Corneal Epithelial–Stromal Fibroblast Constructs to Study Cell–Cell Communication in Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.1.1. Human Corneal Fibroblasts
2.1.2. Human Corneal Epithelial Cells
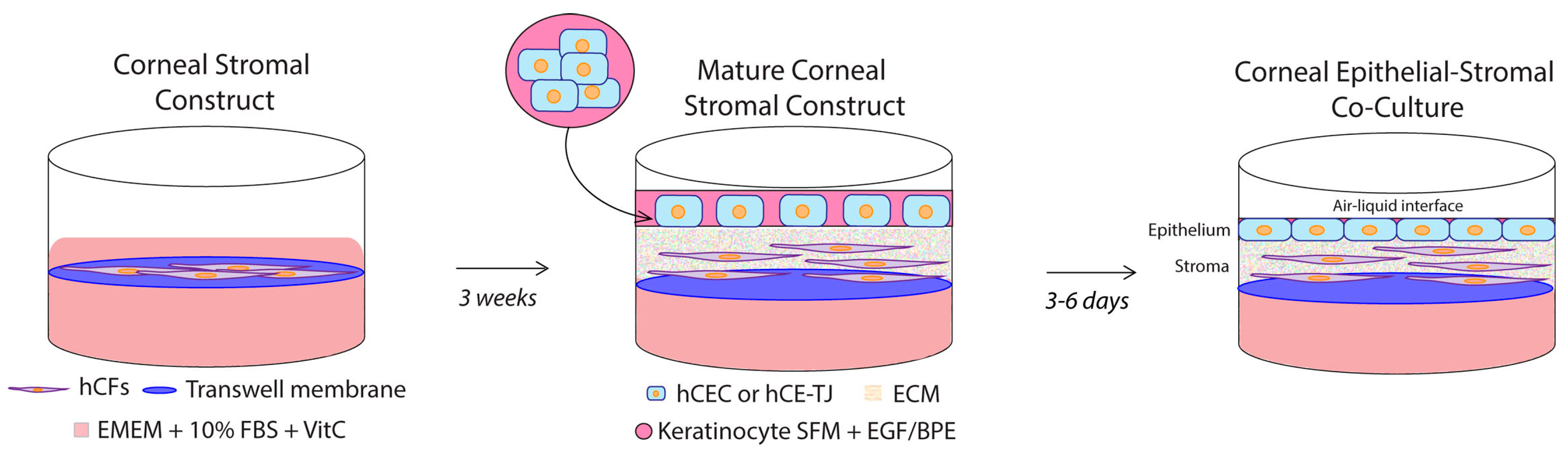
2.1.3. Three-Dimensional (3D) Corneal Stromal Constructs
2.1.4. Co-Culture of Corneal Epithelium and Stromal Fibroblasts
2.2. TEM
2.3. Immunohistochemistry
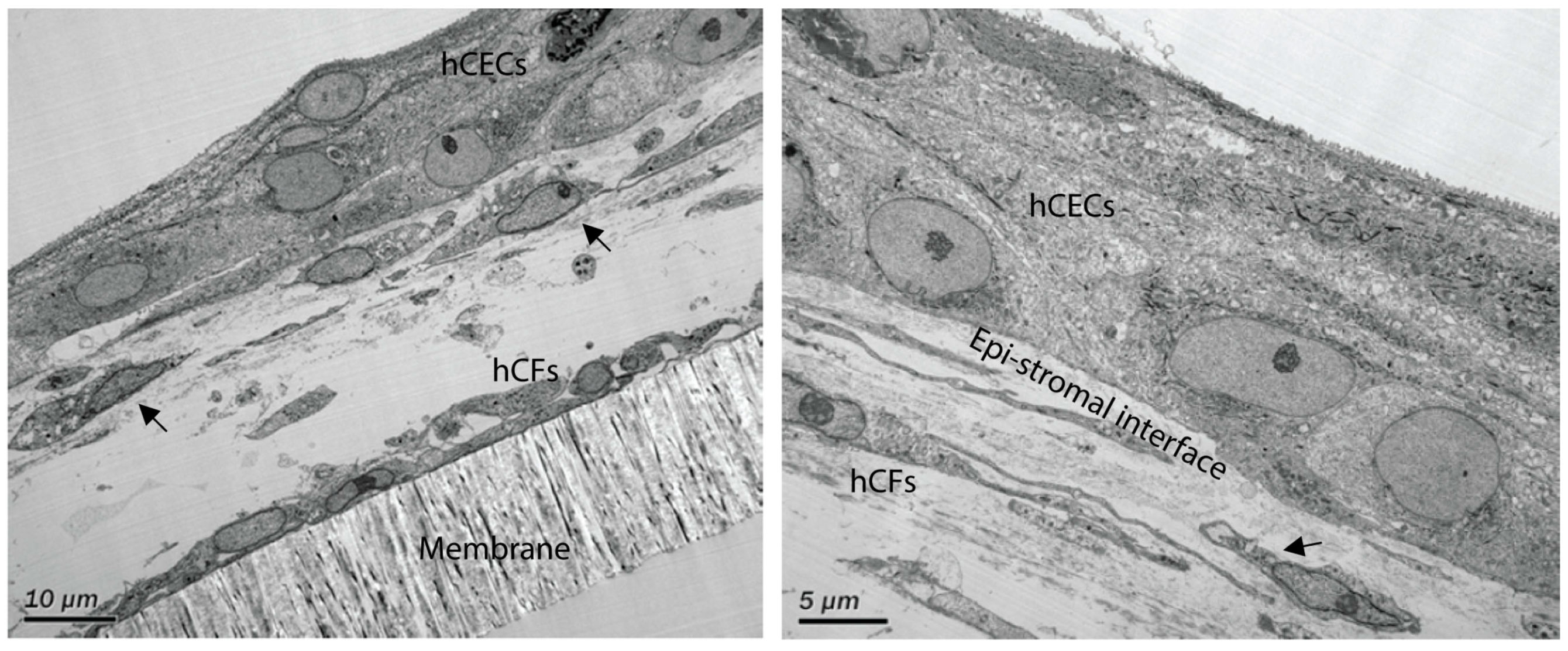
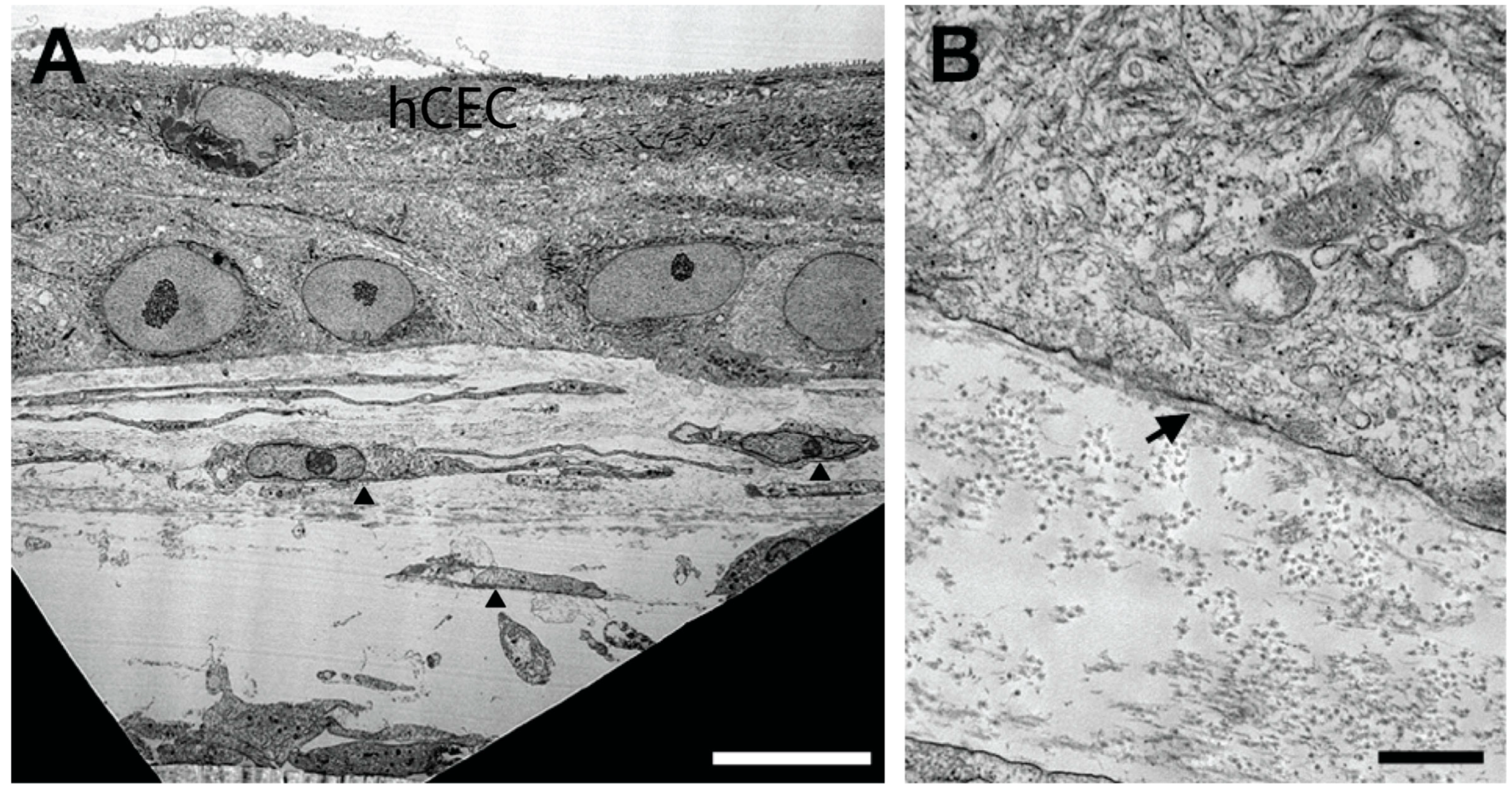
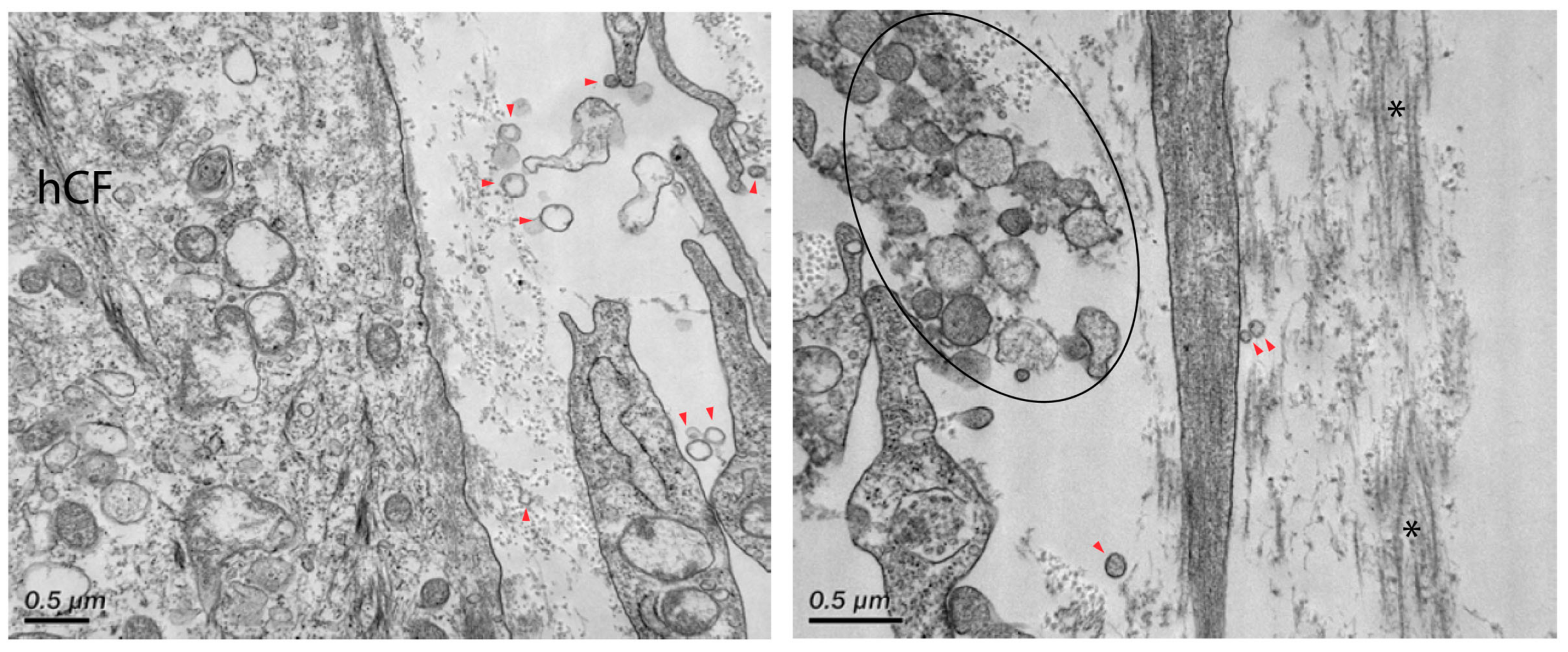
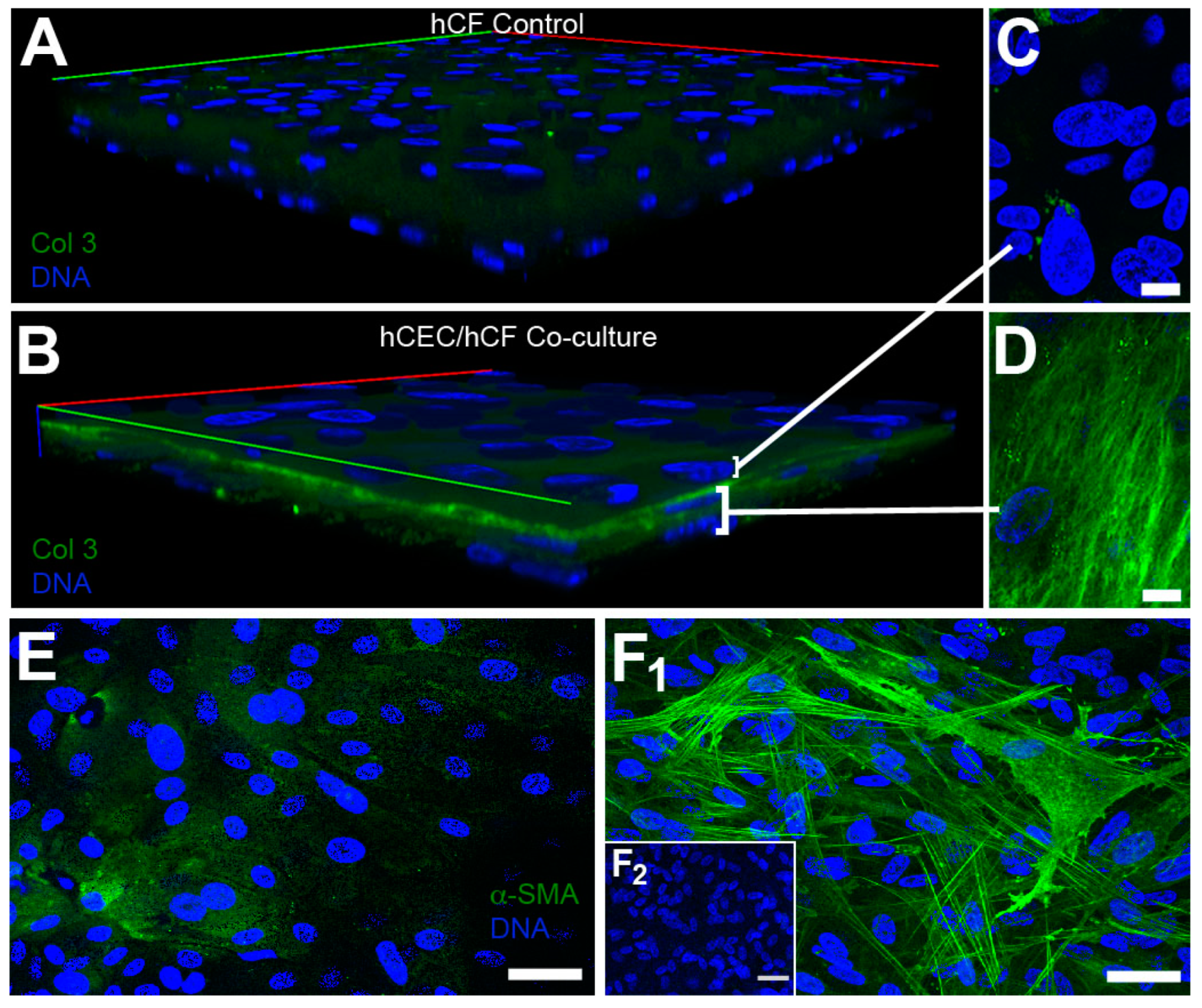
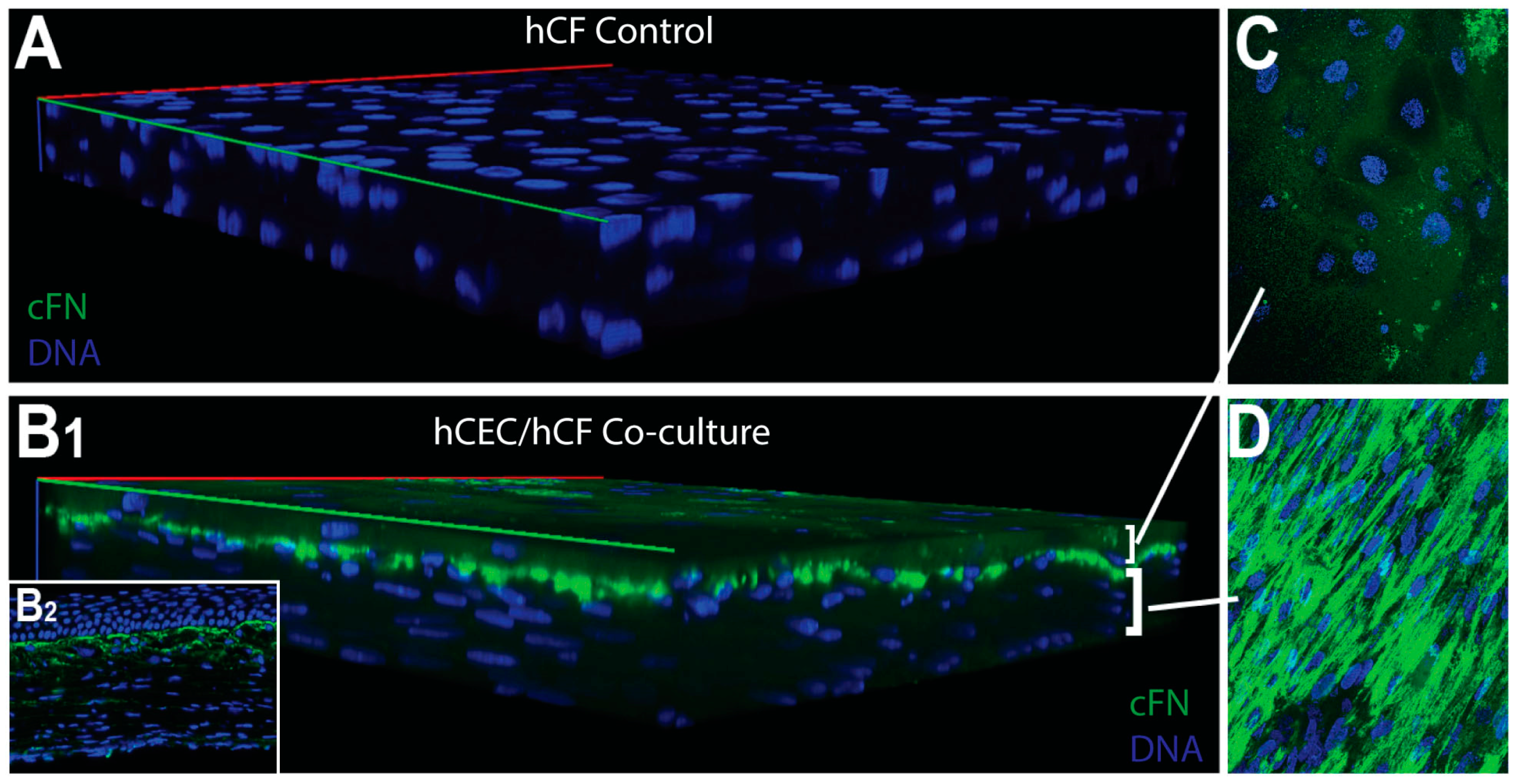
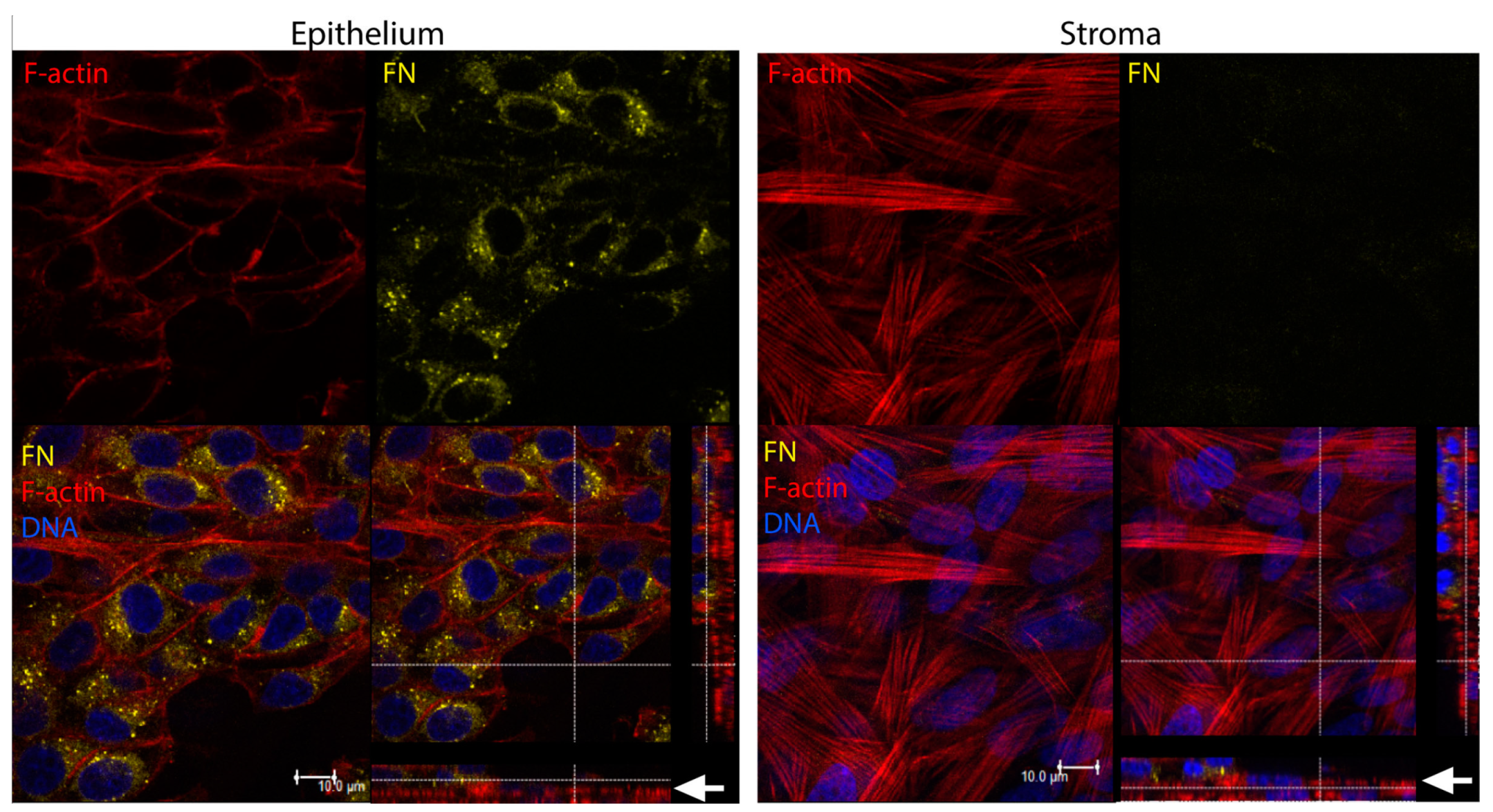
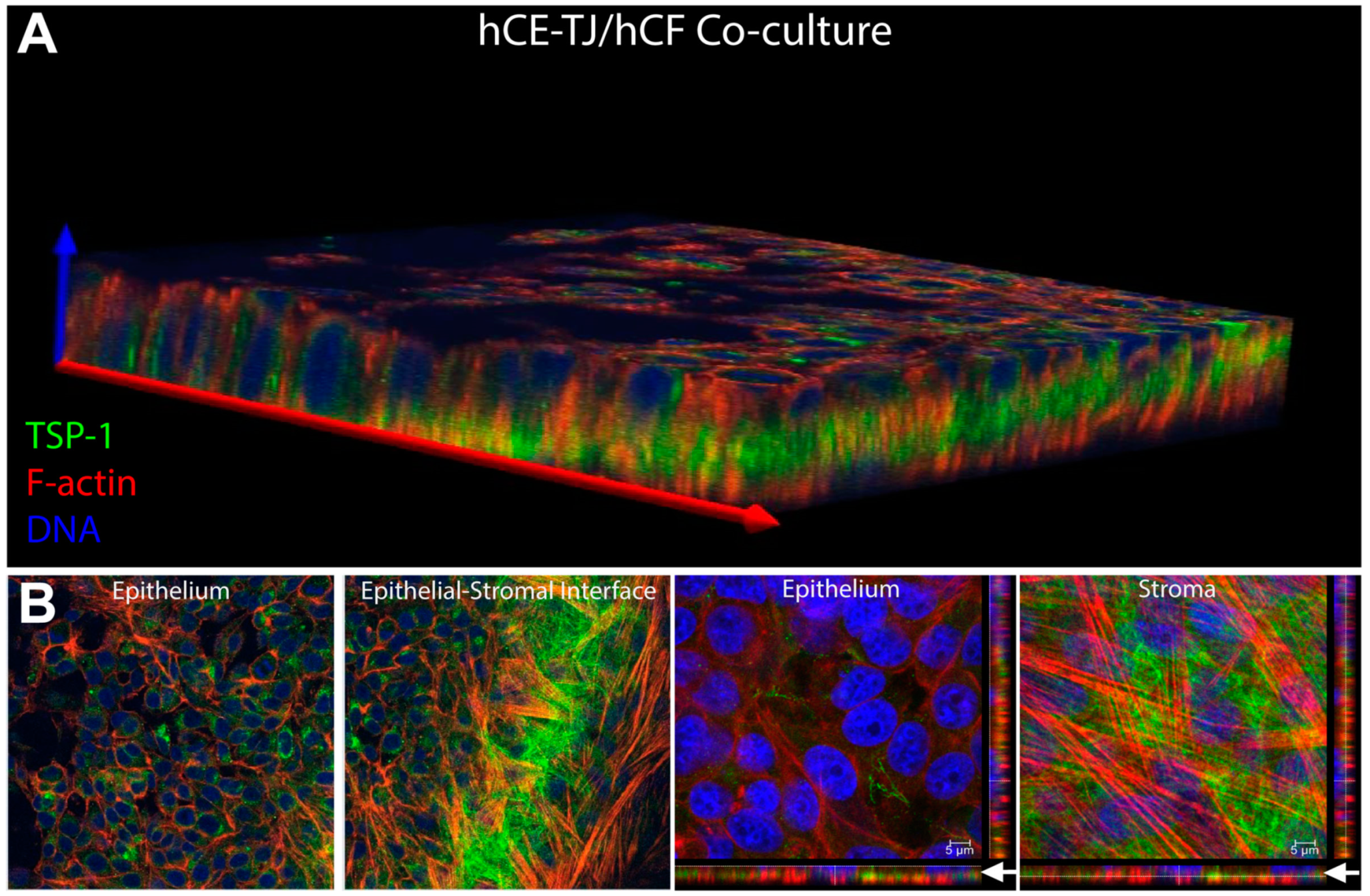
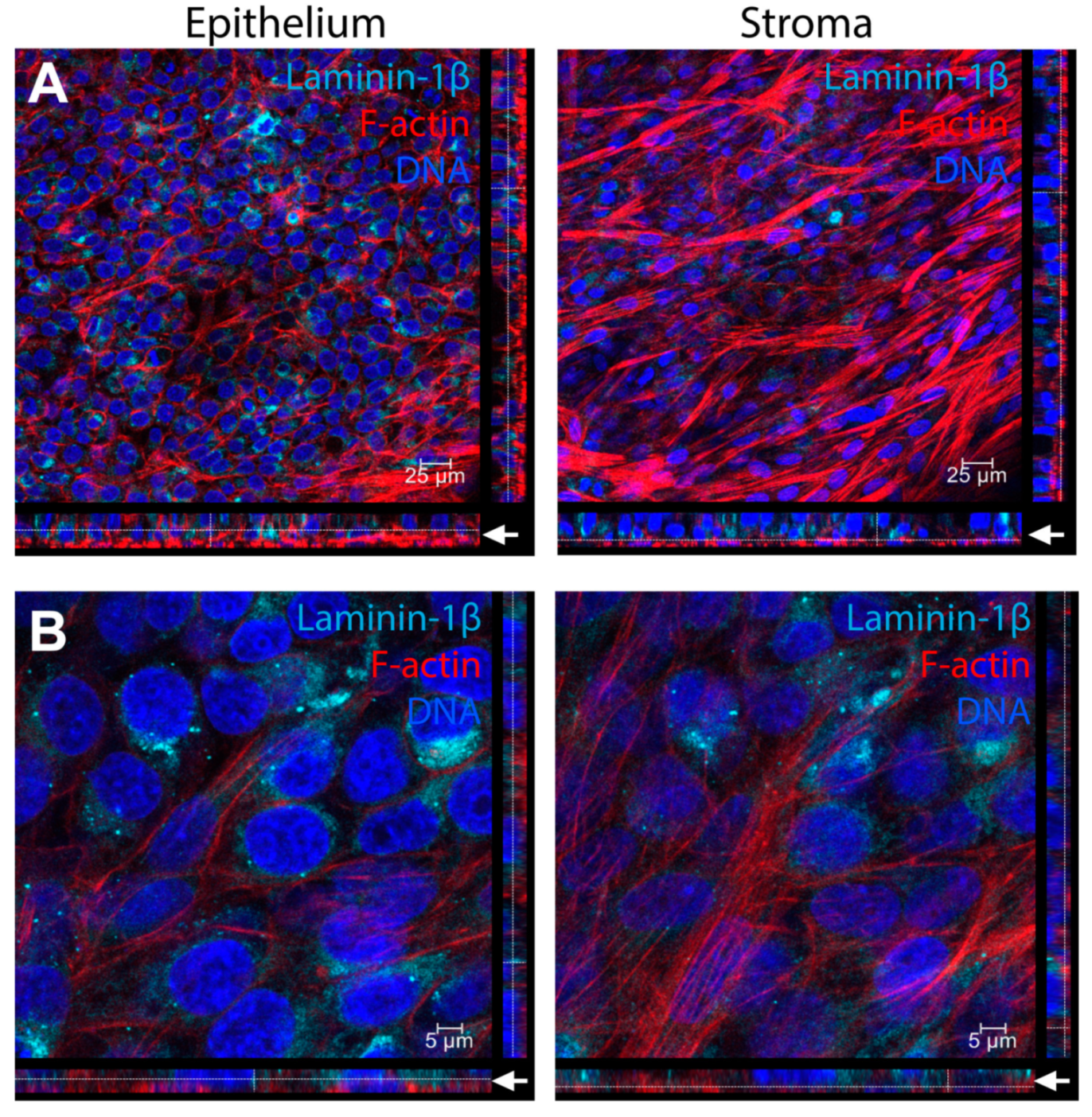
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Construct | Thickness |
|---|---|
| hCE-TJ/hCF co-culture | 21.54 ± 6.58 μm |
| Protein | Relative Fluorescence Intensity | |
|---|---|---|
| Epithelium | Stroma | |
| Fibronectin | 1346.99 ± 228.17 | 812.10 ± 422.80 |
| Thrombospondin-1 | 1477.24 ± 484.14 | 1200.52 ± 600.91 |
References
- Whitcher, J.P.; Srinivasan, M.; Upadhyay, M.P. Corneal blindness: A global perspective. Bull. World Health Organ. 2001, 79, 214–221. [Google Scholar] [PubMed]
- Marino, G.K.; Santhiago, M.R.; Torricelli, A.A.; Santhanam, A.; Wilson, S.E. Corneal Molecular and Cellular Biology for the Refractive Surgeon: The Critical Role of the Epithelial Basement Membrane. J. Refract. Surg. 2016, 32, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.E.; Marino, G.K.; Torricelli, A.A.M.; Medeiros, C.S. Injury and defective regeneration of the epithelial basement membrane in corneal fibrosis: A paradigm for fibrosis in other organs? Matrix Biol. J. Int. Soc. Matrix Biol. 2017, 64, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Han, K.Y.; Tran, J.A.; Chang, J.H.; Azar, D.T.; Zieske, J.D. Potential role of corneal epithelial cell-derived exosomes in corneal wound healing and neovascularization. Sci. Rep. 2017, 7, 40548. [Google Scholar] [CrossRef] [PubMed]
