Stem Cell Niche Microenvironment: Review
Abstract
1. Introduction
2. Human Cornea
3. Identity and Location of CESCs (Corneal Epithelial Stem Cells)
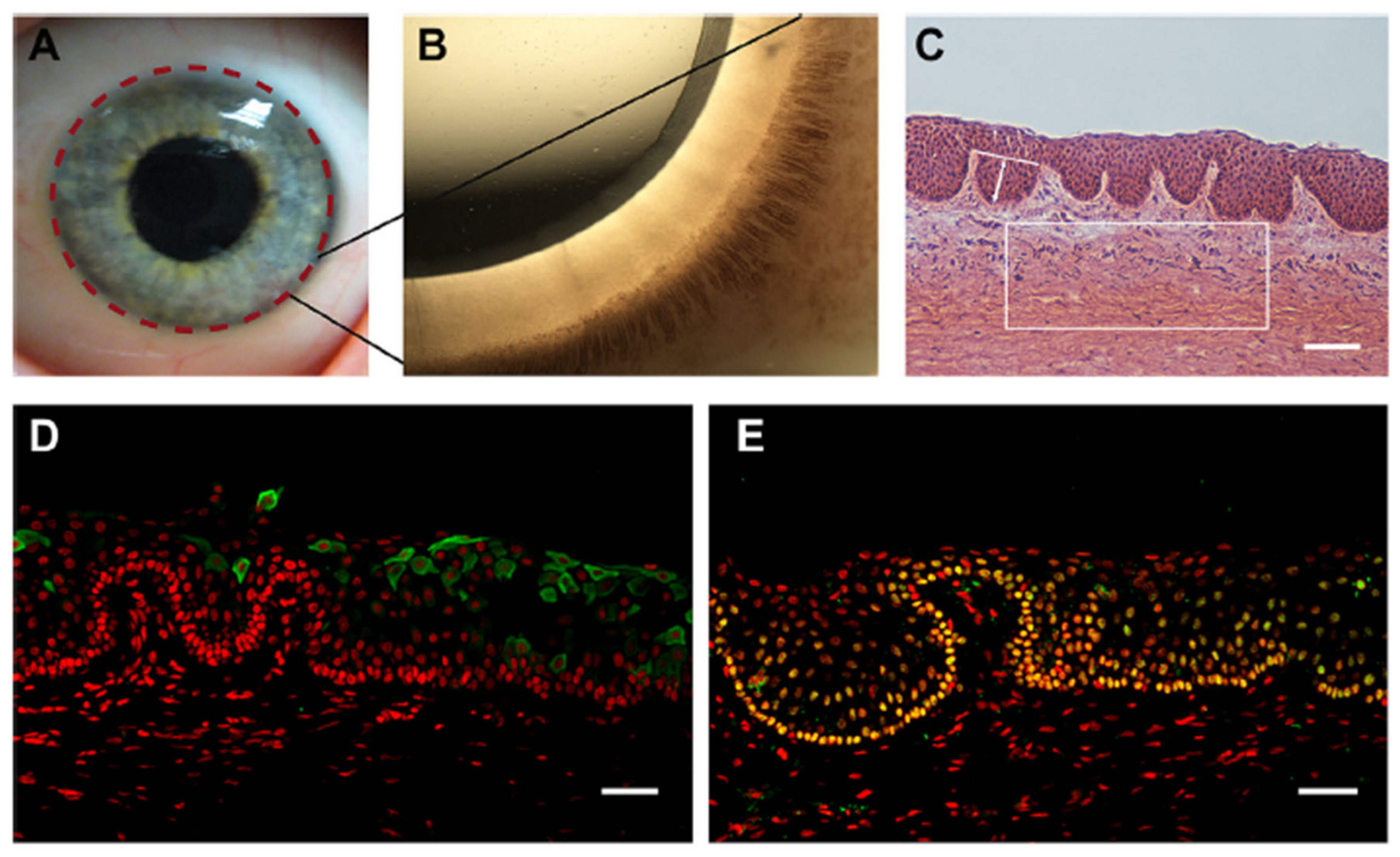
4. The Corneal Stem Cell Niche
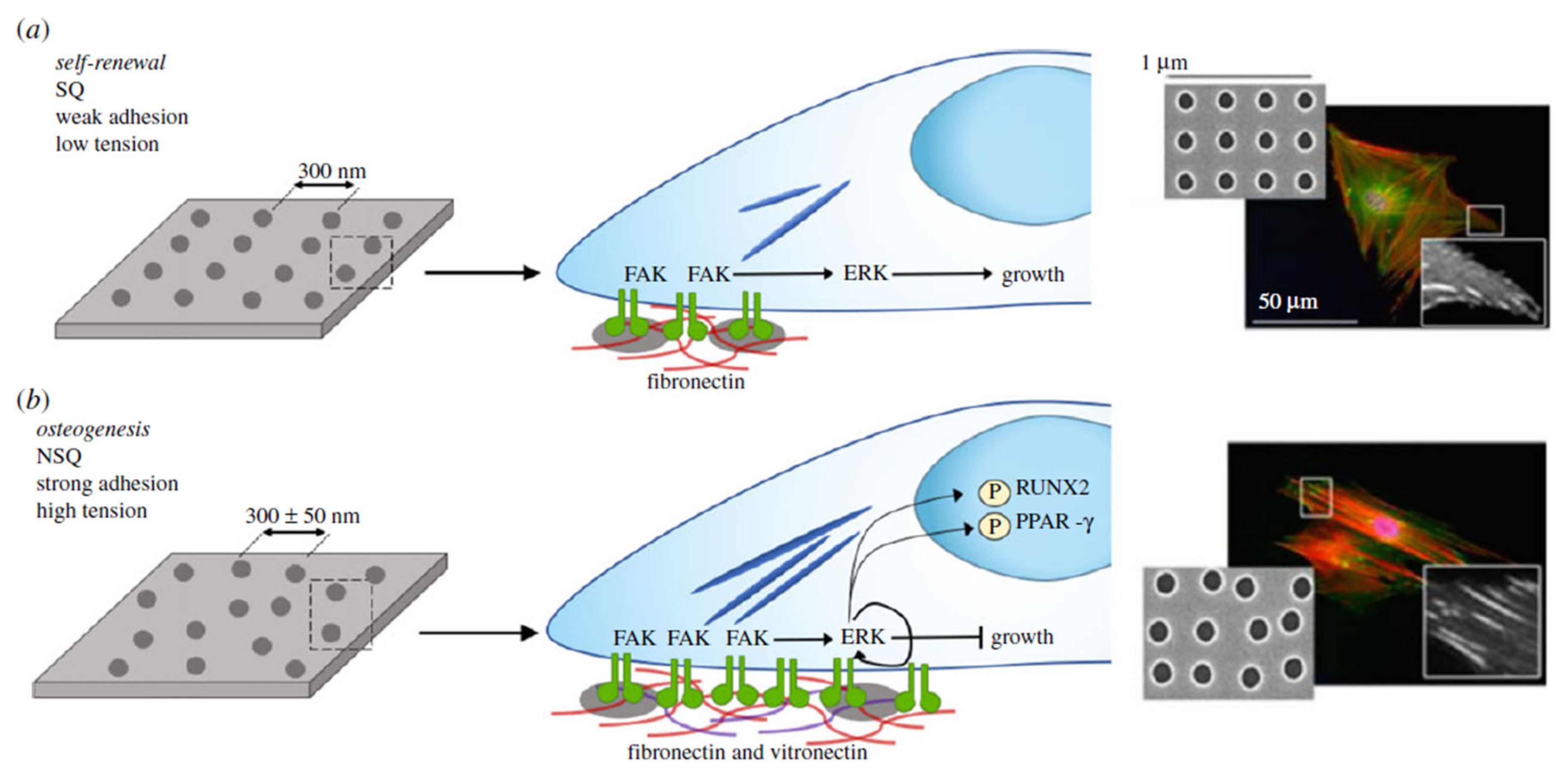
Topography
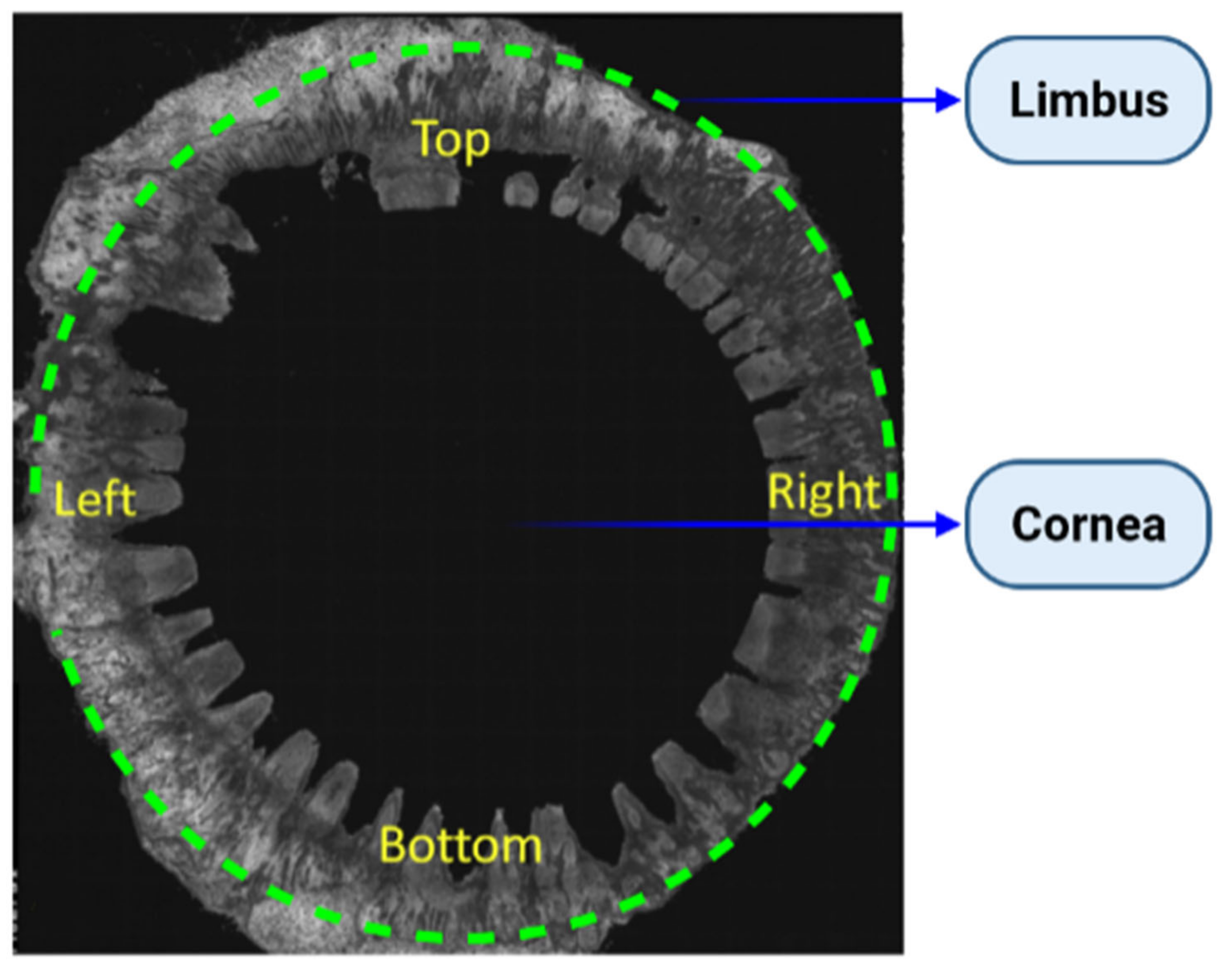
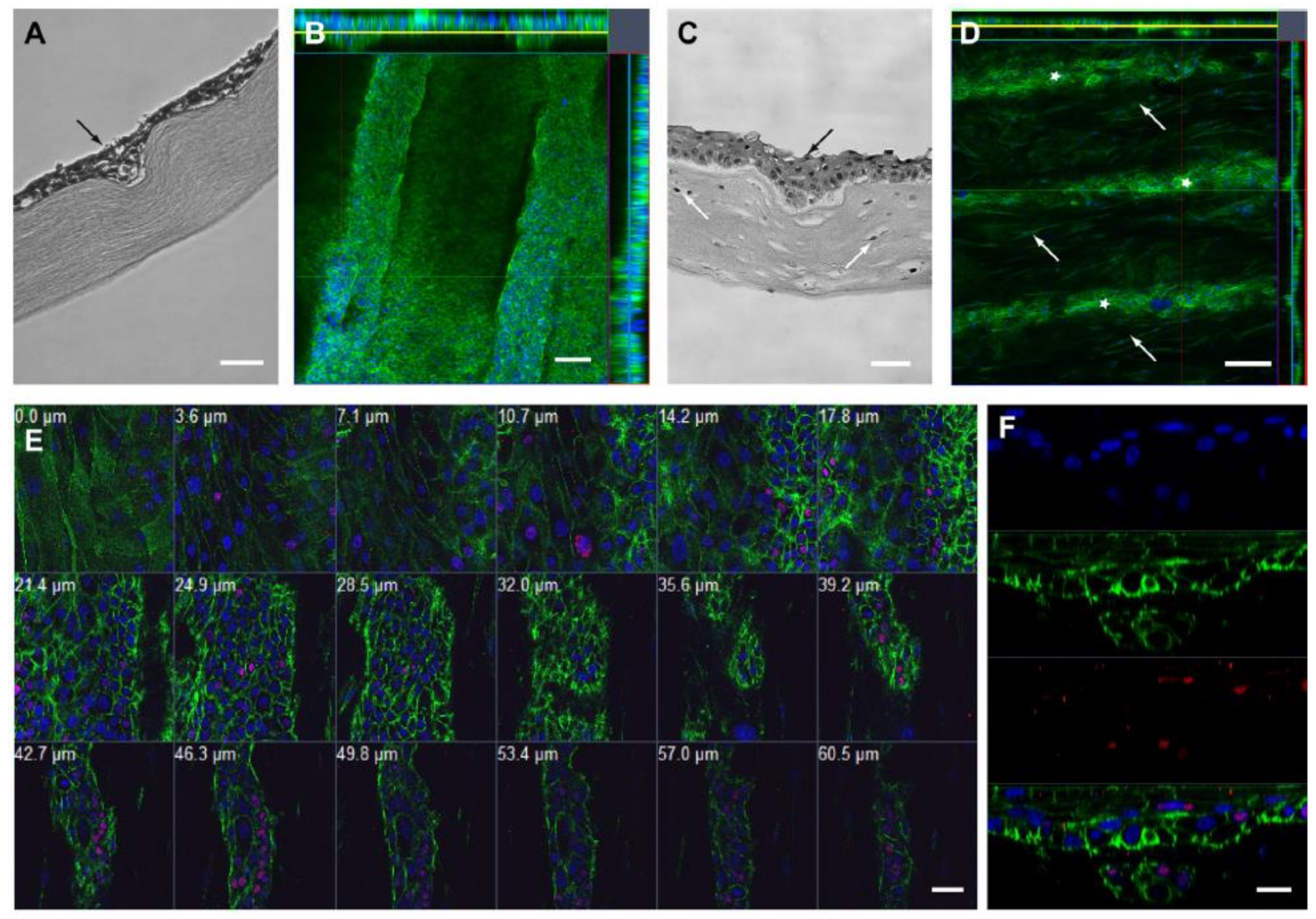
5. The Limbal Stem Cell Niche
Microenvironment Structure
6. The Limbus and Other Stem Cell Niches
7. Reconstruction of the Limbal Stem Cell Niche
7.1. Bio-Active Extracellular Matrix for Limbal Niche Replacement
Fabricating Bio-Active ECMs for Niche Reconstruction
7.2. Cell-Based Approaches for Restoring the Limbal Niche in Mesenchymal Stem Cells
- Plastic conformity.
- Separation of osteocytes, adipocytes and chondrocytes.
- Negative expression of CD14, CD34, CD45 and HLA-DR, and positive expression of CD105, CD73 and CD90.
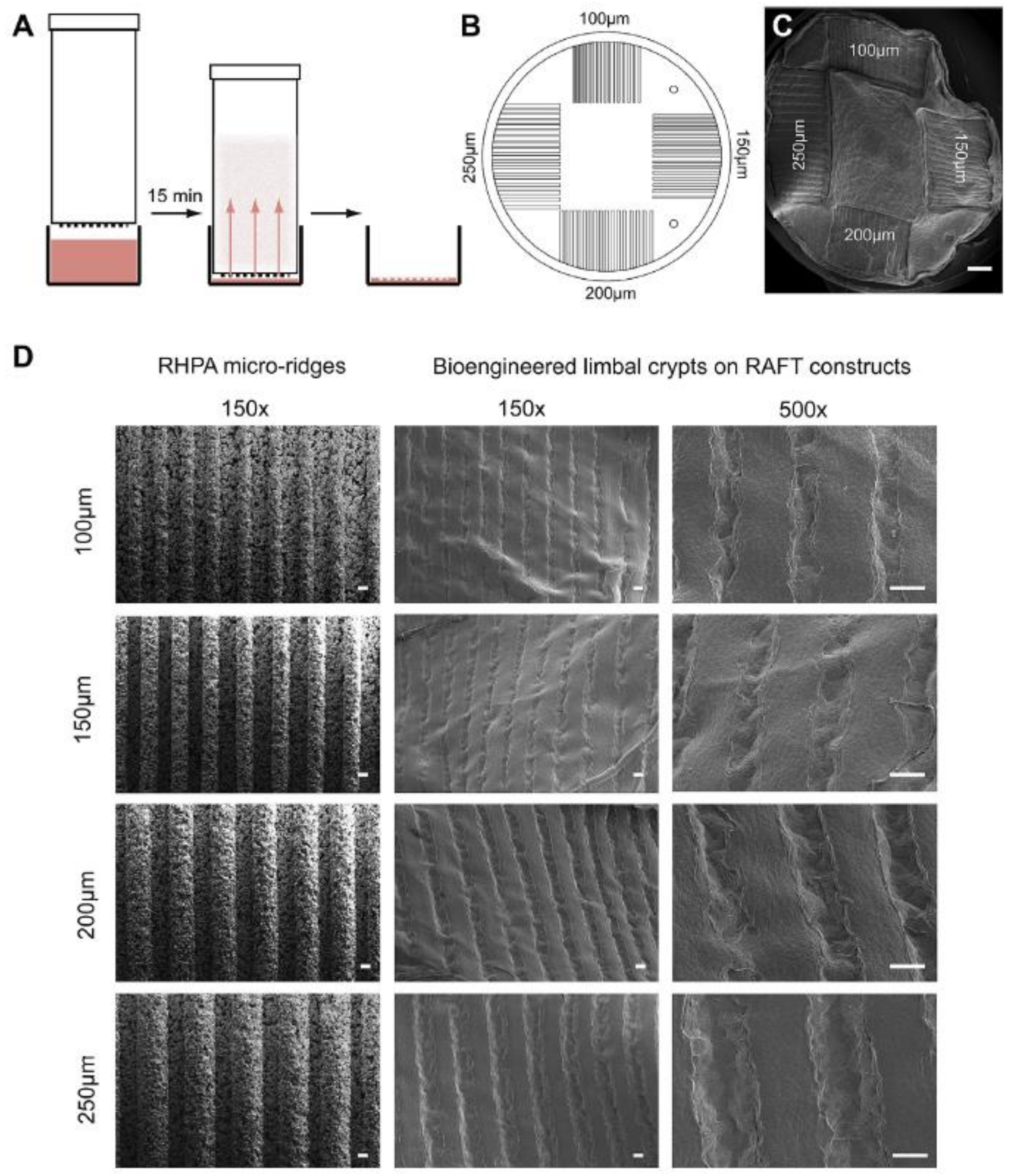
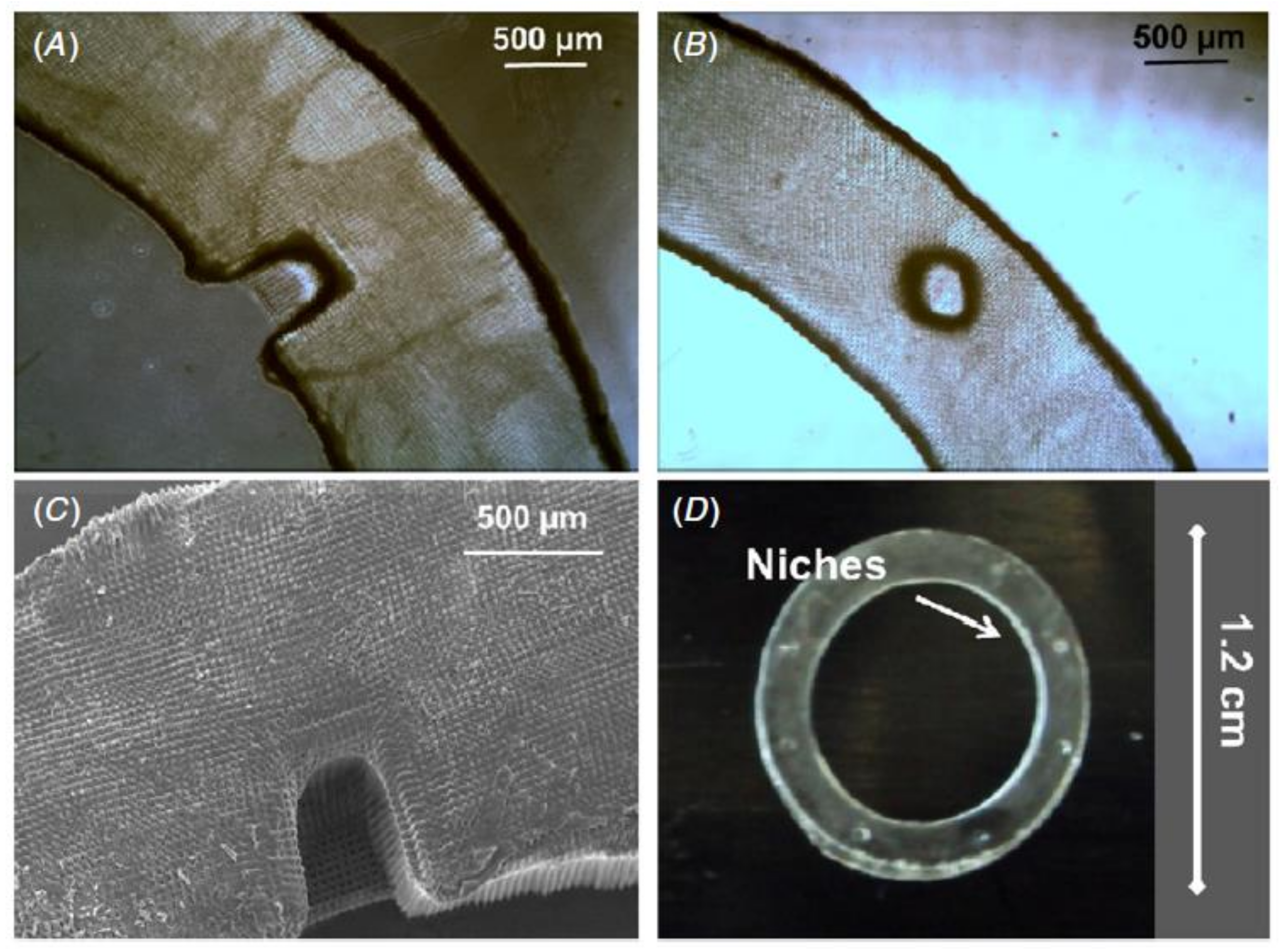
8. Production of BLCs (Bioengineered Limbal Crypts) in RAFT (Real Architecture for 3D Tissue) Constructs
8.1. Characterization of BLCs
8.2. Cell-Filled BLCs
9. Artificial Stem Cell Niches for Self-Renewal
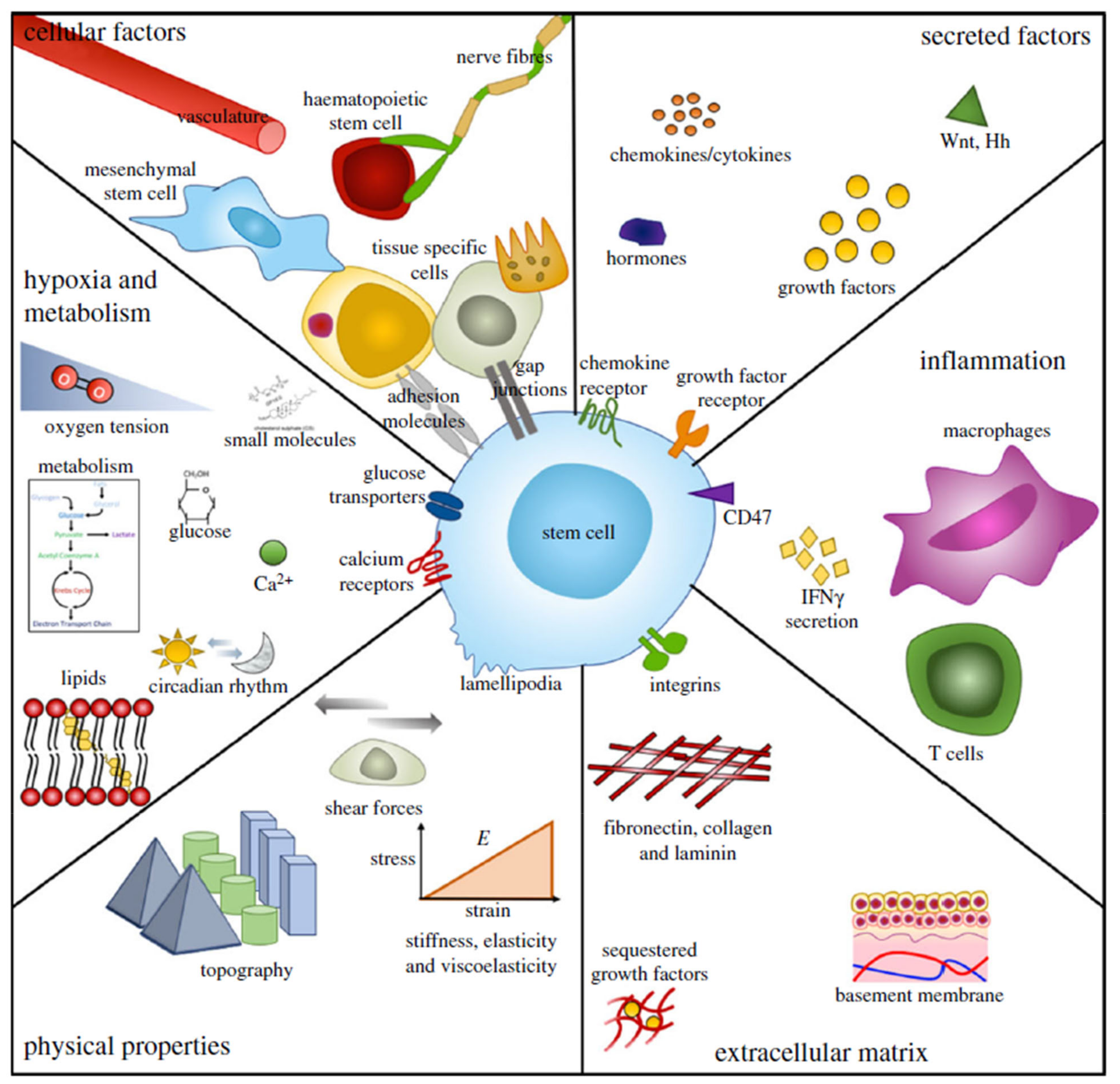
10. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Notara, M.; Alatza, A.; Gilfillan, J.; Harris, A.; Levis, H.; Schrader, S.; Vernon, A.; Daniels, J. In sickness and in health: Corneal epithelial stem cell biology, pathology and therapy. Exp. Eye Res. 2010, 90, 188–195. [Google Scholar] [CrossRef]
- Notara, M.; Shortt, A.; Galatowicz, G.; Calder, V.; Daniels, J.T. IL6 and the human limbal stem cell niche: A mediator of epithelial–stromal interaction. Stem Cell Res. 2010, 5, 188–200. [Google Scholar] [CrossRef]
- Daniels, J.T.; Dart, J.K.G.; Tuft, S.J.; Khaw, P.T. Corneal stem cells in review. Wound Repair. Regen. 2001, 9, 483–494. [Google Scholar] [CrossRef]
- Xuan, M.; Wang, S.; Liu, X.; He, Y.; Li, Y.; Zhang, Y. Proteins of the corneal stroma: Importance in visual function. Cell Tissue Res. 2016, 364, 9–16. [Google Scholar] [CrossRef] [PubMed]
- DelMonte, D.W.; Kim, T. Anatomy and physiology of the cornea. J. Cataract. Refract. Surg. 2011, 37, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Grieve, K.; Ghoubay, D.; Georgeon, C.; Thouvenin, O.; Bouheraoua, N.; Paques, M.; Borderie, V. Three-dimensional structure of the mammalian limbal stem cell niche. Exp. Eye Res. 2015, 140, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Massie, I.; Dziasko, M.; Kureshi, A.; Levis, H.J.; Morgan, L.; Neale, M.; Sheth, R.; Tovell, V.E.; Vernon, A.J.; Funderburgh, J.L. Advanced imaging and tissue engineering of the human limbal epithelial stem cell niche. In Stem Cell Protocols; Humana Press: New York, NY, USA, 2015; pp. 179–202. [Google Scholar]
- Notara, M.; Shortt, A.J.; O’Callaghan, A.R.; Daniels, J.T. The impact of age on the physical and cellular properties of the human limbal stem cell niche. Age 2012, 35, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Nubile, M.; Curcio, C.; Dua, H.S.; Calienno, R.; Lanzini, M.; Iezzi, M.; Mastropasqua, R.; Agnifili, L.; Mastropasqua, L. Pathological changes of the anatomical structure and markers of the limbal stem cell niche due to inflammation. Mol. Vis. 2013, 19, 516–525. [Google Scholar]
- Parfitt, G.J.; Kavianpour, B.; Wu, K.L.; Xie, Y.; Brown, D.J.; Jester, J.V. Immunofluorescence tomography of mouse ocular surface epithelial stem cells and their niche microenvironment. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7338–7344. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, B.E.; Victoria, D.A.; Murillo, G.M.; Herreras, J.M.; Calonge, M. In vivo confocal microscopy assessment of the corneoscleral limbal stem cell niche before and after biopsy for cultivated limbal epithelial transplantation to restore corneal epithelium. Histol. Histopathol. 2015, 30, 183–192. [Google Scholar] [CrossRef]
- Kim, B.Y.; Riaz, K.M.; Bakhtiari, P.; Chan, C.C.; Welder, J.D.; Holland, E.J.; Basti, S.; Djalilian, A.R. Medically reversible limbal stem cell disease: Clinical features and management strategies. Ophthalmology 2014, 121, 2053–2058. [Google Scholar] [CrossRef]
- Notara, M.; Refaian, N.; Braun, G.; Steven, P.; Bock, F.; Cursiefen, C. Short-term uvb-irradiation leads to putative limbal stem cell damage and niche cell-mediated upregulation of macrophage recruiting cytokines. Stem Cell Res. 2015, 15, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Hertsenberg, A.J.; Funderburgh, M.L.; Burrow, M.K.; Mann, M.M.; Du, Y.; Lathrop, K.L.; Syed-Picard, F.N.; Adams, S.M.; Birk, D.E. Human limbal biopsy–derived stromal stem cells prevent corneal scarring. Sci. Transl. Med. 2014, 6, 266ra172. [Google Scholar] [CrossRef]
- Pinnamaneni, N.; Funderburgh, J.L. Concise review: Stem cells in the corneal stroma. Stem Cells 2012, 30, 1059–1063. [Google Scholar] [CrossRef] [PubMed]
- Van Buskirk, E.M. The anatomy of the limbus. Eye 1989, 3, 101–108. [Google Scholar] [CrossRef]
- Schofield, R. The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells 1978, 4, 7–25. [Google Scholar]
- Mcnairn, A.J.; Guasch, G. Epithelial transition zones: Merging microenvironments, niches, and cellular transformation. Eur. J. Dermatol. 2011, 21, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Scadden, D.T. The stem-cell niche as an entity of action. Nature 2006, 441, 1075–1079. [Google Scholar] [CrossRef]
- Davanger, M.; Evensen, A. Role of the pericorneal papillary structure in renewal of corneal epithelium. Nature 1971, 229, 560–561. [Google Scholar] [CrossRef]
- Davis, J.; Duncan, M.K.; Robison Jr, W.G.; Piatigorsky, J. Requirement for Pax6 in corneal morphogenesis: A role in adhesion. J. Cell Sci. 2003, 116, 2157–2167. [Google Scholar] [CrossRef]
- Nakatsu, M.N.; Ding, Z.; Ng, M.Y.; Truong, T.T.; Yu, F.; Deng, S.X. Wnt/β-catenin signaling regulates proliferation of human cornea epithelial stem/progenitor cells. Investig. Ophthalmol. Vis. Sci. 2011, 52, 4734–4741. [Google Scholar] [CrossRef] [PubMed]
- Stepp, M.A.; Zieske, J.D. The corneal epithelial stem cell niche. Ocul. Surf. 2005, 3, 15–26. [Google Scholar] [CrossRef]
