The Fluctuation of Process Gasses Especially of Carbon Monoxide during Aerobic Biostabilization of an Organic Fraction of Municipal Solid Waste under Different Technological Regimes
Abstract
1. Summary
2. Data Description
3. Methods
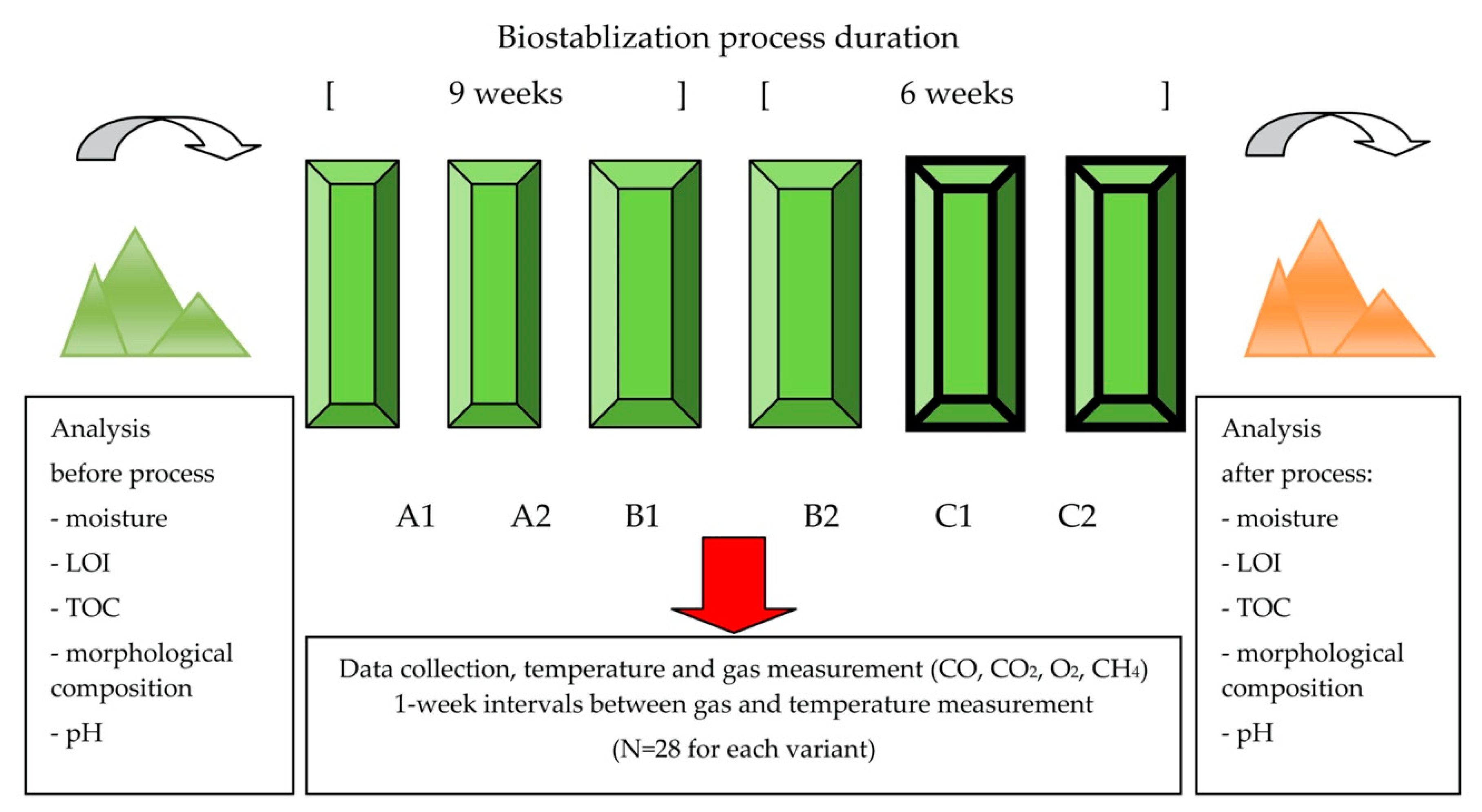
3.1. Waste Characteristics
3.2. Experiment Configuration
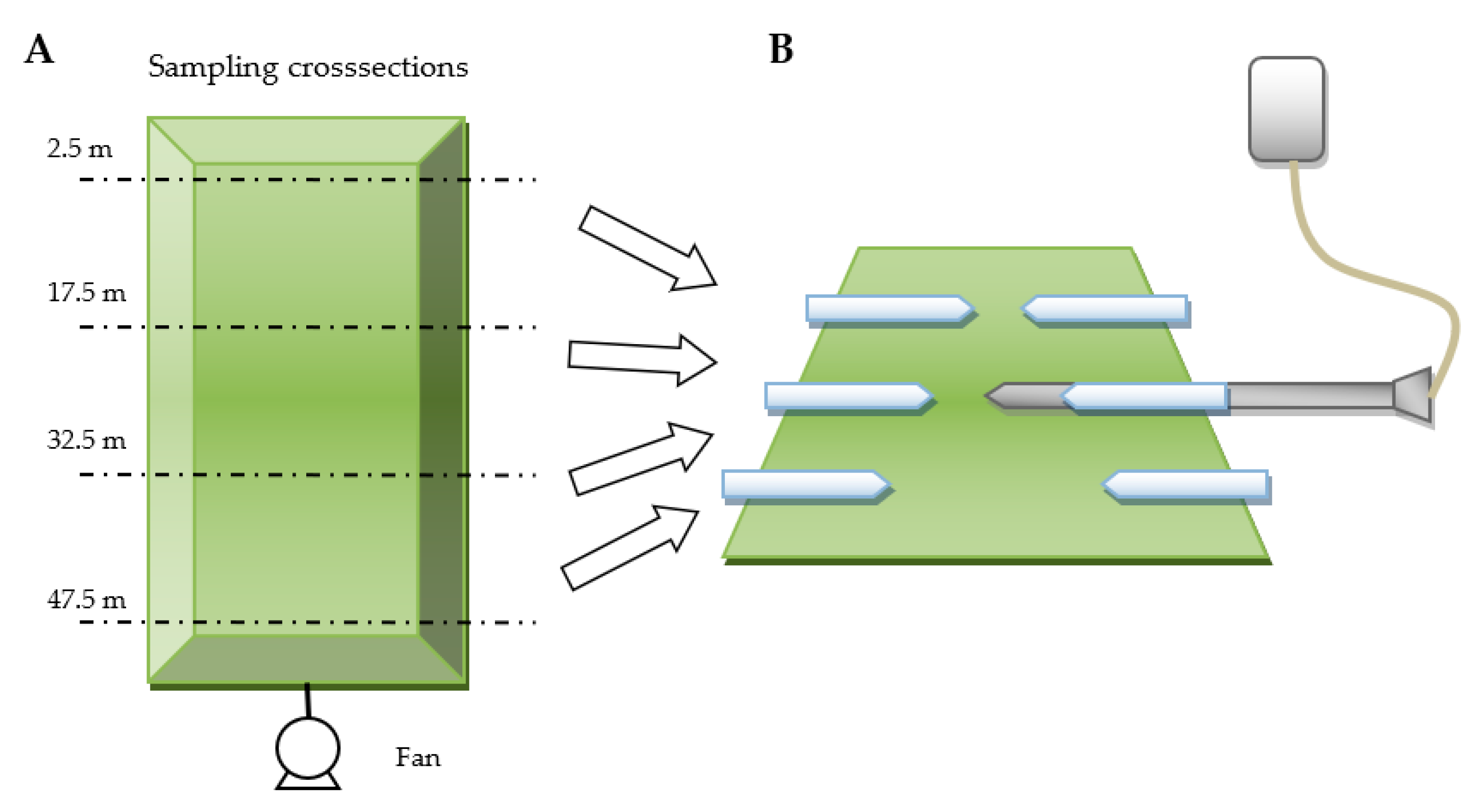
3.3. Measurements of Gas Concentrations in the Reactors
4. User Notes
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chadwick, D.; Sommer, S.G.; Thorman, R.; Fangueiro, D.; Cardenas, L.; Amon, B.; Misselbrook, T. Manure management: Implications for greenhouse gas emissions. Anim. Feed Sci. Technol. 2011, 166, 514–531. [Google Scholar] [CrossRef]
- Bürstel, I.; Siebert, E.; Frielingsdorf, S.; Zebger, I.; Friedrich, B.; Lenz, O. CO synthesized from the central one-carbon pool as source for the iron carbonyl in O2-tolerant [NiFe]-hydrogenase. Proc. Natl. Acad. Sci. USA 2016, 113, 14722–14726. [Google Scholar] [CrossRef] [PubMed]
- Amlinger, F.; Peyr, S.; Cuhls, C. Green house gas emissions from composting and mechanical biological treatment. Waste Manag. Res. 2008, 26, 47–60. [Google Scholar] [CrossRef] [PubMed]
- van Asperen, H.; Warneke, T.; Sabbatini, S.; Nicolini, G.; Papale, D.; Notholt, J. The role of photo- and thermal degradation for CO2 and CO fluxes in an arid ecosystem. Biogeosciences 2015, 12, 4161–4174. [Google Scholar] [CrossRef]
- Su, J.J.; Liu, B.Y.; Chang, Y.C. Emission of greenhouse gas from livestock waste and wastewater treatment in Taiwan. Agric. Ecosyst. Environ. 2003, 95, 253–263. [Google Scholar] [CrossRef]
