Thauera aminoaromatica MZ1T Identified as a Polyhydroxyalkanoate-Producing Bacterium within a Mixed Microbial Consortium
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Chemicals
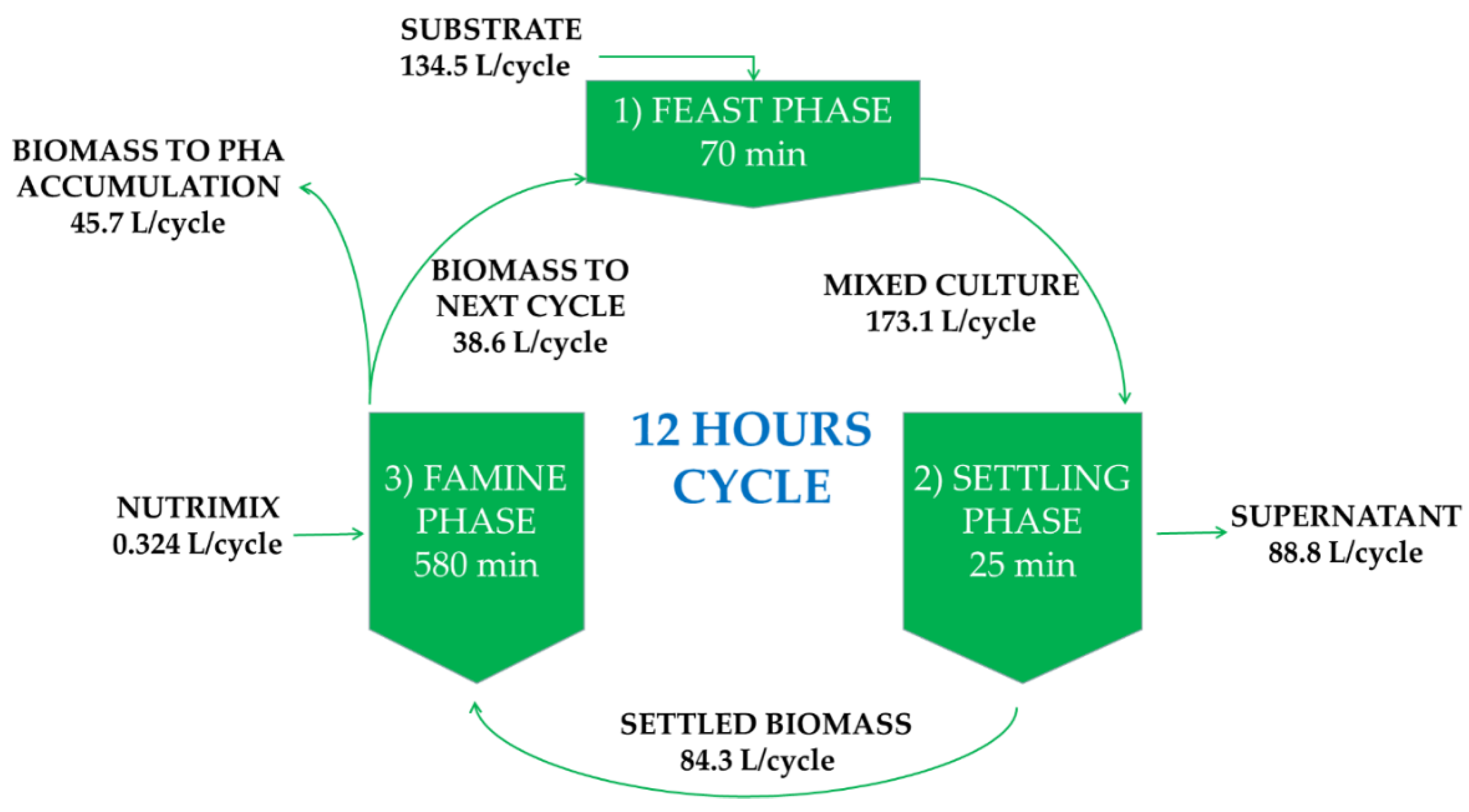
2.2. Operating Conditions Pilot Reactor
2.3. Bacterial Growth Conditions—Lab-Scale Bioreactor
2.3.1. Growth Media
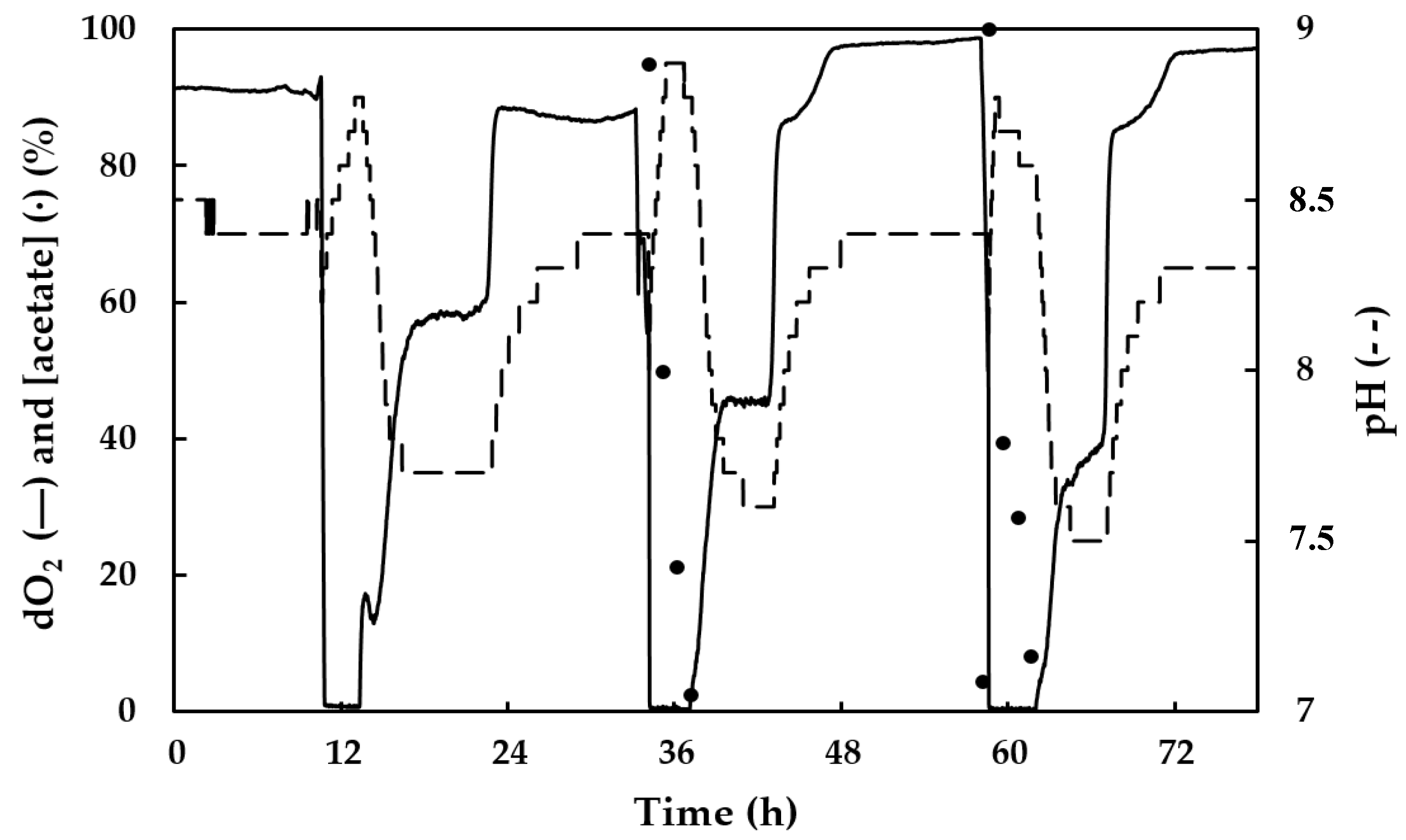
2.3.2. Growth Conditions: 5 L Enrichment Bioreactor under a Feast–Famine Regime
