Polyhydroxyalkanoate Biosynthesis at the Edge of Water Activity-Haloarchaea as Biopolyester Factories
Abstract
:1. Introduction
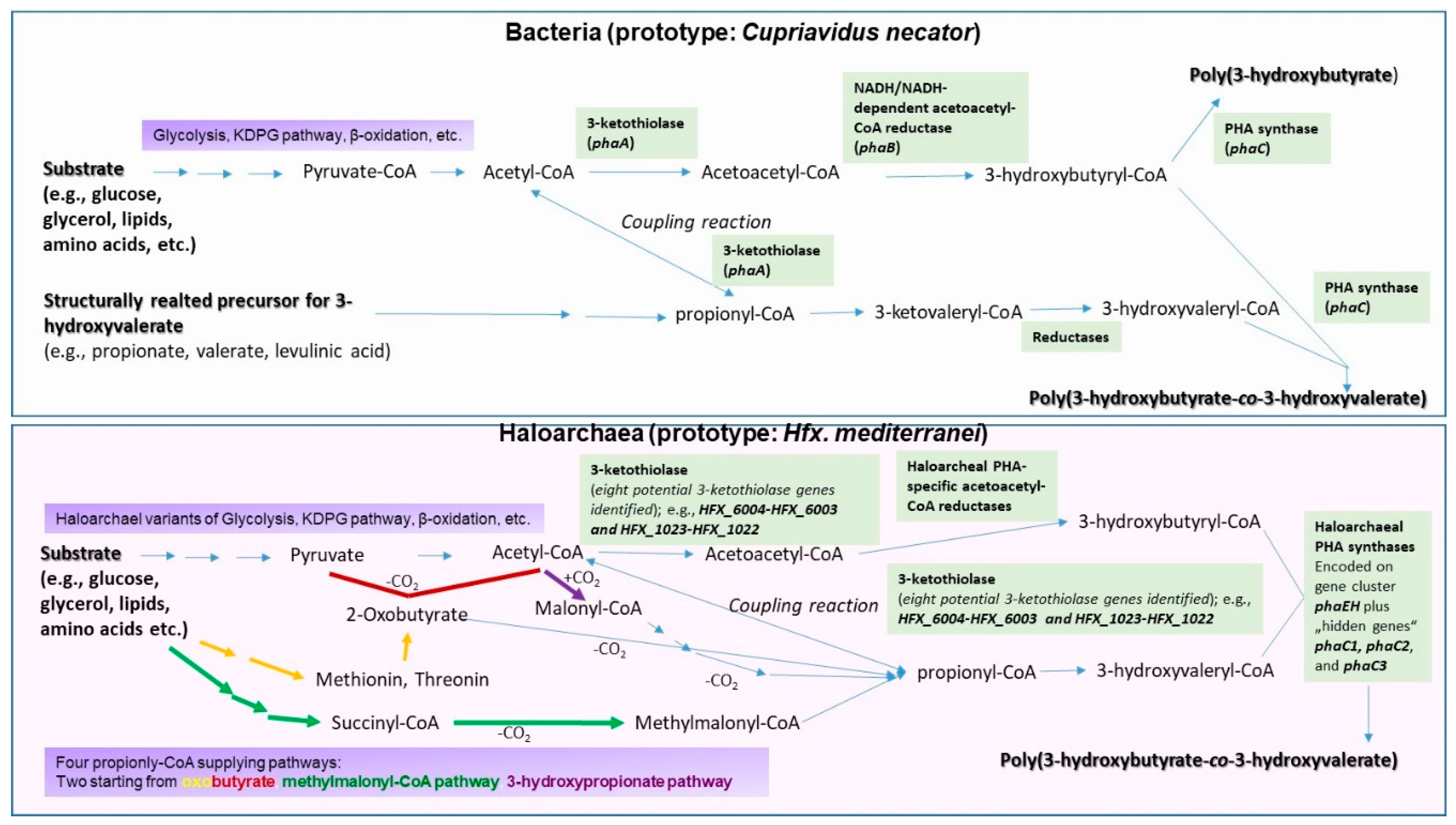
2. Genetic and Enzymatic Particularities of Haloarchaeal PHA Biosynthesis
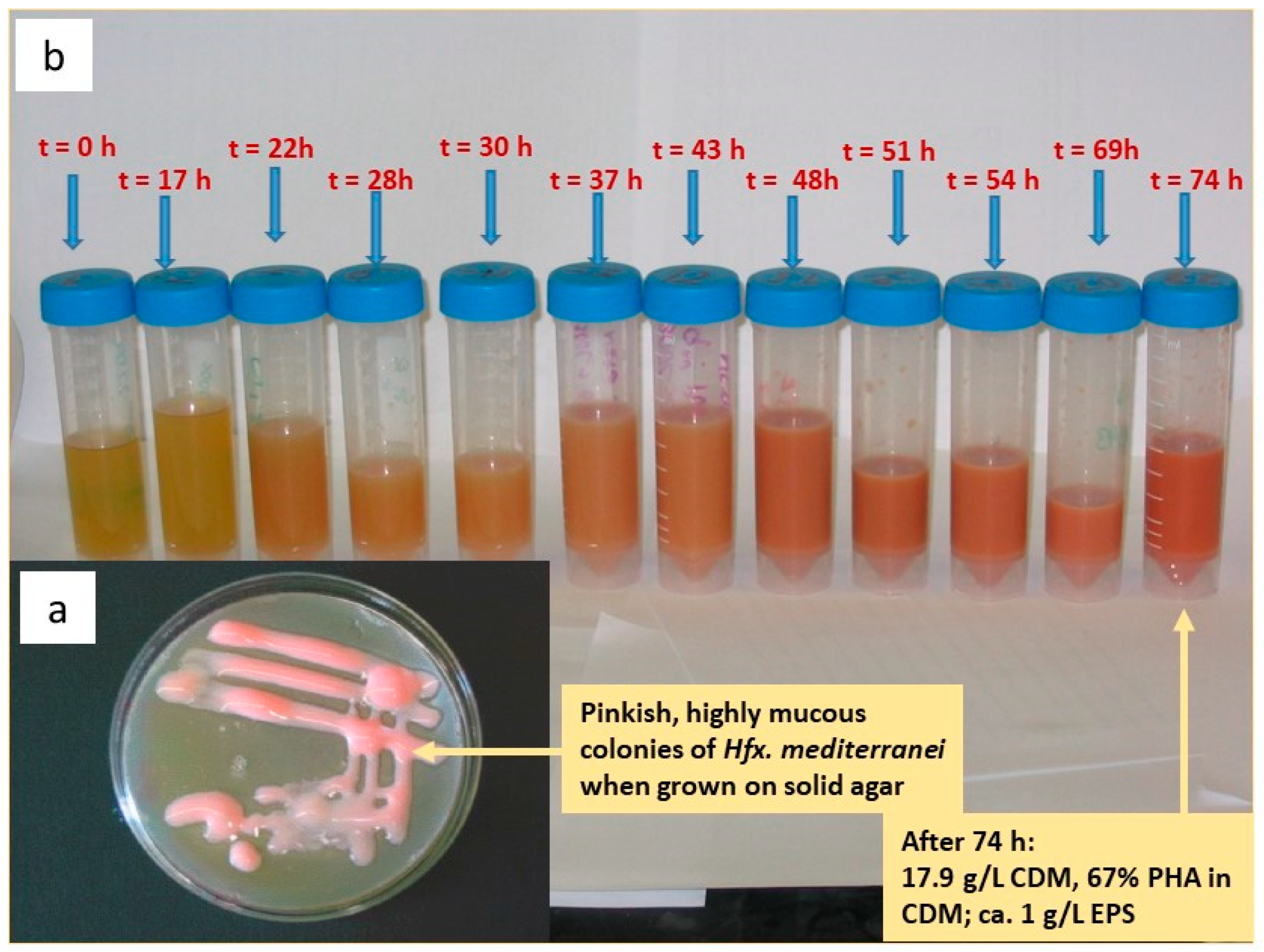
3. Haloferax mediterranei—The Prototype PHA Production Strain among Haloarchaea
4. Process Parameters for Optimized Hfx. mediterranei-Mediated PHA Production
5. Use of Different Feedstocks for PHA Biosynthesis by Hfx. mediterranei
5.1. Hfx. mediterranei on Hydrolyzed Whey Permeate
5.2. Hfx. mediterranei on Crude Glycerol Phase from Biodiesel Industry
5.3. Hfx. mediterranei on Processed Starchy Materials
5.4. Hfx. mediterranei on Waste Streams of Bioethanol Production
5.5. Hfx. mediterranei on Wastewater of Olive Oil Production
5.6. Hfx. mediterranei on Hydrolyzed Macroalgae
6. Microstructure of Hfx. mediterranei PHA Copolyesters
7. Further Haloarchaeal Genera Encompassing PHA Producers
7.1. Haloarcula sp.
7.2. Halogeometricum sp.
7.3. Halopiger sp.
7.4. Halobiforma sp.
7.5. Natrinema sp.
7.6. Haloquadratum sp.
7.7. Halococcus sp.
7.8. Halogranum sp.
7.9. Haloterrigena sp.
7.10. Halorhabdus sp.
8. Conclusions
Funding
Conflicts of Interest
References
- Lemoigne, M. Produits de Deshydration et de Polymerisation de L’acide β = Oxybutyrique. Bull. Soc. Chim. Biol. 1926, 8, 770–782. [Google Scholar]
- Jendrossek, D. Polyhydroxyalkanoate granules are complex subcellular organelles (carbonosomes). J. Bacteriol. 2009, 191, 3195–3202. [Google Scholar] [CrossRef]
- Kourmentza, C.; Plácido, J.; Venetsaneas, N.; Burniol-Figols, A.; Varrone, C.; Gavala, H.N.; Reis, M.A. Recent advances and challenges towards sustainable polyhydroxyalkanoate (PHA) production. Bioengineering 2017, 4, 55. [Google Scholar] [CrossRef]
- Koller, M.; Maršálek, L.; Miranda de Sousa Dias, M.; Braunegg, G. Producing microbial polyhydroxyalkanoate (PHA) biopolyesters in a sustainable manner. New Biotechnol. 2017, 37, 24–38. [Google Scholar] [CrossRef]
- Koller, M. Chemical and biochemical engineering approaches in manufacturing Polyhydroxyalkanoate (PHA) biopolyesters of tailored structure with focus on the diversity of building blocks. Chem. Biochem. Eng. Q. 2018, 32, 413–438. [Google Scholar] [CrossRef]
- Koller, M. A review on established and emerging fermentation schemes for microbial production of Polyhydroxyalkanoate (PHA) biopolyesters. Fermentation 2018, 4, 30. [Google Scholar] [CrossRef]
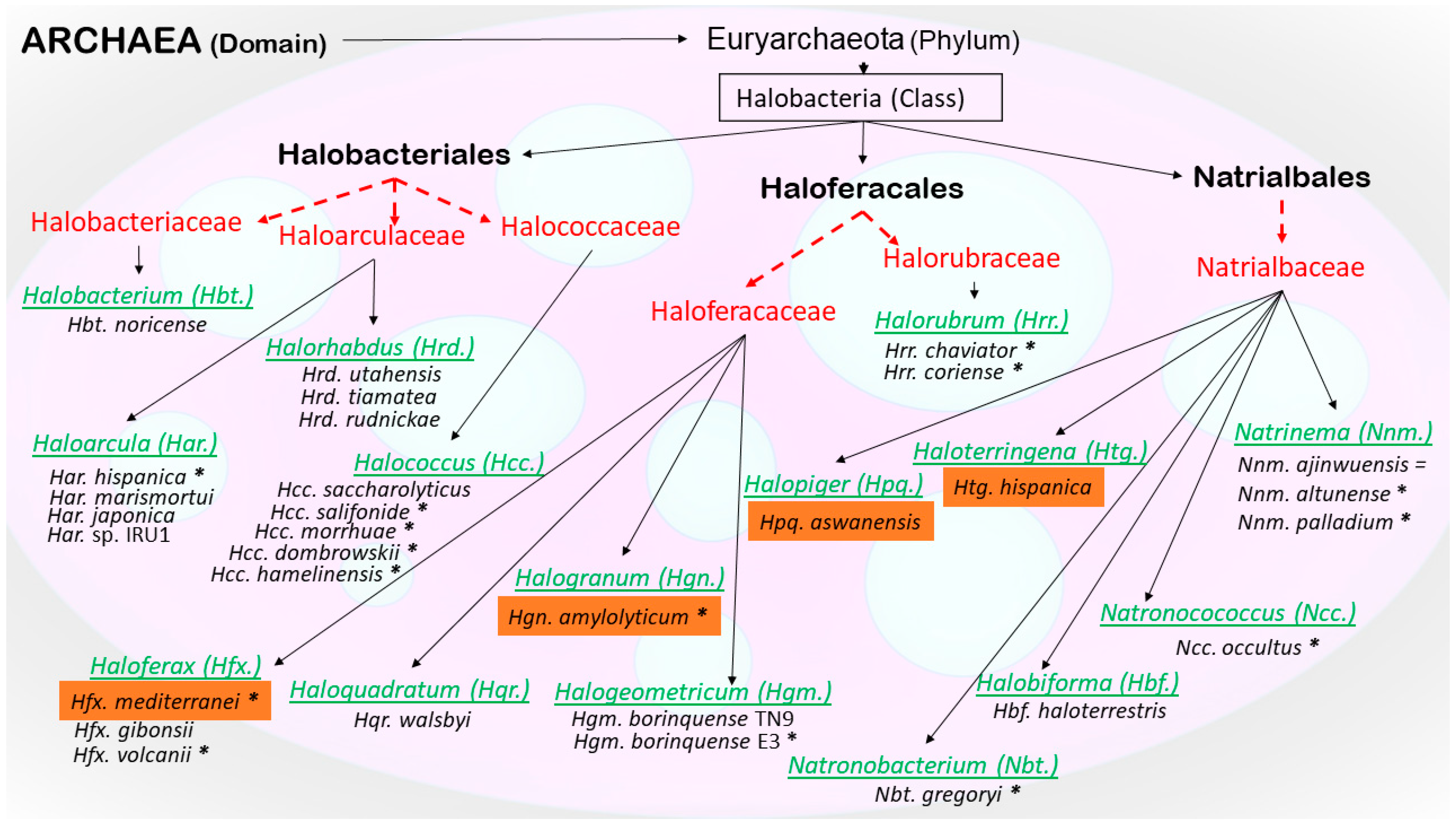
- Oren, A.; Ventosa, A. International Committee on Systematics of Prokaryotes Subcommittee on the taxonomy of Halobacteriaceae and subcommittee on the taxonomy of Halomonadaceae. Minutes of the joint open meeting, 23 May 2016, San Juan, Puerto Rico. Int. J. Syst. Evol. Microbiol. 2016, 66, 4291. [Google Scholar] [CrossRef]
- Gupta, R.S.; Naushad, S.; Baker, S. Phylogenomic analyses and molecular signatures for the class Halobacteria and its two major clades: A proposal for division of the class Halobacteria into an emended order Halobacteriales and two new orders, Haloferacales ord. nov. and Natrialbales ord. nov., containing the novel families Haloferacaceae fam. nov. and Natrialbaceae fam. nov. Int. J. Syst. Evol. Microbiol. 2015, 65, 1050–1069. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.S.; Naushad, S.; Fabros, R.; Adeolu, M. A phylogenomic reappraisal of family-level divisions within the class Halobacteria: Proposal to divide the order Halobacteriales into the families Halobacteriaceae, Haloarculaceae fam. nov., and Halococcaceae fam. nov., and the order Haloferacales into the families, Haloferacaceae and Halorubraceae fam nov. Antonie van Leeuwenhoek 2016, 109, 565–587. [Google Scholar] [CrossRef] [PubMed]
- Kirk, R.G.; Ginzburg, M. Ultrastructure of two species of halobacterium. J. Ultrastruct. Res. 1972, 41, 80–94. [Google Scholar] [CrossRef]
- Nicolaus, B.; Lama, L.; Esposito, E.; Manca, M.C.; Improta, R.; Bellitti, M.R.; Duckworth, A.W.; Grant, W.D.; Gambacorta, A. Haloarcula spp able to biosynthesize exo-and endopolymers. J. Ind. Microbiol. Biotechnol. 1999, 23, 489–496. [Google Scholar] [CrossRef]
- Quillaguamán, J.; Guzmán, H.; Van-Thuoc, D.; Hatti-Kaul, R. Synthesis and production of polyhydroxyalkanoates by halophiles: Current potential and future prospects. Appl. Microbiol. Biotechnol. 2010, 85, 1687–1696. [Google Scholar] [CrossRef] [PubMed]
- Mezzolla, V.; D’Urso, O.; Poltronieri, P. Role of PhaC type I and Type II enzymes during PHA biosynthesis. Polymers 2018, 10, 910. [Google Scholar] [CrossRef]
- Hezayen, F.F.; Steinbüchel, A.; Rehm, B.H. Biochemical and enzymological properties of the polyhydroxybutyrate synthase from the extremely halophilic archaeon strain 56. Arch. Biochem. Biophys. 2002, 403, 284–291. [Google Scholar] [CrossRef]