- Vaverková, M.D.; Elbl, J.; Voběrková, S.; Koda, E.; Adamcová, D.; Mariusz Gusiatin, Z.; Al Rahman, A.; Radziemska, M.; Mazur, Z. Composting versus mechanical–biological treatment: Does it really make a difference in the final product parameters and maturity. Waste Manag. 2020, 106, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.K.; Boldrin, A.; Christensen, T.H.; Scheutz, C. Home composting as an alternative treatment option for organic household waste in Denmark: An environmental assessment using life cycle assessment-modelling. Waste Manag. 2012, 32, 31–40. [Google Scholar] [CrossRef]
- Manfredi, S.; Tonini, D.; Christensen, T.H. Environmental assessment of different management options for individual waste fractions by means of life-cycle assessment modelling. Resour. Conserv. Recycl. 2011, 55, 995–1004. [Google Scholar] [CrossRef]
- Lee, S.H.; Choi, K.I.; Osako, M.; Dong, J.I. Evaluation of environmental burdens caused by changes of food waste management systems in Seoul, Korea. Sci. Total Environ. 2007, 387, 42–53. [Google Scholar] [CrossRef]
- Stamou, I.; Antizar-Ladislao, B. A life cycle assessment of the use of compost from contaminated biodegradable municipal solid waste with silver and titanium dioxide nanoparticles. J. Clean. Prod. 2016, 135, 884–891. [Google Scholar] [CrossRef]
- Wu, L.; Wang, R. Carbon monoxide: Endogenous production, physiological functions, and pharmacological applications. Pharmacol. Rev. 2005, 57, 585–630. [Google Scholar] [CrossRef] [PubMed]
- Kaymak, C.; Basar, H. Carbon monoxide intoxication - Review. Fabad J. Pharm. Sci. 2010, 35, 163–172. [Google Scholar]