2.3.3. Growth Conditions: Polyhydroxyalkanoates (PHA) Production in 2 L Bioreactor
2.4. Bacterial Growth Conditions—Thauera aminoaromatica MZ1(T)
2.5. Chemical Extraction of PHA
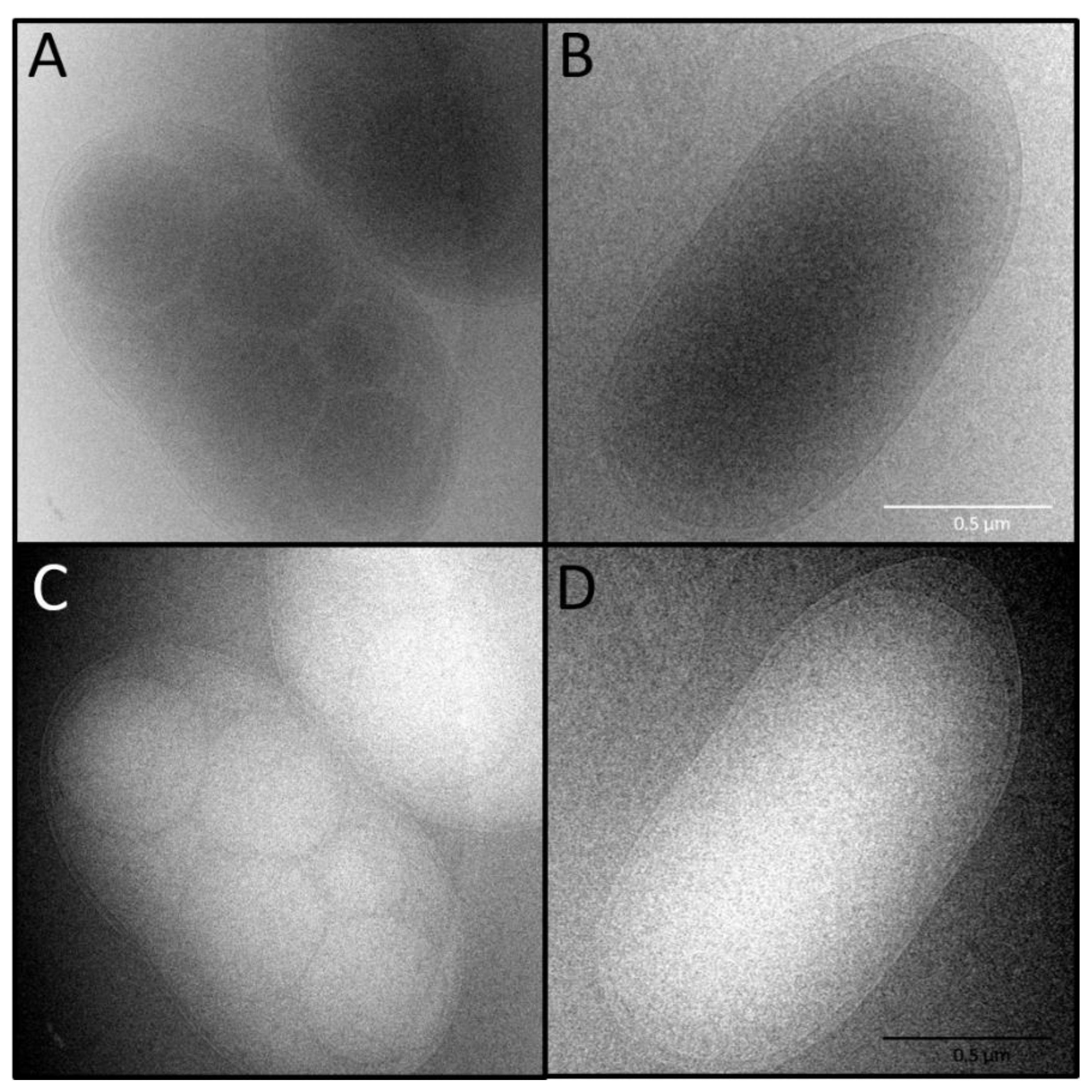
2.6. Microbial Characterization by Microscopy
2.6.1. Phase-Contrast and Fluorescence Microscopy—Nile Blue A Staining
2.6.2. Cryo-Transmission Electron Microscopy (Cryo-TEM)
2.7. Genomic Diversity Analysis
