Glycocalyx Sensing with a Mathematical Model of Acoustic Shear Wave Biosensor
Abstract
:1. Introduction
2. Materials and Methods
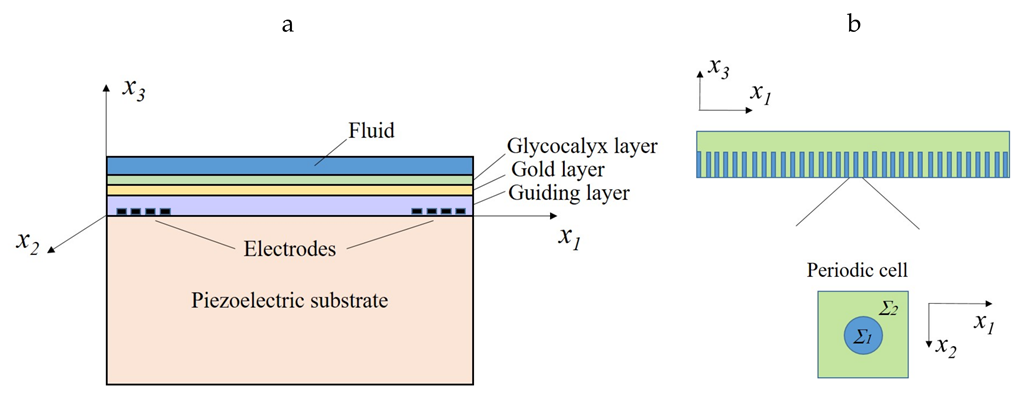
2.1. Mathematical Model of Acoustic Wave Biosensor
- 1.
- On the interfaces between two solid layers, the continuity of the displacements and the equilibrium of normal pressures must hold.
- 2.
- The electric displacement and the tangent component of the electric field must be zero on the interface between the piezoelectric substrate and the guiding layer.
- 3.
- The equilibrium of the pressures and a no-slip condition is required on the interface between the fluid and homogenized bristle structure (glycocalyx).
2.2. Computer Program for the Calculation of the Acoustic Wave Velocity
3. Results
3.1. Geometrical and Physical Characteristics of Glycocalyx
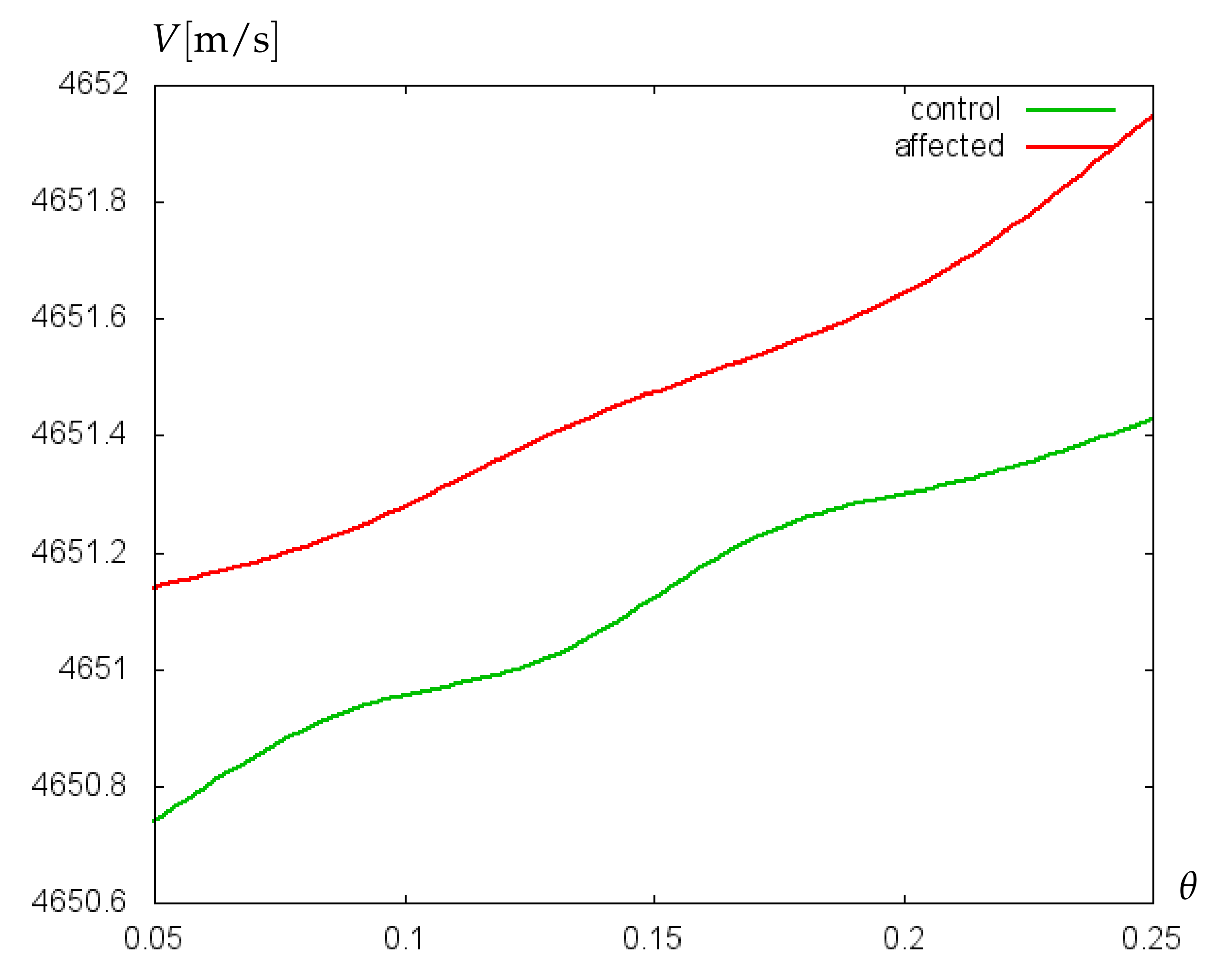
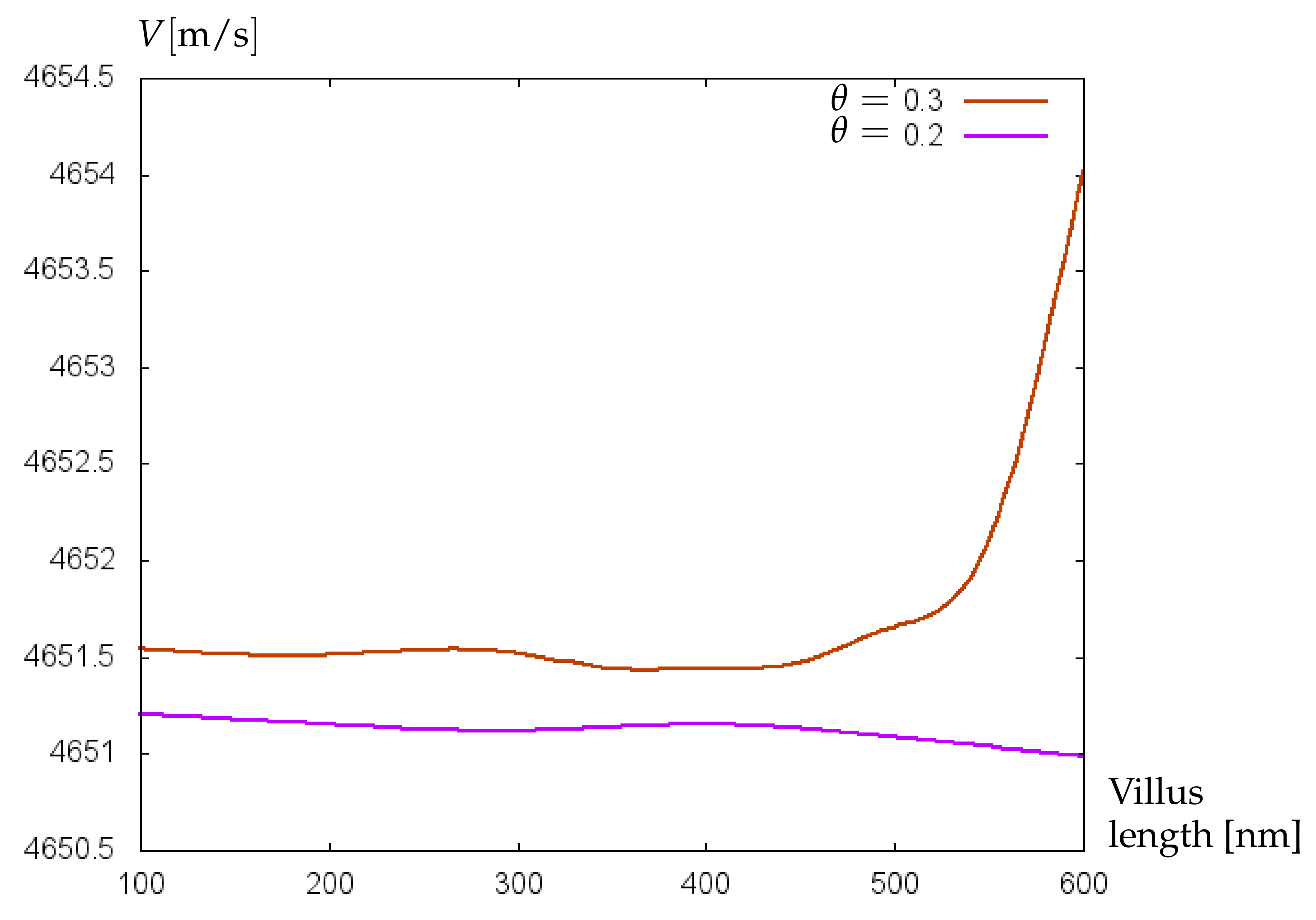
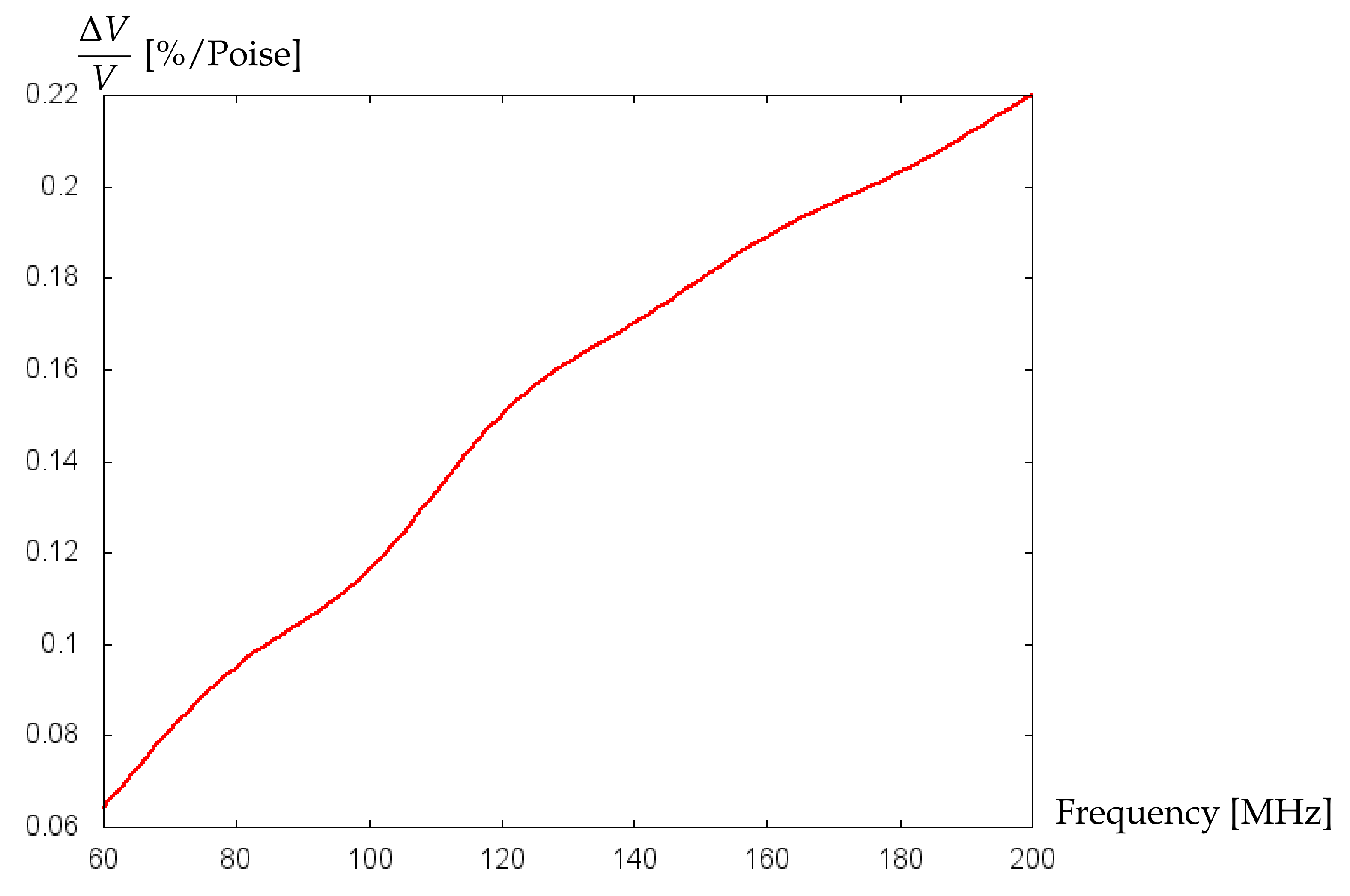
3.2. Numerical Simulations and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gouverneur, M.; van den Berg, B.; Nieuwdorp, M.; Stroes, E.; Vink, H. Vasculoprotective properties of the endothelial glycocalyx: Effects of fluid shear stress. J. Intern. Med. 2006, 259, 393–400. [Google Scholar] [CrossRef]
- Ando, Y.; Okada, H.; Takemura, G.; Suzuki, K.; Takada, C.; Tomita, H.; Zaikokuji, R.; Hotta, Y.; Miyazaki, N.; Yano, H.; et al. Brain-specific ultrastructure of capillary endothelial glycocalyx and its possible contribution for blood brain barrier. Sci. Rep. 2018, 8, 17523. [Google Scholar] [CrossRef]
