Engineering and Monitoring 3D Cell Constructs with Time-Evolving Viscoelasticity for the Study of Liver Fibrosis In Vitro
Abstract
:1. Introduction
2. Materials and Methods
2.1. Re-Engineering the MCTR Bioreactor
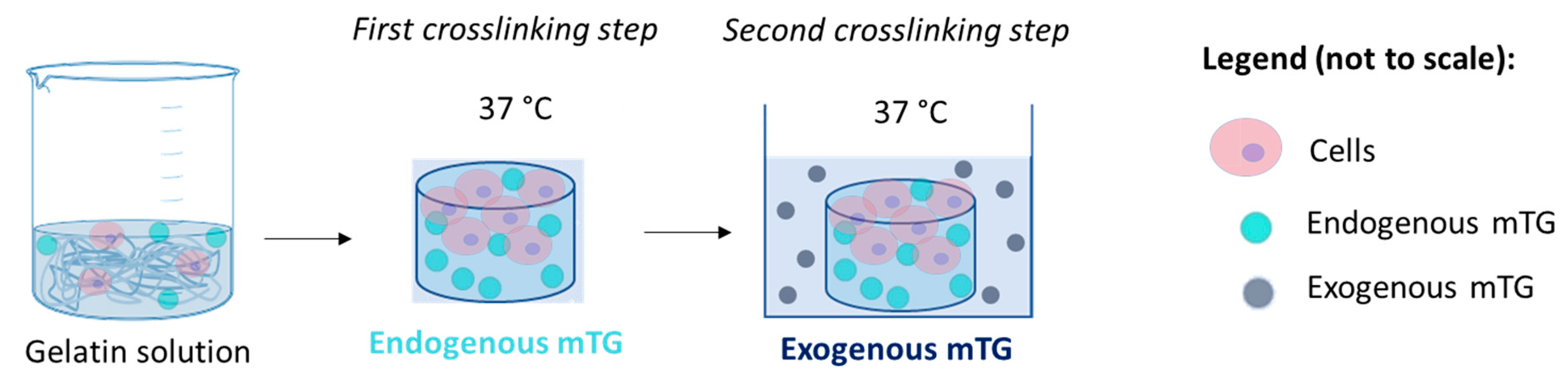
2.2. Gel Fabrication and Cell Encapsulation
2.3. Gel Water Content and Degradation
2.4. Mechanical Testing in the Low-Force MCTR Bioreactor
2.5. Cell Viability and Staining
2.6. Statistical Analysis
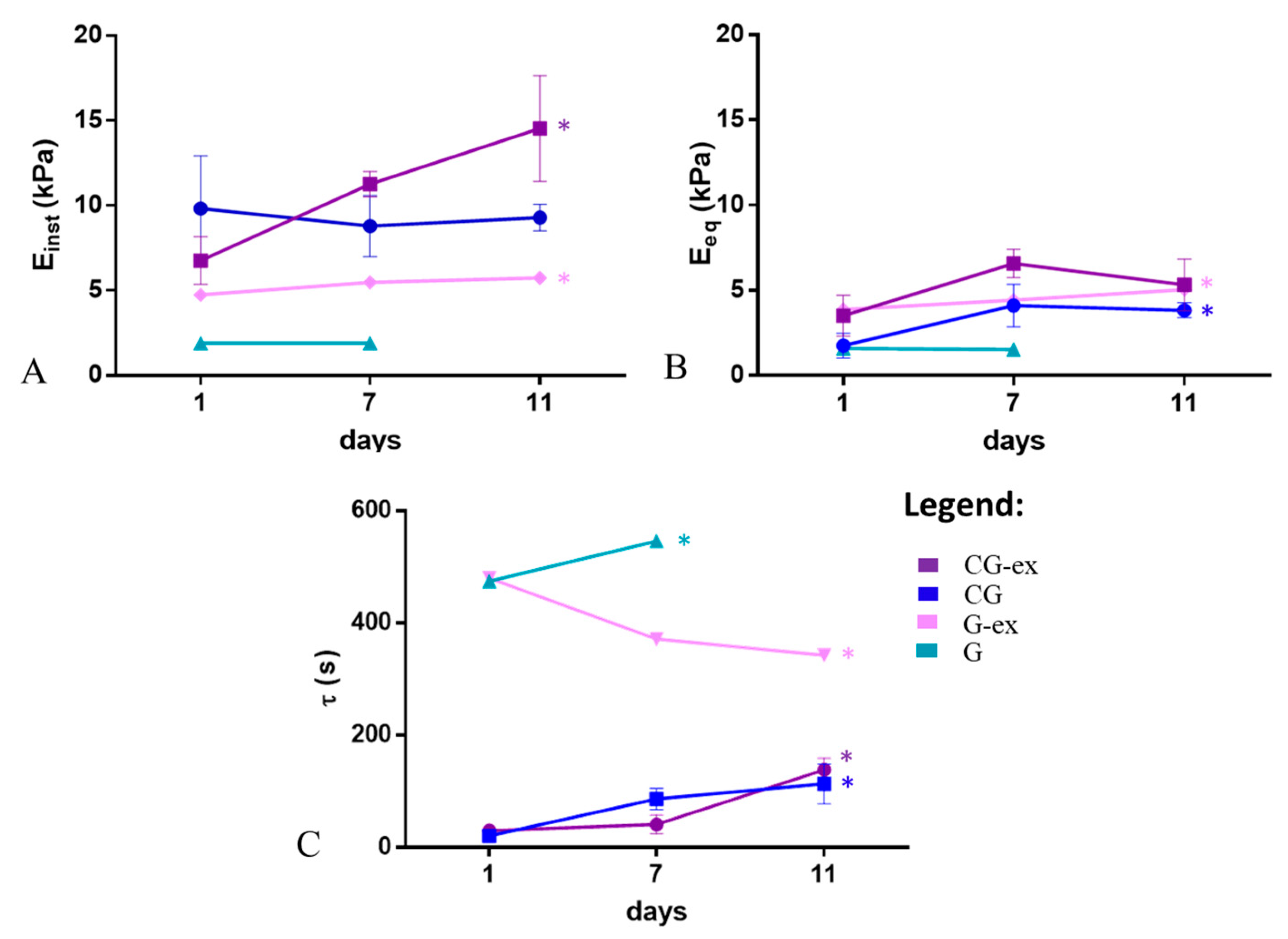
3. Results
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wallace, K.; Burt, A.; Wright, M.C. Liver fibrosis. Biochem. J. 2008, 411, 1–18. [Google Scholar] [CrossRef]
- Van De Bovenkamp, M.; Groothuis, G.; Meijer, D.; Olinga, P. Liver fibrosis in vitro: Cell culture models and precision-cut liver slices. Toxicol. Vitr. 2007, 21, 545–557. [Google Scholar] [CrossRef]
- Bedossa, P.; Paradis, V. Liver extracellular matrix in health and disease. J. Pathol. 2003, 200, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Mattei, G.; Tirella, A.; Gallone, G.; Ahluwalia, A. Viscoelastic characterisation of pig liver in unconfined compression. J. Biomech. 2014, 47, 2641–2646. [Google Scholar] [CrossRef]
- Mattei, G.; Ahluwalia, A. Sample, testing and analysis variables affecting liver mechanical properties: A review. Acta Biomater. 2016, 45, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Iredale, J.P. Models of liver fibrosis: Exploring the dynamic nature of inflammation and repair in a solid organ. J. Clin. Investig. 2007, 117, 539–548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeh, W.C. Elastic moduls measurements of human liver and correlation with pathology. Ultrasound Med. Biol. 2002, 28, 467–474. [Google Scholar] [CrossRef]
- Snedeker, J.G.; Gautieri, A. The role of collagen crosslinks in ageing and diabetes—The good, the bad, and the ugly. Muscle Ligaments Tendons J. 2019, 4, 303–308. [Google Scholar] [CrossRef]
- Cox, T.R.; Erler, J.T. Remodeling and homeostasis of the extracellular matrix: Implications for fibrotic diseases and cancer. Dis. Model. Mech. 2011, 4, 165–178. [Google Scholar] [CrossRef] [Green Version]
