LC–MS/MS and UPLC–UV Evaluation of Anthocyanins and Anthocyanidins during Rabbiteye Blueberry Juice Processing
Abstract
:1. Introduction
2. Materials and Methods
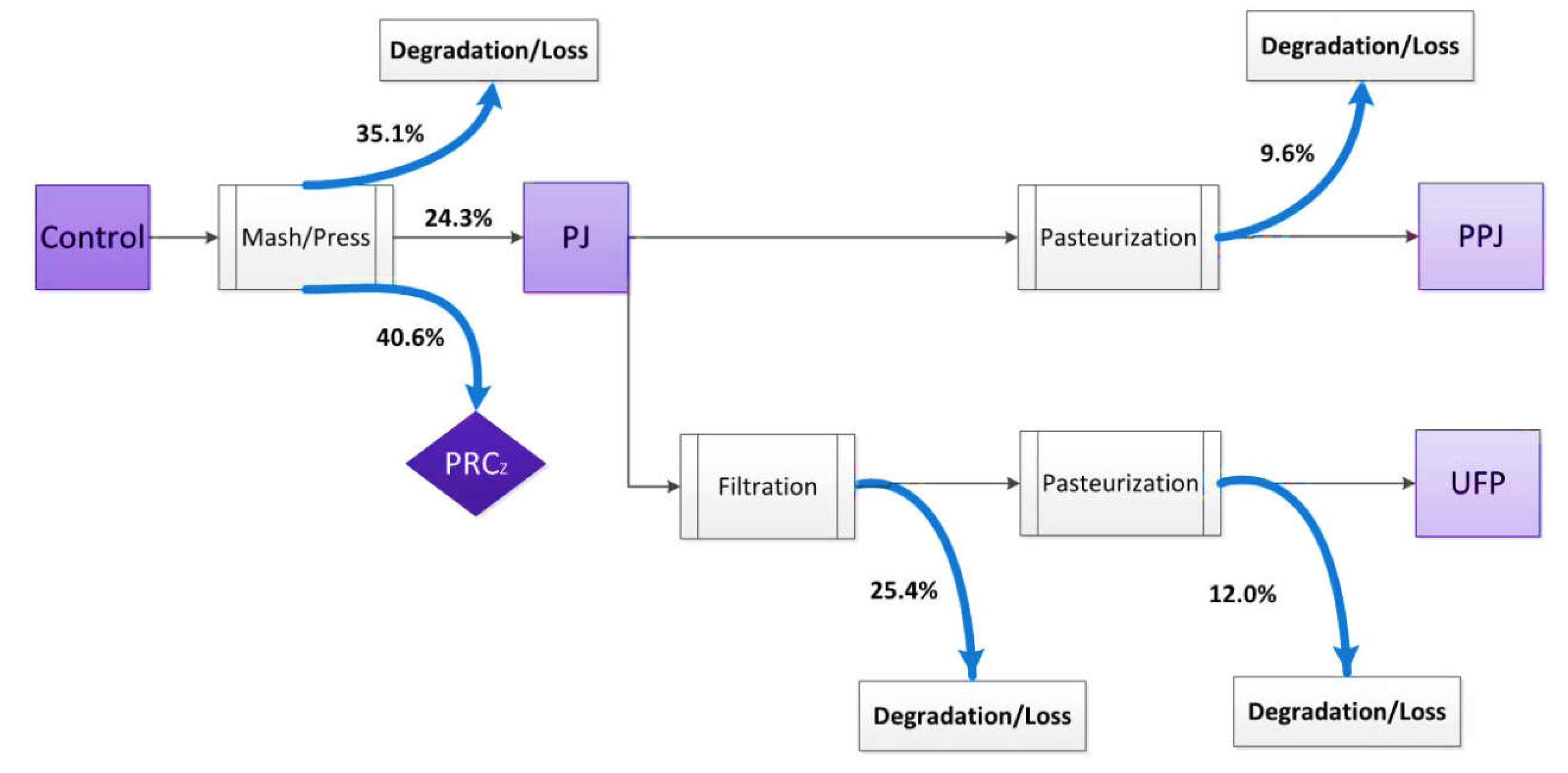
2.1. Juice Processing
2.2. Anthocyanin Analysis
2.2.1. Extraction
2.2.2. HPLC–MS/MS Chromatography
2.3. Anthocyanidin Analysis
2.3.1. Extraction
2.3.2. Acid Hydrolysis
2.3.3. UPLC Chromatography
2.4. Statistical Analysis
3. Results and Discussion
3.1. Identification of Anthocyanins and Anthocyanidins
3.1.1. Anthocyanins
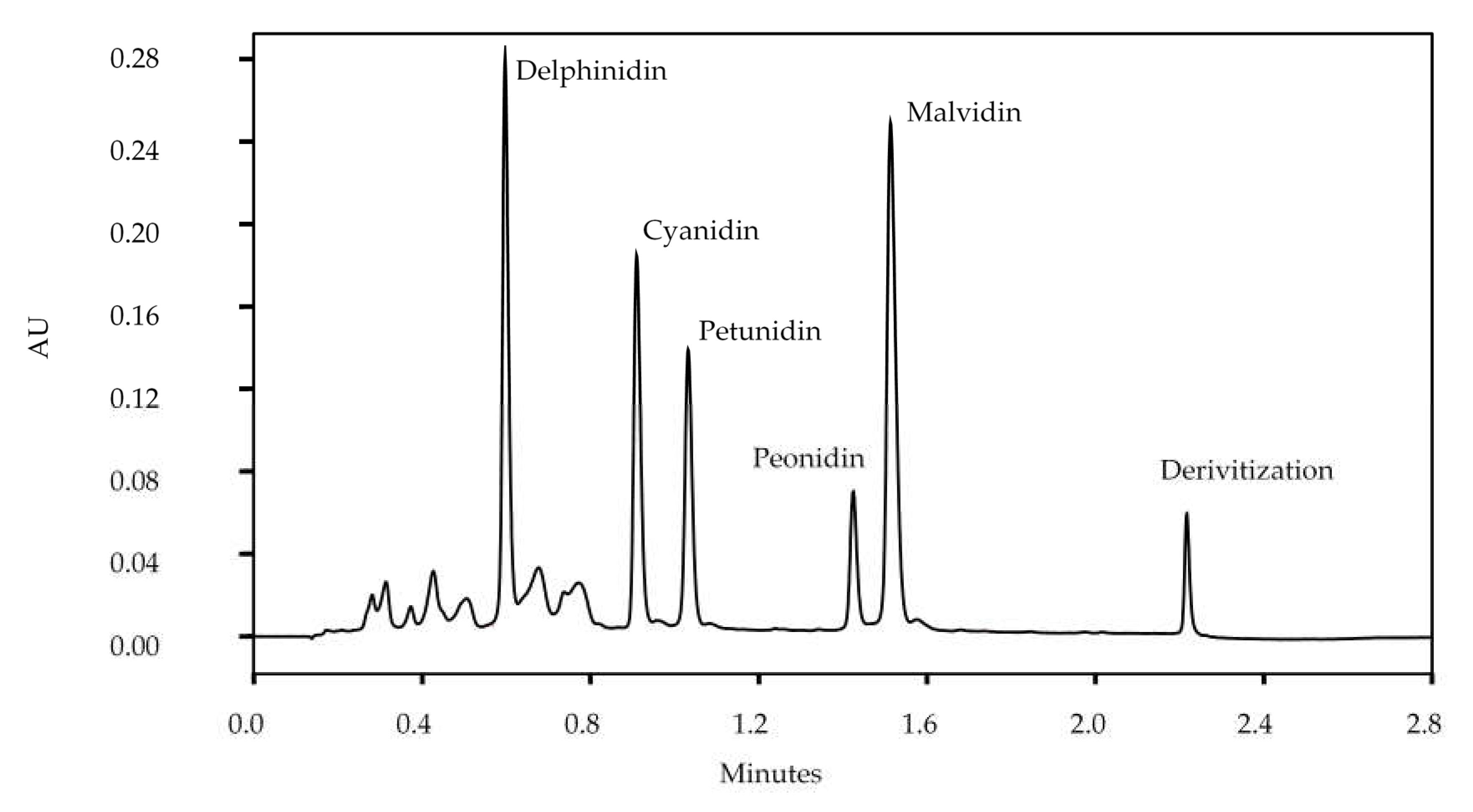
3.1.2. Anthocyanidins
3.2. Changes of Anthocyanins during Blueberry Juice Processing
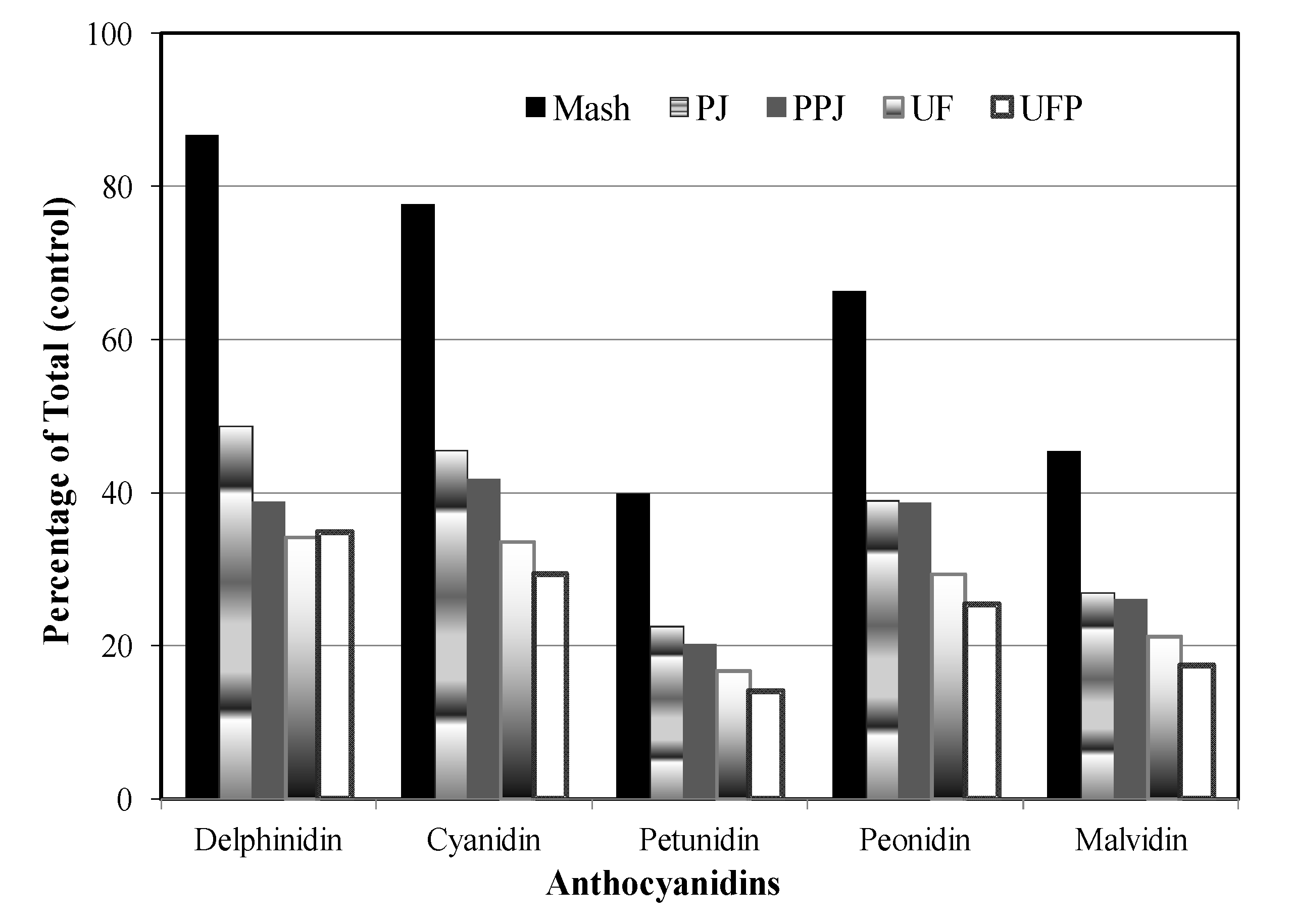
3.3. Changes of Anthocyanidins during Blueberry Juice Processing
3.3.1. Individual Anthocyanidins
3.3.2. Total Anthocyanidins
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Disclaimer
References
- Kalt, W.; Dufour, D. Health functionality of blueberries. HortTechnology 1997, 7, 216–221. [Google Scholar]
- Soto-Vaca, A.; Gutierrez, A.; Losso, J.N.; Xu, Z.; Finley, J.W. Evolution of phenolic compounds from color and flavor problems to health benefits. J. Agric. Food Chem. 2012, 60, 6658–6677. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Kou, X.; Fugal, K.; McLaughlin, J. Comparison of HPLC methods for determination of anthocyanins and anthocyanidins in bilberry extracts. J. Agric. Food Chem. 2004, 52, 688–691. [Google Scholar] [CrossRef] [PubMed]
