Towards Bioengineered Liver Stem Cell Transplantation Studies in a Preclinical Dog Model for Inherited Copper Toxicosis
Abstract
1. Introduction
2. A Roadmap towards a Relevant Preclinical Model Animal for Liver Stem Cell Transplantations
2.1. Requirement 1. Copper Accumulation in Bedlington Terriers Is caused by a Deletion of exon-2 in the COMMD1-gene
2.2. Requirement 2. Longitudinal Studies on COMMD1 Deficient Dogs Highlight Similarities between WD and Canine Copper Toxicosis
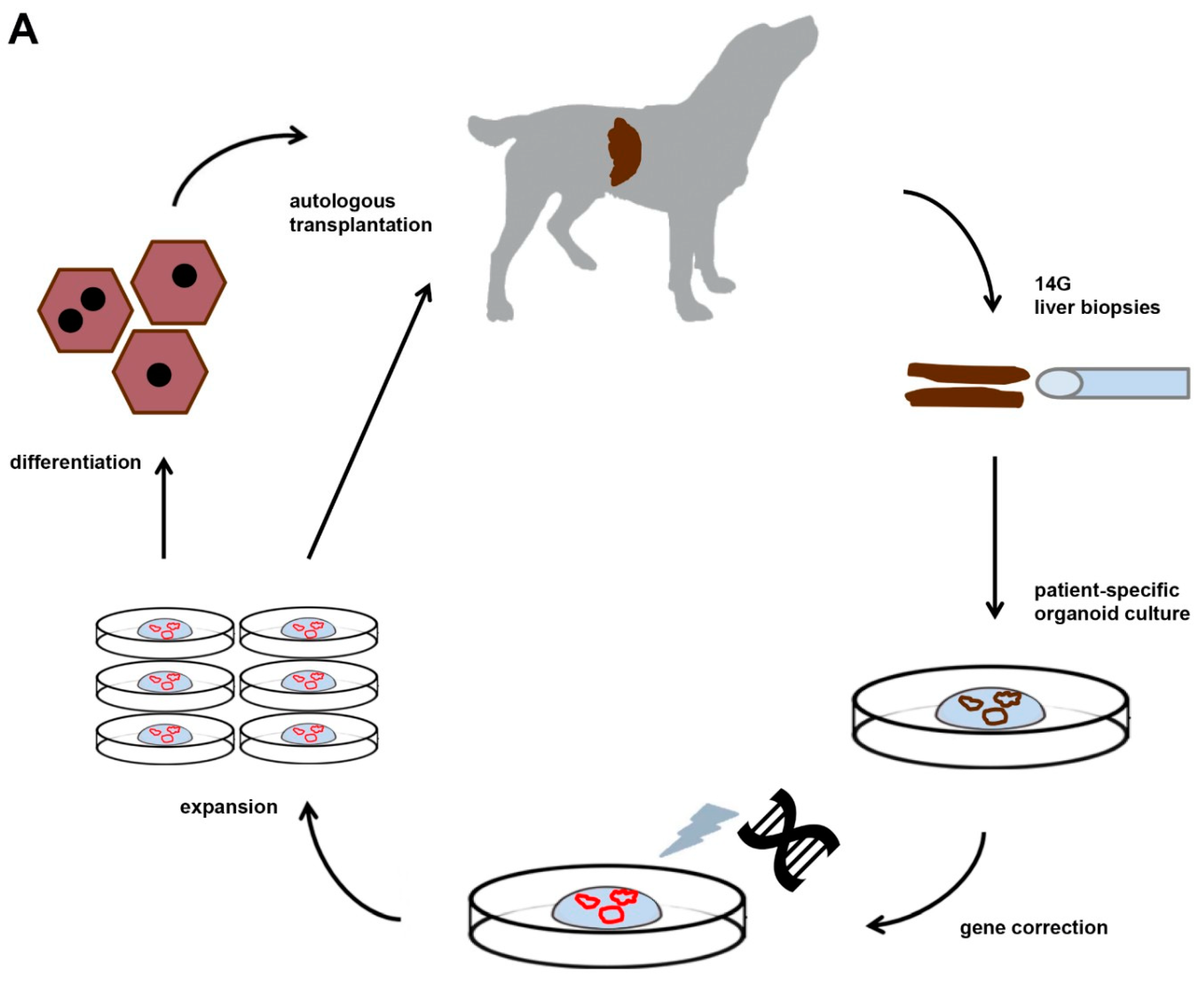
2.3. Requirement 3. Culture of Sufficient Quantities of COMMD1-Functional Autologous Liver Stem Cells
2.4. Requirement 4. Number and Routing of Genetically-Engineered Autologous Transplanted Donor Cells
3. Transplantation of Autologous COMMD1-Positive Liver Organoids into Copper-Laden Livers of COMMD1 Deficient Dogs
4. Bedlington Terriers with a commd1 Mutation or Labrador Retrievers with an ATPB Mutation, Which Is the Preferred Breed to Study WD?