- Collinson, J.M.; Chanas, S.A.; Hill, R.E.; West, J.D. Corneal development, limbal stem cell function, and corneal epithelial cell migration in the Pax6+/− mouse. Investig. Ophthalmol. Vis. Sci. 2004, 45, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Kolli, S.; Lako, M.; Figueiredo, F.; Daniels, J.T. Stem cell therapies for ocular surface disease. Drug Discov. Today 2010, 15, 306–313. [Google Scholar] [CrossRef]
- Espana, E.M.; Grueterich, M.; Ti, S.E.; Tseng, S.C. Phenotypic study of a case receiving a keratolimbal allograft and amniotic membrane for total limbal stem cell deficiency. Ophthalmology 2003, 110, 481–486. [Google Scholar] [CrossRef]
- Keivyon, K.R.; Tseng, S.C. Limbal autograft transplantation for ocular surface disorders. Ophthalmology 1989, 96, 709–723. [Google Scholar] [CrossRef]
- Shortt, A.J.; Secker, G.A.; Notara, M.D.; Limb, G.A.; Khaw, P.T.; Tuft, S.J.; Daniels, J.T. Transplantation of ex vivo cultured limbal epithelial stem cells: A review of techniques and clinical results. Surv. Ophthalmol. 2007, 52, 483–502. [Google Scholar] [CrossRef]
- Echevarria, T.J.; Di Girolamo, N. Tissue-regenerating, vision-restoring corneal epithelial stem cells. Stem Cell Rev. Rep. 2011, 7, 256–268. [Google Scholar] [CrossRef]
- Schlötzer-Schrehardt, U.; Dietrich, T.; Saito, K.; Sorokin, L.; Sasaki, T.; Paulsson, M.; Kruse, F. Characterization of extracellular matrix components in the limbal epithelial stem cell compartment. Exp. Eye Res. 2007, 85, 845–860. [Google Scholar] [CrossRef] [PubMed]
- Shortt, A.J.; Secker, G.A.; Munro, P.M.; Khaw, P.T.; Tuft, S.J.; Daniels, J.T. Characterization of the limbal epithelial stem cell niche: Novel imaging techniques permit in vivo observation and targeted biopsy of limbal epithelial stem cells. Stem Cells 2007, 25, 1402–1409. [Google Scholar] [CrossRef]
- Girolamo, N.D.; Sarris, M.; Chui, J.; Cheema, H.; Coroneo, M.T.; Wakefield, D. Localization of the low-affinity nerve growth factor receptor p75 in human limbal epithelial cells. J. Cell Mol. Med. 2008, 12, 2799–2811. [Google Scholar] [CrossRef]
- Dziasko, M.A.; Daniels, J.T. Anatomical features and cell-cell interactions in the human limbal epithelial stem cell niche. Ocul. Surf. 2016, 14, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Cotsarelis, G.; Cheng, S.-Z.; Dong, G.; Sun, T.-T.; Lavker, R.M. Existence of slow-cycling limbal epithelial basal cells that can be preferentially stimulated to proliferate: Implications on epithelial stem cells. Cell 1989, 57, 201–209. [Google Scholar] [CrossRef]
- Daniels, J.T.; Notara, M.; Shortt, A.J.; Secker, G.; Harris, A.; Tuft, S.J. Limbal epithelial stem cell therapy. Expert Opin. Biol. Ther. 2007, 7, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.; Tseng, S. Corneal epithelial wound healing in the absence of limbal epithelium. Investig. Ophthalmol. Vis. Sci. 1991, 32, 96–105. [Google Scholar]
- Dua, H.S.; Azuara-Blanco, A. Limbal stem cells of the corneal epithelium. Surv. Ophthalmol. 2000, 44, 415–425. [Google Scholar] [CrossRef]
- Majo, F.; Rochat, A.; Nicolas, M.; Abou Jaoudé, G.; Barrandon, Y. Oligopotent stem cells are distributed throughout the mammalian ocular surface. Nature 2008, 456, 250–254. [Google Scholar] [CrossRef]
- Levis, H.J.; Massie, I.; Dziasko, M.A.; Kaasi, A.; Daniels, J.T. Rapid tissue engineering of biomimetic human corneal limbal crypts with 3D niche architecture. Biomaterials 2013, 34, 8860–8868. [Google Scholar] [CrossRef]
- Arwert, E.N.; Hoste, E.; Watt, F.M. Epithelial stem cells, wound healing and cancer. Nat. Rev. Cancer 2012, 12, 170–180. [Google Scholar] [CrossRef]
- Tian, H.; Biehs, B.; Warming, S.; Leong, K.G.; Rangell, L.; Klein, O.D.; de Sauvage, F.J. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 2011, 478, 255–259. [Google Scholar] [CrossRef]
- Wilson, A.; Laurenti, E.; Oser, G.; van der Wath, R.C.; Blanco-Bose, W.; Jaworski, M.; Offner, S.; Dunant, C.F.; Eshkind, L.; Bockamp, E. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell 2008, 135, 1118–1129. [Google Scholar] [CrossRef] [PubMed]
- Watt, F.M.; Hogan, B.L. Out of Eden: Stem cells and their niches. Science 2000, 287, 1427–1430. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Van Es, J.H.; Snippert, H.J.; Stange, D.E.; Vries, R.G.; Van Den Born, M.; Barker, N.; Shroyer, N.F.; Van De Wetering, M.; Clevers, H. Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 2011, 469, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Watt, F.M.; Huck, W.T. Role of the extracellular matrix in regulating stem cell fate. Nat. Rev. Mol. Cell Biol. 2013, 14, 467–473. [Google Scholar] [CrossRef]
- Ouyang, H.; Xue, Y.; Lin, Y.; Zhang, X.; Xi, L.; Patel, S.; Cai, H.; Luo, J.; Zhang, M.; Zhang, M. WNT7A and PAX6 define corneal epithelium homeostasis and pathogenesis. Nature 2014, 511, 358–361. [Google Scholar] [CrossRef]
- Clevers, H.; Loh, K.M.; Nusse, R. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science 2014, 346, 6205. [Google Scholar] [CrossRef]
- Aragona, M.; Panciera, T.; Manfrin, A.; Giulitti, S.; Michielin, F.; Elvassore, N.; Dupont, S.; Piccolo, S. A mechanical checkpoint controls multicellular growth through YAP/TAZ regulation by actin-processing factors. Cell 2013, 154, 1047–1059. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.-C.; Li, L.; Fuchs, E. Emerging interactions between skin stem cells and their niches. Nat. Med. 2014, 20, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Nowell, C.S.; Odermatt, P.D.; Azzolin, L.; Hohnel, S.; Wagner, E.F.; Fantner, G.E.; Lutolf, M.P.; Barrandon, Y.; Piccolo, S.; Radtke, F. Chronic inflammation imposes aberrant cell fate in regenerating epithelia through mechanotransduction. Nat. Cell Biol. 2016, 18, 168–180. [Google Scholar] [CrossRef]
- Rovida, E.; Peppicelli, S.; Bono, S.; Bianchini, F.; Tusa, I.; Cheloni, G.; Marzi, I.; Cipolleschi, M.G.; Calorini, L.; Sbarba, P.D. The metabolically-modulated stem cell niche: A dynamic scenario regulating cancer cell phenotype and resistance to therapy. Cell Cycle 2014, 13, 3169–3175. [Google Scholar] [CrossRef]
- Plikus, M.V.; Mayer, J.A.; de La Cruz, D.; Baker, R.E.; Maini, P.K.; Maxson, R.; Chuong, C.-M. Cyclic dermal BMP signalling regulates stem cell activation during hair regeneration. Nature 2008, 451, 340–344. [Google Scholar] [CrossRef]