- Gould, K.; Davies, K.M.; Winefield, C. Anthocyanins: Biosynthesis, Functions, and Applications, 1st ed.; Springer: New York, NY, USA, 2009; p. 325. [Google Scholar]
- Pina, F.; Melo, M.J.; Laia, C.A.T.; Parola, A.J.; Lima, J.C. Chemistry and applications of flavylium compounds: A handful of colours. Chem. Soc. Rev. 2012, 41, 869–908. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem. 2006, 54, 4069–4075. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Mazza, G. Quantitation and distribution of simple and acylated anthocyanins and other phenolics in blueberries. J. Food Sci. 1994, 59, 1057–1059. [Google Scholar] [CrossRef]
- Baxter, H.; Harborne, J.B.; Moss, G.P. Phytochemical Dictionary: A Handbook of Bioactive Compounds from Plants, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1998; p. 976. [Google Scholar]
- Routray, W.; Orsat, V. Blueberries and their anthocyanins: Factors affecting biosynthesis and properties. Compr. Rev. Food Sci. Food Saf. 2011, 10, 303–320. [Google Scholar] [CrossRef]
- Barnes, J.S.; Nguyen, H.P.; Shen, S.; Schug, K.A. General method for extraction of blueberry anthocyanins and identification using high performance liquid chromatography–electrospray ionization-ion trap-time of flight-mass spectrometry. J. Chromatogr. A 2009, 1216, 4728–4735. [Google Scholar] [CrossRef] [PubMed]
- Tropp, D. Why Local Food Matters: The Rising Importance of Locally-grown Food in the U.S. Food System. In Proceedings of the National Association of Counties Legislative Conference, Washington, DC, USA, 28 July 2014. [Google Scholar]
- Barkla, C. Fruit juices. In Market News Service; International Trade Centre (A joint agency of the World Trade Organization and the United Nations): Geneva, Switzerland, 2011; pp. 1–21. [Google Scholar]
- AIJN. Fruit Juice Matters 2017 Report; European Fruit Juice Association: Brussels, Belgum, 2017. [Google Scholar]
- Rohan, M. The markets for soft drinks and fruit juices change as a constant—Focusing on health. In Food and Drink Business Europe; Rohan, M., Ed.; Premier Publishing Ltd.: Dublin, Ireland, 2013. [Google Scholar]
- Bates, R.P.; Morris, J.R.; Crandall, P.G. Principles and Practices of Small and Medium Scale Fruit Juice Processing. FAO Agricultural Services Bulletin 2001. Available online: http://www.fao.org/docrep/005/y2515e/y2515e00.htm (accessed on 11 November 2017).