- Wells, R.G. The role of matrix stiffness in regulating cell behavior. Hepatology 2008, 47, 1394–1400. [Google Scholar] [CrossRef] [PubMed]
- Sandrin, L.; Fourquet, B.; Hasquenoph, J.-M.; Yon, S.; Fournier, C.; Mal, F.; Christidis, C.; Ziol, M.; Poulet, B.; Kazemi, F.; et al. Transient elastography: A new noninvasive method for assessment of hepatic fibrosis. Ultrasound Med. Biol. 2003, 29, 1705–1713. [Google Scholar] [CrossRef]
- Yin, M.; Talwalkar, J.A.; Glaser, K.J.; Manduca, A.; Grimm, R.C.; Rossman, P.J.; Fidler, J.L.; Ehman, R.L. Assessment of Hepatic Fibrosis with Magnetic Resonance Elastography. Clin. Gastroenterol. Hepatol. 2007, 5, 1207–1213.e2. [Google Scholar] [CrossRef] [Green Version]
- Asbach, P.; Klatt, D.; Schlosser, B.; Biermer, M.; Muche, M.; Rieger, A.; Loddenkemper, C.; Somasundaram, R.; Berg, T.; Hamm, B.; et al. Viscoelasticity-based Staging of Hepatic Fibrosis with Multifrequency MR Elastography. Radiology 2010, 257, 80–86. [Google Scholar] [CrossRef]
- Asbach, P.; Klatt, D.; Hamhaber, U.; Braun, J.; Somasundaram, R.; Hamm, B.; Sack, I. Assessment of liver viscoelasticity using multifrequency MR elastography. Magn. Reson. Med. 2008, 60, 373–379. [Google Scholar] [CrossRef]
- Sun, K.; Kusminski, C.M.; Scherer, P.E. Adipose tissue remodeling and obesity. J. Clin. Investig. 2011, 121, 2094–2101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soldatow, V.Y.; LeCluyse, E.; Griffith, L.; Rusyn, I. In vitro models for liver toxicity testing. Toxicol. Res. 2013, 2, 23–39. [Google Scholar] [CrossRef] [Green Version]
- Wells, R.G. The Role of Matrix Stiffness in Hepatic Stellate Cell Activation and Liver Fibrosis. J. Clin. Gastroenterol. 2005, 39, S158–S161. [Google Scholar] [CrossRef]
- Olsen, A.L.; Bloomer, S.A.; Chan, E.P.; Gaça, M.D.A.; Georges, P.; Sackey, B.; Uemura, M.; Janmey, P.A.; Wells, R.G. Hepatic stellate cells require a stiff environment for myofibroblastic differentiation. Am. J. Physiol. Liver Physiol. 2011, 301, G110–G118. [Google Scholar] [CrossRef] [Green Version]
- Hansen, L.; Wilhelm, J.; Fassett, J. Regulation of Hepatocyte Cell Cycle Progression and Differentiation by Type I Collagen Structure. Curr. Top. Dev. Biol. 2005, 72, 205–236. [Google Scholar] [CrossRef]
