Tissue Level Mechanical Properties and Extracellular Matrix Investigation of the Bovine Jugular Venous Valve Tissue
Abstract
1. Introduction
2. Materials and Methods
2.1. Light Microscopy of the Collagen Microstructure
2.2. Isolation of the Elastin
2.3. Light Microscopy of the Elastin Microstructure
2.4. Scanning Electron Microscopy of the Elastin Microstructure
2.5. Force Control Mechanical Testing
2.6. Statistical Analysis
3. Results and Discussion
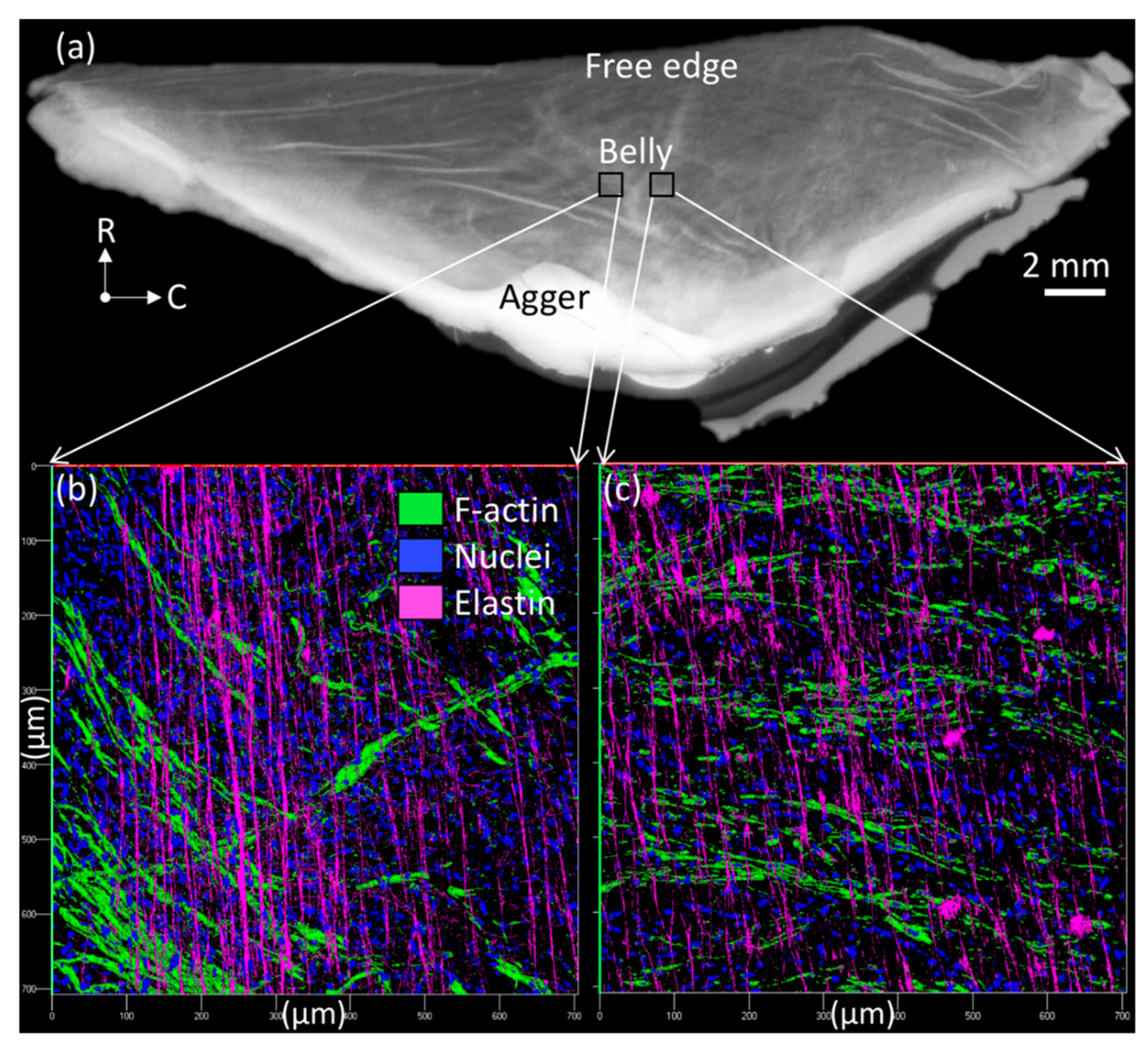
3.1. Anatomical Findings during Dissection
3.2. Light Microscopy of the Collagen Microstructure
3.3. Isolation of the Elastin Microstructure
3.4. Light Microscopy of the Elastin Microstructure
3.5. Scanning Electron Microscopy of the Elastin Microstructure
3.6. Biaxial Mechanical Testing
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Spiegel, D.R.; Smith, J.; Wade, R.R.; Cherukuru, N.; Ursani, A.; Dobruskina, Y.; Crist, T.; Busch, R.F.; Dhanani, R.M.; Dreyer, N. Transient global amnesia: Current perspectives. Neuropsychiatr. Dis. Treat. 2017, 13, 2691–2703. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, T.; Alfke, K.; Stingele, R.; Rohr, A.; Freitag-Wolf, S.; Jansen, O.; Deuschl, G. Selective affection of hippocampal CA-1 neurons in patients with transient global amnesia without long-term sequelae. Brain 2006, 129, 2874–2884. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, D.R.; McCroskey, A.L.; Deyerle, B.A. A Case of Transient Global Amnesia: A Review and How It May Shed Further Insight into the Neurobiology of Delusions. Innov. Clin. Neurosci. 2016, 13, 32–41. [Google Scholar] [PubMed]
- Nedelmann, M.; Kaps, M.; Mueller-Forell, W. Venous obstruction and jugular valve insufficiency in idiopathic intracranial hypertension. J. Neurol. 2009, 256, 964–969. [Google Scholar] [CrossRef]
- Corno, A.F.; Hurni, M.; Griffin, H.; Jeanrenaud, X.; von Segesser, L.K. Glutaraldehyde-fixed bovine jugular vein as a substitute for the pulmonary valve in the Ross operation. J. Thorac. Cardiovasc. Surg. 2001, 122, 493–494. [Google Scholar] [CrossRef][Green Version]