- Richter, R.P.; Payne, G.A.; Ambalavanan, N.; Gaggar, A.; Richter, J.R. The endothelial glycocalyx in critical illness: A pediatric perspective. Matrix Biol. Plus 2022, 14, 100106. [Google Scholar] [PubMed]
- Battiato, I.; Tartakovsky, D.; Cabrales, P.; Intaglietta, M. Role of glycocalyx in attenuation of shear stress on endothelial cells: From in vivo experiments to microfluidic circuits. In Proceedings of the 2017 European Conference on Circuit Theory and Design (ECCTD), Catania, Italy, 4–6 September 2017; IEEE: New York, NY, USA, 2017; pp. 1–4. [Google Scholar]
- Pontrelli, G.; Halliday, I.; Spencer, T.J.; König, C.S.; Collins, M.W. Modelling the glycocalyx–endothelium–erythrocyte interaction in the microcirculation: A computational study. Comput. Methods Biomech. Biomed. Eng. 2015, 18, 351–361. [Google Scholar] [CrossRef]
- Jiang, X.Z.; Lu, Y.; Luo, K.H.; Ventikos, Y. Understanding endothelial glycocalyx function under flow shear stress from a molecular perspective. Biorheology 2019, 56, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Rodriguez, D.; Guillou, L.; Cornat, F.; Lafaurie-Janvore, J.; Babataheri, A.; de Langre, E.; Barakat, A.I.; Husson, J. Mechanical criterion for the rupture of a cell membrane under compression. Biophys. J. 2016, 111, 2711–2721. [Google Scholar] [CrossRef]