- Baliga, N.S.; Bonneau, R.; Facciotti, M.T.; Pan, M.; Glusman, G.; Deutsch, E.W.; Shannon, P.; Chiu, Y.; Weng, R.S.; Gan, R.R.; et al. Genome sequence of Haloarcula marismortui: A halophilic archaeon from the Dead Sea. Genome Res. 2004, 14, 2221–2234. [Google Scholar] [CrossRef]
- Bolhuis, H.; Palm, P.; Wende, A.; Falb, M.; Rampp, M.; Rodriguez-Valera, F.; Pfeiffer, F.; Oesterhelt, D. The genome of the square archaeon Haloquadratum walsbyi: Life at the limits of water activity. BMC Genom. 2006, 7, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Lu, Q.; Zhou, L.; Zhou, J.; Xiang, H. Molecular characterization of the phaECHm genes, required for biosynthesis of poly(3-hydroxybutyrate) in the extremely halophilic archaeon Haloarcula marismortui. Appl. Environ. Microbiol. 2007, 73, 6058–6065. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Han, J.; Zhou, L.; Zhou, J.; Xiang, H. Genetic and biochemical characterization of the poly(3-hydroxybutyrate-co-3-hydroxyvalerate) synthase in Haloferax mediterranei. J. Bacteriol. 2008, 190, 4173–4180. [Google Scholar] [CrossRef]
- Han, J.; Li, M.; Hou, J.; Wu, L.; Zhou, J.; Xiang, H. Comparison of four phaC genes from Haloferax mediterranei and their function in different PHBV copolymer biosyntheses in Haloarcula hispanica. Saline Syst. 2010, 6, 9. [Google Scholar] [CrossRef]
- Han, J.; Zhang, F.; Hou, J.; Liu, X.; Li, M.; Liu, H.; Cai, L.; Zhang, B.; Chen, Y.; Zhou, J.; et al. Complete genome sequence of the metabolically versatile halophilic archaeon Haloferax mediterranei, a poly(3-hydroxybutyrate-co-3-hydroxyvalerate) producer. J. Bacteriol. 2012, 194, 4463–4464. [Google Scholar] [CrossRef]
- Ding, J.Y.; Chiang, P.W.; Hong, M.J.; Dyall-Smith, M.; Tang, S.L. Complete genome sequence of the extremely halophilic archaeon Haloarcula hispanica strain N601. Genome Announc. 2014, 2, e00178-14. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Feng, B.; Han, J.; Liu, H.; Zhao, D.; Zhou, J.; Xiang, H. Haloarchaeal-type beta-ketothiolases involved in Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) synthesis in Haloferax mediterranei. Appl. Environ. Microbiol. 2013, 79, 5104–5111. [Google Scholar] [CrossRef]
- Han, J.; Hou, J.; Liu, H.; Cai, S.; Feng, B.; Zhou, J.; Xiang, H. Wide distribution among halophilic archaea of a novel polyhydroxyalkanoate synthase subtype with homology to bacterial type III synthases. Appl. Environ. Microbiol. 2010, 76, 7811–7819. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Lu, Q.; Zhou, L.; Liu, H.; Xiang, H. Identification of the polyhydroxyalkanoate (PHA)-specific acetoacetyl coenzyme A reductase among multiple FabG paralogs in Haloarcula hispanica and reconstruction of the PHA biosynthetic pathway in Haloferax volcanii. Appl. Environ. Microbiol. 2009, 75, 6168–6175. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.; Cai, S.; Han, J.; Liu, H.; Zhou, J.; Xiang, H. Identification of the phaB genes and analysis of the PHBHV precursor supplying pathway in Haloferax mediterranei. Acta Microbiol. Sin. 2010, 50, 1305–1312. [Google Scholar]
- Xiang, H. PHBHV Biosynthesis by Haloferax mediterranei: From Genetics, Metabolism, and Engineering to Economical Production. In Microbial Biopolyester Production, Performance and Processing. Microbiology, Feedsstocks, and Metabolism; Koller, M., Ed.; Book Series: Recent Advances in Biotechnology; Bentham Science Publishers: Sharjah, United Arab Emirates, 2016; Volume 1, pp. 348–379. [Google Scholar]
- Rodríguez-Valera, F.; Ruiz-Berraquero, F.; Ramos-Cormenzana, A. Behaviour of mixed populations of halophilic bacteria in continuous cultures. Can. J. Microbiol. 1980, 26, 1259–1263. [Google Scholar] [CrossRef]
- Rodriguez-Valera, F.; Juez, G.; Kushner, D.J. Halobacterium mediterranei spec, nov., a new carbohydrate-utilizing extreme halophile. Syst. Appl. Microbiol. 1983, 4, 369–381. [Google Scholar] [CrossRef]
- Fernandez-Castillo, R.; Rodriguez-Valera, F.; Gonzalez-Ramos, J.; Ruiz-Berraquero, F. Accumulation of poly(β-hydroxybutyrate) by halobacteria. Appl. Environ. Microbiol. 1986, 51, 214–216. [Google Scholar]
- Torreblanca, M.; Rodriguez-Valera, F.; Juez, G.; Ventosa, A.; Kamekura, M.; Kates, M. Classification of non-alkaliphilic halobacteria based on numerical taxonomy and polar lipid composition, and description of Haloarcula gen. nov. and Haloferax gen. nov. Syst. Appl. Microbiol. 1986, 8, 89–99. [Google Scholar] [CrossRef]
- Rodriguez-Valera, F.; Lillo, J. Halobacteria as producers of polyhydroxyalkanoates. FEMS Microbiol. Lett. 1982, 103, 181–186. [Google Scholar] [CrossRef]