- Zieske, J.D.; Mason, V.S.; Wasson, M.E.; Meunier, S.F.; Nolte, C.J.M.; Fukai, N.; Olsen, B.R.; Parenteau, N.L. Basement Membrane Assembly and Differentiation of Cultured Corneal Cells: Importance of Culture Environment and Endothelial Cell Interaction. Exp. Cell Res. 1994, 214, 621–633. [Google Scholar] [CrossRef] [PubMed]
- Griffith, M.; Osborne, R.; Munger, R.; Xiong, X.; Doillon, C.J.; Laycock, N.L.; Hakim, M.; Song, Y.; Watsky, M.A. Functional human corneal equivalents constructed from cell lines. Science 1999, 286, 2169–2172. [Google Scholar] [CrossRef]
- Reichl, S.; Bednarz, J.; Müller-Goymann, C.C. Human corneal equivalent as cell culture model for in vitro drug permeation studies. Br. J. Ophthalmol. 2004, 88, 560–565. [Google Scholar] [CrossRef]
- Kobayashi, T.; Shiraishi, A.; Hara, Y.; Kadota, Y.; Yang, L.; Inoue, T.; Shirakata, Y.; Ohashi, Y. Stromal-epithelial interaction study: The effect of corneal epithelial cells on growth factor expression in stromal cells using organotypic culture model. Exp. Eye Res. 2015, 135, 109–117. [Google Scholar] [CrossRef]
- Wilson, S.L.; Yang, Y.; El Haj, A.J. Corneal stromal cell plasticity: In Vitro regulation of cell phenotype through cell-cell interactions in a three-dimensional model. Tissue Eng. Part A 2014, 20, 225–238. [Google Scholar] [CrossRef]
- Zhang, C.; Du, L.; Sun, P.; Shen, L.; Zhu, J.; Pang, K.; Wu, X. Construction of tissue-engineered full-thickness cornea substitute using limbal epithelial cell-like and corneal endothelial cell-like cells derived from human embryonic stem cells. Biomaterials 2017, 124, 180–194. [Google Scholar] [CrossRef]
- Couture, C.; Zaniolo, K.; Carrier, P.; Lake, J.; Patenaude, J.; Germain, L.; Guérin, S.L. The tissue-engineered human cornea as a model to study expression of matrix metalloproteinases during corneal wound healing. Biomaterials 2016, 78, 86–101. [Google Scholar] [CrossRef] [PubMed]
- Levis, H.J.; Brown, R.A.; Daniels, J.T. Plastic compressed collagen as a biomimetic substrate for human limbal epithelial cell culture. Biomaterials 2010, 31, 7726–7737. [Google Scholar] [CrossRef] [PubMed]
