Epidemiology and Genetic Characteristics of Porcine Reproductive and Respiratory Syndrome Virus in the Hunan and Hebei Provinces of China
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimen Collection
2.2. Enzyme-Linked Immunosorbent Assay (ELISA)
2.3. Sample Handling and PRRSV Detection
2.4. Sequencing and Genetic Analysis
2.5. Data Analysis
3. Results
3.1. PRRSV Seroprevalence in Pig Herds in Hunan and Hebei Provinces
3.2. Molecular Detection of PRRSV in Clinical Samples
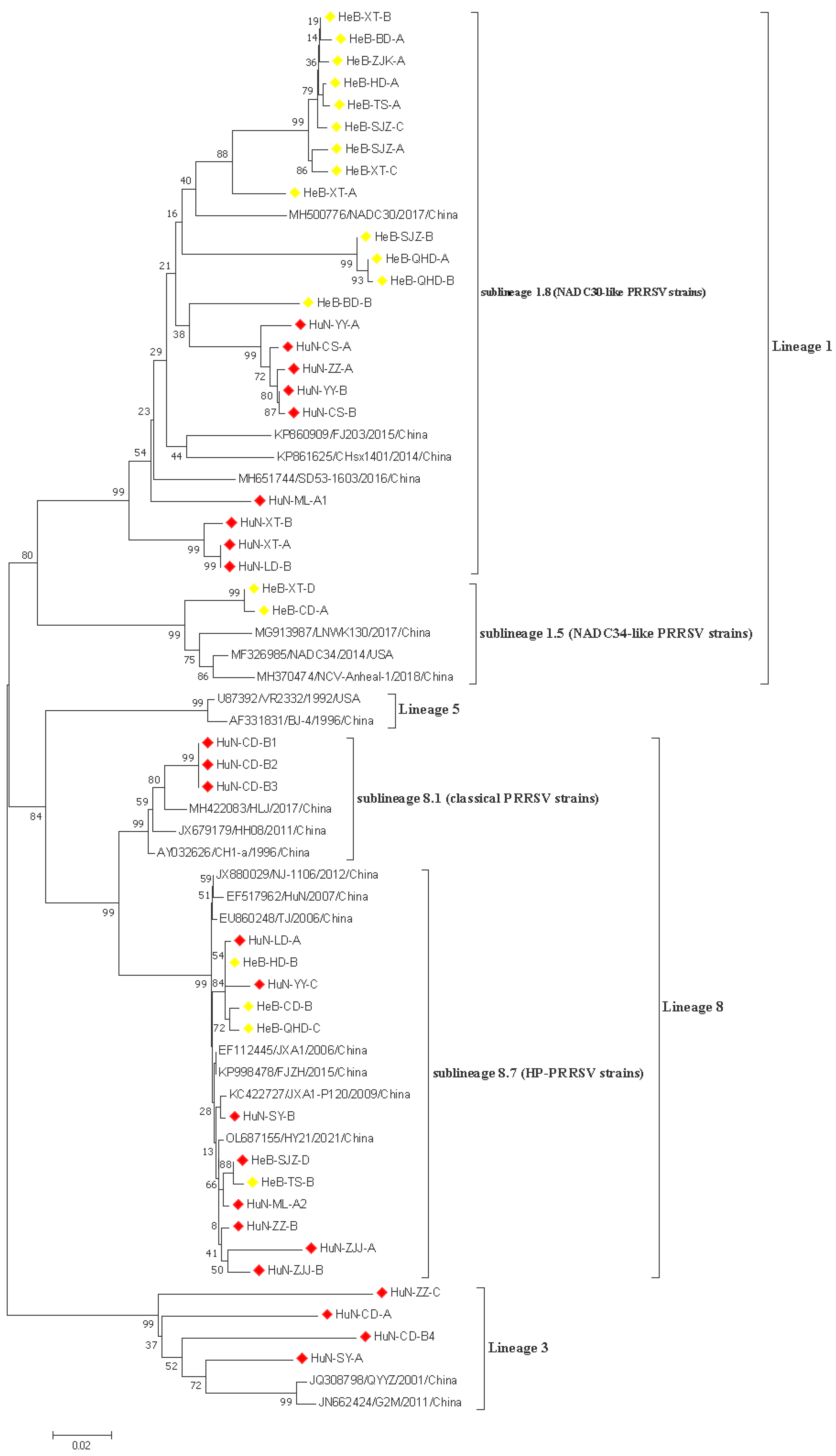
3.3. Phylogenetic Analysis of ORF5 Gene
3.4. Sequence Analysis of ORF5 Gene
3.5. Bioinformatic Analysis of the Complete Genome Sequences of PRRSV Strains
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Benfield, D.A.; Nelson, E.; Collins, J.E.; Harris, L.; Goyal, S.M.; Robison, D.; Christianson, W.T.; Morrison, R.B.; Gorcyca, D.; Chladek, D. Characterization of Swine Infertility and Respiratory Syndrome (SIRS) Virus (Isolate ATCC VR-2332). J. Vet. Diagn. Investig. 1992, 4, 127–133. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Tran, H.A.T.; Nguyen, T.Q.; Nguyen, P.B.T.; Le, T.H.T.; Lai, D.C.; Nguyen, M.N. Phylogenetic analysis of porcine reproductive and respiratory syndrome virus in Vietnam, 2021. Virus Genes 2022, 58, 361–366. [Google Scholar] [CrossRef]
- Fang, K.; Liu, S.; Li, X.; Chen, H.; Qian, P. Epidemiological and Genetic Characteristics of Porcine Reproductive and Respiratory Syndrome Virus in South China Between 2017 and 2021. Front. Vet. Sci. 2022, 9, 853044. [Google Scholar] [CrossRef]
- Zhao, D.; Yang, B.; Yuan, X.; Shen, C.; Zhang, D.; Shi, X.; Zhang, T.; Cui, H.; Yang, J.; Chen, X.; et al. Advanced Research in Porcine Reproductive and Respiratory Syndrome Virus Co-infection With Other Pathogens in Swine. Front. Vet. Sci. 2021, 8, 699561. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Ji, X.; Fu, C.; Hu, D.; Pang, H.; Wang, T.; Li, C.; Wang, G.; Peng, J. Evolution Characterization and Pathogenicity of a Porcine Reproductive and Respiratory Syndrome Virus Isolate from a Pig Farm in Shandong Province, China. Viruses 2022, 14, 1194. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Xiao, S.; Xiao, Y.; Wang, X.; Zhang, C.; Zhao, Q.; Nan, Y.; Huang, B.; Liu, H.; Liu, N.; et al. MYH9 is an Essential Factor for Porcine Reproductive and Respiratory Syndrome Virus Infection. Sci. Rep. 2016, 6, 25120. [Google Scholar] [CrossRef] [PubMed]