- McElhinney, D.B.; Hennesen, J.T. The Melody (R) valve and Ensemble (R) delivery system for transcatheter pulmonary valve replacement. Ann. N.Y. Acad. Sci. 2013, 1291, 77–85. [Google Scholar] [CrossRef]
- de Borst, G.J.; Moll, F.L. Percutaneous venous valve designs for treatment of deep venous insufficiency. J. Endovasc. Ther. 2012, 19, 291–302. [Google Scholar] [CrossRef]
- Jensen, M.O.; Lemmon, J.D.; Gessaghi, V.C.; Conrad, C.P.; Levine, R.A.; Yoganathan, A.P. Harvested porcine mitral xenograft fixation: Impact on fluid dynamic performance. J. Heart Valve Dis. 2001, 10, 111–124. [Google Scholar]
- Gottlob, R.; May, R. Venous Valves: Morphology, Function, Radiology, Surgery; Springer: Wien, Austria; New York, NY, USA, 1986. [Google Scholar]
- Lu, J.; Huang, H.S. Biaxial mechanical behavior of bovine saphenous venous valve leaflets. J. Mech. Behav. Biomed. Mater. 2018, 77, 594–599. [Google Scholar] [CrossRef]
- Ushiki, T. Collagen fibers, reticular fibers and elastic fibers. A comprehensive understanding from a morphological viewpoint. Arch. Histol. Cytol. 2002, 65, 109–126. [Google Scholar] [CrossRef]
- Huang, H.S.; Lu, J. Biaxial mechanical properties of bovine jugular venous valve leaflet tissues. Biomech. Model. Mechanobiol. 2017, 16, 1911–1923. [Google Scholar] [CrossRef] [PubMed]
- Muiznieks, L.D.; Keeley, F.W. Molecular assembly and mechanical properties of the extracellular matrix: A fibrous protein perspective. Biochim. Biophys. Acta 2013, 1832, 866–875. [Google Scholar] [CrossRef]
- de Wolf, M.A.; de Graaf, R.; Kurstjens, R.L.; Penninx, S.; Jalaie, H.; Wittens, C.H. Short-Term Clinical Experience with a Dedicated Venous Nitinol Stent: Initial Results with the Sinus-Venous Stent. Eur. J. Vasc. Endovasc. Surg. 2015, 50, 518–526. [Google Scholar] [CrossRef]
- Mouton, W.G.; Habegger, A.K.; Haenni, B.; Tschanz, S.; Baumgartner, I.; Ochs, M. Valve disease in chronic venous disorders: A quantitative ultrastructural analysis by transmission electron microscopy and stereology. Swiss Med. Wkly. 2013, 143, w13755. [Google Scholar] [CrossRef] [PubMed]
- Budd, T.W.; Meenaghan, M.A.; Wirth, J.; Taheri, S.A. Histopathology of veins and venous valves of patients with venous insufficiency syndrome: Ultrastructure. J. Med. 1990, 21, 181–199. [Google Scholar]
