154/ENSSSCG00000052681 | 15.12 | Del | 0.000101 |
| CNV267 | 15:1259201-1261200 | ENSSSCG00000060497/NMI/TNFAIP6 | 14.56 | Del | 0.000135 |
| CNV234 | 14:6050401-6053200 | ENSSSCG00000055110/GFRA2/XPO7/DOK2/DMTN/FGF17 | 14.26 | Dup | 0.000159 |
| CNV321 | 16:61828001-61833200 | GABRG2/GABRA1/GABRA6/GABRB2 | 12.49 | Dup | 0.00041 |
| CNV330 | 6:135400401-135405200 | FUBP1/DNAJB4/NEXN/MIGA1/USP33/GIPC2/ENSSSCG00000035986/ENSSSCG00000050806 | 11.61 | Del | 0.000657 |
| CNV720 | 9:4729201-4731600 | ENSSSCG00000060217/ENSSSCG00000062640/ENSSSCG00000050624/ssc-mir-10390 | 11.13 | Dup | 0.000849 |
| CNV510 | 4:47520801-47538800 | ENSSSCG00000042115 | 10.35 | Dup | 0.001295 |
| CNV107 | 11:19803201-19810400 | ENSSSCG00000042466/SUCLA2/NUDT15/MED4/ENSSSCG00000053915 | 9.936 | Dup | 0.001621 |
| CNV552 | 5:45503601-45508000 | CCDC91/ENSSSCG00000045661/PTHLH | 9.868 | Dup | 0.001682 |
| CNV390 | 2:47668801-47674000 | NSSSCG00000031143/USP47/DKK3/U6/GALNT18 | 9.819 | Dup | 0.001727 |
| CNV22 | 1:93250401-93252800 | ENSSSCG00000005790/ENSSSCG00000053538 | 9.671 | Dup | 0.001872 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, X.; Tian, M.; Wang, W.; Feng, Y.; Li, Z.; Wang, J.; Song, Y.; Zhang, J.; Liu, D. Identification of Candidate Genes for Min Pig Villi Hair Traits by Genome-Wide Association of Copy Number Variation. Vet. Sci. 2023, 10, 307. https://doi.org/10.3390/vetsci10050307
He X, Tian M, Wang W, Feng Y, Li Z, Wang J, Song Y, Zhang J, Liu D. Identification of Candidate Genes for Min Pig Villi Hair Traits by Genome-Wide Association of Copy Number Variation. Veterinary Sciences. 2023; 10(5):307. https://doi.org/10.3390/vetsci10050307
Chicago/Turabian StyleHe, Xinmiao, Ming Tian, Wentao Wang, Yanzhong Feng, Zhongqiu Li, Jiahui Wang, Yan Song, Jinfeng Zhang, and Di Liu. 2023. "Identification of Candidate Genes for Min Pig Villi Hair Traits by Genome-Wide Association of Copy Number Variation" Veterinary Sciences 10, no. 5: 307. https://doi.org/10.3390/vetsci10050307
APA StyleHe, X., Tian, M., Wang, W., Feng, Y., Li, Z., Wang, J., Song, Y., Zhang, J., & Liu, D. (2023). Identification of Candidate Genes for Min Pig Villi Hair Traits by Genome-Wide Association of Copy Number Variation. Veterinary Sciences, 10(5), 307. https://doi.org/10.3390/vetsci10050307