- Guz, N.; Dokukin, M.; Kalaparthi, V.; Sokolov, I. If cell mechanics can be described by elastic modulus: Study of different models and probes used in indentation experiments. Biophys. J. 2014, 107, 564–575. [Google Scholar] [CrossRef] [PubMed]
- Faucher, K.M.; Sun, X.L.; Chaikof, E.L. Fabrication and characterization of glycocalyx-mimetic surfaces. Langmuir 2003, 19, 1664–1670. [Google Scholar] [CrossRef]
- Liang, X.; Zhang, A.; Sun, W.; Lei, J.; Liu, X.; Tang, Z.; Chen, H. Vascular cell behavior on glycocalyx–mimetic surfaces: Simultaneous mimicking of the chemical composition and topographical structure of the vascular endothelial glycocalyx. Colloids Surfaces B Biointerfaces 2022, 212, 112337. [Google Scholar] [CrossRef] [PubMed]
- Mandal, D.; Banerjee, S. Surface acoustic wave (SAW) sensors: Physics, materials, and applications. Sensors 2022, 22, 820. [Google Scholar] [CrossRef]
- Huang, Y.; Das, P.K.; Bhethanabotla, V.R. Surface acoustic waves in biosensing applications. Sens. Actuators Rep. 2021, 3, 100041. [Google Scholar] [CrossRef]
- Botkin, N.D.; Hoffmann, K.-H.; Starovoitov, V.N.; Turova, V.L. Treatment of the interface between fine elastic structures and fluids. In Proceedings of the 2003 IEEE International Conference “Physics and Control”, St.-Petersburg, Russia, 20–22 August 2003; Fradkov, A.L., Churilov, A.N., Eds.; IEEE: New York, NY, USA, 2003; Volume 4, pp. 1367–1372. [Google Scholar]