- Lillo, J.G.; Rodriguez-Valera, F. Effects of culture conditions on poly(β-hydroxybutyric acid) production by Haloferax mediterranei. Appl. Environ. Microbiol. 1990, 56, 2517–2521. [Google Scholar]
- Antón, J.; Meseguer, I.; Rodriguez-Valera, F. Production of an extracellular polysaccharide by Haloferax mediterranei. Appl. Environ. Microbiol. 1988, 54, 2381–2386. [Google Scholar]
- Hermann-Krauss, C.; Koller, M.; Muhr, A.; Fasl, H.; Stelzer, F.; Braunegg, G. Archaeal production of polyhydroxyalkanoate (PHA) co-and terpolyesters from biodiesel industry-derived by-products. Archaea 2013, 2013, 129268. [Google Scholar] [CrossRef]
- Oren, A. Microbial life at high salt concentrations: Phylogenetic and metabolic diversity. Saline Syst. 2008, 4, 1–13. [Google Scholar] [CrossRef]
- Parolis, H.; Parolis, L.A.; Boán, I.F.; Rodríguez-Valera, F.; Widmalm, G.; Manca, M.C.; Jansson, P.-E.; Sutherland, I.W. The structure of the exopolysaccharide produced by the halophilic Archaeon Haloferax mediterranei strain R4 (ATCC 33500). Carbohydr. Res. 1996, 295, 147–156. [Google Scholar] [CrossRef]
- Koller, M.; Chiellini, E.; Braunegg, G. Study on the production and re-use of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and extracellular polysaccharide by the archaeon Haloferax mediterranei strain DSM 1411. Chem. Biochem. Eng. Q. 2015, 29, 87–98. [Google Scholar] [CrossRef]
- Cui, Y.W.; Gong, X.Y.; Shi, Y.P.; Wang, Z.D. Salinity effect on production of PHA and EPS by Haloferax mediterranei. RSC Adv. 2017, 7, 53587–53595. [Google Scholar] [CrossRef]
- Han, J.; Hou, J.; Zhang, F.; Ai, G.; Li, M.; Cai, S.; Liu, H.; Wang, L.; Wang, Z.; Zhang, S.; Cai, L.; Zhao, D.; Zhou, J.; Xiang, H. Multiple propionyl coenzyme A-supplying pathways for production of the bioplastic poly(3-hydroxybutyrate-co-3-hydroxyvalerate) in Haloferax mediterranei. Appl. Environ. Microbiol. 2013, 79, 2922–2931. [Google Scholar] [CrossRef]
- Koller, M. Switching from petro-plastics to microbial polyhydroxyalkanoates (PHA): The biotechnological escape route of choice out of the plastic predicament? The EuroBiotech J. 2019, 3, 32–44. [Google Scholar] [CrossRef]
- Koller, M.; Hesse, P.; Bona, R.; Kutschera, C.; Atlić, A.; Braunegg, G. Potential of various archae-and eubacterial strains as industrial polyhydroxyalkanoate producers from whey. Macromol. Biosci. 2007, 7, 218–226. [Google Scholar] [CrossRef]
- Cai, S.; Cai, L.; Liu, H.; Liu, X.; Han, J.; Zhou, J.; Xiang, H. Identification of the haloarchaeal phasin (PhaP) that functions in polyhydroxyalkanoate accumulation and granule formation in Haloferax mediterranei. Appl. Environ. Microbiol. 2012, 78, 1946–1952. [Google Scholar] [CrossRef]
- Liu, G.; Hou, J.; Cai, S.; Zhao, D.; Cai, L.; Han, J.; Zhou, J.; Xiang, H. A patatin-like protein associated with the polyhydroxyalkanoate (PHA) granules of Haloferax mediterranei acts as an efficient depolymerase in the degradation of native PHA. Appl. Environ. Microbiol. 2015, 81, 3029–3038. [Google Scholar] [CrossRef]
- Ferre-Güell, A.; Winterburn, J. Production of the copolymer poly(3-hydroxybutyrate-co-3-hydroxyvalerate) with varied composition using different nitrogen sources with Haloferax mediterranei. Extremophiles 2017, 21, 1037–1047. [Google Scholar] [CrossRef]
- Melanie, S.; Winterburn, J.B.; Devianto, H. Production of biopolymer Polyhydroxyalkanoates (PHA) by extreme halophilic marine Archaea Haloferax mediterranei in medium with varying phosphorus concentration. J. Eng. Technol. Sci. 2018, 50, 255–271. [Google Scholar] [CrossRef]
- Cui, Y.W.; Zhang, H.Y.; Ji, S.Y.; Wang, Z.W. Kinetic analysis of the temperature effect on polyhydroxyalkanoate production by Haloferax mediterranei in synthetic molasses wastewater. J. Polym. Environ. 2017, 25, 277–285. [Google Scholar] [CrossRef]
- Koller, M.; Hesse, P.; Bona, R.; Kutschera, C.; Atlić, A.; Braunegg, G. Biosynthesis of high quality polyhydroxyalkanoate co-and terpolyesters for potential medical application by the archaeon Haloferax mediterranei. Macromol. Symp. 2007, 253, 33–39. [Google Scholar] [CrossRef]
- Pais, J.; Serafim, L.S.; Freitas, F.; Reis, M.A. Conversion of cheese whey into poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Haloferax mediterranei. New Biotechnol. 2016, 33, 224–230. [Google Scholar] [CrossRef]
- Chen, C.W.; Don, T.M.; Yen, H.F. Enzymatic extruded starch as a carbon source for the production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Haloferax mediterranei. Process Biochem. 2006, 41, 2289–2296. [Google Scholar] [CrossRef]