5. Future of Novel Preclinical Models, DoGtor Can You Help Me?
Author Contributions
Funding
Conflicts of Interest
References
- Inesi, G. Molecular features of copper binding proteins involved in copper homeostasis. Critical review. sometime be replace by from pipets-2-pets. In remains to be seen how the rational combination of IUBMB Life. IUBMB Life 2017, 69, 211–217. [Google Scholar]
- Kim, B.E.; Nevitt, T.; Thiele, D.J. Mechanisms for copper acquisition, distribution and regulation. Nat. Chem. Biol. 2008, 4, 176–185. [Google Scholar] [PubMed]
- Zhou, B.; Gitschier, J. hCTR1: A human gene for copper uptake identified by complementation in yeast. Proc. Natl. Acad. Sci. USA 1997, 94, 7481–7486. [Google Scholar] [CrossRef] [PubMed]
- Lutsenko, S.; Barnes, N.L.; Bartee, M.Y.; Dmitriev, O.Y. Function and regulation of human copper-transporting ATPases. Physiol. Rev. 2007, 87, 1011–1046. [Google Scholar]
- Tanner, M.S. Role of copper in Indian Childhood Cirrhosis. Am. J. Clin. Nutr. 1998, 67, S1074–S1081. [Google Scholar] [CrossRef] [PubMed]
- Muller, T.; Feichtinger, H.; Berger, H.; Muller, W. Endemic Tyrolean Infantile Cirrhosis: An ecogenetic disorder. Lancet 1996, 347, 877–880. [Google Scholar] [PubMed]
- Scheinberg, I.C.; Sternlieb, I. Wilson Disease and Idiopathic Copper Toxicosis. Am. J. Cin. Nutr. 1996, 63, S842–S845. [Google Scholar]
- Bull, P.C.; Thomas, G.R.; Rommens, J.M.; Forbers, J.R.; Cox, D.W. The Wilson disease gene is a putative copper transporting P-type ATPase similar to the Menkes gene. Nat. Genet. 1993, 5, 327–337. [Google Scholar] [CrossRef]
- Tanzi, R.E.; Petrukhin, K.; Chernov, I.; Pelleguer, J.L.; Wasco, W.; Ross, B.; Romano, D.M.; Parano, E.; Pavone, L.; Bzustowicz, L.M.; et al. The Wilson disease gene is a copper transporting ATPase with homology t the Menkes disease gene. Nat. Genet. 1993, 5, 344–350. [Google Scholar] [CrossRef]
- Ferenci, P. Phenotype-genotype correlations in patients with Wilson’s disease. Ann. N. Y. Acad. Sci. 2014, 1315, 1–5. [Google Scholar]
- Lutsenko, S. Modifying factors and phenotypic diversity in Wilson’s disease. Ann. N. Y. Acad. Sci. 2014, 1315, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Medici, V.; Weiss, K.H. Genetic and environmental modifiers of Wilson disease. In Handbook of Clinical Neurology; Elsevier: Amsterdam, the Netherlands, 2017; Volume 142, pp. 35–41. [Google Scholar]
- Chelly, J.; Tumer, Z.; Tonnesen, T.; Petteson, A.; Ishikawa-Brush, Y.; Tommerup, N.; Norn, N.; Monaco, A.P. Isolation of a candidate gene for Menkes disease that encodes a potential heavy metal binding protein. Nat. Genet. 1993, 3, 14–19. [Google Scholar] [PubMed]
- Mercer, J.F.B.; Livingston, J.; Hall, B.; Paynter, J.A.; Begy, C.; Chandrasekharappa, S.; Lockhart, P.; Grimes, A.; Bhave, M.; Siemieniak, D.; et al. Isolation of a partial candidate gene for Menkes disease by positional cloning. Nat. Genet. 1993, 3, 20–25. [Google Scholar] [PubMed]