- Mi, S.; Chen, B.; Wright, B.; Connon, C.J. Ex vivo construction of an artificial ocular surface by combination of corneal limbal epithelial cells and a compressed collagen scaffold containing keratocytes. Tissue Eng. Part A 2010, 16, 2091–2100. [Google Scholar] [CrossRef] [PubMed]
- McKay, T.B.; Seyed-Razavi, Y.; Ghezzi, C.E.; Dieckmann, G.; Nieland, T.J.F.; Cairns, D.M.; Pollard, R.E.; Hamrah, P.; Kaplan, D.L. Corneal pain and experimental model development. Prog. Retin. Eye Res. 2019, 71, 88–113. [Google Scholar] [CrossRef] [PubMed]
- Gabison, E.E.; Huet, E.; Baudouin, C.; Menashi, S. Direct epithelial-stromal interaction in corneal wound healing: Role of EMMPRIN/CD147 in MMPs induction and beyond. Prog. Retin. Eye Res. 2009, 28, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Ren, R.; Hutcheon, A.E.; Guo, X.Q.; Saeidi, N.; Melotti, S.A.; Ruberti, J.W.; Zieske, J.D.; Trinkaus-Randall, V. Human primary corneal fibroblasts synthesize and deposit proteoglycans in long-term 3-D cultures. Dev. Dyn. 2008, 237, 2705–2715. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Hutcheon, A.E.; Melotti, S.A.; Zieske, J.D.; Trinkaus-Randall, V.; Ruberti, J.W. Morphologic characterization of organized extracellular matrix deposition by ascorbic acid-stimulated human corneal fibroblasts. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4050–4060. [Google Scholar] [CrossRef]
- Wilson, S.L.; Wimpenny, I.; Ahearne, M.; Rauz, S.; El Haj, A.J.; Yang, Y. Chemical and topographical effects on cell differentiation and matrix elasticity in a corneal stromal layer model. Adv. Funct. Mater. 2012, 22, 3641–3649. [Google Scholar] [CrossRef]
- Beales, M.P.; Funderburgh, J.L.; Jester, J.V.; Hassell, J.R. Proteoglycan synthesis by bovine keratocytes and corneal fibroblasts: Maintenance of the keratocyte phenotype in culture. Investig. Ophthalmol. Vis. Sci. 1999, 40, 1658–1663. [Google Scholar]
- Jester, J.V.; Barry-Lane, P.A.; Cavanagh, H.D.; Petroll, W.M. Induction of alpha-smooth muscle actin expression and myofibroblast transformation in cultured corneal keratocytes. Cornea 1996, 15, 505–516. [Google Scholar] [CrossRef]