- Hoffmann, K.-H.; Botkin, N.D.; Starovoitov, V.N. Homogenization of interfaces between rapidly oscillating fine elastic structures and fluids. SIAM J. Appl. Math. 2005, 65, 983–1005. [Google Scholar] [CrossRef]
- Botkin, N.D.; Hoffmann, K.-H.; Pykhteev, O.A.; Turova, V.L. Dispersion relations for acoustic waves in heterogeneous multi-layered structures contacting with fluids. J. Frankl. Inst. 2007, 344, 520–534. [Google Scholar] [CrossRef]
- Haeren, R.H.; Van de Ven, S.E.; Van Zandvoort, M.A.; Vink, H.; van Overbeeke, J.J.; Hoogland, G.; Rijkers, K. Assessment and imaging of the cerebrovascular glycocalyx. Curr. Neurovascular Res. 2016, 13, 249–260. [Google Scholar] [CrossRef]
- Pykhteev, O. Characterization of Acoustic Waves in Multi-Layered Structures. Ph.D. Thesis, Technische Universität München, München, Germany, 2010. [Google Scholar]
- Damiano, E.R. The effect of the endothelial-cell glycocalyx on the motion of red blood cells through capillaries. Microvasc. Res. 1998, 55, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Secomb, T.W.; Hsu, R.; Pries, A.R. Motion of red blood cells in a capillary with an endothelial surface layer: Effect of flow velocity. Am. J. -Physiol.-Heart Circ. Physiol. 2001, 281, H629–H636. [Google Scholar] [CrossRef]
- van Haaren, P.M.; VanBavel, E.; Vink, H.; Spaan, J.A. Localization of the permeability barrier to solutes in isolated arteries by confocal microscopy. Am. J.-Physiol.-Heart Circ. Physiol. 2003, 285, H2848–H2856. [Google Scholar] [CrossRef]