- Markovic, J.N.; Shortell, C.K. Genomics of varicose veins and chronic venous insufficiency. Semin. Vasc. Surg. 2013, 26, 2–13. [Google Scholar] [CrossRef]
- Schoof, P.H.; Takkenberg, J.J.; van Suylen, R.J.; Zondervan, P.E.; Hazekamp, M.G.; Dion, R.A.; Bogers, A.J. Degeneration of the pulmonary autograft: An explant study. J. Thorac. Cardiovasc. Surg. 2006, 132, 1426–1432. [Google Scholar] [CrossRef] [PubMed]
- Matthews, P.B.; Jhun, C.S.; Yaung, S.; Azadani, A.N.; Guccione, J.M.; Ge, L.; Tseng, E.E. Finite element modeling of the pulmonary autograft at systemic pressure before remodeling. J. Heart Valve Dis. 2011, 20, 45–52. [Google Scholar]
- Ozturk, N.; Sucu, N.; Comelekoglu, U.; Yilmaz, B.C.; Aytacoglu, B.N.; Vezir, O. Pressure applied during surgery alters the biomechanical properties of human saphenous vein graft. Heart Vessels 2013, 28, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Ackroyd, J.S.; Pattison, M.; Browse, N.L. A study of the mechanical properties of fresh and preserved human femoral vein wall and valve cusps. Br. J. Surg. 1985, 72, 117–119. [Google Scholar] [CrossRef]
- Kaul, N.; Huang, H.S. Constitutive modeling of jugular vein-derived venous valve leaflet tissues. J. Mech. Behav. Biomed. Mater. 2017, 75, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Saphir, O.; Lev, M. The venous valve in the aged. Am. Heart J. 1952, 44, 843–850. [Google Scholar] [CrossRef]
- Crissman, R.S.; Pakulski, L.A. A rapid digestive technique to expose networks of vascular elastic fibers for SEM observation. Stain Technol. 1984, 59, 171–180. [Google Scholar] [CrossRef]
- Crissman, R.S. Comparison of 2 Digestive Techniques for Preparation of Vascular Elastic Networks for Sem Observation. J. Electron Microsc. Tech. 1987, 6, 335–348. [Google Scholar] [CrossRef]
- Mecham, R.P. Methods in elastic tissue biology: Elastin isolation and purification. Methods 2008, 45, 32–41. [Google Scholar] [CrossRef]
- Jackson, D.; Cleary, G. The Determination of Collagen and Elastin. In Methods of Biochemical Analysis; John Wiley & Sons Inc.: New York, NY, USA, 1967; pp. 25–67. [Google Scholar]