- Greco, V.; Chen, T.; Rendl, M.; Schober, M.; Pasolli, H.A.; Stokes, N.; dela Cruz-Racelis, J.; Fuchs, E. A two-step mechanism for stem cell activation during hair regeneration. Cell Stem Cell 2009, 4, 155–169. [Google Scholar] [CrossRef]
- Oshimori, N.; Fuchs, E. Paracrine TGF-β signaling counterbalances BMP-mediated repression in hair follicle stem cell activation. Cell Stem Cell 2012, 10, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Al, M.; Zaernia, A.; Sefat, F. Biomaterials for breast reconstruction: Promises, advances, and challenges. J. Tissue Eng. Regen. Med. 2020, 14, 1549–1569. [Google Scholar] [CrossRef]
- Mozafari, M.; Sefat, F. Scaffolds for Bone Tissue Engineering. In Handbook of Tissue Engineering Scaffolds; Mozafari, M., Sefat, F., Atala, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 1. [Google Scholar]
- Zhang, X.; Wang, C.; Liao, M.; Dai, L.; Tang, Y.; Zhang, H.; Coates, P.; Sefat, F.; Zheng, L.; Song, J. Aligned electrospun cellulose scaffolds coated with rhBMP-2 for both in vitro and in vivo bone tissue engineering. Carbohydr. Polym. 2019, 213, 27–38. [Google Scholar] [CrossRef]
- Mohamadi, F.; Ebrahimi-Barough, S.; Nourani, M.R.; Mansoori, K.; Salehi, M.; Alizadeh, A.A.; Tavangar, S.M.; Sefat, F.; Sharifi, S.; Ai, J. Enhanced sciatic nerve regeneration by human endometrial stem cells in an electrospun poly (ε-caprolactone)/collagen/NBG nerve conduit in rat. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1731–1743. [Google Scholar] [CrossRef]
- Hancox, Z.; Yousaf, S.; Khurshid, Z.; Zafar, M.S.; Asencio, I.O.; Youseffi, M.; Mozafari, M.; Sefat, F. Scaffolds for Dental Cementum. In Handbook of Tissue Engineering Scaffolds; Mozafari, M., Sefat, F., Atala, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Zafar, M.; Najeeb, S.; Khurshid, Z.; Vazirzadeh, M.; Zohaib, S.; Najeeb, B.; Sefat, F. Potential of electrospun nanofibers for biomedical and dental applications. Materials 2016, 9, 73. [Google Scholar] [CrossRef]
- Bye, F.J.; Bullock, A.J.; Singh, R.; Sefat, F.; Roman, S.; MacNeil, S. Development of a basement membrane substitute incorporated into an electrospun scaffold for 3D skin tissue engineering. J. Biomater. Tissue Eng. 2014, 4, 686–692. [Google Scholar] [CrossRef]
- Mahjour, S.B.; Fu, X.; Yang, X.; Fong, J.; Sefat, F.; Wang, H. Rapid creation of skin substitutes from human skin cells and biomimetic nanofibers for acute full-thickness wound repair. Burns 2015, 41, 1764–1774. [Google Scholar] [CrossRef]
- Mahjour, S.B.; Sefat, F.; Polunin, Y.; Wang, L.; Wang, H. Improved cell infiltration of electrospun nanofiber mats for layered tissue constructs. J. Biomed. Mater. Res. Part A 2016, 104, 1479–1488. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, P.; Ortega, Í.; Sefat, F.; Sangwan, V.S.; Green, N.; Claeyssens, F.; MacNeil, S. Rocking media over ex vivo corneas improves this model and allows the study of the effect of proinflammatory cytokines on wound healing. Investig. Ophthalmol. Vis. Sci. 2015, 56, 1553–1561. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Deshpande, P.; Ramachandran, C.; Sefat, F.; Mariappan, I.; Johnson, C.; McKean, R.; Hannah, M.; Sangwan, V.S.; Claeyssens, F.; Ryan, A.J. Simplifying corneal surface regeneration using a biodegradable synthetic membrane and limbal tissue explants. Biomaterials 2013, 34, 5088–5106. [Google Scholar] [CrossRef] [PubMed]
- Hancox, Z.; Keshel, S.H.; Yousaf, S.; Saeinasab, M.; Shahbazi, M.-A.; Sefat, F. The progress in corneal translational medicine. Biomater. Sci. 2020, 8, 6469–6504. [Google Scholar] [CrossRef] [PubMed]
- Ortega, Í.; Sefat, F.; Deshpande, P.; Paterson, T.; Ramachandran, C.; Ryan, A.J.; MacNeil, S.; Claeyssens, F. Combination of microstereolithography and electrospinning to produce membranes equipped with niches for corneal regeneration. J. Vis. Exp. 2014, 12, 51826. [Google Scholar] [CrossRef] [PubMed]
- Sefat, F.; McKean, R.; Deshpande, P.; Ramachandran, C.; Hill, C.J.; Sangwan, V.S.; Ryan, A.J.; MacNeil, S. Production, sterilisation and storage of biodegradable electrospun PLGA membranes for delivery of limbal stem cells to the cornea. Procedia Eng. 2013, 59, 101–116. [Google Scholar] [CrossRef]
- Yousaf, S.; Heidari Keshal, S.; Farzi, G.A.; Momeni-Moghadam, M.; Daghigh Ahmadi, E.; Mozafari, M.; Sefat, F. Scaffolds for Intraocular Lens. In Handbook of Tissue Engineering Scaffolds; Mozafari, M., Sefat, F., Atala, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Yousaf, S.; Heidari Keshal, S.; Farzi, G.A.; Momeni-Moghadam, M.; Daghigh Ahmadi, E.; Mozafari, M.; Sefat, F. Scaffolds for Corneal Tissue Engineering. In Handbook of Tissue Engineering Scaffolds; Mozafari, M., Sefat, F., Atala, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Bazgir, M.; Raja, T.; Hancox, Z.; Gentile, P.; Ferreira, A.M.; Zhang, W.; Mozafari, M.; Youseffi, M.; Coates, P.; Sefat, F. Scaffolds for Blood Vessels Tissue Engineering. In Handbook of Tissue Engineering Scaffolds; Mozafari, M., Sefat, F., Atala, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Oluwadamilola, A.; Zare, M.; Mozafari, M.; Youseffi, M.; Twigg, P.; Sefat, F. Scaffolds for Ligament Tissue Engineering. In Handbook of Tissue Engineering Scaffolds; Mozafari, M., Sefat, F., Atala, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Hancox, Z.; Yousaf, S.; Shah, T.; Soon, C.F.; Zhang, W.; Karthik, N.; Ahmed, N.; Mozafari, M.; Sefat, F. Scaffolds for Reconstruction of the Diaphragm. In Handbook of Tissue Engineering Scaffolds; Mozafari, M., Sefat, F., Atala, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Hancox, Z.; Yousaf, S.; Farzi, G.A.; Momeni-Moghadam, M.; Shakeri, S.; Youseffi, M.; Mozafari, M.; Sefat, F. Scaffolds for Tracheal Tissue Engineering. In Handbook of Tissue Engineering Scaffolds; Mozafari, M., Sefat, F., Atala, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Raja, T.I.; Mozafari, M.; Milan, P.B.; Samadikuchaksaraei, A.; Sefat, F. Nanoengineered biomaterials for tracheal replacement. In Nanoengineered Biomaterials for Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2019; pp. 285–303. [Google Scholar]
- Moztarzadeh, S.; Mottaghy, K.; Sefat, F.; Samadikuchaksaraei, A.; Mozafari, M. Nanoengineered biomaterials for lung regeneration. In Nanoengineered Biomaterials for Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2019; pp. 305–323. [Google Scholar]