- Marsh, G.; Waugh, R.E. Quantifying the mechanical properties of the endothelial glycocalyx with atomic force microscopy. JoVE (J. Vis. Exp.) 2013, 72, e50163. [Google Scholar] [CrossRef] [PubMed]
- Vink, H.; Duling, B.R. Identification of distinct luminal domains for macromolecules, erythrocytes, and leukocytes within mammalian capillaries. Circ. Res. 1996, 79, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Pries, A.R.; Kuebler, W.M. Normal endothelium. In The Vascular Endothelium I; Springer: Berlin/Heidelberg, Germany, 2006; pp. 1–40. [Google Scholar]
- Bai, K.; Wang, W. Spatio-temporal development of the endothelial glycocalyx and its mechanical property in vitro. J. R. Soc. Interface 2012, 9, 2290–2298. [Google Scholar] [CrossRef] [Green Version]
- Perazzo, A.; Nunes, J.K.; Guido, S.; Stone, H.A. Flow-induced gelation of microfiber suspensions. Proc. Natl. Acad. Sci. USA 2017, 114, E8557–E8564. [Google Scholar] [CrossRef]
- Kabedev, A.; Lobaskin, V. Structure and elasticity of bush and brush-like models of the endothelial glycocalyx. Sci. Rep. 2018, 8, 240. [Google Scholar] [CrossRef]
- Zhu, J.; Li, X.; Yin, J.; Hu, Y.; Gu, Y.; Pan, S. Glycocalyx degradation leads to blood–brain barrier dysfunction and brain edema after asphyxia cardiac arrest in rats. J. Cereb. Blood Flow Metab. 2018, 38, 1979–1992. [Google Scholar] [CrossRef] [PubMed]