- Guvendiren, M.; Perepelyuk, M.; Wells, R.G.; Burdick, J.A. Hydrogels with differential and patterned mechanics to study stiffness-mediated myofibroblastic differentiation of hepatic stellate cells. J. Mech. Behav. Biomed. Mater. 2014, 38, 198–208. [Google Scholar] [CrossRef] [Green Version]
- Hui, E.; Gimeno, K.I.; Guan, G.; Caliari, S.R. Spatial control of viscoelasticity in phototunable hyaluronic acid hydrogels. bioRxiv 2019, 646778. [Google Scholar] [CrossRef]
- Carberry, B.J.; Rao, V.V.; Anseth, K.S. Phototunable Viscoelasticity in Hydrogels Through Thioester Exchange. Ann. Biomed. Eng. 2020, 48, 2053–2063. [Google Scholar] [CrossRef] [PubMed]
- Caliari, S.; Perepelyuk, M.; Cosgrove, B.D.; Tsai, S.J.; Lee, G.Y.; Mauck, R.; Wells, R.G.; Burdick, J.A. Stiffening hydrogels for investigating the dynamics of hepatic stellate cell mechanotransduction during myofibroblast activation. Sci. Rep. 2016, 6, 21387. [Google Scholar] [CrossRef] [Green Version]
- Mattei, G.; Cacopardo, L.; Ahluwalia, A.A. Engineering Gels with Time-Evolving Viscoelasticity. Materials 2020, 13, 438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravi, M.; Paramesh, V.; Kaviya, S.; Anuradha, E.; Solomon, F.P. 3D Cell Culture Systems: Advantages and Applications. J. Cell. Physiol. 2015, 230, 16–26. [Google Scholar] [CrossRef]
- Mastrorocco, A.; Cacopardo, L.; Martino, N.A.; Fanelli, D.; Camillo, F.; Ciani, E.; Roelen, B.A.J.; Ahluwalia, A.; Dell’Aquila, M.E. One-step automated bioprinting-based method for cumulus-oocyte complex microencapsulation for 3D in vitro maturation. PLoS ONE 2020, 15, e0238812. [Google Scholar] [CrossRef]
- Yung, C.; Wu, L.; Tullman, J.; Payne, G.; Bentley, W.; Barbari, T. Transglutaminase crosslinked gelatin as a tissue engineering scaffold. J. Biomed. Mater. Res. Part A 2007, 83, 1039–1046. [Google Scholar] [CrossRef]
- Broderick, E.P.; O’Halloran, D.M.; Rochev, Y.A.; Griffin, M.; Collighan, R.J.; Pandit, A.S. Enzymatic stabilization of gelatin-based scaffolds. J. Biomed. Mater. Res. 2004, 72, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Selfe, J. Fundamentals of Biomechanics. Physiotherapy 2000, 86, 163. [Google Scholar] [CrossRef] [Green Version]