- Sefat, F.; Raja, T.I.; Zafar, M.S.; Khurshid, Z.; Najeeb, S.; Zohaib, S.; Ahmadi, E.D.; Rahmati, M.; Mozafari, M. Nanoengineered biomaterials for cartilage repair. In Nanoengineered Biomaterials for Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2019; pp. 39–71. [Google Scholar]
- Sefat, F.; Raja, T.I.; Moghadam, Z.S.; Milan, P.B.; Samadikuchaksaraei, A.; Mozafari, M. Nanoengineered biomaterials for bladder regeneration. In Nanoengineered Biomaterials for Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2019; pp. 459–474. [Google Scholar]
- Urbanska, A.M.; Sefat, F.; Yousaf, S.; Kargozar, S.; Milan, P.B.; Mozafari, M. Nanoengineered biomaterials for intestine regeneration. In Nanoengineered Biomaterials for Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2019; pp. 363–378. [Google Scholar]
- Curtis, A.; Varde, M. Control of cell behavior: Topological factors. J. Natl. Cancer Inst. 1964, 33, 15–26. [Google Scholar]
- Dalby, M.J.; Gadegaard, N.; Oreffo, R.O. Harnessing nanotopography and integrin–matrix interactions to influence stem cell fate. Nat. Mater. 2014, 13, 558–569. [Google Scholar] [CrossRef]
- Kasemo, B.; Lausmaa, J. Biomaterial and implant surfaces: A surface science approach. Int. J. Oral Maxillofac. Implant. 1988, 3, 45–68. [Google Scholar]
- Keselowsky, B.G.; Collard, D.M.; García, A.J. Surface chemistry modulates fibronectin conformation and directs integrin binding and specificity to control cell adhesion. J. Biomed. Mater. Res. Part A 2003, 66, 247–259. [Google Scholar] [CrossRef]
- Pierschbacher, M.D.; Ruoslahti, E. Cell attachment activity of fibronectin can be duplicated by small synthetic fragments of the molecule. Nature 1984, 309, 30–33. [Google Scholar] [CrossRef]
- Kilian, K.A.; Bugarija, B.; Lahn, B.T.; Mrksich, M. Geometric cues for directing the differentiation of mesenchymal stem cells. Proc. Natl. Acad. Sci. USA 2010, 107, 4872–4877. [Google Scholar] [CrossRef] [PubMed]
- McBeath, R.; Pirone, D.M.; Nelson, C.M.; Bhadriraju, K.; Chen, C.S. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev. Cell 2004, 6, 483–495. [Google Scholar] [CrossRef]
- Dalby, M.; Riehle, M.; Johnstone, H.; Affrossman, S.; Curtis, A. Investigating the limits of filopodial sensing: A brief report using SEM to image the interaction between 10 nm high nano-topography and fibroblast filopodia. Cell Biol. Int. 2004, 28, 229–236. [Google Scholar] [CrossRef]
- McNamara, L.E.; Sjöström, T.; Seunarine, K.; Meek, R.D.; Su, B.; Dalby, M.J. Investigation of the limits of nanoscale filopodial interactions. J. Tissue Eng. 2014, 5, 2041731414536177. [Google Scholar] [CrossRef]
- Watari, S.; Hayashi, K.; Wood, J.A.; Russell, P.; Nealey, P.F.; Murphy, C.J.; Genetos, D.C. Modulation of osteogenic differentiation in hMSCs cells by submicron topographically-patterned ridges and grooves. Biomaterials 2012, 33, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Dalby, M.J.; Gadegaard, N.; Tare, R.; Andar, A.; Riehle, M.O.; Herzyk, P.; Wilkinson, C.D.; Oreffo, R.O. The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat. Mater. 2007, 6, 997–1003. [Google Scholar] [CrossRef]
- McMurray, R.J.; Gadegaard, N.; Tsimbouri, P.M.; Burgess, K.V.; McNamara, L.E.; Tare, R.; Murawski, K.; Kingham, E.; Oreffo, R.O.; Dalby, M.J. Nanoscale surfaces for the long-term maintenance of mesenchymal stem cell phenotype and multipotency. Nat. Mater. 2011, 10, 637–644. [Google Scholar] [CrossRef]
- Tsimbouri, P.M.; McMurray, R.J.; Burgess, K.V.; Alakpa, E.V.; Reynolds, P.M.; Murawski, K.; Kingham, E.; Oreffo, R.O.; Gadegaard, N.; Dalby, M.J. Using nanotopography and metabolomics to identify biochemical effectors of multipotency. ACS Nano 2012, 6, 10239–10249. [Google Scholar] [CrossRef] [PubMed]
- Bucaro, M.A.; Vasquez, Y.; Hatton, B.D.; Aizenberg, J. Fine-tuning the degree of stem cell polarization and alignment on ordered arrays of high-aspect-ratio nanopillars. ACS Nano 2012, 6, 6222–6230. [Google Scholar] [CrossRef]
- Biggs, M.J.; Richards, R.G.; Gadegaard, N.; Wilkinson, C.D.; Oreffo, R.O.; Dalby, M.J. The use of nanoscale topography to modulate the dynamics of adhesion formation in primary osteoblasts and ERK/MAPK signalling in STRO-1+ enriched skeletal stem cells. Biomaterials 2009, 30, 5094–5103. [Google Scholar] [CrossRef]
- Elosegui-Artola, A.; Andreu, I.; Beedle, A.E.; Lezamiz, A.; Uroz, M.; Kosmalska, A.J.; Oria, R.; Kechagia, J.Z.; Rico-Lastres, P.; Le Roux, A.-L. Force triggers YAP nuclear entry by regulating transport across nuclear pores. Cell 2017, 171, 1397–1410.e1314. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Tytell, J.D.; Ingber, D.E. Mechanotransduction at a distance: Mechanically coupling the extracellular matrix with the nucleus. Nat. Rev. Mol. Cell Biol. 2009, 10, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Dalby, M.J.; Biggs, M.J.; Gadegaard, N.; Kalna, G.; Wilkinson, C.D.; Curtis, A.S. Nanotopographical stimulation of mechanotransduction and changes in interphase centromere positioning. J. Cell. Biochem. 2007, 100, 326–338. [Google Scholar] [CrossRef]
- Dalby, M.J.; Gadegaard, N.; Herzyk, P.; Sutherland, D.; Agheli, H.; Wilkinson, C.D.; Curtis, A.S. Nanomechanotransduction and interphase nuclear organization influence on genomic control. J. Cell. Biochem. 2007, 102, 1234–1244. [Google Scholar] [CrossRef]
- McNamara, L.E.; Burchmore, R.; Riehle, M.O.; Herzyk, P.; Biggs, M.J.; Wilkinson, C.D.; Curtis, A.S.; Dalby, M.J. The role of microtopography in cellular mechanotransduction. Biomaterials 2012, 33, 2835–2847. [Google Scholar] [CrossRef]
- Tsimbouri, P.M.; Murawski, K.; Hamilton, G.; Herzyk, P.; Oreffo, R.O.; Gadegaard, N.; Dalby, M.J. A genomics approach in determining nanotopographical effects on MSC phenotype. Biomaterials 2013, 34, 2177–2184. [Google Scholar] [CrossRef]
- Ngandu Mpoyi, E.; Cantini, M.; Reynolds, P.M.; Gadegaard, N.; Dalby, M.J.; Salmerón-Sánchez, M. Protein adsorption as a key mediator in the nanotopographical control of cell behavior. ACS Nano 2016, 10, 6638–6647. [Google Scholar] [CrossRef]
- Wood, M.A.; Bagnaninchi, P.; Dalby, M.J. The β integrins and cytoskeletal nanoimprinting. Exp. Cell Res. 2008, 314, 927–935. [Google Scholar] [CrossRef]