2.8. Analytical Analysis of PHA
2.8.1. Determination of the Acetic Acid Concentration by High-Performance Liquid Chromatography (HPLC) or Kit
2.8.2. Gas Chromatography–Mass Spectroscopy (GC–MS)
2.8.3. Nuclear Magnetic Resonance (NMR)
3. Results and Discussion
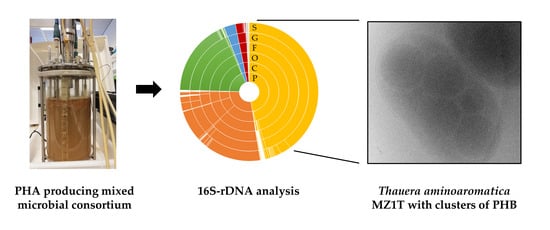
3.1. Description of the Mixed Microbial Consortium
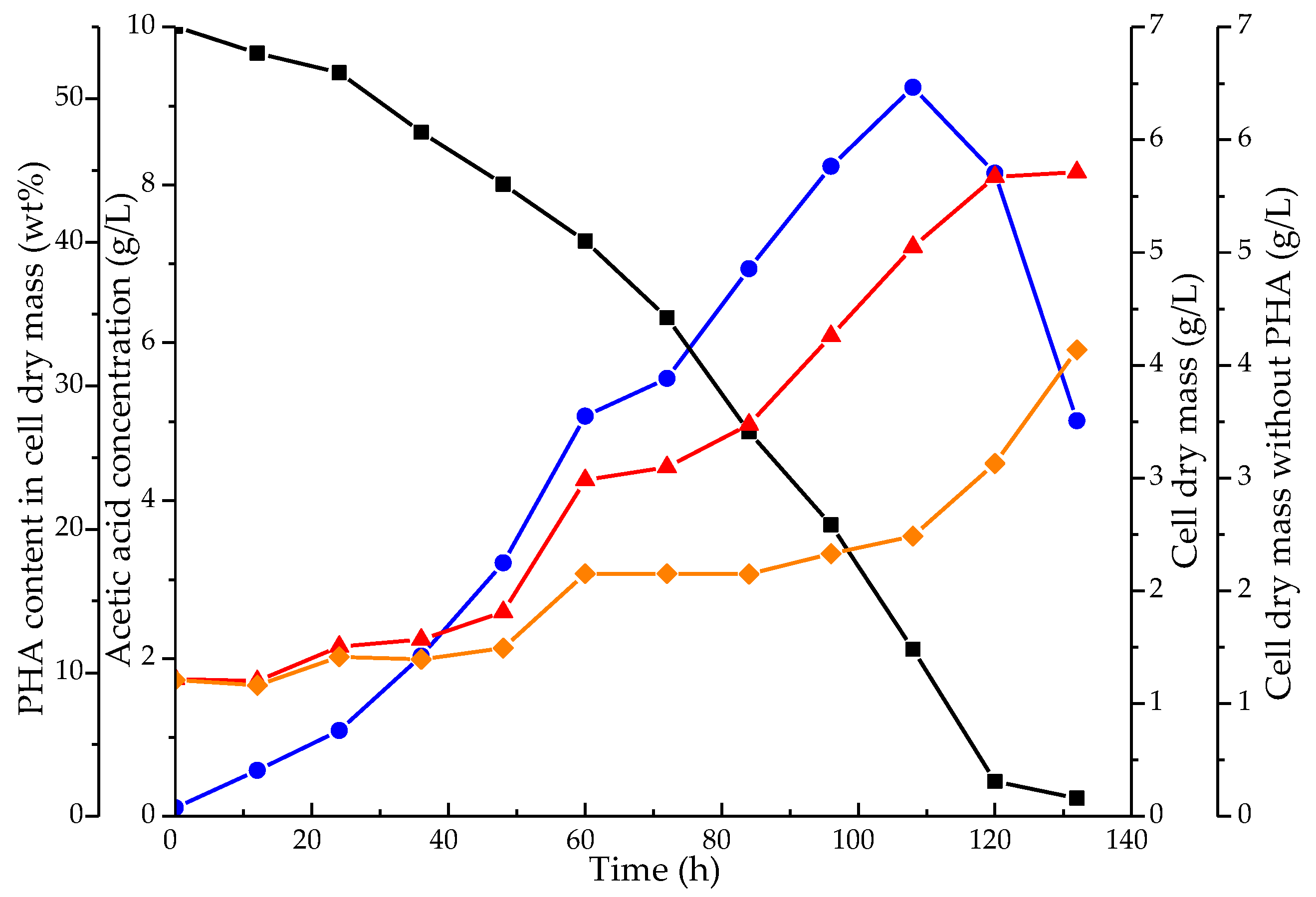
3.2. Culture Growth and PHA Production by the Mixed Microbial Consortium
3.3. Extraction and Identification of PHA
3.4. Genomic (Diversity) Analysis of the Mixed Microbial Consortium
3.5. Does T. aminoaromatica MZ1T Produce PHA?
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Plastics—The Facts 2019: An Analysis of European Plastic Production, Demand and Waste Data. Available online: https://www.plasticseurope.org/en/resources/publications/1804-plastics-facts-2019 (accessed on 20 February 2020).