- Hennink, W.E.; Nostrum, C.F. Van Novel crosslinking methods to design hydrogels. Adv. Drug Deliv. Rev. 2012, 64, 223–236. [Google Scholar] [CrossRef]
- Charrier, E.E.; Pogoda, K.; Wells, R.G.; Janmey, P.A. Control of cell morphology and differentiation by substrates with independently tunable elasticity and viscous dissipation. Nat. Commun. 2018, 9, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Cameron, A.; Frith, J.; Cooper-White, J.J. The influence of substrate creep on mesenchymal stem cell behaviour and phenotype. Biomaterials 2011, 32, 5979–5993. [Google Scholar] [CrossRef]
- Chaudhuri, O.; Gu, L.; Darnell, M.; Klumpers, D.D.; Bencherif, S.A.; Weaver, J.C.; Huebsch, N.; Mooney, D.J. Substrate stress relaxation regulates cell spreading. Nat. Commun. 2015, 6, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cacopardo, L.; Guazzelli, N.; Nossa, R.; Mattei, G.; Ahluwalia, A. Engineering hydrogel viscoelasticity. J. Mech. Behav. Biomed. Mater. 2019, 89, 162–167. [Google Scholar] [CrossRef]
- Giusti, S.; Mazzei, D.; Cacopardo, L.; Mattei, G.; Domenici, C.; Ahluwalia, A. Environmental Control in Flow Bioreactors. Processes 2017, 5, 16. [Google Scholar] [CrossRef] [Green Version]
- Popp, J.R.; Roberts, J.J.; Gallagher, D.V.; Anseth, K.S.; Bryant, S.J.; Quinn, T.P. An Instrumented Bioreactor for Mechanical Stimulation and Real-Time, Nondestructive Evaluation of Engineered Cartilage Tissue. J. Med. Devices 2012, 6, 021006. [Google Scholar] [CrossRef] [Green Version]
- Lujan, T.J.; Wirtz, K.M.; Bahney, C.S.; Madey, S.M.; Johnstone, B.; Bottlang, M. A Novel Bioreactor for the Dynamic Stimulation and Mechanical Evaluation of Multiple Tissue-Engineered Constructs. Tissue Eng. Part C Methods 2011, 17, 367–374. [Google Scholar] [CrossRef] [Green Version]