- Chen, C.S.; Mrksich, M.; Huang, S.; Whitesides, G.M.; Ingber, D.E. Geometric control of cell life and death. Science 1997, 276, 1425–1428. [Google Scholar] [CrossRef]
- Von Erlach, T.C.; Bertazzo, S.; Wozniak, M.A.; Horejs, C.-M.; Maynard, S.A.; Attwood, S.; Robinson, B.K.; Autefage, H.; Kallepitis, C.; del Río Hernández, A. Cell-geometry-dependent changes in plasma membrane order direct stem cell signalling and fate. Nat. Mater. 2018, 17, 237–242. [Google Scholar] [CrossRef]
- Cavalcanti-Adam, E.A.; Volberg, T.; Micoulet, A.; Kessler, H.; Geiger, B.; Spatz, J.P. Cell spreading and focal adhesion dynamics are regulated by spacing of integrin ligands. Biophys. J. 2007, 92, 2964–2974. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti-Adam, E.A.; Aydin, D.; Hirschfeld-Warneken, V.C.; Spatz, J.P. Cell adhesion and response to synthetic nanopatterned environments by steering receptor clustering and spatial location. HFSP J. 2008, 2, 276–285. [Google Scholar] [CrossRef]
- Schvartzman, M.; Palma, M.; Sable, J.; Abramson, J.; Hu, X.; Sheetz, M.P.; Wind, S.J. Nanolithographic control of the spatial organization of cellular adhesion receptors at the single-molecule level. Nano Lett. 2011, 11, 1306–1312. [Google Scholar] [CrossRef]
- Yang, J.; McNamara, L.E.; Gadegaard, N.; Alakpa, E.V.; Burgess, K.V.; Meek, R.D.; Dalby, M.J. Nanotopographical induction of osteogenesis through adhesion, bone morphogenic protein cosignaling, and regulation of microRNAs. ACS Nano 2014, 8, 9941–9953. [Google Scholar] [CrossRef]
- Malmstroöm, J.; Lovmand, J.; Kristensen, S.; Sundh, M.; Duch, M.; Sutherland, D.S. Focal complex maturation and bridging on 200 nm vitronectin but not fibronectin patches reveal different mechanisms of focal adhesion formation. Nano Lett. 2011, 11, 2264–2271. [Google Scholar] [CrossRef]
- Eberwein, P.; Reinhard, T. Concise reviews: The role of biomechanics in the limbal stem cell niche: New insights for our understanding of this structure. Stem Cells 2015, 33, 916–924. [Google Scholar] [CrossRef]
- Rama, P.; Matuska, S.; Paganoni, G.; Spinelli, A.; De Luca, M.; Pellegrini, G. Limbal stem-cell therapy and long-term corneal regeneration. N. Engl. J. Med. 2010, 363, 147–155. [Google Scholar] [CrossRef]
- Dua, H.S.; Shanmuganathan, V.; Powell-Richards, A.; Tighe, P.; Joseph, A. Limbal epithelial crypts: A novel anatomical structure and a putative limbal stem cell niche. Br. J. Ophthalmol. 2005, 89, 529–532. [Google Scholar] [CrossRef]
- Goldberg, M.F.; Bron, A.J. Limbal palisades of Vogt. Trans. Am. Ophthalmol. Soc. 1982, 80, 155. [Google Scholar] [PubMed]
- Townsend, W.M. The limbal palisades of Vogt. Trans. Am. Ophthalmol. Soc. 1991, 89, 721. [Google Scholar]
- Morrison, S.J.; Shah, N.M.; Anderson, D.J. Regulatory mechanisms in stem cell biology. Cell 1997, 88, 287–298. [Google Scholar] [CrossRef]
- Schlötzer-Schrehardt, U.; Kruse, F.E. Identification and characterization of limbal stem cells. Exp. Eye Res. 2005, 81, 247–264. [Google Scholar] [CrossRef] [PubMed]
- Tseng, S.C. Regulation and clinical implications of corneal epithelial stem cells. Mol. Biol. Rep. 1996, 23, 47–58. [Google Scholar] [CrossRef]
- Dziasko, M.A.; Armer, H.E.; Levis, H.J.; Shortt, A.J.; Tuft, S.; Daniels, J.T. Localisation of epithelial cells capable of holoclone formation in vitro and direct interaction with stromal cells in the native human limbal crypt. PLoS ONE 2014, 9, e94283. [Google Scholar] [CrossRef]
- Lathrop, K.L.; Gupta, D.; Kagemann, L.; Schuman, J.S.; SundarRaj, N. Optical coherence tomography as a rapid, accurate, noncontact method of visualizing the palisades of Vogt. Investig. Ophthalmol. Vis. Sci. 2012, 53, 1381–1387. [Google Scholar] [CrossRef]
- Miri, A.; Al-Aqaba, M.; Otri, A.M.; Fares, U.; Said, D.G.; Faraj, L.A.; Dua, H.S. In vivo confocal microscopic features of normal limbus. Br. J. Ophthalmol. 2012, 96, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Molvær, R.K.; Andreasen, A.; Heegaard, S.; Thomsen, J.S.; Hjortdal, J.; Urbak, S.F.; Nielsen, K. Interactive 3D computer model of the human corneolimbal region: Crypts, projections and stem cells. Acta Ophthalmol. 2013, 91, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Shanmuganathan, V.A.; Foster, T.; Kulkarni, B.B.; Hopkinson, A.; Gray, T.; Powe, D.G.; Lowe, J.; Dua, H.S. Morphological characteristics of the limbal epithelial crypt. Br. J. Ophthalmol. 2007, 91, 514–519. [Google Scholar] [CrossRef][Green Version]
- Bizheva, K.; Hutchings, N.; Sorbara, L.; Moayed, A.A.; Simpson, T. In vivo volumetric imaging of the human corneo-scleral limbus with spectral domain OCT. Biomed. Opt. Express 2011, 2, 1794–1802. [Google Scholar] [CrossRef]
- Yeung, A.M.-H.; Schlötzer-Schrehardt, U.; Kulkarni, B.; Tint, N.L.; Hopkinson, A.; Dua, H.S. Limbal epithelial crypt: A model for corneal epithelial maintenance and novel limbal regional variations. Arch. Ophthalmol. 2008, 126, 665–669. [Google Scholar] [CrossRef][Green Version]
- Deng, S.X.; Sejpal, K.D.; Tang, Q.; Aldave, A.J.; Lee, O.L.; Yu, F. Characterization of limbal stem cell deficiency by in vivo laser scanning confocal microscopy: A microstructural approach. Arch. Ophthalmol. 2012, 130, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, B.B.; Tighe, P.J.; Mohammed, I.; Yeung, A.M.; Powe, D.G.; Hopkinson, A.; Shanmuganathan, V.A.; Dua, H.S. Comparative transcriptional profiling of the limbal epithelial crypt demonstrates its putative stem cell niche characteristics. BMC Genom. 2010, 11, 1–18. [Google Scholar] [CrossRef]
- Polisetti, N.; Zenkel, M.; Menzel-Severing, J.; Kruse, F.E.; Schlötzer-Schrehardt, U. Cell adhesion molecules and stem cell-niche-interactions in the limbal stem cell niche. Stem Cells 2016, 34, 203–219. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Young, R.D.; Lewis, P.N.; Shinomiya, K.; Meek, K.M.; Kinoshita, S.; Caterson, B.; Quantock, A.J. Mesenchymal–epithelial cell interactions and proteoglycan matrix composition in the presumptive stem cell niche of the rabbit corneal limbus. Mol. Vis. 2015, 21, 1328. [Google Scholar] [PubMed]
- Huang, M.; Wang, B.; Wan, P.; Liang, X.; Wang, X.; Liu, Y.; Zhou, Q.; Wang, Z. Roles of limbal microvascular net and limbal stroma in regulating maintenance of limbal epithelial stem cells. Cell Tissue Res. 2015, 359, 547–563. [Google Scholar] [CrossRef]