- Marcucio, R.S.; Qin, L.; Alsberg, E.; Boerckel, J.D. Reverse engineering development: Crosstalk opportunities between developmental biology and tissue engineering. J. Orthop. Res. 2017, 35, 2356–2368. [Google Scholar] [CrossRef] [PubMed]
- Ruberti, J.W.; Zieske, J.D. Prelude to corneal tissue engineering-gaining control of collagen organization. Prog. Retin. Eye Res. 2008, 27, 549–577. [Google Scholar] [CrossRef] [PubMed]
- Zieske, J.D.; Hutcheon, A.E.K.; Guo, X.; Chung, E.-H.; Joyce, N.C. TGF-β Receptor Types I and II Are Differentially Expressed during Corneal Epithelial Wound Repair. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1465–1471. [Google Scholar]
- Guo, X.; Hutcheon, A.E.K.; Tran, J.A.; Zieske, J.D. TGF-beta-target genes are differentially regulated in corneal epithelial cells and fibroblasts. New Front. Ophthalmol. 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.P.; Zoukhri, D.; Zieske, J.D.; Dartt, D.A.; Sergheraert, C.; Loing, E.; Yu, F.S. A role for MAP kinase in regulating ectodomain shedding of APLP2 in corneal epithelial cells. Am. J. Physiol. Cell Physiol. 2001, 281, C603–C614. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.K. A formaldehyde glutaraldehyde fixative of high osmolality for use in electron microscopy. J. Cell Biol. 1965, 27, 1A–149A. [Google Scholar]
- Gipson, I.K.; Grill, S.M.; Spurr, S.J.; Brennan, S.J. Hemidesmosome formation in vitro. J. Cell Biol. 1983, 97, 849–857. [Google Scholar] [CrossRef]
- Kumano, Y.; Sakamoto, T.; Egawa, M.; Tanaka, M.; Yamamoto, I. Enhancing effect of 2-o-α-d-glucopyranosyl-L-ascorbic acid, a stable ascorbic acid derivative, on collagen synthesis. Biol. Pharm. Bull. 1998, 21, 662–666. [Google Scholar] [CrossRef][Green Version]
- Jester, J.V.; Barry, P.A.; Lind, G.J.; Petroll, W.M.; Garana, R.; Cavanagh, H.D. Corneal keratocytes: In Situ and In Vitro organization of cytoskeletal contractile proteins. Investig. Ophthalmol. Vis. Sci. 1994, 35, 730–743. [Google Scholar]
- Hutcheon, A.E.K.; Zieske, J.D.; Guo, X. 3D in vitro model for human corneal endothelial cell maturation. Exp. Eye Res. 2019, 184, 183–191. [Google Scholar] [CrossRef]