- Vulpe, C.; Levinson, B.; Whitney, S.; Packman, S.; Gitschier, J. Isolation of a candidate gene for Menkes disease and evidence that it encodes a copper-transporting ATPase. Nat. Genet. 1993, 3, 7–13. [Google Scholar] [PubMed]
- Moller, L.B.; Mogensen, M.; Horn, N. Molecular diagnosis of Menkes disease: Genotype-phenotype correlation. Biochimie 2009, 91, 1273–1277. [Google Scholar] [CrossRef] [PubMed]
- Tumer, Z. An overview and update of ATP7A mutations leading to Menkes disease and occipital horn syndrome. Hum. Mutat. 2013, 34, 417–429. [Google Scholar] [PubMed]
- Larson, G.; Karlsson, E.K.; Perri, A.; Webster, M.T.; Ho, S.Y.; Peters, J.; Stahl, P.W.; Lingaas, F.; Fredholm, M.; Comstock, K.E.; et al. Rethinking dog domestication by integrating genetics, archeology, and biogeography. Proc. Natl. Acad. Sci. USA 2012, 109, 8878–8883. [Google Scholar] [CrossRef]
- Parker, H.G.; Shearin, A.L.; Ostrander, E.A. Man’s best friend becomes biology’s best in show: Genome analyses in the domestic dog. Annu. Rev. Genet. 2010, 44, 309–336. [Google Scholar]
- Starzl, T.E.; Kaupp, H.A., Jr.; Brock, D.R.; Linman, J.W. Studies on the rejection of the transplanted homologous dog liver. Surg. Gynecol. Obstet. 1961, 112, 135–144. [Google Scholar]
- Haywood, S.; Muller, T.; Muller, W.; Heinz-Erian, P.; Tanner, M.S.; Ross, G. Copper-associated liver disease in north ronaldsay sheep: A possible animal model for non-wilsonian hepatic copper toxicosis of infancy and childhood. J. Pathol. 2001, 195, 264–269. [Google Scholar] [CrossRef]
- Fuentalba, I.C.; Aburto, E.M. Animal models of copper-associated liver disease. Comp. Hepatol. 2003, 2, 5. [Google Scholar] [CrossRef]
- Fieten, H.; Penning, L.C.; Leegwater, P.A.; Rothuizen, J. New canine models of copper toxicosis: Diagnosis, treatment, and genetics. Ann. N. Y. Acad. Sci. 2014, 1314, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Reed, E.; Lutsenko, S.; Bandmann, O. Animal models of Wilson disease. J. Neurochem. 2018, 146, 356–373. [Google Scholar] [CrossRef] [PubMed]
- Twedt, D.C.; Sternlieb, I.; Gilbertson, S.R. Clinical, morphologic, and chemical studies on copper toxicosis of Bedlington Terriers. J. Am. Vet. Med. Assoc. 1979, 175, 269–275. [Google Scholar] [PubMed]
- Haywood, S.; Rutgers, H.C.; Christian, M.K. Hepatitis and copper accumulation in Skye terriers. Vet. Pathol. 1988, 25, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Thornburg, L.P.; Rottinghaus, G.; Dennis, G.; Crawford, S. The relationship between hepatic copper content and morphologic changes in the liver of West Highland White Terriers. Vet. Pathol. 1996, 33, 656–661. [Google Scholar] [CrossRef]
- Thornburg, L.P. Histomorphological and immunohistochemical studies of chronic active hepatitis in Doberman Pinschers. Vet. Pathol. 1998, 35, 380–385. [Google Scholar] [CrossRef]
- Webb, C.B.; Twedt, D.C.; Meyer, D.J. Copper-associated liver disease in Dalmatians: A review of 10 dogs (1998-2001). J. Vet. Intern. Med. 2002, 16, 665–668. [Google Scholar]
- Hoffmann, G.; van den Ingh, T.S.; Bode, P.; Rothuizen, J. Copper-associated chronic hepatitis in Labrador Retrievers. J. Vet. Intern. Med. 2006, 20, 856–861. [Google Scholar] [CrossRef]