- Kourmentza, C.; Plácido, J.; Venetsaneas, N.; Burniol-Figols, A.; Varrone, C.; Gavala, H.N.; Reis, M.A.M. Recent advances and challenges towards sustainable polyhydroxyalkanoate (PHA) production. Bioengineering 2017, 4, 55. [Google Scholar] [CrossRef] [PubMed]
- Puppi, D.; Pecorini, G.; Chiellini, F. Biomedical processing of polyhydroxyalkanoates. Bioengineering 2019, 6, 108. [Google Scholar] [CrossRef] [PubMed]
- Amelia, T.S.M.; Govindasamy, S.; Tamothran, A.M.; Vigneswari, S.; Bhubalan, K. Applications of PHA in agriculture. In Biotechnological Applications of Polyhydroxyalkanoates; Kalia, V.C., Ed.; Springer: Singapore, 2019; pp. 347–361. [Google Scholar]
- Castro-Sowinski, S.; Burdman, S.; Matan, O.; Okon, Y. Natural functions of bacterial polyhydroxyalkanoates. In Plastics from Bacteria; Chen, G.Q., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 39–61. [Google Scholar]
- Beun, J.J.; Dircks, K.; van Loosdrecht, M.C.M.; Heijnen, J.J. Poly-β-hydroxybutyrate metabolism in dynamically fed mixed microbial cultures. Water Res. 2002, 36, 1167–1180. [Google Scholar] [CrossRef]
- Tamis, J.; Marang, L.; Jiang, Y.; van Loosdrecht, M.C.M.; Kleerebezem, R. Modeling PHA-producing microbial enrichment cultures—Towards a generalized model with predictive power. New Biotechnol. 2014, 31, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Koller, M. Advances in polyhydroxyalkanoate (PHA) production. Bioengineering 2017, 4, 88. [Google Scholar] [CrossRef]
- Huang, L.; Chen, Z.; Wen, Q.; Zhao, L.; Lee, D.-J.; Yang, L.; Wang, Y. Insights into feast-famine polyhydroxyalkanoate (PHA)-producer selection: Microbial community succession, relationships with system function and underlying driving forces. Water Res. 2018, 131, 167–176. [Google Scholar] [CrossRef]
- Oliveira, C.S.S.; Silva, C.E.; Carvalho, G.; Reis, M.A. Strategies for efficiently selecting PHA producing mixed microbial cultures using complex feedstocks: Feast and famine regime and uncoupled carbon and nitrogen availabilities. New Biotechnol. 2017, 37, 69–79. [Google Scholar] [CrossRef]
- Tan, G.-Y.A.; Chen, C.-L.; Li, L.; Ge, L.; Wang, L.; Razaad, I.M.N.; Li, Y.; Zhao, L.; Mo, Y.; Wang, J.-Y. Start a research on biopolymer polyhydroxyalkanoate (PHA): A review. Polymers 2014, 6, 706–754. [Google Scholar] [CrossRef]
- Yan, Q.; Zhao, M.; Miao, H.; Ruan, W.; Song, R. Coupling of the hydrogen and polyhydroxyalkanoates (PHA) production through anaerobic digestion from Taihu blue algae. Bioresour. Technol. 2010, 101, 4508–4512. [Google Scholar] [CrossRef]
- Obruca, S.; Benesova, P.; Kucera, D.; Petrik, S.; Marova, I. Biotechnological conversion of spent coffee grounds into polyhydroxyalkanoates and carotenoids. New Biotechnol. 2015, 32, 569–574. [Google Scholar] [CrossRef]
- Kourmentza, C.; Costa, J.; Azevedo, Z.; Servin, C.; Grandfils, C.; De Freitas, V.; Reis, M.A.M. Burkholderia thailandensis as a microbial cell factory for the bioconversion of used cooking oil to polyhydroxyalkanoates and rhamnolipids. Bioresour. Technol. 2018, 247, 829–837. [Google Scholar] [CrossRef]
- Altaee, N.; Fahdil, A.; Yousif, E.; Sudesh, K. Recovery and subsequent characterization of polyhydroxybutyrate from Rhodococcus equi cells grown on crude palm kernel oil. J. Taibah Univ. Sci. 2016, 10, 543–550. [Google Scholar] [CrossRef]