- Katikireddy, K.R.; Jurkunas, U.V. Limbal stromal tissue specific stem cells and their differentiation potential to corneal epithelial cells. Methods Mol. Biol. 2016, 437–444. [Google Scholar] [CrossRef]
- Higa, K.; Shimmura, S.; Miyashita, H.; Shimazaki, J.; Tsubota, K. Melanocytes in the corneal limbus interact with K19-positive basal epithelial cells. Exp. Eye Res. 2005, 81, 218–223. [Google Scholar] [CrossRef]
- Chidambaranathan, G.P.; Mathews, S.; Panigrahi, A.K.; Mascarenhas, J.; Prajna, N.V.; Muthukkaruppan, V. In vivo confocal microscopic analysis of limbal stroma in patients with limbal stem cell deficiency. Cornea 2015, 34, 1478–1486. [Google Scholar] [CrossRef]
- Mathews, S.; Chidambaram, J.D.; Lanjewar, S.; Mascarenhas, J.; Prajna, N.V.; Muthukkaruppan, V.; Chidambaranathan, G.P. In vivo confocal microscopic analysis of normal human anterior limbal stroma. Cornea 2015, 34, 464. [Google Scholar] [CrossRef] [PubMed]
- Higa, K.; Kato, N.; Yoshida, S.; Ogawa, Y.; Shimazaki, J.; Tsubota, K.; Shimmura, S. Aquaporin 1-positive stromal niche-like cells directly interact with N-cadherin-positive clusters in the basal limbal epithelium. Stem Cell Res. 2013, 10, 147–155. [Google Scholar] [CrossRef]
- Xie, H.T.; Chen, S.Y.; Li, G.G.; Tseng, S.C. Limbal epithelial stem/progenitor cells attract stromal niche cells by SDF-1/CXCR4 signaling to prevent differentiation. Stem Cells 2011, 29, 1874–1885. [Google Scholar] [CrossRef]
- Han, B.; Chen, S.-Y.; Zhu, Y.-T.; Tseng, S.C. Integration of BMP/Wnt signaling to control clonal growth of limbal epithelial progenitor cells by niche cells. Stem Cell Res. 2014, 12, 562–573. [Google Scholar] [CrossRef]
- Chen, S.-Y.; Hayashida, Y.; Chen, M.-Y.; Xie, H.T.; Tseng, S.C. A new isolation method of human limbal progenitor cells by maintaining close association with their niche cells. Tissue Eng. Part C Methods 2011, 17, 537–548. [Google Scholar] [CrossRef]
- González, S.; Deng, S.X. Presence of native limbal stromal cells increases the expansion efficiency of limbal stem/progenitor cells in culture. Exp. Eye Res. 2013, 116, 169–176. [Google Scholar] [CrossRef]
- Polisetti, N.; Agarwal, P.; Khan, I.; Kondaiah, P.; Sangwan, V.S.; Vemuganti, G.K. Gene expression profile of epithelial cells and mesenchymal cells derived from limbal explant culture. Mol. Vis. 2010, 16, 1227. [Google Scholar] [PubMed]
- Mathan, J.J.; Ismail, S.; McGhee, J.J.; McGhee, C.N.J.; Sherwin, T. Sphere-forming cells from peripheral cornea demonstrate the ability to repopulate the ocular surface. Stem Cell Res. Ther. 2016, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.L.; Wagers, A.J. No place like home: Anatomy and function of the stem cell niche. Nat. Rev. Mol. Cell Biol. 2008, 9, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Morris, R.; Potten, C.S. Slowly cycling (label-retaining) epidermal cells behave like clonogenic stem cells in vitro. Cell Prolif. 1994, 27, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Niu, C.; Ye, L.; Huang, H.; He, X.; Tong, W.-G.; Ross, J.; Haug, J.; Johnson, T.; Feng, J.Q. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 2003, 425, 836–841. [Google Scholar] [CrossRef]
- Calvi, L.; Adams, G.; Weibrecht, K.; Weber, J.; Olson, D.; Knight, M.; Martin, R.; Schipani, E.; Divieti, P.; Bringhurst, F.R. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 2003, 425, 841–846. [Google Scholar] [CrossRef]
- Adams, G.B.; Chabner, K.T.; Alley, I.R.; Olson, D.P.; Szczepiorkowski, Z.M.; Poznansky, M.C.; Kos, C.H.; Pollak, M.R.; Brown, E.M.; Scadden, D.T. Stem cell engraftment at the endosteal niche is specified by the calcium-sensing receptor. Nature 2006, 439, 599–603. [Google Scholar] [CrossRef]
- Hsu, H.-C.; Ema, H.; Osawa, M.; Nakamura, Y.; Suda, T.; Nakauchi, H. Hematopoietic stem cells express Tie-2 receptor in the murine fetal liver. Blood 2000, 96, 3757–3762. [Google Scholar] [CrossRef]
- Thoft, R.A.; Friend, J. The X, Y, Z hypothesis of corneal epithelial maintenance. Investig. Ophthalmol. Vis. Sci. 1983, 24, 1442–1443. [Google Scholar]
- Mort, R.L.; Ramaesh, T.; Kleinjan, D.A.; Morley, S.D.; West, J.D. Mosaic analysis of stem cell function and wound healing in the mouse corneal epithelium. BMC Dev. Biol. 2009, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Sureka, S.P.; Shanbhag, S.S.; Kethiri, A.R.; Singh, V.; Sangwan, V.S. Simple limbal epithelial transplantation: Long-term clinical outcomes in 125 cases of unilateral chronic ocular surface burns. Ophthalmology 2016, 123, 1000–1010. [Google Scholar] [CrossRef]
- Djalilian, A.R.; Mahesh, S.P.; Koch, C.A.; Nussenblatt, R.B.; Shen, D.; Zhuang, Z.; Holland, E.J.; Chan, C.-C. Survival of donor epithelial cells after limbal stem cell transplantation. Investig. Ophthalmol. Vis. Sci. 2005, 46, 803–807. [Google Scholar] [CrossRef] [PubMed]
- Ganger, A.; Vanathi, M.; Mohanty, S.; Tandon, R. Long-term outcomes of cultivated limbal epithelial transplantation: Evaluation and comparison of results in children and adults. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef]
- Shen, C.; Chan, C.C.; Holland, E.J. Limbal Stem Cell Transplantation for Soft Contact Lens Wear–Related Limbal Stem Cell Deficiency. Am. J. Ophthalmol. 2015, 160, 1142–1149.e1141. [Google Scholar] [CrossRef]
- Vazirani, J.; Ali, M.H.; Sharma, N.; Gupta, N.; Mittal, V.; Atallah, M.; Amescua, G.; Chowdhury, T.; Abdala-Figuerola, A.; Ramirez-Miranda, A. Autologous simple limbal epithelial transplantation for unilateral limbal stem cell deficiency: Multicentre results. Br. J. Ophthalmol. 2016, 100, 1416–1420. [Google Scholar] [CrossRef]
- Zakaria, N.; Possemiers, T.; Dhubhghaill, S.N.; Leysen, I.; Rozema, J.; Koppen, C.; Timmermans, J.-P.; Berneman, Z.; Tassignon, M.-J. Results of a phase I/II clinical trial: Standardized, non-xenogenic, cultivated limbal stem cell transplantation. J. Transl. Med. 2014, 12, 1–13. [Google Scholar] [CrossRef]
- Levis, H.J.; Daniels, J.T. Recreating the human limbal epithelial stem cell niche with bioengineered limbal crypts. Curr. Eye Res. 2016, 41, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Dehghani, S.; Rasoulianboroujeni, M.; Ghasemi, H.; Keshel, S.H.; Nozarian, Z.; Hashemian, M.N.; Zarei-Ghanavati, M.; Latifi, G.; Ghaffari, R.; Cui, Z. 3D-Printed membrane as an alternative to amniotic membrane for ocular surface/conjunctival defect reconstruction: An in vitro & in vivo study. Biomaterials 2018, 174, 95–112. [Google Scholar] [CrossRef]