- Van De Sluis, B.; Rothuizen, J.; Pearson, P.L.; van Oost, B.A.; Wijmenga, C. Identification of a new copper metabolism gene by positional cloning in a purebred dog population. Hum. Mol. Genet. 2002, 11, 165–173. [Google Scholar] [CrossRef]
- Van de Sluis, B.J.; Breen, M.; Nanji, M.; van Wolferen, M.; de Jong, P.; Binns, M.M.; Pearson, P.L.; Kuipers, J.; Rothuizen, J.; Cox, D.W.; et al. Genetic mapping of the copper toxicosis locus in Bedlington terriers to dog chromosome 10, in a region syntenic to human chromosome region 2p13-p16. Hum. Mol. Genet. 1999, 8, 501–507. [Google Scholar] [CrossRef]
- Müller, T.; van de Sluis, B.; Zhernakova, A.; van Binsbergen, E.; Janecke, A.R.; Bavdekar, A.; Pandit, A.; Weirich-Schwaiger, H.; Witt, H.; Ellemunter, H.; et al. The canine copper toxicosis gene MURR1 does not cause non-Wilsonian hepatic copper toxicosis. J. Hepatol. 2003, 38, 164–168. [Google Scholar] [PubMed]
- Stuehler, B.; Reichert, J.; Stremmel, W.; Schaefer, M. Analysis of the human homologue of the canine copper toxicosis gene MURR1 in Wilson disease patients. J. Mol. Med. 2004, 82, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Lovicu, M.; Dessi, V.; Lepori, M.B.; Zappu, A.; Zancan, L.; Giacchino, R.; Marazzi, M.G.; Iorio, R.; Vegnente, A.; Vajro, P.; et al. The canine copper toxicosis gene MURR1 is not implicated in the pathogenesis of Wilson disease. J. Gastroenterol. 2006, 41, 582–586. [Google Scholar] [PubMed]
- Wu, Z.Y.; Zhao, G.X.; Chen, W.J.; Wang, N.; Wan, B.; Lin, M.T.; Murong, S.X.; Yu, L. Mutation analysis of 218 Chinese patients with Wilson diseases revealed no correlation between the canine copper toxicosis gene MURR1 And Wilson disease. J. Mol. Med. 2006, 84, 438442. [Google Scholar] [CrossRef] [PubMed]
- De Bie, P.; van de Sluis, B.; Klomp, L.; Wijmenga, C. The many faces of the copper metabolism protein MURR1/COMMD1. J. Hered. 2005, 97, 803–811. [Google Scholar] [CrossRef]
- Maine, G.N.; Burstein, E. COMMD proteins: COMMing to the scene. Cell. Mol. Life Sci. 2007, 64, 1997–2005. [Google Scholar]
- Fedoseienko, A.; Bartuzi, P.; van de Sluis, B. Functional understanding of the versatile protein copper metabolism MURR1 domain 1 (COMMD1) in copper homeostasis. Ann. N. Y. Acad. Sci. 2014, 1314, 6–14. [Google Scholar]
- Riera-Romo, M. COMMD1: A Multifunctional Regulatory Protein. J. Cell. Biochem. 2018, 119, 34–51. [Google Scholar]
- Ganesh, L.; Burstein, E.; Guha-Niyogi, A.; Louder, M.K.; MAscola, J.R.; Klomp, L.W.; Wijmenga, C.; Duckett, C.S.; Nabel, G.J. The gene product Murr1 restricts HIV-1 replication in resting CD4+ lymphocytes. Nature 2003, 426, 853–857. [Google Scholar]
- Bartuzi, P.; Billadeau, D.D.; Favier, R.; Rong, S.; Dekker, D.; Fedoseienko, A.; Fieten, H.; Wijers, M.; Levels, J.H.; Huijkman, N.; et al. CCC- and WASH-mediated endosomal sorting of LDLR is required for normal clearance of circulating LDL. Nat. Commun. 2016, 7, 10961. [Google Scholar] [CrossRef] [PubMed]