- Huang, T.Y.; Duan, K.J.; Huang, S.Y.; Chen, C.W. Production of polyhydroxyalkanoates from inexpensive extruded rice bran and starch by Haloferax mediterranei. J. Ind. Microbiol. Biotechnol. 2006, 33, 701–706. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Saha, J.; Haldar, S.; Bhowmic, A.; Mukhopadhyay, U.K.; Mukherjee, J. Production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) by Haloferax mediterranei using rice-based ethanol stillage with simultaneous recovery and re-use of medium salts. Extremophiles 2014, 18, 463–470. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Jana, K.; Haldar, S.; Bhowmic, A.; Mukhopadhyay, U.K.; De, S.; Mukherjee, J. Integration of poly-3-(hydroxybutyrate-co-hydroxyvalerate) production by Haloferax mediterranei through utilization of stillage from rice-based ethanol manufacture in India and its techno-economic analysis. World J. Microbiol. Biotechnol. 2015, 31, 717–727. [Google Scholar] [CrossRef]
- Alsafadi, D.; Al-Mashaqbeh, O. A one-stage cultivation process for the production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) from olive mill wastewater by Haloferax mediterranei. New Biotechnol. 2017, 34, 47–53. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Pramanik, A.; Maji, S.K.; Haldar, S.; Mukhopadhyay, U.K.; Mukherjee, J. Utilization of vinasse for production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) by Haloferax mediterranei. AMB Express 2012, 2, 34. [Google Scholar] [CrossRef]
- Ghosh, S.; Gnaim, R.; Greiserman, S.; Fadeev, L.; Gozin, M.; Golberg, A. Macroalgal biomass subcritical hydrolysates for the production of polyhydroxyalkanoate (PHA) by Haloferax mediterranei. Bioresour. Technol. 2019, 271, 166–173. [Google Scholar] [CrossRef]
- Koller, M.; Puppi, D.; Chiellini, F.; Braunegg, G. Comparing chemical and enzymatic Hydrolysis of whey lactose to generate feedstocks for haloarchaeal poly(3-hydroxybutyrate-co-3-hydroxyvalerate) biosynthesis. Int. J. Pharm. Sci. Res. 2016, 3, 112. [Google Scholar] [CrossRef]
- Koller, M.; Atlić, A.; Gonzalez-Garcia, Y.; Kutschera, C.; Braunegg, G. Polyhydroxyalkanoate (PHA) biosynthesis from whey lactose. Macromol. Symp. 2008, 27, 287–292. [Google Scholar] [CrossRef]
- Koller, M. Recycling of waste streams of the biotechnological poly(hydroxyalkanoate) production by Haloferax mediterranei on whey. Int. J. Polym. Sci. 2015, 2015, 370164. [Google Scholar] [CrossRef]
- Koller, M.; Sandholzer, D.; Salerno, A.; Braunegg, G.; Narodoslawsky, M. Biopolymer from industrial residues: Life cycle assessment of poly(hydroxyalkanoates) from whey. Resour. Conserv. Recycl. 2013, 73, 64–71. [Google Scholar] [CrossRef]
- Narodoslawsky, M.; Shazad, K.; Kollmann, R.; Schnitzer, H. LCA of PHA production–Identifying the ecological potential of bio-plastic. Chem. Biochem. Eng. Q. 2015, 29, 299–305. [Google Scholar] [CrossRef]
- Mahansaria, R.; Dhara, A.; Saha, A.; Haldar, S.; Mukherjee, J. Production enhancement and characterization of the polyhydroxyalkanoate produced by Natrinema ajinwuensis (as synonym)≡ Natrinema altunense strain RM-G10. Int. J. Biol. Macromol. 2018, 107, 1480–1490. [Google Scholar] [CrossRef]
- Don, T.M.; Chen, C.W.; Chan, T.H. Preparation and characterization of poly(hydroxyalkanoate) from the fermentation of Haloferax mediterranei. J. Biomater. Sci. Polym. E 2016, 17, 1425–1438. [Google Scholar] [CrossRef]
- Koller, M.; Bona, R.; Chiellini, E.; Braunegg, G. Extraction of short-chain-length poly-[(R)-hydroxyalkanoates] (scl-PHA) by the “anti-solvent” acetone under elevated temperature and pressure. Biotechnol. Lett. 2013, 35, 1023–1028. [Google Scholar] [CrossRef]
- Han, J.; Wu, L.P.; Hou, J.; Zhao, D.; Xiang, H. Biosynthesis, characterization, and hemostasis potential of tailor-made poly(3-hydroxybutyrate-co-3-hydroxyvalerate) produced by Haloferax mediterranei. Biomacromolecules 2015, 16, 578–588. [Google Scholar] [CrossRef]
- Ferre-Güell, A.; Winterburn, J. Increased production of polyhydroxyalkanoates with controllable composition and consistent material properties by fed-batch fermentation. Biochem. Eng. J. 2019, 141, 35–42. [Google Scholar] [CrossRef]
- Ferre-Güell, A.; Winterburn, J. Biosynthesis and characterization of Polyhydroxyalkanoates with controlled composition and microstructure. Biomacromolecules 2018, 19, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Altekar, W.; Rajagopalan, R. Ribulose bisphosphate carboxylase activity in halophilic Archaebacteria. Arch. Microbiol. 1990, 153, 169–174. [Google Scholar] [CrossRef]
- Legat, A.; Gruber, C.; Zangger, K.; Wanner, G.; Stan-Lotter, H. Identification of polyhydroxyalkanoates in Halococcus and other haloarchaeal species. Appl. Microbiol. Biotechnol. 2010, 87, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Taran, M.; Amirkhani, H. Strategies of poly (3-hydroxybutyrate) synthesis by Haloarcula sp. IRU1 utilizing glucose as carbon source: Optimization of culture conditions by Taguchi methodology. Int. J. Biol. Macromol. 2010, 47, 632–634. [Google Scholar] [CrossRef]