- Valentino, F.; Morgan-Sagastume, F.; Campanari, S.; Villano, M.; Werker, A.; Majone, M. Carbon recovery from wastewater through bioconversion into biodegradable polymers. New Biotechnol. 2017, 37, 9–23. [Google Scholar] [CrossRef]
- Cho, K.S.; Ryu, H.W.; Park, C.H.; Goodrich, P.R. Poly(hydroxybutyrate-co-hydroxyvalerate) from swine waste liquor by Azotobacter vinelandii UWD. Biotechnol. Lett. 1997, 19, 7–10. [Google Scholar] [CrossRef]
- Moretto, G.; Russo, I.; Bolzonella, D.; Pavan, P.; Majone, M.; Valentino, F. An urban biorefinery for food waste and biological sludge conversion into polyhydroxyalkanoates and biogas. Water Res. 2020, 170, 1–12. [Google Scholar] [CrossRef]
- Albuquerque, M.G.E.; Torres, C.A.V.; Reis, M.A.M. Polyhydroxyalkanoate (PHA) production by a mixed microbial culture using sugar molasses: Effect of the influent substrate concentration on culture selection. Water Res. 2010, 44, 3419–3433. [Google Scholar] [CrossRef]
- Jiang, Y.; Hebly, M.; Kleerebezem, R.; Muyzer, G.; van Loosdrecht, M.C.M. Metabolic modeling of mixed substrate uptake for polyhydroxyalkanoate (PHA) production. Water Res. 2011, 45, 1309–1321. [Google Scholar] [CrossRef]
- Kourmentza, C.; Kornaros, M. Biotransformation of volatile fatty acids to polyhydroxyalkanoates by employing mixed microbial consortia: The effect of pH and carbon source. Bioresour. Technol. 2016, 222, 388–398. [Google Scholar] [CrossRef]
- Ling, C.; Qiao, G.-Q.; Shuai, B.-W.; Olavarria, K.; Yin, J.; Xiang, R.-J.; Song, K.-N.; Shen, Y.-H.; Guo, Y.; Chen, G.-Q. Engineering NADH/NAD+ ratio in Halomonas bluephagenesis for enhanced production of polyhydroxyalkanoates (PHA). Metab. Eng. 2018, 49, 275–286. [Google Scholar] [CrossRef]
- Hanson, A.J.; Guho, N.M.; Paszczynski, A.J.; Coats, E.R. Community proteomics provides functional insight into polyhydroxyalkanoate production by a mixed microbial culture cultivated on fermented dairy manure. Appl. Microbiol. Biotechnol. 2016, 100, 7957–7976. [Google Scholar] [CrossRef]
- Johnson, K.; Jiang, Y.; Kleerebezem, R.; Muyzer, G.; van Loosdrecht, M.C.M. Enrichment of a mixed bacterial culture with a high polyhydroxyalkanoate storage capacity. Biomacromolecules 2009, 10, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Vishniac, W.; Santer, M. The Thiobacilli. Bacteriol. Rev. 1957, 21, 195–213. [Google Scholar] [PubMed]
- Ramsay, J.A.; Berger, E.; Voyer, R.; Chavarie, C.; Ramsay, B.A. Extraction of poly-3-hydroxybutyrate using chlorinated solvents. Biotechnol. Tech. 1994, 8, 589–594. [Google Scholar] [CrossRef]
- Kumar, M.; Ghosh, P.; Khosla, K.; Thakur, I.S. Recovery of polyhydroxyalkanoates from municipal secondary wastewater sludge. Bioresour. Technol. 2018, 255, 111–115. [Google Scholar] [CrossRef]
- Ostle, A.G.; Holt, J. Nile blue A as a fluorescent stain for poly-β-hydroxybutyrate. Appl. Environ. Microbiol. 1982, 44, 238–241. [Google Scholar] [CrossRef]
- Guo, W.; Heeres, H.J.; Yue, J. Continuous synthesis of 5-hydroxymethylfurfural from glucose using a combination of AlCl3 and HCl as catalyst in a biphasic slug flow capillary microreactor. Chem. Eng. J. 2020, 381, 122754. [Google Scholar] [CrossRef]