- Nikdel, S.; Chen, C.S.; Parish, M.E.; MacKellar, D.G.; Friedrich, L.M. Pasteurization of citrus juice with microwave energy in a continuous-flow unit. J. Agric. Food Chem. 1993, 41, 2116–2119. [Google Scholar] [CrossRef]
- Perera, C.O.; Smith, B. Technology of processing of horticultural crops. In Handbook of Farm, Dairy, and Food Machinery Engineering, 2nd ed.; Kutz, M., Ed.; Academic Press: Cambridge, MA, USA, 2013; pp. 259–315. [Google Scholar]
- Brambilla, A.; Maffi, D.; Rizzolo, A. Study of the influence of berry-blanching on syneresis in blueberry purées. Procedia Food Sci. 2011, 1, 1502–1508. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Impact of juice processing on blueberry anthocyanins and polyphenolics: Comparison of two pretreatments. J. Food Sci. 2002, 67, 1660–1667. [Google Scholar] [CrossRef]
- Brambilla, A.; Lo Scalzo, R.; Bertolo, G.; Torreggiani, D. Steam-blanched highbush blueberry (Vaccinium corymbosum L.) juice: Phenolic profile and antioxidant capacity in relation to cultivar selection. J. Agric. Food Chem. 2008, 56, 2643–2648. [Google Scholar] [CrossRef] [PubMed]
- Landbo, A.-K.; Kaack, K.; Meyer, A.S. Statistically designed two step response surface optimization of enzymatic prepress treatment to increase juice yield and lower turbidity of elderberry juice. Innov. Food Sci. Emerg. Technol. 2007, 8, 135–142. [Google Scholar] [CrossRef]
- Alper, N.; Bahceci, K.S.; Acar, J. Influence of processing and pasteurization on color values and total phenolic compounds of pomegranate juice. J. Food Process. Preserv. 2005, 29, 357–368. [Google Scholar] [CrossRef]
- Brooks, R.M.; Olmo, H.P. The Brooks and Olmo Register of Fruit & Nut Varieties; ASHS Press: Alexandria, VA, USA, 1997. [Google Scholar]
- Marshall, D.A.; Spiers, J.M.; Braswell, J.H. Splitting severity among rabbiteye (Vaccinium ashei ‘Reade’) blueberry cultivars in Mississippi and Louisiana. Intl. J. Fruit Sci. 2006, 6, 77–81. [Google Scholar] [CrossRef]
- USDA. Germplasm Resources Information Network (GRIN) Online Database; United States Department of Agriculture, Agricultural Research Service, National Genetic Resources Program, Ag Data Commons, 2014. Available online: http://dx.doi.org/10.15482/USDA.ADC/1212393 (accessed on 11 November 2017).
- Srivastava, A.; Akoh, C.C.; Yi, W.; Fischer, J.; Krewer, G. Effect of storage conditions on the biological activity of phenolic compounds of blueberry extract packed in glass bottles. J. Agric. Food Chem. 2007, 55, 2705–2713. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, J.C.; Stein-Chisholm, R.E.; Lloyd, S.W.; Bett-Garber, K.L.; Grimm, C.C.; Watson, M.A.; Lea, J.M. Volatile, anthocyanidin, quality and sensory changes in rabbiteye blueberry from whole fruit through pilot plant juice processing. J. Sci. Food Agric. 2017, 97, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Hynes, M.; Aubin, A. Acquity UPLC for the Rapid Analysis of Anthocyainidins in Berries; Waters Corporation: Milford, MA, USA, 2006. [Google Scholar]
- Cho, M.J.; Howard, L.R.; Prior, R.L.; Clark, J.R. Flavonoid glycosides and antioxidant capacity of various blackberry, blueberry and red grape genotypes determined by high-performance liquid chromatography/mass spectrometry. J. Sci. Food Agric. 2004, 84, 1771–1782. [Google Scholar] [CrossRef]
- Fanali, C.; Dugo, L.; D’Orazio, G.; Lirangi, M.; Dachà, M.; Dugo, P.; Mondello, L. Analysis of anthocyanins in commercial fruit juices by using nano-liquid chromatography-electrospray-mass spectrometry and high-performance liquid chromatography with UV-vis detector. J. Sep. Sci. 2011, 34, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Kalt, W.; McDonald, J.E.; Ricker, R.D.; Lu, X. Anthocyanin content and profile within and among blueberry species. Can. J. Plant Sci. 1999, 79, 617–623. [Google Scholar] [CrossRef]
- Lee, J.; Finn, C.E.; Wrolstad, R.E. Comparison of anthocyanin pigment and other phenolic compounds of Vaccinium membranaceum and Vaccinium ovatum native to the pacific northwest of North America. J. Agric. Food Chem. 2004, 52, 7039–7044. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, J.-I.; Tanaka, I.; Seo, S.; Yamazaki, M.; Saito, K. LC/PDA/ESI-MS profiling and radical scavenging activity of anthocyanins in various berries. J. Biomed. Biotechnol. 2004, 5, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Lohachoompol, V.; Mulholland, M.; Srzednicki, G.; Craske, J. Determination of anthocyanins in various cultivars of highbush and rabbiteye blueberries. Food Chem. 2008, 111, 249–254. [Google Scholar] [CrossRef]
- Prior, R.L.; Lazarus, S.A.; Cao, G.; Muccitelli, H.; Hammerstone, J.F. Identification of procyanidins and anthocyanins in blueberries and cranberries (Vaccinium spp.) using high-performance liquid chromatography/mass spectrometry. J. Agric. Food Chem. 2001, 49, 1270–1276. [Google Scholar] [CrossRef] [PubMed]
- Skrede, G.; Wrolstad, R.E.; Durst, R.W. Changes in anthocyanins and polyphenolics during juice processing of highbush blueberries (Vaccinium corymbosum L.). J. Food Sci. 2000, 65, 357–364. [Google Scholar] [CrossRef]