- Taran, M. Synthesis of poly(3-hydroxybutyrate) from different carbon sources by Haloarcula sp. IRU1. Polym.-Plast. Technol. 2011, 50, 530–532. [Google Scholar] [CrossRef]
- Taran, M. Utilization of petrochemical wastewater for the production of poly(3-hydroxybutyrate) by Haloarcula sp. IRU1. J. Hazard. Mater. 2011, 188, 26–28. [Google Scholar] [CrossRef]
- Taran, M. Poly(3-hydroxybutyrate) production from crude oil by Haloarcula sp. IRU1: Optimization of culture conditions by Taguchi method. Pet. Sci. Technol. 2011, 29, 1264–1269. [Google Scholar] [CrossRef]
- Taran, M.; Sharifi, M.; Bagheri, S. Utilization of textile wastewater as carbon source by newly isolated Haloarcula sp. IRU1: Optimization of conditions by Taguchi methodology. Clean Technol. Environ. 2011, 13, 535–538. [Google Scholar] [CrossRef]
- Pramanik, A.; Mitra, A.; Arumugam, M.; Bhattacharyya, A.; Sadhukhan, S.; Ray, A.; Haldar, S.; Mukhopadhyay, U.K.; Mukherjee, J. Utilization of vinasse for the production of polyhydroxybutyrate by Haloarcula marismortui. Folia Microbiol. 2012, 57, 71–79. [Google Scholar] [CrossRef]
- Oren, A.; Ventosa, A.; Gutiérrez, M.C.; Kamekura, M. Haloarcula quadrata sp. nov., a square, motile archaeon isolated from a brine pool in Sinai (Egypt). Int. J. Syst. Evol. Microbiol. 1999, 49, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Salgaonkar, B.B.; Mani, K.; Bragança, J.M. Accumulation of polyhydroxyalkanoates by halophilic archaea isolated from traditional solar salterns of India. Extremophiles 2013, 17, 787–795. [Google Scholar] [CrossRef]
- Salgaonkar, B.B.; Bragança, J.M. Biosynthesis of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Halogeometricum borinquense strain E3. Int. J. Biol. Macromol. 2015, 78, 339–346. [Google Scholar] [CrossRef]
- Salgaonkar, B.B.; Bragança, J.M. Utilization of sugarcane bagasse by Halogeometricum borinquense strain E3 for biosynthesis of poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Bioengineering 2017, 4, 50. [Google Scholar] [CrossRef] [PubMed]
- Salgaonkar, B.B.; Mani, K.; Bragança, J.M. Sustainable bioconversion of cassava waste to Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Halogeometricum borinquense strain E3. J. Polym. Environ. 2019, 27, 299–308. [Google Scholar] [CrossRef]
- Hezayen, F.F.; Rehm, B.H.A.; Eberhardt, R.; Steinbüchel, A. Polymer production by two newly isolated extremely halophilic archaea: Application of a novel corrosion-resistant bioreactor. Appl. Microbiol. Biotechnol. 2000, 54, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Hezayen, F.F.; Gutiérrez, M.C.; Steinbüchel, A.; Tindall, B.J.; Rehm, B.H.A. Halopiger aswanensis sp. nov., a polymer-producing and extremely halophilic archaeon isolated from hypersaline soil. Int. J. Syst. Evol. Microbiol. 2010, 60, 633–637. [Google Scholar] [CrossRef]
- Hezayen, F.F.; Tindall, B.J.; Steinbüchel, A.; Rehm, B.H.A. Characterization of a novel halophilic archaeon, Halobiforma haloterrestris gen. nov., sp. nov., and transfer of Natronobacterium nitratireducens to Halobiforma nitratireducens comb. nov. Int. J. Syst. Evol. Microbiol. 2002, 52, 2271–2280. [Google Scholar] [CrossRef]
- Xu, X.W.; Wu, M.; Zhou, P.J.; Liu, S.J. Halobiforma lacisalsi sp. nov., isolated from a salt lake in China. Int. J. Syst. Evol. Microbiol. 2005, 55, 1949–1952. [Google Scholar] [CrossRef]
- Danis, O.; Ogan, A.; Tatlican, P.; Attar, A.; Cakmakci, E.; Mertoglu, B.; Birbir, M. Preparation of poly(3-hydroxybutyrate-co-hydroxyvalerate) films from halophilic archaea and their potential use in drug delivery. Extremophiles 2015, 19, 515–524. [Google Scholar] [CrossRef]
- Walsby, A.E. A square bacterium. Nature 1980, 283, 69. [Google Scholar] [CrossRef]
- Walsby, A.E. Archaea with square cells. Trends Microbiol. 2005, 13, 193–195. [Google Scholar] [CrossRef] [PubMed]
- Burns, D.G.; Janssen, P.H.; Itoh, T.; Kamekura, M.; Li, Z.; Jensen, G.; Rodríguez-Valera, F.; Bolhuis, H.; Dyall-Smith, M.L. Haloquadratum walsbyi gen. nov., sp. nov., the square haloarchaeon of Walsby, isolated from saltern crystallizers in Australia and Spain. Int. J. Syst. Evol. Microbiol. 2007, 57, 387–392. [Google Scholar] [CrossRef]
- Saponetti, M.S.; Bobba, F.; Salerno, G.; Scarfato, A.; Corcelli, A.; Cucolo, A. Morphological and structural aspects of the extremely halophilic archaeon Haloquadratum walsbyi. PLoS ONE 2011, 6, e18653. [Google Scholar] [CrossRef]
- Zhao, Y.X.; Rao, Z.M.; Xue, Y.F.; Gong, P.; Ji, Y.Z.; Ma, Y.H. Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) production by Haloarchaeon Halogranum amylolyticum. Appl. Microbiol. Biotechnol. 2015, 99, 7639–7649. [Google Scholar] [CrossRef] [PubMed]