- Kuss, N.; Bauknecht, E.; Felbinger, C.; Gehm, J.; Gehm, L.; Pöschl, J.; Ruef, P. Whole blood viscosity of preterm infants–differences to term neonates. Clin. Hemorheol. Microcirc. 2015, 61, 397–405. [Google Scholar] [PubMed]
- Setyawan, A.; Badarudin, A. Effects of liquid viscosity on the wave velocity and wave frequency in horizontal annular flow. Appl. Mech. Mater. 2015, 758, 7–12. [Google Scholar] [CrossRef]
- Lampe, R.; Turova, V.; Botkin, N.; Eckardt, L.; Felderhoff-Müser, U.; Rieger-Fackeldey, E.; Alves-Pinto, A.; Kovtanyuk, A.; Sidorenko, I. Postnatal paraclinical parameters associated to occurrence of intracerebral hemorrhage in preterm infants. Neuropediatrics 2019, 50, 103–110. [Google Scholar]
- Turova, V.; Sidorenko, I.; Eckardt, L.; Rieger-Fackeldey, E.; Felderhoff-Müser, U.; Alves-Pinto, A.; Lampe, R. Machine learning models for identifying preterm infants at risk of cerebral hemorrhage. PLoS ONE 2020, 15, e0227419. [Google Scholar] [CrossRef] [PubMed]
- Weinbaum, S.; Tarbell, J.M.; Damiano, E.R. The structure and function of the endothelial glycocalyx layer. Annu. Rev. Biomed. Eng. 2007, 9, 121–167. [Google Scholar] [CrossRef]
- Meuwese, M.C.; Mooij, H.L.; Nieuwdorp, M.; van Lith, B.; Marck, R.; Vink, H.; Kastelein, J.P.; Stroes, E.S. Partial recovery of the endothelial glycocalyx upon rosuvastatin therapy in patients with heterozygous familial hypercholesterolemia. J. Lipid Res. 2009, 50, 148–153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