- Cowan, N.; Helfter, C.; Langford, B.; Coyle, M.; Levy, P.; Moxley, J.; Simmons, I.; Leeson, S.; Nemitz, E.; Skiba, U. Seasonal fluxes of carbon monoxide from an intensively grazed grassland in Scotland. Atmos. Environ. 2018, 194, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhou, E.; Jiang, H.; Li, W.; Wu, G.; Huang, L.; Hedlund, B.P.; Dong, H. Distribution and diversity of aerobic carbon monoxide-oxidizing bacteria in geothermal springs of China, the Philippines, and the United States. Geomicrobiol. J. 2015, 32, 903–913. [Google Scholar] [CrossRef]
- Haarstad, K.; Bergersen, O.; Sorheim, R. Occurrence of carbon monoxide during organic waste degradation. J. Air Waste Manag. Assoc. 2006, 56, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Hellebrand, H.J. Carbon monoxide emissions during composting. Berichte Uber Landwirtschaft 1999, 77, 109–112. [Google Scholar]
- Hellebrand, H.J.; Kalk, W.D. Emission of carbon monoxide during composting of dung and green waste. Nutr. Cycl. Agroecosyst. 2001, 60, 79–82. [Google Scholar] [CrossRef]
- Stegenta, S.; Sobieraj, K.; Pilarski, G.; Koziel, J.A.; Białowiec, A. Analysis of the spatial and temporal distribution of process gases within municipal biowaste compost. Sustainability 2019, 11, 2340. [Google Scholar] [CrossRef]
- Stegenta-Dąbrowska, S.; Drabczyński, G.; Sobieraj, K.; Koziel, J.A.; Białowiec, A. The Biotic and abiotic carbon monoxide formation during aerobic co-digestion of dairy cattle manure with green waste and sawdust. Front. Bioeng. Biotechnol. 2019, 7, 283. [Google Scholar] [CrossRef]
- Phillip, E.A.; Clark, O.G.; Londry, K.; Yu, S.; Leonard, J. Emission of carbon monoxide during composting of municipal solid waste. Compost Sci. Util. 2011, 19, 170–177. [Google Scholar] [CrossRef]