- Yoshimura, Y.; Enomoto, H.; Moriyama, T.; Kawamura, Y.; Setou, M.; Zaima, N. Visualization of anthocyanin species in rabbiteye blueberry Vaccinium ashei by matrix-assisted laser desorption/ionization imaging mass spectrometry. Anal. Bioanal. Chem. 2012, 403, 1885–1895. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Prior, R.L. Systematic identification and characterization of anthocyanins by HPLC-ESI-MS/MS in common foods in the United States: Fruits and berries. J. Agric. Food Chem. 2005, 53, 2589–2599. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, J.C.; Lloyd, S.W.; Preece, J.E.; Moersfelder, J.W.; Stein-Chisholm, R.E.; Obando-Ulloa, J.M. Physicochemical properties and aroma volatile profiles in a diverse collection of California-grown pomegranate (Punica granatum L.) germplasm. Food Chem. 2015, 181, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Havlíková, L.; Míková, K. Heat stability of anthocyanins. Z. Lebensm. Forch. 1985, 181, 427–432. [Google Scholar] [CrossRef]
- Burns, J.; Mullen, W.; Landrault, N.; Teissedre, P.-L.; Lean, M.E.J.; Crozier, A. Variations in the profile and content of anthocyanins in wines made from Cabernet Sauvignon and hybrid grapes. J. Agric. Food Chem. 2002, 50, 4096–4102. [Google Scholar] [CrossRef] [PubMed]
- Howard, L.R.; Clark, J.R.; Brownmiller, C. Antioxidant capacity and phenolic content in blueberries as affected by genotype and growing season. J. Sci. Food Agric. 2003, 83, 1238–1247. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jimenez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L.; Knox, C.; Elisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Giusti, M.M.; Wrolstad, R.E. Acylated anthocyanins from edible sources and their applications in food systems. Biochem. Eng. J. 2003, 14, 217–225. [Google Scholar] [CrossRef]
- Castañeda-Ovando, A.; de Lourdes Pacheco-Hernández, M.; Páez-Hernández, M.E.; Rodríguez, J.A.; Galán-Vidal, C.A. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Sadilova, E.; Stintzing, F.C.; Carle, R. Thermal degradation of acylated and nonacylated anthocyanins. J. Food Sci. 2006, 71, C504–C512. [Google Scholar] [CrossRef]
- Patras, A.; Brunton, N.P.; O’Donnell, C.; Tiwari, B.K. Effect of thermal processing on anthocyanin stability in foods; mechanisms and kinetics of degradation. Trends Food Sci. Technol. 2010, 21, 3–11. [Google Scholar] [CrossRef]
- Brownmiller, C.; Howard, L.R.; Prior, R.L. Processing and storage effects on monomeric anthocyanins, percent polymeric color, and antioxidant capacity of processed blueberry products. J. Food Sci. 2008, 73, H72–H79. [Google Scholar] [CrossRef] [PubMed]
- Kalbasi, A.; Cisneros-Zevallos, L. Fractionation of monomeric and polymeric anthocyanins from concord grape (Vitis labrusca L.) juice by membrane ultrafiltration. J. Agric. Food Chem. 2007, 55, 7036–7042. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.Y.; Hwang, L.S.; Chiang, B.H. Concentration of perilla anthocyanins by ultrafiltration. J. Food Sci. 1986, 51, 1494–1497. [Google Scholar] [CrossRef]
- Cissé, M.; Vaillant, F.; Pallet, D.; Dornier, M. Selecting ultrafiltration and nanofiltration membranes to concentrate anthocyanins from roselle extract (Hibiscus sabdariffa L.). Food Res. Int. 2011, 44, 2607–2614. [Google Scholar] [CrossRef]
- Pap, N.; Mahosenaho, M.; Pongrácz, E.; Mikkonen, H.; Jaakkola, M.; Virtanen, V.; Myllykoski, L.; Horváth-Hovorka, Z.; Hodúr, C.; Vatai, G. Effect of ultrafiltration on anthocyanin and flavonol content of black currant juice (Ribes nigrum L.). Food Bioprocess Technol. 2012, 5, 921–928. [Google Scholar] [CrossRef]
- Patil, G.; Raghavarao, K.S. Integrated membrane process for the concentration of anthocyanin. J. Food Eng. 2007, 78, 1233–1239. [Google Scholar] [CrossRef]
- Fischer, U.A.; Carle, R.; Kammerer, D.R. Identification and quantification of phenolic compounds from pomegranate (Punica granatum L.) peel, mesocarp, aril and differently produced juices by HPLC-DAD–ESI/MS. Food Chem. 2011, 127, 807–821. [Google Scholar] [CrossRef] [PubMed]
- Rwabahizi, S.; Wrolstad, R.E. Effects of mold contamination and ultrafiltration on the color stability of strawberry juice and concentrate. J. Food Sci. 1988, 53, 857–861. [Google Scholar] [CrossRef]
- Rossi, M.; Giussani, E.; Morelli, R.; Lo Scalzo, R.; Nani, R.C.; Torreggiani, D. Effect of fruit blanching on phenolics and radical scavenging activity of highbush blueberry juice. Food Res. Int. 2003, 36, 999–1005. [Google Scholar] [CrossRef]
- Sablani, S.S.; Andrews, P.K.; Davies, N.M.; Walters, T.; Saez, H.; Syamaladevi, R.M.; Mohekar, P.R. Effect of thermal treatments on phytochemicals in conventionally and organically grown berries. J. Sci. Food Agric. 2010, 90, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Stein-Chisholm, R.E.; Losso, J.N.; Finley, J.W.; Beaulieu, J.C. Not from concentrate blueberry juice extraction utilizing frozen fruit, heated mash and enzyme processes. HortTechnology 2017, 27, 30–36. [Google Scholar] [CrossRef]
- Giusti, M.M.; Wrolstad, R.E. Characterization and measurement of anthocyanins by UV-visible spectroscopy. In Current Protocols in Food Analytical Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2001; Available online: http://onlinelibrary.wiley.com/doi/10.1002/0471142913.faf0102s00/abstract (accessed on 22 November 2017).