- Spee, B.; Arends, B.; van Wees, A.M.; Bode, P.; Penning, L.C.; Rothuizen, J. Functional consequences of RNA interference targeting COMMD1 in a canine hepatic cell line in relation to copper toxicosis. Anim. Genet. 2007, 38, 168–170. [Google Scholar] [PubMed]
- Vonk, W.I.; Bartuzi, P.; de Bie, P.; Kloosterhuis, N.; Wichers, C.G.; Berger, R.; Haywood, S.; Klomp, L.W.; Wijmenga, C.; van de Sluis, B. Liver-specific Commd1 knockout mice are susceptible to hepatic copper accumulation. PLoS ONE 2011, 6, e29183. [Google Scholar]
- De Bie, P.; van de Sluis, B.; Burstein, E.; van den Berghe, P.V.; Muller, P.; Berger, R.; Gitlin, J.D.; Wijmenga, C.; Klomp, L.W. Distinct Wilson’s disease mutations in ATP7B are associated with enhanced binding to COMMD1 and reduced stability of ATP7B. Gastroenterology 2007, 133, 1316–1326. [Google Scholar] [CrossRef] [PubMed]
- Weiss, K.H.; Lozoya, J.C.; Tuma, S.; Gotthardt, D.; Reichert, J.; Ehehalt, R.; Stremmel, W.; Fullekrug, J. Copper-induced translocation of the Wilson disease protein ATP7B independent of Murr1/COMMD1 and Rab7. Am. J. Pathol. 2008, 173, 1783–1794. [Google Scholar] [CrossRef] [PubMed]
- Vonk, W.I.; de Bie, P.; Wichers, C.G.; van den Berghe, P.V.; van der Plaats, R.; Berger, R.; Wijmenga, C.; Klomp, L.W.; van de Sluis, B. The copper-transporting capacity of ATP7A mutants associated with Menkes disease is ameliorated by COMMD1 as a result of improved protein expression. Cell. Mol. Life Sci. 2012, 69, 149–163. [Google Scholar] [PubMed]
- Favier, R.P.; Spee, B.; Penning, L.C.; Rothuizen, J. Copper-induced hepatitis: The COMMD1 deficient dog as a translational animal model for human chronic hepatitis. Vet. Q. 2011, 31, 49–60. [Google Scholar]
- Favier, R.P.; Spee, B.; Schotanus, B.A.; van den Ingh, T.S.; Fieten, H.; Brinkhof, B.; Viebahn, C.S.; Penning, L.C.; Rothuizen, J. COMMD1-deficient dogs accumulate copper in hepatocytes and provide a good model, for chronic hepatitis and fibrosis. PLoS ONE 2012, 7, e42158. [Google Scholar] [CrossRef]
- Favier, R.P.; Spee, B.; Fieten, H.; van den Ingh, T.S.; Schotanus, B.A.; Brinkhof, B.; Rothuizen, J.; Penning, L.C. Aberrant expression of copper associated genes after copper accumulation in COMMD1-deficient dogs. J. Trace Elem. Med. Biol. 2015, 29, 347–353. [Google Scholar] [CrossRef]
- Schaub, J.R.; Malato, Y.; Gormond, C.; Willenbring, H. Evidence against a stem cell origin of new hepatocytes in a common mouse model of chronic liver injury. Cell Rep. 2014, 8, 933–939. [Google Scholar] [CrossRef]
- Yanger, K.; Knigin, D.; Zong, Y.; Maggs, L.; Gu, G.; Akiyama, H.; Pikarsky, E.; Stanger, B.Z. Adults hepatocytes are generated by self-duplication rather than stem cell differentiation. Cell Stem Cell 2014, 15, 340–349. [Google Scholar]
- Kordes, C.; Sawitza, I.; Goetze, S.; Herebian, D.; Haussinger, D. Hepatic stellate cells contribute to progenitor cells and liver regeneration. J. Clin. Investig. 2014, 124, 5503–5515. [Google Scholar] [CrossRef] [PubMed]