- Borrero-de Acuña, J.M.; Hidalgo-Dumont, C.; Pacheco, N.; Cabrera, A.; Poblete-Castro, I. A novel programmable lysozyme-based lysis system in Pseudomonas putida for biopolymer production. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Kumar, M.; Gupta, A.; Thakur, I.S. Carbon dioxide sequestration by chemolithotrophic oleaginous bacteria for production and optimization of polyhydroxyalkanoate. Bioresour. Technol. 2015, 213, 249–256. [Google Scholar] [CrossRef]
- Cai, L.; Yuan, M.-Q.; Liu, F.; Jian, J.; Chen, G.-Q. Enhanced production of medium-chain-length polyhydroxyalkanoates (PHA) by PHA depolymerase knockout mutant of Pseudomonas putida KT2442. Bioresour. Technol. 2009, 100, 2265–2270. [Google Scholar] [CrossRef]
- Kobayashi, J.; Kondo, A. Disruption of poly (3-hydroxyalkanoate) depolymerase gene and overexpression of three poly (3-hydroxybutyrate) biosynthetic genes improve poly (3-hydroxybutyrate) production from nitrogen rich medium by Rhodobacter sphaeroides. Microb. Cell Fact. 2019, 18, 1–13. [Google Scholar] [CrossRef]
- Lajoie, C.A.; Layton, A.C.; Gregory, I.R.; Sayler, G.S.; Taylor, D.E.; Meyers, A.J. Zoogleal clusters and sludge dewatering potential in an industrial activated-sludge wastewater treatment plant. Water Environ. Res. 2000, 72, 56–64. [Google Scholar] [CrossRef]
- Jiang, K.; Sanseverino, J.; Chauhan, A.; Lucas, S.; Copeland, A.; Lapidus, A.; Del Rio, T.G.; Dalin, E.; Tice, H.; Bruce, D.; et al. Complete genome sequence of Thauera aminoaromatica strain MZ1T. Stand. Genom. Sci. 2012, 6, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, S.; Jogler, M.; Jogler, C. On the maverick Planctomycetes. FEMS Microbiol. Rev. 2018, 42, 739–760. [Google Scholar] [CrossRef] [PubMed]
- Fukunaga, Y.; Kurahashi, M.; Sakiyama, Y.; Ohuchi, M.; Yokota, A.; Harayama, S. Phycisphaera mikurensis gen. nov., sp. nov., isolated from a marine alga, and proposal of Phycisphaeraceae fam. nov., Phycisphaerales ord. nov. and Phycisphaerae classis nov. in the phylum Planctomycetes. J. Gen. Appl. Microbiol. 2009, 55, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.S.; Welch, K.T.; Prebyl, B.S.; Baker, D.C.; Meyers, A.J.; Sayler, G.S. Analysis and glycosyl composition of the exopolysaccharide isolated from the floc-forming wastewater bacterium Thauera sp. MZ1T. Environ. Microbiol. 2004, 6, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Seyfried, B.; Tschech, A.; Fuchs, G. Anaerobic degradation of pheylacetate and 4-hydroxyphenylacetate by denitrifying bacteria. Arch. Microbiol. 1991, 155, 249–255. [Google Scholar] [CrossRef]
- Mechichi, T.; Stackebrandt, E.; Gad’on, N.; Fuchs, G. Phylogenetic and metabolic diversity of bacteria degrading aromatic compounds under denitrifying conditions, and description of Thauera phenylacetica sp. nov., Thauera aminoaromatica sp. nov., and Azoarcus buckelii sp. nov. Arch. Microbiol. 2002, 178, 26–35. [Google Scholar] [CrossRef]
- Kutralam-Muniasamy, G.; Marsch, R.; Pérez-Guevara, F. Investigation on the evolutionary relation of diverse polyhydroxyalkanoate gene clusters in Betaproteobacteria. J. Mol. Evol. 2018, 86, 470–483. [Google Scholar] [CrossRef]
- Li, T.; Elhadi, D.; Chen, G.-Q. Co-production of microbial polyhydroxyalkanoates with other chemicals. Metab. Eng. 2017, 43, 29–36. [Google Scholar] [CrossRef]
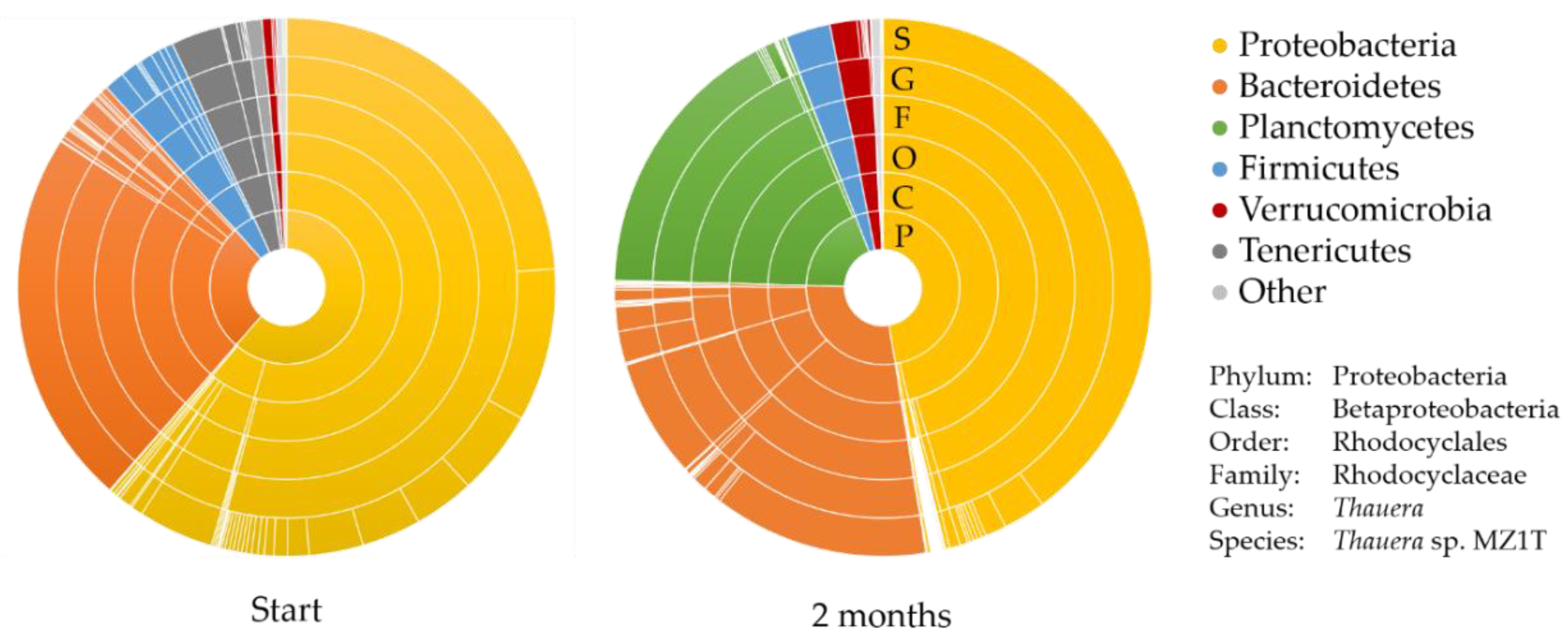
| Species | Phylum | Relative Abundance (%) | |