- Lee, J.; Rennaker, C.; Wrolstad, R.E. Correlation of two anthocyanin quantification methods: HPLC and spectrophotometric methods. Food Chem. 2008, 110, 782–786. [Google Scholar] [CrossRef]
- You, Q.; Wang, B.; Chen, F.; Huang, Z.; Wang, X.; Luo, P.G. Comparison of anthocyanins and phenolics in organically and conventionally grown blueberries in selected cultivars. Food Chem. 2011, 125, 201–208. [Google Scholar] [CrossRef]
- Wang, S.Y.; Chen, C.-T.; Sciarappa, W.; Wang, C.Y.; Camp, M.J. Fruit quality, antioxidant capacity, and flavonoid content of organically and conventionally grown blueberries. J. Agric. Food Chem. 2008, 56, 5788–5794. [Google Scholar] [CrossRef] [PubMed]
- Nicoué, E.É.; Savard, S.; Belkacemi, K. Anthocyanins in wild blueberries of Quebec: Extraction and identification. J. Agric. Food Chem. 2007, 55, 5626–5635. [Google Scholar] [CrossRef] [PubMed]
- Plaza, M.; Turner, C. Pressurized hot water extraction of bioactives. Trend. Anal. Chem. 2015, 71, 39–54. [Google Scholar] [CrossRef]
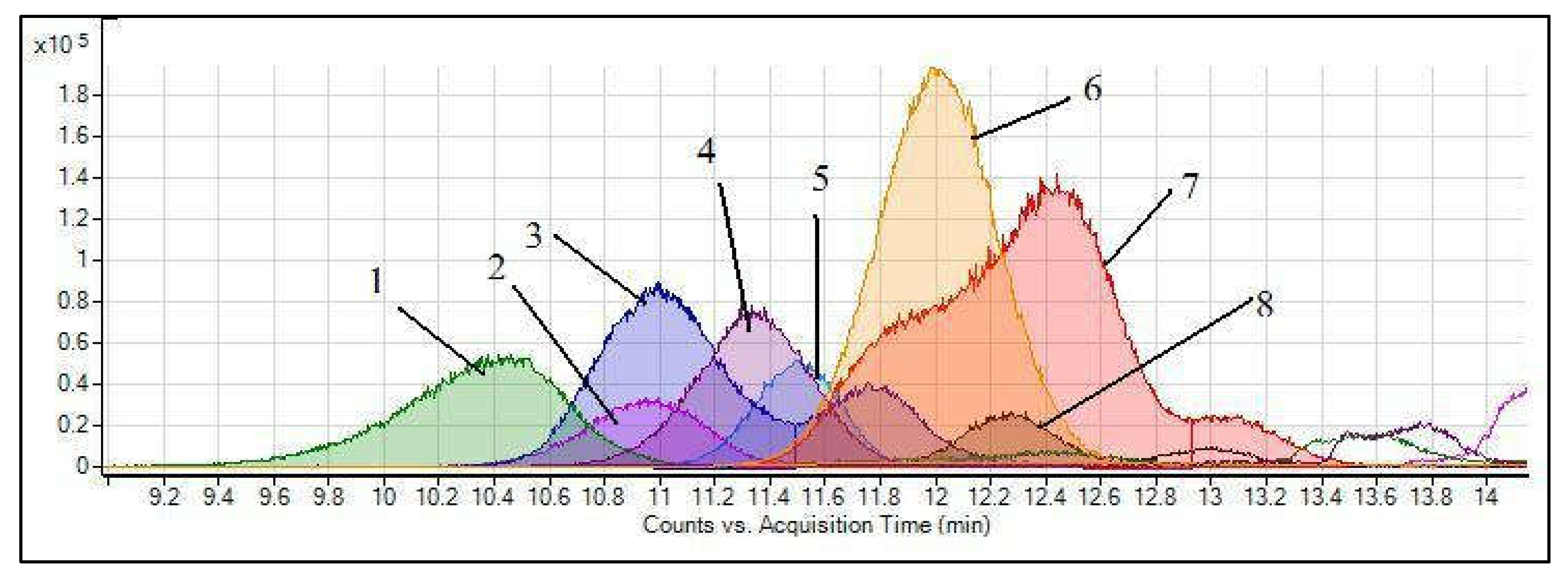
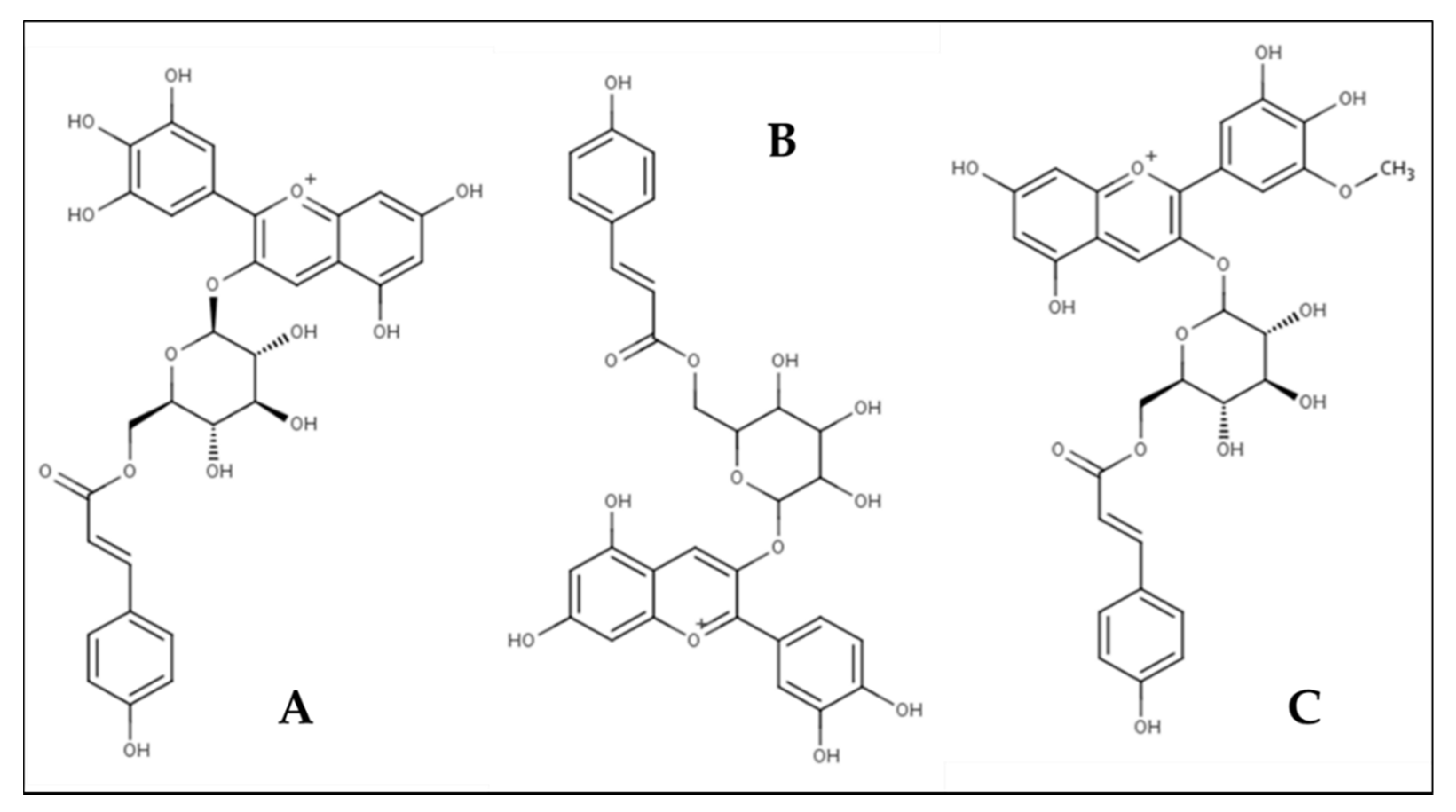

| Major Anthocyanin z | Sugar Moiety | Molecular Formula | [M + H]+ (m/z) y | MS/MS (m/z) x | Rt (min) HPLC w |
| Delphinidin-3-arabinoside | Arabinose | C20H19O11 | 435.0922 | 303.0500 | 10.94 |
| Cyanidin-3-arabinoside | Arabinose | C20H19O10 | 419.0928 | 287.0550 | 11.47 |
| Petunidin-3-arabinoside | Arabinose | C21H21O11 | 449.1078 | 317.0700 | 11.66 |
| Peonidin-3-arabinoside | Arabinose | C21H21O11 | 433.1129 | 301.0700 | 12.20 |
| Malvidin-3-arabinoside | Arabinose | C22H23O11 | 463.1235 | 331.0800 | 12.34 |
| Delphinidin-3-pyranoside | Galactose/Glucose | C21H21O12 | 465.1027 | 303.0500 | 10.38 |
| Cyanidin-3-pyranoside | Galactose/Glucose | C21H21O11 | 449.1078 | 287.0550 | 10.99 |
| Petunidin-3-pyranoside | Galactose/Glucose | C22H23O12 | 479.1184 | 317.0700 | 11.29 |
| Peonidin-3-pyranoside | Galactose/Glucose | C22H23O11 | 463.1235 | 301.0700 | 12.99 |
| Malvidin-3-pyranoside | Galactose/Glucose | C23H25O12 | 493.1340 | 331.0800 | 11.98 |
| Minor Anthocyanin z | Sugar Moiety | Molecular Formula | [M + H]+ (m/z) | MS/MS (m/z) | Rt (min) HPLC w |
| Delphinidin-3-(p-coumaroyl-glucoside) | Glucose | C30H27O14 | 611.1395 | 303.0500 | 13.11 |