- Tarlow, B.D.; Pelz, C.; Naugler, W.E.; Wakefield, L.; Wilson, E.M.; Finegold, M.J.; Grompe, M. Bipotent adult liver progenitors are derived from chronically injured mature hepatocytes. Cell Stem Cell 2014, 15, 605–618. [Google Scholar] [PubMed]
- Wang, B.; Zhao, L.; Fish, M.; Logan, C.Y.; Nusse, R. Self-renewing diploid axin2+ cells fuel homeostatic renewal of the liver. Nature 2015, 524, 180–185. [Google Scholar] [PubMed]
- Huch, M.; Dolle, L. The plastic cellular states of liver cells: Are EpCAM and Lgr5 fit for purpose? Hepatology 2016, 64, 652–662. [Google Scholar]
- Huch, M. The versatile and plastic liver. Nature 2015, 517, 155–156. [Google Scholar] [PubMed]
- De Vos, R.; Desmet, V. Ultrastructural characteristics of novel epithelial cell types identified in human pathologic liver specimens with chronic ductular reaction. Am. J. Pathol. 1992, 140, 1441–1450. [Google Scholar] [PubMed]
- Hsia, C.C.; Evarts, R.P.; Nakatsukasa, H.; Marsden, E.R.; Thorgeirsson, S.S. Occurrence of oval-type cells in hepatitis B virus-associated human hepatocarcinogenesis. Hepatology 1992, 16, 1327–1333. [Google Scholar]
- Roskams, T.; de Vos, R.; Desmet, V. “Undifferentiated progenitor cells” in focal nodular hyperplasia of the liver. Histopathology 1996, 28, 291–299. [Google Scholar] [CrossRef]
- Crosby, H.A.; Hubscher, S.; Fabris, L.; Joplin, R.; Sell, S.; Kelly, D.; Strain, A.J. Immunolocalization of putative human liver progenitor cells in livers from patients with end-stage primary biliary cirrhosis and sclerosing cholangitis using the monoclonal antibody OV-6. Am. J. Pathol. 1998, 152, 771–779. [Google Scholar]
- Lowes, K.N.; Brennan, B.A.; Yeoh, G.C.; Olynyk, J.K. Oval cell numbers in human chronic liver diseases are directly related to disease severity. Am. J. Pathol. 1999, 154, 537–541. [Google Scholar] [PubMed]
- Libbrecht, L.; Desmet, V.; Van Damme, B.; Roskams, T. The immunohistochemical phenotype of dysplastic foci in human liver: Correlation with putative progenitor cells. J. Hepatol. 2000, 33, 76–84. [Google Scholar] [CrossRef]
- Schotanus, B.A.; van den Ingh, T.S.; Penning, L.C.; Rothuizen, J.; Roskams, T.A.; Spee, B. Cross-species immunohistochemical investigation of the activation of the liver progenitor cell niche in different types of liver disease. Liver Int. 2009, 29, 1241–1252. [Google Scholar] [CrossRef] [PubMed]
- Ijzer, J.; Schotanus, B.A.; Vander Borght, S.; Roskams, T.A.; Kisjes, R.; Penning, L.C.; Rothuizen, J.; van den Ingh, T.S. Characterisation of the hepatic progenitor cell compartment in normal liver and in hepatitis: An immunohistochemical comparison between dog and man. Vet. J. 2010, 184, 308–314. [Google Scholar] [PubMed]
- Kruitwagen, H.S.; Spee, B.; Viebahn, C.S.; Venema, H.B.; Penning, L.C.; Grinwis, G.C.; Favier, R.P.; van den Ingh, T.S.; Rothuizen, J.; Schotanus, B.A. The canine hepatic progenitor cell niche: Molecular characterisation in health and disease. Vet. J. 2014, 201, 345–532. [Google Scholar] [CrossRef] [PubMed]
- Kruitwagen, H.S.; Spee, B.; Fieten, H.; van Steenbeek, F.G.; Schotanus, B.A.; Penning, L.C. Translation from mice to men: Are dogs a dodgy intermediate? Eur. Med. J. Hepatol. 2014, 1, 48–54. [Google Scholar]