- Akers, J.C.; Gonda, D.; Kim, R.; Carter, B.S.; Chen, C.C. Biogenesis of extracellular vesicles (EV): Exosomes, microvesicles, retrovirus-like vesicles, and apoptotic bodies. J. Neuro-Oncol. 2013, 113, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zieske, J.D.; Higashijima, S.C.; Spurr-Michaud, S.J.; Gipson, I.K. Biosynthetic responses of the rabbit cornea to a keratectomy wound. Investig. Ophthalmol. Vis. Sci. 1987, 28, 1668–1677. [Google Scholar]
- Nickeleit, V.; Kaufman, A.H.; Zagachin, L.; Dutt, J.E.; Foster, C.S.; Colvin, R.B. Healing corneas express embryonic fibronectin isoforms in the epithelium, subepithelial stroma, and endothelium. Am. J. Pathol. 1996, 149, 549–558. [Google Scholar] [PubMed]
- Nishida, T.; Nakagawa, S.; Awata, T.; Ohashi, Y.; Watanabe, K.; Manabe, R. Fibronectin promotes epithelial migration of cultured rabbit cornea in situ. J. Cell Biol. 1983, 97, 1653–1657. [Google Scholar] [CrossRef] [PubMed]
- Tervo, K.; Van Setten, G.; Beuerman, R.; Virtanen, I.; Tarkkanen, A.; Tervo, T. Expression of tenascin and cellular fibronectin in the rabbit cornea after anterior keratectomy. Immunohistochemical study of wound healing dynamics. Investig. Ophthalmol. Vis. Sci. 1991, 32, 2912–2918. [Google Scholar]
- Uno, K.; Hayashi, H.; Kuroki, M.; Uchida, H.; Yamauchi, Y.; Kuroki, M.; Oshima, K. Thrombospondin-1 accelerates wound healing of corneal epithelia. Biochem. Biophys. Res. Commun. 2004, 315, 928–934. [Google Scholar] [CrossRef]
- Torricelli, A.A.; Singh, V.; Santhiago, M.R.; Wilson, S.E. The corneal epithelial basement membrane: Structure, function, and disease. Investig. Ophthalmol. Vis. Sci. 2013, 54, 6390–6400. [Google Scholar] [CrossRef]
- Ljubimov, A.V.; Burgeson, R.E.; Butkowski, R.J.; Michael, A.F.; Sun, T.T.; Kenney, M.C. Human corneal basement membrane heterogeneity: Topographical differences in the expression of type IV collagen and laminin isoforms. Lab. Investig. J. Tech. Methods Pathol. 1995, 72, 461–473. [Google Scholar]
- Torricelli, A.A.; Marino, G.K.; Santhanam, A.; Wu, J.; Singh, A.; Wilson, S.E. Epithelial basement membrane proteins perlecan and nidogen-2 are up-regulated in stromal cells after epithelial injury in human corneas. Exp. Eye Res. 2015, 134, 33–38. [Google Scholar] [CrossRef]