- Stegenta, S.; Dębowski, M.; Bukowski, P.; Randerson, P.F.; Białowiec, A. The influence of perforation of foil reactors on greenhouse gas emission rates during aerobic biostabilization of the undersize fraction of municipal wastes. J. Environ. Manag. 2018, 207, 355–365. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stegenta-Dąbrowska, S.; Rogosz, J.; Bukowski, P.; Dębowski, M.; Randerson, P.F.; Bieniek, J.; Białowiec, A. The Fluctuation of Process Gasses Especially of Carbon Monoxide during Aerobic Biostabilization of an Organic Fraction of Municipal Solid Waste under Different Technological Regimes. Data 2020, 5, 40. https://doi.org/10.3390/data5020040
Stegenta-Dąbrowska S, Rogosz J, Bukowski P, Dębowski M, Randerson PF, Bieniek J, Białowiec A. The Fluctuation of Process Gasses Especially of Carbon Monoxide during Aerobic Biostabilization of an Organic Fraction of Municipal Solid Waste under Different Technological Regimes. Data. 2020; 5(2):40. https://doi.org/10.3390/data5020040
Chicago/Turabian StyleStegenta-Dąbrowska, Sylwia, Jakub Rogosz, Przemysław Bukowski, Marcin Dębowski, Peter F. Randerson, Jerzy Bieniek, and Andrzej Białowiec. 2020. "The Fluctuation of Process Gasses Especially of Carbon Monoxide during Aerobic Biostabilization of an Organic Fraction of Municipal Solid Waste under Different Technological Regimes" Data 5, no. 2: 40. https://doi.org/10.3390/data5020040
APA StyleStegenta-Dąbrowska, S., Rogosz, J., Bukowski, P., Dębowski, M., Randerson, P. F., Bieniek, J., & Białowiec, A. (2020). The Fluctuation of Process Gasses Especially of Carbon Monoxide during Aerobic Biostabilization of an Organic Fraction of Municipal Solid Waste under Different Technological Regimes. Data, 5(2), 40. https://doi.org/10.3390/data5020040