- Arends, B.; Vankelecom, H.; Vander Borght, S.; Roskams, T.; Penning, L.C.; Rothuizen, J.; Spee, B. The dog liver contains a “side population” of cells with hepatic progenitor-like characteristics. Stem Cells Dev. 2009, 18, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Arends, B.; Spee, B.; Schotanus, B.A.; Roskams, T.; van den Ingh, T.S.; Penning, L.C.; Rothuizen, J. In vitro differentiation of liver progenitor cells derived from healthy dog livers. Stem Cells Dev. 2009, 18, 351–358. [Google Scholar] [PubMed]
- Kruitwagen, H.S.; Westendorp, B.; Viebahn, C.S.; Post, K.; van Wolferen, M.E.; Oosterhoff, L.A.; Egan, D.A.; Delabar, J.M.; Toussaint, M.J.; Schotanus, B.A.; et al. DYRK1A is a regulator of S-phase entry in hepatic progenitor cells. Stem Cell Dev. 2018, 27, 133–146. [Google Scholar]
- Huch, M.; Dorrell, C.; Boj, S.F.; van Es, J.H.; Li, V.S.; van de Wetering, M.; Sato, T.; Hamer, K.; Sasaki, N.; Finegold, M.J.; et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature 2013, 494, 247–250. [Google Scholar] [CrossRef]
- Huch, M.; Gehart, H.; van Boxtel, R.; Hamer, K.; Blokzijl, F.; Verstegen, M.M.A.; Ellis, E.; van Wenum, M.; Fuchs, S.A.; de Ligt, J.; et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 2015, 160, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Nantasanti, S.; Spee, B.; Kruitwagen, H.S.; Chen, C.; Geijsen, N.; Oosterhoff, L.A.; van Wolferen, M.E.; Palaez, N.; Fieten, H.; Wubbolts, R.W.; et al. Disease Modeling and Gene Therapy of Copper Storage Disease in Canine Hepatic Organoids. Stem Cell Rep. 2015, 5, 895–907. [Google Scholar] [CrossRef] [PubMed]
- Kuijk, E.W.; Rasmussen, S.; Blokzijl, F.; Huch, M.; Gehart, H.; Toonen, P.; Begthel, H.; Clevers, H.; Geurts, A.M.; Cuppen, E. Generation and characterization of rat liver stem cell lines and their engraftment in a rat model of liver failure. Sci. Rep. 2016, 6, 22154. [Google Scholar] [CrossRef] [PubMed]
- Kruitwagen, H.S.; Oosterhoff, L.A.; Vernooij, I.G.W.H.; Schrall, I.M.; van Wolferen, M.E.; Bannink, F.; Roesch, C.; van Uden, L.; Molenaar, M.R.; Helms, J.B.; et al. Long-term adult feline liver organoids cultures for disease modeling of hepatic steatosis. Stem Cell Rep. 2017, 8, 822–830. [Google Scholar]
- Kocken, J.M.; Borel Rinkes, I.H.; Bijma, A.M.; de Roos, W.K.; Bouwman, E.; Terpstra, O.T.; Sinaasappel, M. Correction of an inborn error of metabolism by intraportal hepatocyte transplantation in a dog model. Transplantation 1996, 62, 358–364. [Google Scholar] [CrossRef]
- Fox, I.J.; Chowdhury, J.R.; Kaufman, S.S.; Goertzen, T.C.; Chowdhury, N.R.; Warkentin, P.I.; Dorko, K.; Sauter, B.V.; Strom, S.C. Treatment of the Crigler-Najjar syndrome type I with hepatocyte transplantation. N. Eng. J. Med. 1998, 338, 1422–1426. [Google Scholar]