| Cyanidin-3-(p-coumaroyl-glucoside) | Glucose | C30H27O13 | 595.1446 | 287.0550 | 13.86 |
| Petunidin-3-(p-coumaroyl-glucoside) | Glucose | C31H29O14 | 625.1552 | 317.0700 | 14.02 |
| Peonidin-3-(p-coumaroyl-glucoside) | Glucose | C31H29O13 | 609.1603 | 301.0700 | ND v |
| Malvidin-3-(p-coumaroyl-glucoside) | Glucose | C32H31O14 | 639.1708 | 331.0800 | ND |
| Delphinidin-3-(6″-acetyl-pyranoside) | Galactose/Glucose | C23H23O13 | 507.1133 | 303.0500 | ND |
| Cyanidin-3-(6″-acetyl-pyranoside) | Galactose/Glucose | C23H23O12 | 491.1184 | 287.0550 | ND |
| Petunidin-3-(6″-acetyl-pyranoside) | Galactose/Glucose | C24H25O13 | 521.1289 | 317.0700 | ND |
| Peonidin-3-(6″-acetyl-pyranoside) | Galactose/Glucose | C24H25O12 | 505.1340 | 301.0700 | ND |
| Malvidin-3-(6″-acetyl-pyranoside) | Galactose/Glucose | C25H27O13 | 535.1446 | 331.0800 | ND |
| Anthocyanin | Literature z |
|---|---|
| Delphinidin-3-arabinoside | [3,10,19,28,29,30,31,32,33,34,35,36,37,38] |
| Cyanidin-3-arabinoside | [3,7,10,19,29,30,31,32,33,34,35,37,38] |
| Petunidin-3-arabinoside | [7,10,19,28,29,30,31,32,33,35,36,37,38] |
| Peonidin-3-arabinoside | [10,26,28,29,32,33,34,35,37,38] |
| Malvidin-3-arabinoside | [7,10,19,28,29,31,32,33,34,35,36,37] |
| Delphinidin-3-pyranoside | [3,7,10,19,26,28,29,30,31,32,33,34,35,36] |
| Cyanidin-3-pyranoside | [3,7,10,19,26,28,29,30,31,32,33,34,35,36,38] |
| Petunidin-3-pyranoside | [3,7,10,19,26,28,29,30,31,32,33,34,35,36,38] |
| Peonidin-3-pyranoside | [3,7,10,19,26,28,29,30,31,32,33,34,35,36,38] |
| Malvidin-3-pyranoside | [3,7,10,19,26,28,29,30,31,32,34,35,36,38] |
| Delphinidin-3-(p-coumaroyl-glucoside) y | [10] |
| Cyanidin-3-(p-coumaroyl-glucoside) | [10] |
| Petunidin-3-(p-coumaroyl-glucoside) | [10] |
| Peonidin-3-(p-coumaroyl-glucoside) | [10] |
| Malvidin-3-(p-coumaroyl-glucoside) | [10] |
| Delphinidin-3-(6″-acetyl-pyranoside) | [7,10,28,29,31,36] |
| Cyanidin-3-(6″-acetyl-pyranoside) | [7,10,28,31] |
| Petunidin-3-(6″-acetyl-pyranoside) | [7,10,28,29,31] |
| Peonidin-3-(6″-acetyl-pyranoside) | [7,10,28,31] |
| Malvidin-3-(6″-acetyl-pyranoside) | [7,10,28,29,31,36] |
| Treatment z | Del-3-ara y | Cya-3-ara | Pet-3-ara/Cya-3-pyr | Peo-3-ara |
|---|---|---|---|---|
| PRC | 0.2 ± 0.05 d x,w | 0.04 ± 0.03 d | 0.2 ± 0.03 d | ND |
| Control | 8.8 ± 1.8 a | 8.4 ± 1.2 a | 22.8 ± 2.9 a | 5.1 ± 0.9 a |