- Xue, R.-X.; Sun, S.-F.; Li, Y.-G.; Wang, M.-L.; Wang, G.-S.; Li, Y.-J.; Zhang, Y.; Wei, X.-H.; Chen, F.; Ma, J.-J.; et al. Diversity of porcine reproductive and respiratory syndrome virus in Shandong, China. Acta Virol. 2021, 65, 303–306. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, Q.; Cao, Z.; Tang, Y.-D.; Xia, D.; Wang, G.; Shan, H. Recent Advances in Porcine Reproductive and Respiratory Syndrome Virus NADC30-like Research in China: Molecular Characterization, Pathogenicity, and Control. Front. Microbiol. 2022, 12, 791313. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Lam, T.T.; Hon, C.C.; Murtaugh, M.P.; Davies, P.R.; Hui, R.K.; Li, J.; Wong, L.T.; Yip, C.W.; Jiang, J.W.; et al. Phylogeny-Based Evolutionary, Demographical, and Geographical Dissection of North American Type 2 Porcine Reproductive and Respiratory Syndrome Viruses. J. Virol. 2010, 84, 8700–8711. [Google Scholar] [CrossRef] [PubMed]
- Jakab, S.; Kaszab, E.; Marton, S.; Bányai, K.; Bálint, Á.; Nemes, I.; Szabó, I. Genetic diversity of imported PRRSV-2 strains, 2005–2020, Hungary. Front. Vet. Sci. 2022, 9, 986850. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Chen, X.-X.; Li, R.; Qiao, S.; Zhang, G. The prevalent status and genetic diversity of porcine reproductive and respiratory syndrome virus in China: A molecular epidemiological perspective. Virol. J. 2018, 15, 2. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Feng, Y.; Martin, D.P.; Chen, J.; Ma, S.; Xia, P.; Zhang, G. Genetic diversity and phylogenetic analysis of the ORF5 gene of PRRSV from central China. Res. Vet. Sci. 2017, 115, 226–234. [Google Scholar] [CrossRef]
- Tian, K.; Yu, X.; Zhao, T.; Feng, Y.; Cao, Z.; Wang, C.; Hu, Y.; Chen, X.; Hu, D.; Tian, X.; et al. Emergence of Fatal PRRSV Variants: Unparalleled Outbreaks of Atypical PRRS in China and Molecular Dissection of the Unique Hallmark. PLoS ONE 2007, 2, e526. [Google Scholar] [CrossRef]
- Zhang, H.; Xia, M.; Wang, W.; Ju, D.; Cao, L.; Wu, B.; Wang, X.; Wu, Y.; Song, N.; Hu, J.; et al. An Attenuated Highly Pathogenic Chinese PRRS Viral Vaccine Confers Cross Protection to Pigs against Challenge with the Emerging PRRSV NADC30-like Strain. Virol. Sin. 2018, 33, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wei, C.; Lin, Z.; Xia, W.; Ma, Y.; Dai, A.; Yang, X. Full genome sequence analysis of a 1-7-4-like PRRSV strain in Fujian Province, China. PeerJ 2019, 7, e7859. [Google Scholar] [CrossRef]
- Li, C.; Gong, B.; Sun, Q.; Xu, H.; Zhao, J.; Xiang, L.; Tang, Y.-D.; Leng, C.; Li, W.; Guo, Z.; et al. First Detection of NADC34-like PRRSV as a Main Epidemic Strain on a Large Farm in China. Pathogens 2021, 11, 32. [Google Scholar] [CrossRef]
- Cheng, T.-Y.; Campler, M.R.; Schroeder, D.C.; Yang, M.; Mor, S.K.; Ferreira, J.B.; Arruda, A.G. Detection of Multiple Lineages of PRRSV in Breeding and Growing Swine Farms. Front. Vet. Sci. 2022, 9, 884733. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Tan, L.; Wang, C.; He, S.; Fang, L.; Wang, Z.; Zhong, Y.; Zhang, K.; Liu, D.; Yang, Q.; et al. Serological Investigation and Genetic Characteristics of Pseudorabies Virus in Hunan Province of China from 2016 to 2020. Front. Vet. Sci. 2021, 8, 762326. [Google Scholar] [CrossRef]