- Tseng, H.; Grande-Allen, K.J. Elastic fibers in the aortic valve spongiosa: A fresh perspective on its structure and role in overall tissue function. Acta Biomater. 2011, 7, 2101–2108. [Google Scholar] [CrossRef]
- Huang, H.-Y.S.; Balhouse, B.N.; Huang, S. Application of simple biomechanical and biochemical tests to heart valve leaflets: Implications for heart valve characterization and tissue engineering. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2012, 226, 868–876. [Google Scholar] [CrossRef]
- Huang, S.; Huang, H.-Y.S. Biaxial Stress Relaxation of Semilunar Heart Valve Leaflets during Simulated Collagen Catabolism: Effects of Collagenase Concentration and Equibiaxial Strain-State. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2015, 229, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-Y.S.; Huang, S.; Frazier, C.P.; Prim, P.; Harrysson, O. Directional Mechanical Property of Porcine Skin Tissues. J. Mech. Med. Biol. 2014, 14, 14500699. [Google Scholar] [CrossRef]
- Buchanan, R.M.; Sacks, M.S. Interlayer micromechanics of the aortic heart valve leaflet. Biomech. Model. Mechanobiol. 2014, 13, 813–826. [Google Scholar] [CrossRef][Green Version]
- Edwards, E. The orientation of venous valves in relation to body surfaces. Anat. Rec. 1936, 64, 369–385. [Google Scholar] [CrossRef]
- Budras, K.-D.; Habel, R. Chapter 5: Vertebral Column, Thoracic Skeleton and Neck. In Bovine Anatomy; Budras, K.-D., Ed.; Die Deutsche Bibliothek: Hannover, Germany, 2003; pp. 56–61. [Google Scholar]
- Gelse, K.; Poschl, E.; Aigner, T. Collagens—Structure, function and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546. [Google Scholar] [CrossRef] [PubMed]
- Wiltz, D.; Arevalos, A.; Balaoing, L.; Blancas, A.; Sapp, M.; Zhang, X.; Grande-Allen, J. Extracellular Matrix Organization, Structure and Function. In Calcific Aortic Valve Disease; Elena, A., Ed.; InTech: Rijeka, Croatia, 2013; pp. 3–30. [Google Scholar]
- Chen, Q.; Thouas, G. Histology and Tissue Properties II Connective Tissues. In Biomaterials A Basic Introduction; CRC Press: Boca Raton, FL, USA, 2015; pp. 609–632. [Google Scholar]
- Scott, M.; Vesely, I. Aortic valve cusp microstructure: The role of elastin. Ann. Thorac. Surg. 1995, 60, S391–S394. [Google Scholar] [CrossRef]