- Gallego-Muñoz, P.; Lorenzo-Martín, E.; Fernández, I.; Herrero-Pérez, C.; Martínez-García, M.C. Nidogen-2: Location and expression during corneal wound healing. Exp. Eye Res. 2019, 178, 1–9. [Google Scholar] [CrossRef]
- Sharif, R.; Priyadarsini, S.; Rowsey, T.G.; Ma, J.X.; Karamichos, D. Corneal Tissue Engineering: An In Vitro Model of the Stromal-nerve Interactions of the Human Cornea. J. Vis. Exp. 2018, e56308. [Google Scholar] [CrossRef] [PubMed]
- McKay, T.B.; Hjortdal, J.; Priyadarsini, S.; Karamichos, D. Acute hypoxia influences collagen and matrix metalloproteinase expression by human keratoconus cells in vitro. PLoS ONE 2017, 12, e0176017. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Karamichos, D.; Onochie, O.E.; Hutcheon, A.E.K.; Rich, C.B.; Zieske, J.D.; Trinkaus-Randall, V. Hypoxia modulates the development of a corneal stromal matrix model. Exp. Eye Res. 2018, 170, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Karamichos, D.; Zareian, R.; Guo, X.; Hutcheon, A.E.; Ruberti, J.W.; Zieske, J.D. Novel in Vitro Model for Keratoconus Disease. J. Funct. Biomater. 2012, 3, 760–775. [Google Scholar] [CrossRef]
- Priyadarsini, S.; Rowsey, T.G.; Ma, J.X.; Karamichos, D. Unravelling the stromal-nerve interactions in the human diabetic cornea. Exp. Eye Res. 2017, 164, 22–30. [Google Scholar] [CrossRef]
- Zieske, J.D.; Hutcheon, A.E.K.; Guo, X. Extracellular Vesicles and Cell-Cell Communication in the Cornea. Anat. Record 2019. [Google Scholar] [CrossRef]
- Koskela, A.; Engström, K.; Hakelius, M.; Nowinski, D.; Ivarsson, M. Regulation of fibroblast gene expression by keratinocytes in organotypic skin culture provides possible mechanisms for the antifibrotic effect of reepithelialization. Wound Repair Regen. 2010, 18, 452–459. [Google Scholar] [CrossRef]
- DelMonte, D.W.; Kim, T. Anatomy and physiology of the cornea. J. Cataract. Refract. Surg. 2011, 37, 588–598. [Google Scholar] [CrossRef]