- Sorkio, A.; Koch, L.; Koivusalo, L.; Deiwick, A.; Miettinen, S.; Chichkov, B.; Skottman, H. Human stem cell based corneal tissue mimicking structures using laser-assisted 3D bioprinting and functional bioinks. Biomaterials 2018, 171, 57–71. [Google Scholar] [CrossRef]
- Shafiq, M.A.; Gemeinhart, R.A.; Yue, B.Y.; Djalilian, A.R. Decellularized human cornea for reconstructing the corneal epithelium and anterior stroma. Tissue Eng. Part C Methods 2012, 18, 340–348. [Google Scholar] [CrossRef]
- Yoeruek, E.; Bayyoud, T.; Maurus, C.; Hofmann, J.; Spitzer, M.S.; Bartz-Schmidt, K.U.; Szurman, P. Decellularization of porcine corneas and repopulation with human corneal cells for tissue-engineered xenografts. Acta Ophthalmol. 2012, 90, e125–e131. [Google Scholar] [CrossRef]
- Lynch, A.P.; Ahearne, M. Strategies for developing decellularized corneal scaffolds. Exp. Eye Res. 2013, 108, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Xie, L. Focus on the clinical application of the first artificial bioengineered cornea in China. Chin. J. Ophthalmol. 2016, 52, 161–163. [Google Scholar] [CrossRef]
- Shi, Y.; Bikkuzin, T.; Song, Z.; Jin, X.; Jin, H.; Li, X.; Zhang, H. Comprehensive evaluation of decellularized porcine corneal after clinical transplantation. Xenotransplantation 2017, 24, e12338. [Google Scholar] [CrossRef] [PubMed]
- Ahearne, M.; Lynch, A.P. Early observation of extracellular matrix-derived hydrogels for corneal stroma regeneration. Tissue Eng. Part C Methods 2015, 21, 1059–1069. [Google Scholar] [CrossRef]
- Lu, Y.; Yao, Q.K.; Feng, B.; Yan, C.X.; Zhu, M.Y.; Chen, J.Z.; Fu, W.; Fu, Y. Characterization of a hydrogel derived from decellularized corneal extracellular matrix. J. Biomater. Tissue Eng. 2015, 5, 951–960. [Google Scholar] [CrossRef]
- Friedenstein, A.J.; Petrakova, K.V.; Kurolesova, A.I.; Frolova, G.P. Heterotopic transplants of bone marrow. Transplantation 1968, 6, 230–247. [Google Scholar] [CrossRef]
- Neofytou, E.; Deuse, T.; Beygui, R.E.; Schrepfer, S. Mesenchymal stromal cell therapy: Different sources exhibit different immunobiological properties. Transplantation 2015, 99, 1113–1118. [Google Scholar] [CrossRef]
- McCorry, M.C.; Puetzer, J.L.; Bonassar, L.J. Characterization of mesenchymal stem cells and fibrochondrocytes in three-dimensional co-culture: Analysis of cell shape, matrix production, and mechanical performance. Stem Cell Res. Ther. 2016, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Najar, M.; Raicevic, G.; Fayyad-Kazan, H.; Bron, D.; Toungouz, M.; Lagneaux, L. Mesenchymal stromal cells and immunomodulation: A gathering of regulatory immune cells. Cytotherapy 2016, 18, 160–171. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Omoto, M.; Katikireddy, K.R.; Rezazadeh, A.; Dohlman, T.H.; Chauhan, S.K. Mesenchymal stem cells home to inflamed ocular surface and suppress allosensitization in corneal transplantation. Investig. Ophthalmol. Vis. Sci. 2014, 55, 6631–6638. [Google Scholar] [CrossRef] [PubMed]
- Cejkova, J.; Trosan, P.; Cejka, C.; Lencova, A.; Zajicova, A.; Javorkova, E.; Kubinova, S.; Sykova, E.; Holan, V. Suppression of alkali-induced oxidative injury in the cornea by mesenchymal stem cells growing on nanofiber scaffolds and transferred onto the damaged corneal surface. Exp. Eye Res. 2013, 116, 312–323. [Google Scholar] [CrossRef]
- Ye, J.; Yao, K.; Kim, J. Mesenchymal stem cell transplantation in a rabbit corneal alkali burn model: Engraftment and involvement in wound healing. Eye 2006, 20, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Ko, A.Y.; Ko, J.H.; Lee, H.J.; Kim, M.K.; Wee, W.R.; Khwarg, S.I.; Oh, J.Y. Mesenchymal stem/stromal cells protect the ocular surface by suppressing inflammation in an experimental dry eye. Mol. Ther. 2015, 23, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Amirjamshidi, H.; Milani, B.; Sagha, H.; Movahedan, A.; Shafiq, M.; Lavker, R.; Yue, B.; Djalilian, A. Limbal fibroblast conditioned media: A non-invasive treatment for limbal stem cell deficiency. Mol. Vis. 2011, 17, 658. [Google Scholar]
- Acar, U.; Pinarli, F.A.; Acar, D.E.; Beyazyildiz, E.; Sobaci, G.; Ozgermen, B.B.; Sonmez, A.A.; Delibasi, T. Effect of allogeneic limbal mesenchymal stem cell therapy in corneal healing: Role of administration route. Ophthalmic Res. 2015, 53, 82–89. [Google Scholar] [CrossRef]
- Holan, V.; Trosan, P.; Cejka, C.; Javorkova, E.; Zajicova, A.; Hermankova, B.; Chudickova, M.; Cejkova, J. A comparative study of the therapeutic potential of mesenchymal stem cells and limbal epithelial stem cells for ocular surface reconstruction. Stem Cells Transl. Med. 2015, 4, 1052–1063. [Google Scholar] [CrossRef]
- Espandar, L.; Caldwell, D.; Watson, R.; Blanco-Mezquita, T.; Zhang, S.; Bunnell, B. Application of adipose-derived stem cells on scleral contact lens carrier in an animal model of severe acute alkaline burn. Eye Contact Lens 2014, 40, 243. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Li, Y.; Zeng, G.; Yang, B.; Zhu, Y. Transplantation with cultured stem cells derived from the human amniotic membrane for corneal alkali burns: An experimental study. Ann. Clin. Lab. Sci. 2014, 44, 73–81. [Google Scholar] [PubMed]
- Bu, P.; Vin, A.P.; Sethupathi, P.; Ambrecht, L.A.; Zhai, Y.; Nikolic, N.; Qiao, L.; Bouchard, C.S. Effects of activated omental cells on rat limbal corneal alkali injury. Exp. Eye Res. 2014, 121, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Zhang, Y.-Y.; Gu, H.-W.; Guan, H.-J. Effects of bone marrow mesenchymal stem cells on cell proliferation and growth factor expression of limbal epithelial cells in vitro. Ophthalmic Res. 2012, 48, 82–88. [Google Scholar] [CrossRef]
- Lan, Y.; Kodati, S.; Lee, H.S.; Omoto, M.; Jin, Y.; Chauhan, S.K. Kinetics and function of mesenchymal stem cells in corneal injury. Investig. Ophthalmol. Vis. Sci. 2012, 53, 3638–3644. [Google Scholar] [CrossRef]
- Wen, L.; Zhu, M.; Madigan, M.C.; You, J.; King, N.J.; Billson, F.A.; McClellan, K.; Sutton, G.; Petsoglou, C. Immunomodulatory effects of bone marrow-derived mesenchymal stem cells on pro-inflammatory cytokine-stimulated human corneal epithelial cells. PLoS ONE 2014, 9, e101841. [Google Scholar] [CrossRef]