- Puppi, J.; Tan, N.; Mitry, R.R.; Highes, R.D.; Lehec, S.; Miele-Vergani, G.; Karani, J.; Champion, M.P.; Heaton, N.; Mohamed, R.; et al. Hepatocyte transplantation followed by auxiliary liver transplantation; a novel treatment of ornithine transcarbamylase deficiency. Am. J. Transplant. 2008, 8, 452–457. [Google Scholar] [CrossRef]
- Stephenne, X.; Najimi, M.; Sibille, C.; Nasogne, M.C.; Smets, F.; Sokal, E.M. Sustained engraftment and tissue enzyme activity after liver cell transplantation for arginosuccinate lyase deficiency. Gastroenterology 2006, 130, 137–1323. [Google Scholar] [CrossRef]
- Stephenne, X.; Debray, F.G.; Smets, F.; Jazouli, N.; Sana, G.; Tondreau, T.; Menten, R. Hepatocyte transplantation using the domino concept in a child with tetrabiopterin non responsive phenylketonuria. Cell Transplant. 2012, 21, 2765–2770. [Google Scholar]
- Guha, C.; Deb, N.J.; Sappal, B.S.; Ghosh, S.S.; Roy-Chowdhury, N.; Roy-Chowdhury, J. Amplification of engrafted hepatocytes by preparative manipulation of the hoist liver. Artif. Organs 2001, 25, 522–528. [Google Scholar] [CrossRef]
- Darwish, A.A.; Sokal, E.; Stephenne, X.; Najimi, M.; de Goyet, J.D.V.; Reding, R. Permanent access to the portal system for cellular transplantation using an implantable port device. Liver Transplant. 2004, 10, 1213–1215. [Google Scholar] [CrossRef] [PubMed]
- Fieten, H.; Biourge, V.C.; Watson, A.L.; Leegwater, P.A.; van den Ingh, T.S.; Rothuizen, J. Dietary management of Labrador retrievers with subclinical hepatic copper accumulation. J. Vet. Intern. Med. 2015, 29, 822–827. [Google Scholar] [CrossRef] [PubMed]
| WD | BT | LR | |
|---|---|---|---|
| Gene | ATP7B | COMMD1 | ATP7B/ATP7A |
| Mode of inheritance | autosomal recessive | autosomal recessive | complex |
| Age of onset | variable | adolescence-mid age | adolescence-mid age |
| Liver pathology | cirrhosis | cirrhosis | cirrhosis |
| Hepatic Cu (mg/dwl) | <1000 | <12,000 | <1000 |
| Neurology | impaired | not reported | not reported |
| Population | rare | rare * | very frequent |
| Kayser–Fleischer rings | present in 50% | not reported | not reported |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kruitwagen, H.S.; Fieten, H.; Penning, L.C. Towards Bioengineered Liver Stem Cell Transplantation Studies in a Preclinical Dog Model for Inherited Copper Toxicosis. Bioengineering 2019, 6, 88. https://doi.org/10.3390/bioengineering6040088
Kruitwagen HS, Fieten H, Penning LC. Towards Bioengineered Liver Stem Cell Transplantation Studies in a Preclinical Dog Model for Inherited Copper Toxicosis. Bioengineering. 2019; 6(4):88. https://doi.org/10.3390/bioengineering6040088
Chicago/Turabian StyleKruitwagen, Hedwig S., Hille Fieten, and Louis C. Penning. 2019. "Towards Bioengineered Liver Stem Cell Transplantation Studies in a Preclinical Dog Model for Inherited Copper Toxicosis" Bioengineering 6, no. 4: 88. https://doi.org/10.3390/bioengineering6040088
APA StyleKruitwagen, H. S., Fieten, H., & Penning, L. C. (2019). Towards Bioengineered Liver Stem Cell Transplantation Studies in a Preclinical Dog Model for Inherited Copper Toxicosis. Bioengineering, 6(4), 88. https://doi.org/10.3390/bioengineering6040088