- Romano, I.; Poli, A.; Finore, I.; Huertas, F.J.; Gambacorta, A.; Pelliccione, S.; Nicolaus, G.; Lama, L.; Nicolaus, B. Haloterrigena hispanica sp. nov., an extremely halophilic archaeon from Fuente de Piedra, southern Spain. Int. J. Syst. Evol. Microbiol. 2007, 57, 1499–1503. [Google Scholar] [CrossRef] [PubMed]
- Di Donato, P.; Fiorentino, G.; Anzelmo, G.; Tommonaro, G.; Nicolaus, B.; Poli, A. Re-use of vegetable wastes as cheap substrates for extremophile biomass production. Waste Biomass Valoriz. 2011, 2, 103–111. [Google Scholar] [CrossRef]
- Wainø, M.; Tindall, B.J.; Ingvorsen, K. Halorhabdus utahensis gen. nov., sp. nov., an aerobic, extremely halophilic member of the Archaea from Great Salt Lake, Utah. Int. J. Syst. Evol. Microbiol. 2000, 50, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Antunes, A.; Taborda, M.; Huber, R.; Moissl, C.; Nobre, M.F.; da Costa, M.S. Halorhabdus tiamatea sp. nov., a non-pigmented, extremely halophilic archaeon from a deep-sea, hypersaline anoxic basin of the Red Sea, and emended description of the genus Halorhabdus. Int. J. Syst. Evol. Microbiol. 2008, 58, 215–220. [Google Scholar] [CrossRef]
- Antunes, A.; Alam, I.; Bajic, V.B.; Stingl, U. Genome sequence of Halorhabdus tiamatea, the first archaeon isolated from a deep-sea anoxic brine lake. J. Bacteriol. 2011, 193, 4553–4554. [Google Scholar] [CrossRef]
- Albuquerque, L.; Kowalewicz-Kulbat, M.; Drzewiecka, D.; Stączek, P.; d’Auria, G.; Rosselló-Móra, R.; da Costa, M.S. Halorhabdus rudnickae sp. nov., a halophilic archaeon isolated from a salt mine borehole in Poland. Syst. Appl. Microbiol. 2016, 39, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Huang, W.; Wang, D.; Chen, F.; Yin, J.; Li, T.; Zhang, H.; Chen, G.Q. Pilot Scale-up of Poly(3-hydroxybutyrate-co-4-hydroxybutyrate) Production by Halomonas bluephagenesis via Cell Growth Adapted Optimization Process. Biotechnol. J. 2018, 13, 1800074. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yin, J.; Ye, J.; Zhang, H.; Che, X.; Ma, Y.; Li, M.; Wu, L.-P.; Chen, G.Q. Engineering Halomonas bluephagenesis TD01 for non-sterile production of poly(3-hydroxybutyrate-co-4-hydroxybutyrate). Bioresour. Technol. 2017, 244, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Hu, D.; Che, X.; Jiang, X.; Li, T.; Chen, J.; Zhang, H.M.; Chen, G.Q. Engineering of Halomonas bluephagenesis for low cost production of poly (3-hydroxybutyrate-co-4-hydroxybutyrate) from glucose. Metab. Eng. 2018, 47, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Kucera, D.; Pernicová, I.; Kovalcik, A.; Koller, M.; Mullerova, L.; Sedlacek, P.; Mravec, F.; Nebesarova, J.; Kalina, M.; Marova, I.; et al. Characterization of the promising poly(3-hydroxybutyrate) producing halophilic bacterium Halomonas halophila. Bioresour. Technol. 2018, 256, 552–556. [Google Scholar] [CrossRef]
- Yue, H.; Ling, C.; Yang, T.; Chen, X.; Chen, Y.; Deng, H.; Wu, Q.; Chen, J.; Chen, G.Q. A seawater-based open and continuous process for polyhydroxyalkanoates production by recombinant Halomonas campaniensis LS21 grown in mixed substrates. Biotechnol. Biofuels 2014, 7, 108. [Google Scholar] [CrossRef]
| Species | Strain Isolation | Salinity in Medium, Substrates, T | Product | Production Scale/Productivity | Ref. |
|---|---|---|---|---|---|
| Hfx. mediterranei | Salt pond at the coast near Alicante, Spain | 150 g/L NaCl Molasses wastewater T = 15, 20, 25, and 35 °C | PHBHV (16.7 mol.% 3HV) | 2.5 L aerated and stirred flasks 0.62 (g/L·h), qPmax. = 0.037 1/h (35 °C) | [46] |
| ´´ | ´´ | 200 g/L NaCl; T = 37 °C 25–50% pre-treated vinasse | PHBHV (12.4 mol.% 3HV using 25% vinasse) (14.1 mol.% 3HV using 50% vinasse) | Shaking flask scale; 19.7 g/L PHA, 70 wt.% PHA in CDM, 0.21 g/(L·h) | [54] |
| ´´ | ´´ | 200 g/L NaCl; T = 37 °C Rice-based stillage | PHBHV (15.3 mol.% 3HV) | Shaking flask scale; 16.4 g/L PHA, 70 wt.% PHA in CDM, 0.17 g/(L·h) | [51] |
| ´´ | ´´ | 190 g/L total salts; 144 g/L NaCl; Alkaline hydrolyzed Ulva sp. (macroalgae) as substrate T = 42 °C | PHBHV (3 mol.% 3HV) | Shaking flask scale; batch cultivation; 2.2 g/L, 58% PHA in CDM, 0.035 g/(L·h) | [55] |
| ´´ | ´´ | 220 g/L NaCl Dephenolized and native olive mill waste water (OMW) T = 37 °C | PHBHV (6.5 mol.% 3HV) | Shaking flask scale, batch cultivation; 43 wt.% PHA in CDM (concentration and productivity data inconsistent in publication) | [53] |
| ´´ | ´´ | 156 g/L NaCl; Glucose; nitrate or ammonia as N-source T = 37 °C | PHBHV (12.5 mol.% 3HV using nitrate) (16.9 mol.% 3HV using ammonia) | Shaking flask scale, batch cultivation; 0.63 g/L, 4.6% PHA in CDM, 0.035 g/(L·h) with ammonia (C/N = 8) 0.80 g/L, 9.3% PHA in CDM, 0.035 g/(L·h) with ammonia (C/N =8) | [44] |