- Zhao, H.-Z.; Wang, F.-X.; Han, X.-Y.; Guo, H.; Liu, C.-Y.; Hou, L.-N.; Wang, Y.-X.; Zheng, H.; Wang, L.; Wen, Y.-J. Recent advances in the study of NADC34-like porcine reproductive and respiratory syndrome virus in China. Front. Microbiol. 2022, 13, 950402. [Google Scholar] [CrossRef]
- Zhang, H.; Leng, C.; Ding, Y.; Zhai, H.; Li, Z.; Xiang, L.; Zhang, W.; Liu, C.; Li, M.; Chen, J.; et al. Characterization of newly emerged NADC30-like strains of porcine reproductive and respiratory syndrome virus in China. Arch. Virol. 2019, 164, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.H.; Su, F.; Yu, B.; Li, J.X.; Deng, L.R.; Mao, H.M.; Wu, Y.G.; Yang, F.W.; Yuan, X.F. Epidemiological investigation and analysis of main viral infectious diseases in pigs in Zhejiang Province from 2017 to 2020. Chin. J. Vet. Sci. 2022, 42, 233–239. (In Chinese) [Google Scholar]
- Yin, B.; Qi, S.; Sha, W.; Qin, H.; Liu, L.; Yun, J.; Zhu, J.; Li, G.; Sun, D. Molecular Characterization of the Nsp2 and ORF5 (ORF5a) Genes of PRRSV Strains in Nine Provinces of China During 2016–2018. Front. Vet. Sci. 2021, 8, 605832. [Google Scholar] [CrossRef]
- Chen, N.; Ye, M.; Huang, Y.; Li, S.; Xiao, Y.; Li, X.; Li, S.; Li, X.; Yu, X.; Tian, K.; et al. Identification of Two Porcine Reproductive and Respiratory Syndrome Virus Variants Sharing High Genomic Homology but with Distinct Virulence. Viruses 2019, 11, 875. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Zhao, T.; Peng, Z.; Sun, Y.; Stratton, C.W.; Zhou, D.; Tang, X.; Tian, Y.; Chen, H.; Wu, B. Epidemiological and genetic characteristics of porcine reproductive and respiratory syndrome virus circulating in central and South China in 2016. Acta Trop. 2019, 190, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.-C.; Xiong, J.-Y.; Ye, C.; Chang, X.-B.; Guo, J.-C.; Jiang, C.-G.; Zhang, G.-H.; Tian, Z.-J.; Cai, X.-H.; Tong, G.-Z.; et al. Genotypic and geographical distribution of porcine reproductive and respiratory syndrome viruses in mainland China in 1996–2016. Vet. Microbiol. 2017, 208, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Han, Q.; Zhang, L.; Zhang, Z.; Wu, Y.; Shen, H.; Jiang, P. Emergence of mosaic recombinant strains potentially associated with vaccine JXA1-R and predominant circulating strains of porcine reproductive and respiratory syndrome virus in different provinces of China. Virol. J. 2017, 14, 67. [Google Scholar] [CrossRef]