- Araki-Sasaki, K.; Ohashi, Y.; Sasabe, T.; Hayashi, K.; Watanabe, H.; Tano, Y.; Handa, H. An SV40-immortalized human corneal epithelial cell line and its characterization. Investig. Ophthalmol. Vis. Sci. 1995, 36, 614–621. [Google Scholar]
- Li, D.Q.; Wang, Z.; Yoon, K.C.; Bian, F. Characterization, isolation, expansion and clinical therapy of human corneal epithelial stem/progenitor cells. J. Stem Cells 2014, 9, 79–91. [Google Scholar]
- Fernández-Pérez, J.; Ahearne, M. Influence of Biochemical Cues in Human Corneal Stromal Cell Phenotype. Curr. Eye Res. 2019, 44, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Pei, Y.; Reins, R.Y.; McDermott, A.M. Aldehyde dehydrogenase (ALDH) 3A1 expression by the human keratocyte and its repair phenotypes. Exp. Eye Res. 2006, 83, 1063–1073. [Google Scholar] [CrossRef] [PubMed]
- Funderburgh, J.L.; Mann, M.M.; Funderburgh, M.L. Keratocyte phenotype mediates proteoglycan structure: A role for fibroblasts in corneal fibrosis. J. Biol. Chem. 2003, 278, 45629–45637. [Google Scholar] [CrossRef] [PubMed]
- Berryhill, B.L.; Kader, R.; Kane, B.; Birk, D.E.; Feng, J.; Hassell, J.R. Partial Restoration of the Keratocyte Phenotype to Bovine Keratocytes Made Fibroblastic by Serum. Investig. Ophthalmol. Vis. Sci. 2002, 43, 3416–3421. [Google Scholar]
- Ljubimov, A.V.; Saghizadeh, M. Progress in corneal wound healing. Prog. Retin. Eye Res. 2015, 49, 17–45. [Google Scholar] [CrossRef]
- Myrna, K.E.; Pot, S.A.; Murphy, C.J. Meet the corneal myofibroblast: The role of myofibroblast transformation in corneal wound healing and pathology. Vet. Ophthalmol. 2009, 12 (Suppl. 1), 25–27. [Google Scholar] [CrossRef]
- Shu, D.Y.; Lovicu, F.J. Myofibroblast transdifferentiation: The dark force in ocular wound healing and fibrosis. Prog. Retin. Eye Res. 2017, 60, 44–65. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676. [Google Scholar] [CrossRef]

| Antibody/Labeling Agent | Host Species | Concentration | Manufacturer (City, State; Catalog Number) |
|---|---|---|---|
| Anti-cellular fibronectin (cFN) | Mouse | 1:400 | Sigma Aldrich (F6140) |
| Anti-collagen type III | Goat | 1:25–1:50 | Southern Biotech (Birmingham, AL, USA; 1330-01) |