| ´´ | ´´ | 156 g/L NaCl; T = 37 °C Glucose; varying phosphate concentrations | PHBHV (22.4 mol.% 3HV) | 500 mL shaking flasks, batch 0.95 g/L PHA, 15.6 % PHA in CDM; 0.007 g/(L·h) with optimum phosphate concentration 0.5 g/L KH2PO4 | [45] |
| ´´ | ´´ | 156 g/L NaCl; Different even- or add-numbered fatty acids T = 37 °C | PHBHV (random or b-PHBHV) (˂10 mol.% 3HV using even-numbered acids) (>87 mol.% 3HV using odd-numbered acids) | Shaking flask scale; batch and fed-batch 0.4–1.5 g/L PHA, 10.3–27.1 wt.% PHA in CDM, 0.003–0.010 g/(L·h) (fed-batch, dependent on C-source) | [66] |
| Hfx. volcanii | Dead Sea | 200 g/L NaCl; T = 37 °C Glucose | ? | Shaking flask scale; Below detection limit | [68] |
| ´´ | ´´ | 250 g/L NaCl; T = 37 °C Glucose + yeast extract | “PHB” | Shaking flask scale; 7 wt.% PHA in CDM | [29] |
| ´´ (strain BBK2) | Solar salterns of Ribandar in Goa, India | 200 g/L NaCl; T = 37 °C Sugarcane bagasse hydrolysate | ? (not identified) | Shaking flask scale; Not quantified | [78] |
| Hfx. gibbonsii | Salt pond at the coast near Alicante, Spain | “PHB” | Shaking flask scale; 1.2 wt.% PHA in CDM | [29] | |
| Har. marismortui | Dead Sea | 200 g/L NaCl; Raw and charcoal-pretreated vinasse from bioethanol production | PHB | Shaking flak scale; PHA content in CDM between 23 wt.% (10% non-detoxified vinasse) and 30 wt.% (100% charcoal-detoxified vinasse); 0.015 (non-detoxified) and 0.02 (detoxified) g/(L·h) PHB (2.8 and 4.5 g/L PHB, respectively) | [74] |
| Har. hispanica | Salt pond at the coast near Alicante, Spain | 250 g/L NaCl; T = 37 °C Glucose + yeast extract | “PHB” | Shaking flask scale; PHA content: 2.6 wt.% PHA in CDM | [29] |
| ´´ | ´´ | 200 g/L NaCl; T = 37 °C Glucose | PHBHV | 0.09 wt.% PHA in CDM | [68] |
| Har.sp. IRU1 | Hypersaline Urmia lake, Iran | 250 g/L NaCl 42 °C (other T tested) Glucose (other substrates tested) | PHB | Shaking flask scale; 66 wt.% PHB in CDM | [69] |
| ´´ | ´´ | 250 g/L NaCl 42 °C (other T tested) Glucose (other substrates tested) | PHB | Shaking flask scale; 62 (glucose), 57 (starch), 56 (sucrose), 55 (fructose), 40 (acetate), 39 (palmitic acid) wt.% PHB in CDM Max. PHA concentration and productivity: 0.98 g/L, 0.016 g/(L·h) (glucose) | [70] |
| 250 g/L NaCl 47 °C (other T tested) Petrochemical wastewater, tryptone | PHB | Shaking flask scale; Max. 46.6 wt.% PHB in CDM (2% petrochemical wastewater, yeast extract, 47 °C | [71] | ||
| ´´ | ´´ | 250 g/L NaCl 47 °C (other T tested) Crude oil, yeast extract (other N-sources tested) | PHB | Shaking flask scale; Max. 41.3 wt.% PHB in CDM (2% crude oil, yeast extract, 47 °C) | [72] |
| Har. Japonica (strain BS2) | Solar salterns of Ribandar in Goa, India | 200 g/L NaCl; T = 37 °C Sugarcane bagasse hydrolysate | ? (not identified) | Shaking flask scale; Not quantified | [78] |
| Hgm. borinquense (strain TN9) | Solar salterns of Marakkanam in Tamil Nadu, India | 200 g/L NaCl; T = 37 °C Glucose | PHB | Shaking flask scale; PHA content in CDM 14 wt.%; ca. 3 mg/(L·h) PHA | [76] |
| Hgm. Borinquense (strain E3) | Solar salterns of Marakkanam in Tamil Nadu, India | 200 g/L NaCl; T = 37 °C Glucose | PHBHV (21.5 mol.% 3HV) | Shaking flask scale; PHA content in CDM 74 wt.%; 0.21 g/(L·h) PHA | [77] |
| ´´ | ´´ | 200 g/L NaCl; T = 37 °C 25% and 50% hydrolyzed sugarcane bagasse | PHBHV (13.3 mol.% 3HV) | Shaking flak scale; PHA content in CDM between 45 and 50 wt.%; 0.0113 g/(L·h) PHBHV on 25% | [78] |
| ´´ | ´´ | 200 g/L NaCl; T = 37 °C Starch and carbon-rich fibrous waste (cassava bagasse) | PHBHV (13.1% 3HV with starch, 19.7% 3HV with cassava waste) | Shaking flask cultivations in batch mode; Starch: 4.6 g/L PHA, 0.02 g/(L·h), 74.2% PHA in CDM, Cassava bagasse: 1.52 g/L, 0.006 g/(L·h), 44.7% PHA in CDM | [79] |
| Hbt. noricense | Bore core of an Austrian Permian salt deposit | 200 g/L NaCl; T = 37 °C Glucose | PHBHV | Shaking flask scale; 0.11 wt.% PHA in CDM | [68] |
| Hcc. dombrowskii | Dry rock salt from Austrian alpine salt mine | Complex saline medium; T = 37 °C | PHBHV | Shaking flask scale; 0.16 wt.% PHA in CDM | [68] |
| Hcc. hamelinensis | Stromatolites from the Hamelin pool in the Australian Shark Bay | Complex saline medium; T = 37 °C | PHBHV | Shaking flask scale; Not quantified | [68] |
| Hcc. morrhuae | Dead Sea | Complex saline medium; T = 37 °C | PHBHV | Shaking flask scale; Not quantified | [68] |
| Hcc. qingdaonensis | Crude sea-salt sample collected near Qingdao, PR China | Complex saline medium; T = 37 °C | PHBHV | Shaking flask scale; Not quantified | [69] |
| Hcc. saccharolyticus | Salt; Cadiz, Spain | Complex saline medium; T = 37 °C | PHB | Shaking flask scale; 1.2 wt.% PHA in CDM | [68] |