- Ke, Y.; Wu, Y.; Cui, X.; Liu, X.; Yu, M.; Yang, C.; Li, X. Polysaccharide hydrogel combined with mesenchymal stem cells promotes the healing of corneal alkali burn in rats. PLoS ONE 2015, 10, e0119725. [Google Scholar] [CrossRef]
- Cejka, C.; Cejkova, J.; Trosan, P.; Zajicova, A.; Sykova, E.; Holan, V. Transfer of mesenchymal stem cells and cyclosporine A on alkali-injured rabbit cornea using nanofiber scaffolds strongly reduces corneal neovascularization and scar formation. Histol. Histopathol. 2016, 31, 969–980. [Google Scholar] [CrossRef]
- Ma, Y.; Xu, Y.; Xiao, Z.; Yang, W.; Zhang, C.; Song, E.; Du, Y.; Li, L. Reconstruction of chemically burned rat corneal surface by bone marrow–derived human mesenchymal stem cells. Stem Cells 2006, 24, 315–321. [Google Scholar] [CrossRef]
- Ehtesham, M.; Kabos, P.; Kabosova, A.; Neuman, T.; Black, K.L.; John, S.Y. The use of interleukin 12-secreting neural stem cells for the treatment of intracranial glioma. Cancer Res. 2002, 62, 5657–5663. [Google Scholar] [PubMed]
- Reagan, M.R.; Kaplan, D.L. Concise review: Mesenchymal stem cell tumor-homing: Detection methods in disease model systems. Stem Cells 2011, 29, 920–927. [Google Scholar] [CrossRef]
- Chen, C.; Hou, J. Mesenchymal stem cell-based therapy in kidney transplantation. Stem Cell Res. Ther. 2016, 7, 1–7. [Google Scholar] [CrossRef]
- Ito, T.; Itakura, S.; Todorov, I.; Rawson, J.; Asari, S.; Shintaku, J.; Nair, I.; Ferreri, K.; Kandeel, F.; Mullen, Y. Mesenchymal stem cell and islet co-transplantation promotes graft revascularization and function. Transplantation 2010, 89, 1438–1445. [Google Scholar] [CrossRef] [PubMed]
- Pileggi, A.; Xu, X.; Tan, J.; Ricordi, C. Mesenchymal stromal (stem) cells to improve solid organ transplant outcome: Lessons from the initial clinical trials. Curr. Opin. Organ. Transplant. 2013, 18, 672. [Google Scholar] [CrossRef]
- Gilbert, P.M.; Havenstrite, K.L.; Magnusson, K.E.; Sacco, A.; Leonardi, N.A.; Kraft, P.; Nguyen, N.K.; Thrun, S.; Lutolf, M.P.; Blau, H.M. Substrate elasticity regulates skeletal muscle stem cell self-renewal in culture. Science 2010, 329, 1078–1081. [Google Scholar] [CrossRef]
- Curran, J.M.; Chen, R.; Hunt, J.A. The guidance of human mesenchymal stem cell differentiation in vitro by controlled modifications to the cell substrate. Biomaterials 2006, 27, 4783–4793. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; DelRio, F.W.; Ma, H.; Killaars, A.R.; Basta, L.P.; Kyburz, K.A.; Anseth, K.S. Spatially patterned matrix elasticity directs stem cell fate. Proc. Natl. Acad. Sci. USA 2016, 113, E4439–E4445. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.E.L.; Wheadon, H.; Lewis, N.; Yang, J.; Mullin, M.; Hursthouse, A.; Stirling, D.; Dalby, M.J.; Berry, C.C. A quiescent, regeneration-responsive tissue engineered mesenchymal stem cell bone marrow niche model via magnetic levitation. ACS Nano 2016, 10, 8346–8354. [Google Scholar] [CrossRef]
- Dalby, M.J.; García, A.J.; Salmeron-Sanchez, M. Receptor control in mesenchymal stem cell engineering. Nat. Rev. Mater. 2018, 3, 1–14. [Google Scholar] [CrossRef]
- Lane, S.W.; Williams, D.A.; Watt, F.M. Modulating the stem cell niche for tissue regeneration. Nat. Biotechnol. 2014, 32, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Flaim, C.J.; Chien, S.; Bhatia, S.N. An extracellular matrix microarray for probing cellular differentiation. Nat. Methods. 2005, 2, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Gobaa, S.; Hoehnel, S.; Roccio, M.; Negro, A.; Kobel, S.; Lutolf, M.P. Artificial niche microarrays for probing single stem cell fate in high throughput. Nat. Methods. 2011, 8, 949–955. [Google Scholar] [CrossRef]
- Soen, Y.; Mori, A.; Palmer, T.D.; Brown, P.O. Exploring the regulation of human neural precursor cell differentiation using arrays of signaling microenvironments. Mol. Syst. Biol. 2006, 2, 37. [Google Scholar] [CrossRef] [PubMed]
- LaBarge, M.A.; Nelson, C.M.; Villadsen, R.; Fridriksdottir, A.; Ruth, J.R.; Stampfer, M.R.; Petersen, O.W.; Bissell, M.J. Human mammary progenitor cell fate decisions are products of interactions with combinatorial microenvironments. Integr. Biol. 2009, 1, 70–79. [Google Scholar] [CrossRef]
- Roch, A.; Giger, S.; Girotra, M.; Campos, V.; Vannini, N.; Naveiras, O.; Gobaa, S.; Lutolf, M.P. Single-cell analyses identify bioengineered niches for enhanced maintenance of hematopoietic stem cells. Nat. Commun. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, H.B.; Andrade, J.D. Blood compatibility of polyethylene oxide surfaces. Prog. Polym. Sci. 1995, 20, 1043–1079. [Google Scholar] [CrossRef]
- Lutolf, M.P.; Blau, H.M. Artificial stem cell niches. Adv. Mater. 2009, 21, 3255–3268. [Google Scholar] [CrossRef] [PubMed]
- Moeller, H.-C.; Mian, M.K.; Shrivastava, S.; Chung, B.G.; Khademhosseini, A. A microwell array system for stem cell culture. Biomaterials 2008, 29, 752–763. [Google Scholar] [CrossRef]
- Fuchs, E.; Tumbar, T.; Guasch, G. Socializing with the neighbors: Stem cells and their niche. Cell 2004, 116, 769–778. [Google Scholar] [CrossRef]
- Ortega, Í.; Deshpande, P.; Gill, A.A.; MacNeil, S.; Claeyssens, F. Development of a microfabricated artificial limbus with micropockets for cell delivery to the cornea. Biofabrication 2013, 5, 025008. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdul-Al, M.; Kyeremeh, G.K.; Saeinasab, M.; Heidari Keshel, S.; Sefat, F. Stem Cell Niche Microenvironment: Review. Bioengineering 2021, 8, 108. https://doi.org/10.3390/bioengineering8080108
Abdul-Al M, Kyeremeh GK, Saeinasab M, Heidari Keshel S, Sefat F. Stem Cell Niche Microenvironment: Review. Bioengineering. 2021; 8(8):108. https://doi.org/10.3390/bioengineering8080108
Chicago/Turabian StyleAbdul-Al, Mohamed, George Kumi Kyeremeh, Morvarid Saeinasab, Saeed Heidari Keshel, and Farshid Sefat. 2021. "Stem Cell Niche Microenvironment: Review" Bioengineering 8, no. 8: 108. https://doi.org/10.3390/bioengineering8080108
APA StyleAbdul-Al, M., Kyeremeh, G. K., Saeinasab, M., Heidari Keshel, S., & Sefat, F. (2021). Stem Cell Niche Microenvironment: Review. Bioengineering, 8(8), 108. https://doi.org/10.3390/bioengineering8080108