| PJ | 2.8 ± 0.5 b | 2.4 ± 0.4 bc | 7.0 ± 1.0 b | 1.3 ± 0.2 bc |
| PPJ | 2.7 ± 0.9 bc | 2.6 ± 0.8 bc | 7.3 ± 1.0 b | 1.6 ± 0.5 ab |
| UF | 3.6 ± 0.7 b | 3.1 ± 0.6 b | 8.3 ± 0.8 b | 1.9 ± 0.2 b |
| UFP | 1.7 ± 0.4 c | 1.8 ± 0.4 c | 4.4 ± 0.4 c | 0.9 ± 0.2 c |
| Mal-3-ara/Peo-3-pyr | Del-3-pyr | Pet-3-pyr | Mal-3-pyr | |
| PRC | 0.3 ± 0.1 d | 0.5 ± 0.09 d | 0.3 ± 0.03 d | 0.5 ± 0.1 d |
| Control | 49.2 ± 8.3 a | 20.5 ± 4.1 a | 18.8 ± 3.8 a | 49.5 ± 9.7 a |
| PJ | 16.5 ± 2.8 bc | 7.1 ± 1.5 b | 7.1 ± 1.6 b | 24.7 ± 4.4 bc |
| PPJ | 17.7 ± 5.6 b | 6.0 ± 1.7 bc | 7.0 ± 2.2 b | 25.6 ± 7.9 b |
| UF | 22.1 ± 2.3 b | 8.4 ± 1.3 b | 8.7 ± 1.9 b | 30.5 ± 4.6 b |
| UFP | 12.2 ± 3.6 c | 4.0 ± 0.6 c | 4.3 ± 0.9 c | 18.2 ± 4.6 c |
| Del-3-cou | Cya-3-cou | Pet-3-cou | Total Abundance | |
| PRC | 0.05 ± 0.01 c | ND | 0.1 ± 0.05 c | 2.2 ± 0.4 d |
| Control | 0.5 ± 0.1 b | 0.3 ± 0.05 b | 1.4 ± 0.5 b | 185.2 ± 34.0 a |
| PJ | 1.1 ± 0.3 a | 0.4 ± 0.1 a | 2.1 ± 0.7 ab | 72.5 ± 13.4 b |
| PPJ | 0.9 ± 0.4 a | 0.4 ± 0.1 ab | 2.2 ± 1.1 a | 74.0 ± 21.2 b |
| UF | 1.1 ± 0.4 a | 0.5 ± 0.2 a | 2.9 ± 1.1 a | 85.2 ± 21.2 b |
| UFP | 1.0 ± 0.3 a | 0.4 ± 0.08 ab | 2.1 ± 0.4 ab | 50.9 ± 11.5 c |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stein-Chisholm, R.E.; Beaulieu, J.C.; Grimm, C.C.; Lloyd, S.W. LC–MS/MS and UPLC–UV Evaluation of Anthocyanins and Anthocyanidins during Rabbiteye Blueberry Juice Processing. Beverages 2017, 3, 56. https://doi.org/10.3390/beverages3040056
Stein-Chisholm RE, Beaulieu JC, Grimm CC, Lloyd SW. LC–MS/MS and UPLC–UV Evaluation of Anthocyanins and Anthocyanidins during Rabbiteye Blueberry Juice Processing. Beverages. 2017; 3(4):56. https://doi.org/10.3390/beverages3040056
Chicago/Turabian StyleStein-Chisholm, Rebecca E., John C. Beaulieu, Casey C. Grimm, and Steven W. Lloyd. 2017. "LC–MS/MS and UPLC–UV Evaluation of Anthocyanins and Anthocyanidins during Rabbiteye Blueberry Juice Processing" Beverages 3, no. 4: 56. https://doi.org/10.3390/beverages3040056
APA StyleStein-Chisholm, R. E., Beaulieu, J. C., Grimm, C. C., & Lloyd, S. W. (2017). LC–MS/MS and UPLC–UV Evaluation of Anthocyanins and Anthocyanidins during Rabbiteye Blueberry Juice Processing. Beverages, 3(4), 56. https://doi.org/10.3390/beverages3040056