| Anti-fibronectin | Goat | 1:50 | Santa Cruz Biotechnologies (Dallas, TX, USA; SC-90) |
| Anti-laminin-1β | Rabbit | 1:50 | Abcam (Cambridge, MA, USA; ab108536) |
| Anti-α-smooth muscle actin | Mouse | 1:25–1:50 | Dako North America (Carpinteria, CA, USA; M0851) |
| Anti-thrombospondin-1 | Rabbit | 1:50 | Abcam (ab85762) |
| Fluorescein (FITC) AffiniPure Donkey Anti-Goat IgG (H+L) | Donkey | 1:100 | Jackson ImmunoResearch (Philadelphia, PA, USA; 705-095-147) |
| Fluorescein (FITC) AffiniPure Donkey Anti-Mouse IgG (H+L) | Donkey | 1:100 | Jackson ImmunoResearch (715-095-151) |
| Fluorescein (FITC) AffiniPure Donkey Anti-Mouse IgM | Donkey | 1:100 | Jackson ImmunoResearch (715-095-140) |
| Fluorescein (FITC)-phalloidin 1 | n.a. | 1:40 | ThermoFisher (Waltham, MA, USA; F432) |
| Rhodamine (TRITC)-AffiniPure Donkey Anti-Rabbit IgG (H+L) | Donkey | 1:100 | Jackson ImmunoResearch (711-025-152) |
| Rhodamine (TRITC)-phalloidin 1 | n.a. | 1:40 | Invitrogen (Carlsbad, CA, USA; 1001302) |
| TOPRO-3-iodide 2 | n.a. | 1:100 | Life Technologies (Carlsbad, CA, USA; T-3605) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McKay, T.B.; Karamichos, D.; Hutcheon, A.E.K.; Guo, X.; Zieske, J.D. Corneal Epithelial–Stromal Fibroblast Constructs to Study Cell–Cell Communication in Vitro. Bioengineering 2019, 6, 110. https://doi.org/10.3390/bioengineering6040110
McKay TB, Karamichos D, Hutcheon AEK, Guo X, Zieske JD. Corneal Epithelial–Stromal Fibroblast Constructs to Study Cell–Cell Communication in Vitro. Bioengineering. 2019; 6(4):110. https://doi.org/10.3390/bioengineering6040110
Chicago/Turabian StyleMcKay, Tina B., Dimitrios Karamichos, Audrey E. K. Hutcheon, Xiaoqing Guo, and James D. Zieske. 2019. "Corneal Epithelial–Stromal Fibroblast Constructs to Study Cell–Cell Communication in Vitro" Bioengineering 6, no. 4: 110. https://doi.org/10.3390/bioengineering6040110
APA StyleMcKay, T. B., Karamichos, D., Hutcheon, A. E. K., Guo, X., & Zieske, J. D. (2019). Corneal Epithelial–Stromal Fibroblast Constructs to Study Cell–Cell Communication in Vitro. Bioengineering, 6(4), 110. https://doi.org/10.3390/bioengineering6040110