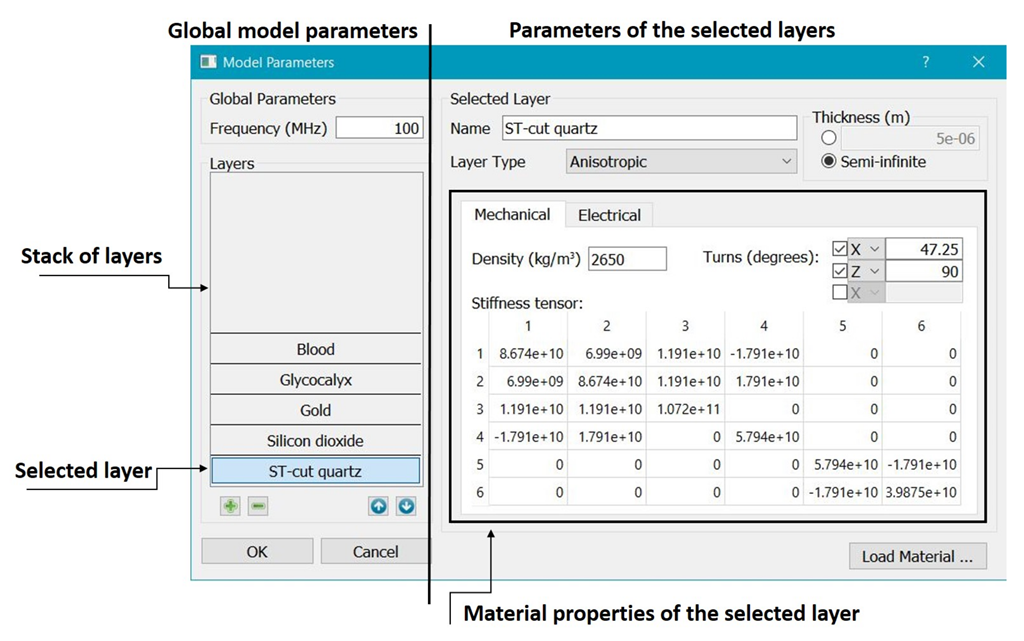







| Layer | Material | Density [kg/m] | Other Parameters |
|---|---|---|---|
| Fluid | Blood | = 1060 | Dynamic viscosity, = 0.0034 [N·s/m] |
| Volume viscosity, = 0.00384 [N·s/m] | |||
| Compressibility, = 3.787e-10 [1/Pa] | |||
| Glycocalyx | Homogenized substance | = 1280 | Young’s modulus = 500 [Pa] |
| Poisson’s ratio = 0.3 | |||
| Shielding | Gold | = 19300 | Young’s modulus E = 78 [GPa] |
| Poisson’s ratio = 0.44 | |||
| Guiding | SiO | = 2200 | Young’s modulus E = 72 [GPa] |
| Poisson’s ratio = 0.17 | |||
| Substrate | -quarz | = 2650 | Stiffness tensor C [ N/m]: |
| Piezoelectric tensor e [C/m]: | |||
| Dielectric tensor [ F/m]: | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turova, V.; Kovtanyuk, A.; Pykhteev, O.; Sidorenko, I.; Lampe, R. Glycocalyx Sensing with a Mathematical Model of Acoustic Shear Wave Biosensor. Bioengineering 2022, 9, 462. https://doi.org/10.3390/bioengineering9090462
Turova V, Kovtanyuk A, Pykhteev O, Sidorenko I, Lampe R. Glycocalyx Sensing with a Mathematical Model of Acoustic Shear Wave Biosensor. Bioengineering. 2022; 9(9):462. https://doi.org/10.3390/bioengineering9090462
Chicago/Turabian StyleTurova, Varvara, Andrey Kovtanyuk, Oleg Pykhteev, Irina Sidorenko, and Renée Lampe. 2022. "Glycocalyx Sensing with a Mathematical Model of Acoustic Shear Wave Biosensor" Bioengineering 9, no. 9: 462. https://doi.org/10.3390/bioengineering9090462