- Salvetti, D.J.; Pino, C.J.; Manuel, S.G.; Dallmeyer, I.; Rangarajan, S.V.; Meyer, T.; Kotov, M.; Shastri, V.P. Design and Validation of a Compressive Tissue Stimulator with High-Throughput Capacity and Real-Time Modulus Measurement Capability. Tissue Eng. Part C Methods 2012, 18, 205–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meinert, C.; Schrobback, K.; Hutmacher, D.W.; Klein, T.J. A novel bioreactor system for biaxial mechanical loading enhances the properties of tissue-engineered human cartilage. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Mohanraj, B.; Hou, C.; Meloni, G.; Cosgrove, B.; George, D.; Mauck, R. NIH Public Access. J. Biomech. 2013, 6, 2130–2136. [Google Scholar]
- Correia, V.M.G.; Panadero, J.A.; Ribeiro, C.; Sencadas, V.; Rocha, J.G.; Ribelles, J.L.G.; Lanceros-Méndez, S. Design and validation of a biomechanical bioreactor for cartilage tissue culture. Biomech. Model. Mechanobiol. 2016, 15, 471–478. [Google Scholar] [CrossRef]
- Selby, J.C.; Shannon, M.A. Apparatus for measuring the finite load-deformation behavior of a sheet of epithelial cells cultured on a mesoscopic freestanding elastomer membrane. Rev. Sci. Instruments 2007, 78, 094301. [Google Scholar] [CrossRef] [PubMed]
- Van Kelle, M.A.; Oomen, P.; Bulsink, J.A.; Broek, M.W.J.-V.D.; Lopata, R.G.; Rutten, M.C.; Loerakker, S.; Bouten, C. A Bioreactor to Identify the Driving Mechanical Stimuli of Tissue Growth and Remodeling. Tissue Eng. Part C Methods 2017, 23, 377–387. [Google Scholar] [CrossRef] [Green Version]
- Walter, B.; Illien-Junger, S.; Nasser, P.; Hecht, A.; Iatridis, J. Development and validation of a bioreactor system for dynamic loading and mechanical characterization of whole human intervertebral discs in organ culture. J. Biomech. 2014, 47, 2095–2101. [Google Scholar] [CrossRef] [Green Version]
- Cacopardo, L.; Mattei, G.; Ahluwalia, A. A new load-controlled testing method for viscoelastic characterisation through stress-rate measurements. Materialia 2020, 9, 100552. [Google Scholar] [CrossRef]
- Caliari, S.; Burdick, J.A. A practical guide to hydrogels for cell culture. Nat. Methods 2016, 13, 405–414. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.-C.; Anseth, K.S. PEG Hydrogels for the Controlled Release of Biomolecules in Regenerative Medicine. Pharm. Res. 2009, 26, 631–643. [Google Scholar] [CrossRef] [Green Version]