- Li, L.; Zhang, L.; Hu, Q.; Zhao, L.; Nan, Y.; Hou, G.; Chen, Y.; Han, X.; Ren, X.; Zhao, Q.; et al. MYH9 Key Amino Acid Residues Identified by the Anti-Idiotypic Antibody to Porcine Reproductive and Respiratory Syndrome Virus Glycoprotein 5 Involve in the Virus Internalization by Porcine Alveolar Macrophages. Viruses 2019, 12, 40. [Google Scholar] [CrossRef]
- Lee, C.; Rogan, D.; Erickson, L.; Zhang, J.; Yoo, D. Characterization of the porcine reproductive and respiratory syndrome virus glycoprotein 5 (GP5) in stably expressing cells. Virus Res. 2004, 104, 33–38. [Google Scholar] [CrossRef]
- Young, J.E.; Dvorak, C.M.T.; Graham, S.P.; Murtaugh, M.P. Isolation of Porcine Reproductive and Respiratory Syndrome Virus GP5-Specific, Neutralizing Monoclonal Antibodies from Hyperimmune Sows. Front. Immunol. 2021, 12, 638493. [Google Scholar] [CrossRef]
- Vashisht, K.; Goldberg, T.L.; Husmann, R.J.; Schnitzlein, W.; Zuckermann, F.A. Identification of immunodominant T-cell epitopes present in glycoprotein 5 of the North American genotype of porcine reproductive and respiratory syndrome virus. Vaccine 2008, 26, 4747–4753. [Google Scholar] [CrossRef]
- Li, Y.; Xu, G.; Du, X.; Xu, L.; Ma, Z.; Li, Z.; Feng, Y.; Jiao, D.; Guo, W.; Xiao, S. Genomic characteristics and pathogenicity of a new recombinant strain of porcine reproductive and respiratory syndrome virus. Arch. Virol. 2021, 166, 389–402. [Google Scholar] [CrossRef] [PubMed]

| Regions | Development Stages | Tested Samples | Positive Samples | Positive Rate (%) | 95%CI | p-Value |
|---|---|---|---|---|---|---|
| Hunan | Gilts | 534 | 413 | 77.34 | 73.79–80.89 | <0.01 |
| Sows | 591 | 477 | 80.71 | 77.53–83.89 | <0.01 | |
| Fattening pigs | 436 | 206 | 47.25 | 42.56–51.94 | Reference | |
| Nursing pigs | 511 | 314 | 61.45 | 57.23–65.67 | <0.01 | |
| Subtotal | 2072 | 1410 | 68.05 | 65.34–70.76 | - | |
| Hebei | Gilts | 421 | 369 | 87.64 | 84.28–91.00 | <0.01 |
| Sows | 655 | 570 | 87.02 | 84.26–89.78 | <0.01 | |
| Fattening pigs | 507 | 291 | 57.40 | 53.09–61.70 | Reference | |
| Nursing pigs | 352 | 290 | 82.39 | 78.41–86.37 | <0.01 | |
| Subtotal | 1935 | 1520 | 78.55 | 76.72–80.37 | - | |
| Total | 4007 | 2930 | 73.12 | 71.74–74.49 | - |
| Region | Clinical Samples | Samples | Positive Samples | Positive Rate (%) | 95%CI | p-Value |
|---|---|---|---|---|---|---|
| Hunan | Healthy pigs in slaughterhouses | 2818 | 217 | 7.70 | 7.54–7.86 | <0.01 |
| Healthy pigs in farms | 2072 | 47 | 2.70 | 2.00–3.40 | Reference | |
| Diseased pigs in non-hazardous disposal sites | 359 | 65 | 18.11 | 14.13–22.09 | <0.01 | |
| Hebei | Diseased pigs with clinical symptoms of PRRS | 550 | 153 | 27.82 | 24.07–31.56 | <0.01 |
| Total | 5799 | 482 | 8.31 | 7.60–9.02 | - |