- Robert, L.; Hornebeck, W. Elastin and Elastases; CRC Press: Boca Raton, FL, USA, 1989. [Google Scholar]
- Barbour, K.; Huang, H.-Y.S. Strain Effects on Collagen Proteolysis in Heart Valve Tissues. Mech. Time-Depend. Mater. 2019, 23, 1–16. [Google Scholar] [CrossRef]
- De Hart, J.; Peters, G.W.; Schreurs, P.J.; Baaijens, F.P. Collagen fibers reduce stresses and stabilize motion of aortic valve leaflets during systole. J. Biomech. 2004, 37, 303–311. [Google Scholar] [CrossRef]
- Vallentine, J.F. Grazing Management; Academic Press: San Diego, CA, USA, 2001. [Google Scholar]
- Valecchi, D.; Bacci, D.; Gulisano, M.; Sgambati, E.; Sibilio, M.; Lipomas, M.; Macchi, C. Internal jugular vein valves: An assessment of prevalence, morphology and competence by color Doppler echography in 240 healthy subjects. Ital. J. Anat. Embryol. 2010, 115, 185–189. [Google Scholar]


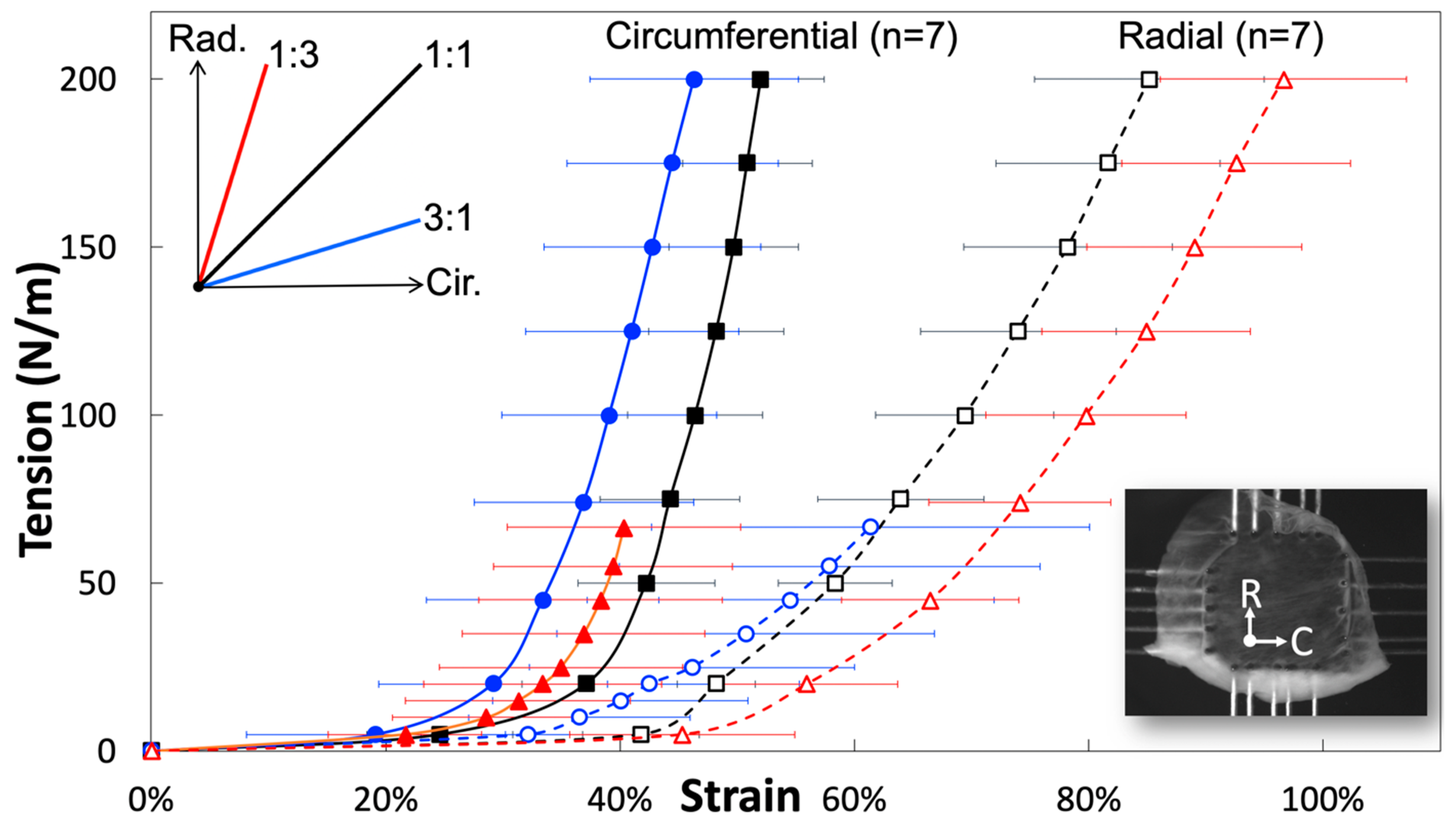
| Modulus | 1:1 | 1:3 | 3:1 |
|---|---|---|---|
| Circumferential | 29.34 ± 1.72 MPa | 33.15 ± 3.29 MPa | 30.19 ± 2.78 MPa |
| Radial | 11.38 ± 0.84 MPa | 11.16 ± 0.98 MPa | 8.41 ± 1.54 MPa |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benson, A.A.; Huang, H.-Y.S. Tissue Level Mechanical Properties and Extracellular Matrix Investigation of the Bovine Jugular Venous Valve Tissue. Bioengineering 2019, 6, 45. https://doi.org/10.3390/bioengineering6020045
Benson AA, Huang H-YS. Tissue Level Mechanical Properties and Extracellular Matrix Investigation of the Bovine Jugular Venous Valve Tissue. Bioengineering. 2019; 6(2):45. https://doi.org/10.3390/bioengineering6020045
Chicago/Turabian StyleBenson, Adam A., and Hsiao-Ying Shadow Huang. 2019. "Tissue Level Mechanical Properties and Extracellular Matrix Investigation of the Bovine Jugular Venous Valve Tissue" Bioengineering 6, no. 2: 45. https://doi.org/10.3390/bioengineering6020045
APA StyleBenson, A. A., & Huang, H.-Y. S. (2019). Tissue Level Mechanical Properties and Extracellular Matrix Investigation of the Bovine Jugular Venous Valve Tissue. Bioengineering, 6(2), 45. https://doi.org/10.3390/bioengineering6020045