- Vinci, B.; Cavallone, D.; Vozzi, G.; Mazzei, D.; Domenici, C.; Brunetto, M.R.; Ahluwalia, A. In vitro liver model using microfabricated scaffolds in a modular bioreactor. Biotechnol. J. 2009, 5, 232–241. [Google Scholar] [CrossRef] [Green Version]
- Carter, D.; van der Meulen, M.C.H.; Beaupre, G. Mechanical factors in bone growth and development. Bone 1996, 18, S5–S10. [Google Scholar] [CrossRef]
- Wu, M.; Fannin, J.; Rice, K.; Wang, B.; Blough, E.R. Effect of aging on cellular mechanotransduction. Ageing Res. Rev. 2011, 10, 1–15. [Google Scholar] [CrossRef] [Green Version]
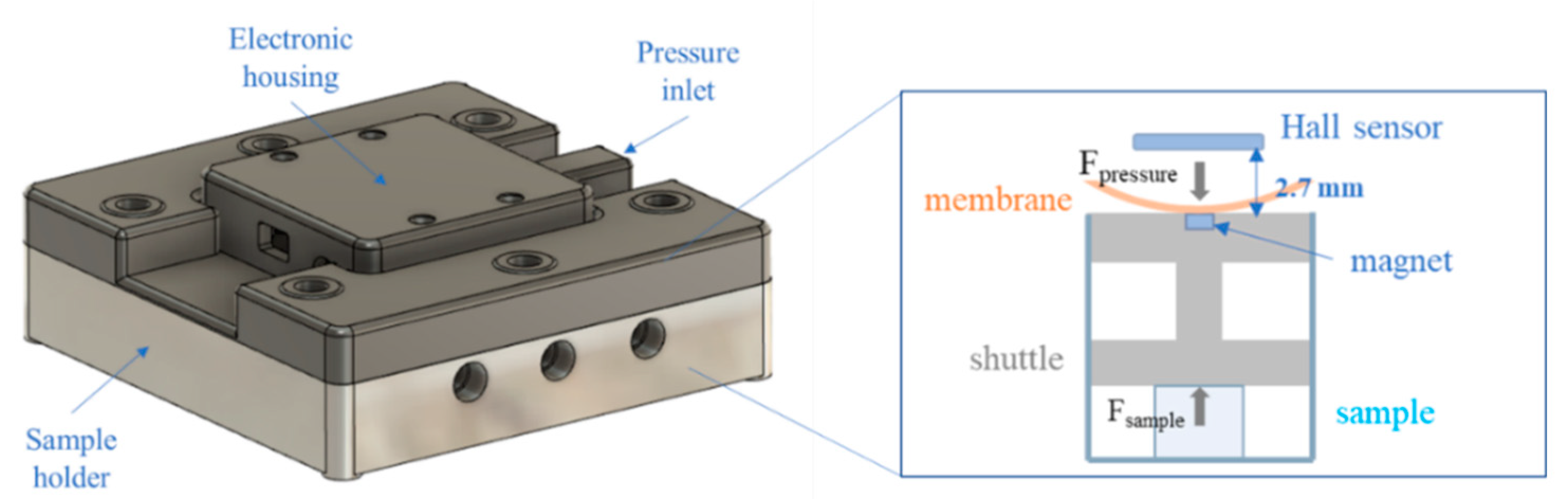





| Low-Force Version | Commercial Version | |
|---|---|---|
| Pressure regulator | Range: 0–7 kPa Resolution: 0.35 mPa | Range: 5–500 kPa Resolution: 0.1 kPa |
| Membrane Hardness | 10A | 60A |
| Magnet dimensions (diameter × height) | 2.5 × 1 mm | 10 × 2.5 mm |
| Shuttle material | PLA | Stainless steel |
| Low-Force Version | Commercial Version | |
|---|---|---|
| Force range | 0.02–1.4 N | 2–100 N |
| Force resolution | 0.7 mN | 0.02 N |
| Shuttle weight | 15 mN | 133 mN |
| Viscoelastic testing | yes | yes [46] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cacopardo, L.; Ahluwalia, A. Engineering and Monitoring 3D Cell Constructs with Time-Evolving Viscoelasticity for the Study of Liver Fibrosis In Vitro. Bioengineering 2021, 8, 106. https://doi.org/10.3390/bioengineering8080106
Cacopardo L, Ahluwalia A. Engineering and Monitoring 3D Cell Constructs with Time-Evolving Viscoelasticity for the Study of Liver Fibrosis In Vitro. Bioengineering. 2021; 8(8):106. https://doi.org/10.3390/bioengineering8080106
Chicago/Turabian StyleCacopardo, Ludovica, and Arti Ahluwalia. 2021. "Engineering and Monitoring 3D Cell Constructs with Time-Evolving Viscoelasticity for the Study of Liver Fibrosis In Vitro" Bioengineering 8, no. 8: 106. https://doi.org/10.3390/bioengineering8080106
APA StyleCacopardo, L., & Ahluwalia, A. (2021). Engineering and Monitoring 3D Cell Constructs with Time-Evolving Viscoelasticity for the Study of Liver Fibrosis In Vitro. Bioengineering, 8(8), 106. https://doi.org/10.3390/bioengineering8080106