| Identified Strain | Reference Strain | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sublineage 8.1 | Sublineage 8.7 | Lineage 3 | Sublineage 1.8 | Sublineage 1.5 | VR-2332 | CH-1a | JXA1 | NADC30 | NADC34 | ||
| Sublineage 8.1 (n = 3) | nt | 100.0 | 90.2 | 97.2 | 93.5 | 84.3 | 86.7 | ||||
| aa | 100.0 | 92.0 | 96.5 | 91.0 | 83.1 | 86.5 | |||||
| Sublineage 8.7 (n = 12) | nt | 91.5–94.2 | 96.7–99.8 | 87.2–89.2 | 92.7–95.2 | 97.0–99.5 | 81.5–87.0 | 86.6–87.7 | |||
| aa | 89.0–91.5 | 89.5–98.0 | 86.1–88.1 | 89.6–93.0 | 96.5–99.5 | 82.1–84.6 | 86.1–87.1 | ||||
| Lineage 3 (n = 4) | nt | 83.0–84.8 | 82.4–84.7 | 87.6–91.4 | 81.6–85.9 | 83.9–85.9 | 82.9–84.6 | 79.3–83.5 | 81.6–85.2 | ||
| aa | 79.5–82.5 | 78.6–83.6 | 88.1–92.5 | 81.1–84.6 | 81.5–86.6 | 80.6–84.1 | 79.6–84.1 | 81.6–85.6 | |||
| Sublineage 1.8 (n = 22) | nt | 83.2–87.2 | 81.4–87.1 | 79.6–84.9 | 89.2–99.5 | 83.3–87.1 | 84.1–88.4 | 83.3–87.1 | 90.5–94.2 | 86.1–92.5 | |
| aa | 81.5–85.5 | 80.6–86.1 | 77.6–87.1 | 88.1–99.5 | 81.6–86.6 | 82.1–87.1 | 82.6–87.6 | 91.5–94.5 | 86.1–92.5 | ||
| Sublineage 1.5 (n = 2) | nt | 85.1–85.4 | 86.2–87.1 | 80.8–84.4 | 86.1–87.9 | 99.7 | 81.1–87.7 | 86.6–86.9 | 86.6–86.9 | 84.8–85.5 | 95.9–96.2 |
| aa | 84.0–85.0 | 84.6–87.1 | 79.0–84.1 | 84.6–89.6 | 99.0 | 85.1–86.1 | 84.6–85.6 | 85.6–86.6 | 87.1–88.1 | 93.5–94.5 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhai, W.; Yu, S.; Zhang, P.; Lin, Y.; Ge, S.; Zhang, T.; Zhang, K.; He, S.; Hu, Q.; Tang, X.; et al. Epidemiology and Genetic Characteristics of Porcine Reproductive and Respiratory Syndrome Virus in the Hunan and Hebei Provinces of China. Vet. Sci. 2023, 10, 63. https://doi.org/10.3390/vetsci10010063
Zhai W, Yu S, Zhang P, Lin Y, Ge S, Zhang T, Zhang K, He S, Hu Q, Tang X, et al. Epidemiology and Genetic Characteristics of Porcine Reproductive and Respiratory Syndrome Virus in the Hunan and Hebei Provinces of China. Veterinary Sciences. 2023; 10(1):63. https://doi.org/10.3390/vetsci10010063
Chicago/Turabian StyleZhai, Wang, Siyu Yu, Pengxuan Zhang, Yuan Lin, Shenghu Ge, Taojie Zhang, Kun Zhang, Shicheng He, Qiaoyun Hu, Xiaomin Tang, and et al. 2023. "Epidemiology and Genetic Characteristics of Porcine Reproductive and Respiratory Syndrome Virus in the Hunan and Hebei Provinces of China" Veterinary Sciences 10, no. 1: 63. https://doi.org/10.3390/vetsci10010063
APA StyleZhai, W., Yu, S., Zhang, P., Lin, Y., Ge, S., Zhang, T., Zhang, K., He, S., Hu, Q., Tang, X., Peng, Z., & Wang, C. (2023). Epidemiology and Genetic Characteristics of Porcine Reproductive and Respiratory Syndrome Virus in the Hunan and Hebei Provinces of China. Veterinary Sciences, 10(1), 63. https://doi.org/10.3390/vetsci10010063