|---|---|---|---|
| 1 | Thauera sp. MZ1T * | Proteobacteria | 23.9 |
| 2 | Uncultured Haliscomenobacter sp. | Bacteroidetes | 22.7 |
| 3 | Uncultured Thauera sp. | Proteobacteria | 9.2 |
| 4 | Thauera aminoaromatica | Proteobacteria | 5.1 |
| 5 | Micavibrio aeruginosavorus arl_13 | Proteobacteria | 4.5 |
| 6 | Thauera aminoaromatica | Proteobacteria | 3.6 |
| 7 | Thauera phenylacetica | Proteobacteria | 3.6 |
| 8 | Uncultured Thauera | Proteobacteria | 3.3 |
| 9 | Uncultured Asteroleplasma sp. | Tenericutes | 3.1 |
| 10 | Uncultured Thauera sp. | Proteobacteria | 1.3 |
| Species | Phylum | Relative Abundance (%) | |
|---|---|---|---|
| 1 | Thauera sp. MZ1T * | Proteobacteria | 39.9 |
| 2 | Algisphaera agarilytica | Planctomycetes | 16.8 |
| 3 | Uncultured Ohtaekwangia sp. | Bacteroidetes | 13.0 |
| 4 | Uncultured Alkaliflexus sp. | Bacteroidetes | 7.1 |
| 5 | Uncultured Thermodesulfobium sp. | Firmicutes | 2.7 |
| 6 | Thauera aminoaromatica | Proteobacteria | 2.5 |
| 7 | Sediminibacterium salmoneum | Bacteroidetes | 1.9 |
| 8 | Prosthecobacter vanneervenii str. dsm 12252 | Verrucomicrobia | 1.6 |
| 9 | Uncultured ferruginibacter sp. | Bacteroidetes | 1.4 |
| 10 | Thauera aminoaromatica | Proteobacteria | 1.3 |
| Category | Genes | Description and Function | NCBI/GenBank Code C. necator H16 [41] | NCBI/GenBank Code T. aminoaromatica MZ1T |
|---|---|---|---|---|
| Synthesis | phaA | Acetyl-CoA acetyltransferase | CAJ92573.1 | ACK53504.1 ACK53575.1 ACK53579.1 ACR01093.1 |
| phaB | Acetoacetyl-CoA reductase | CAJ92574.1 | ACK53788.1 ACR01719.1 | |
| phaC | PHA synthase | CAJ92572.1 CAJ93103.1 | ACK53500.1 Class I ACK53786.1 Domain protein ACK53908.1 Domain protein | |
| Surface proteins | phaP | Phasin | CAJ92517.1 | ACR02450.1 ACR01124.1 ACK54768.1 ACK54704.1 ACK53642.1 |
| Gene regulation | phaR | PHA repressor, regulates phasin expression | CAJ92575.1 | ACR01718.1 |
| Degradation | phaZ | PHA depolymerase | CAJ92291.1 | ACK52971.1 PHA depolymerase ACK53308.1 Esterase |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Colpa, D.I.; Zhou, W.; Wempe, J.P.; Tamis, J.; Stuart, M.C.A.; Krooneman, J.; Euverink, G.-J.W. Thauera aminoaromatica MZ1T Identified as a Polyhydroxyalkanoate-Producing Bacterium within a Mixed Microbial Consortium. Bioengineering 2020, 7, 19. https://doi.org/10.3390/bioengineering7010019
Colpa DI, Zhou W, Wempe JP, Tamis J, Stuart MCA, Krooneman J, Euverink G-JW. Thauera aminoaromatica MZ1T Identified as a Polyhydroxyalkanoate-Producing Bacterium within a Mixed Microbial Consortium. Bioengineering. 2020; 7(1):19. https://doi.org/10.3390/bioengineering7010019
Chicago/Turabian StyleColpa, Dana I., Wen Zhou, Jan Pier Wempe, Jelmer Tamis, Marc C. A. Stuart, Janneke Krooneman, and Gert-Jan W. Euverink. 2020. "Thauera aminoaromatica MZ1T Identified as a Polyhydroxyalkanoate-Producing Bacterium within a Mixed Microbial Consortium" Bioengineering 7, no. 1: 19. https://doi.org/10.3390/bioengineering7010019
APA StyleColpa, D. I., Zhou, W., Wempe, J. P., Tamis, J., Stuart, M. C. A., Krooneman, J., & Euverink, G.-J. W. (2020). Thauera aminoaromatica MZ1T Identified as a Polyhydroxyalkanoate-Producing Bacterium within a Mixed Microbial Consortium. Bioengineering, 7(1), 19. https://doi.org/10.3390/bioengineering7010019