| Hcc. salifodinae | Austrian alpine rock salt | Complex saline medium; T = 37 °C | PHBHV | Shaking flask scale; 0.06 wt.% PHA in CDM | [68] |
| ´´ (strain BK6) | Solar salterns of Ribandar in Goa, India | 200 g/L NaCl; T = 37 °C Sugarcane bagasse hydrolysate | n.d. | Shaking flask scale; Below detection limit | [78] |
| Hrr. chaviator | Sea salt in Baja California, Mexico, Western Australia and Greece | 200 g/L NaCl; T = 37 °C Glucose | PHBHV | Shaking flask scale; Not quantified | [68] |
| Hrr. coriense | Dead Sea | 200 g/L NaCl; T = 37 °C Glucose | PHBHV | Shaking flask scale; Not quantified | [68] |
| Hbf. Haloterrestris (“strain 135(T)”) | Samples collected from surface of hypersaline soil collected in Aswan, Egypt | 220 g/L NaCl; T = 42 °C (other T tested) Acetate + butyric acid or complex media | PHB | Shaking flask scale; 40 wt.% PHB in CDM on butyric acid, 15 wt.% PHB in CDM on complex medium | [83] |
| Nnm. ajinwuensis (=altunense) | Indian salt production pans | 200 g/L NaCl (other salinities tested); T = 37 °C Glucose | PHBHV (13.9 mol.% 3HV) | Repeated batch cultivations in shaking flaks PHA content in CDM 61 wt.%; ca. 15 g/L PHA; 0.21 g/(L·h) PHA | [61] |
| Nnm. Palladium (strain JCM 8980, =isolate 1KYS1) | Kayacik saltern, Turkey | 250 g/L NaCl; Starch | PHBHV (25 mol.% 3HV) | Shaking flak cultivations; PHA content in CDM 53 wt.%; 0.3 mg/(L·h) PHA | [84] |
| Nbt. gregoryi | Soda slat lake liquors from the East African Magadi soda lake | 200 g/L NaCl; T = 37 °C alkaliphile; Carbohydrates | PHB | Shaking flask scale; 0.62 wt.% PHB | [68] |
| Ncc. occultus | Magadi Lake, Kenia | 200 g/L NaCl; T = 37 °C alkaliphile; Glucose | PHBHV | Shaking flask scale; 3.1 wt.% PHB | [68] |
| Hrd. utahensis | Sediments of the Great Salt Lake in Utah | 270 g/L NaCl (maximum described salinity optimum for living beings!); T = 50 °C Limited number of carbohydrates | Not specified (“PHB is produced”) | Shaking flask scale; No quantitative data | [93] |
| Hrd. tiamatea | Hypersaline, anoxic deep-sea brine-sediment interface of the Red Sea | 270 g/L NaCl (maximum described salinity optimum for living beings!); T = 45 °C Starch | Not specified (“PHB is produced”) | Shaking flask scale; No quantitative data | [94] |
| Hrd. rudnickae | Borehole at Polish salt mine | 200 g/L NaCl; T = 40 °C | Not specified (“PHB is produced”) | Shaking flask scale; No quantitative data | [96] |
| Hqr. walsbyi | Sinai peninsula and saltern crystallizers in Australia and Spain | 140–360 g/L NaCl for growth (optimum: >180 g/L); T = 25–45 °C | PHB | Shaking flaks scale; ˂1 wt.% PHA in CDM | [68,88,89] |
| Species | Strain Isolation | Salinity in Medium, Substrates, T | Product | Production Scale/Productivity | Ref. |
|---|---|---|---|---|---|
| Hfx. mediterranei | Salt pond at the coast near Alicante, Spain | 250 g/L marine salts Starch (20 g/L) Glucose (10 g/L) T = 38 °C (other T tested) | PHBHV (in publication: “PHB”) | Stable (monoseptic) continuous cultivation over 3 months in 1.5 L bioreactor; 6.5 g/L PHA on starch 3.5 g/L on glucose | [32] |
| ´´ | ´´ | 150 g/L NaCl; T = 37 °C Glucose plus yeast extract | PHBHV (10 mol.% 3HV) | 10 L bioreactor; fed-batch feeding; 0.21 g/(L·h), 13 g/L PHA, 0.7 g PHA in CDM | [37] |
| ´´ | ´´ | 200 g/L NaCl; T = 37 °C Hydrolyzed whey permeate Hydrolyzed whey permeate plus GBL | PHBHV (6 mol.% 3HV) P(3HB-co-3HV-co-4HB) (21.8 mol.% 3HV, 5.1 mol.% 4HB) | 42 L bioreactor fed-batch process; 0.09 g/(L·h), 12.2 g/L PHBHV 0.14 g/(L·h), 14.7 g/L poly(3HB-co-3HV-co-4HB) | [47] |
| ´´ | ´´ | 150 g/L NaCl; T = 37 °C Hydrolyzed whey permeate | PHBHV (10 mol.% 3HV) | 200 L fed-batch pilot process (300 L bioreactor); techno-economic assessment 7.2 g/L PHA, 66 wt.% PHA in CDM, 0.11 g/(L·h) | [58,59] |
| ´´ | ´´ | 200 g/L NaCl; T = 37 °C Hydrolyzed whey permeate, spent fermentation broth and saline cell debris from previous whey-based processes | PHBHV (10 mol.% 3HV) | 10 L bioreactor batch process 0.04 g/(L·h), 2.28 g/L PHA | [58] |
| ´´ | ´´ | 156 g/L NaCl; T = 37 °C Hydrolyzed whey permeate, elevated trace element concentration | PHBHV (˂2 mol.% 3HV) | 2 L bioreactor batch process 8 g/L PHBHV, 0.17 g/(L·h), 53 wt.% PHA in CDM | [48] |
| ´´ | ´´ | 150 g/L NaCl; T = 37 °C CGP; CGP plus GBL | PHBHV (10 mol.% 3HV) P(3HB-co-3HV-co-4HB) (11 mol.% 3HV, 5 mol.% 4HB) | 42 L/10 L bioreactor fed-batch process; 0.12 g/(L·h), 16.2 g/L PHA 0.10 g/(L·h), 11.1 g/L PHA | [33] |
| ´´ | ´´ | 200–230 g/L NaCl; T = 37 °C Native cornstarch treated via enzymatic reactive extrusion | PHBHV (10.4 mol.% 3HV) | 6 L bioreactor pH-stat fed-batch process; 0.28 g/(L·h), 0.508 g PHA in CDM; 20 g/L PHA | [49] |
| ´´ | ´´ | 234 g/L NaCl; T = 37 °C Mixtures of extruded rice bran plus extruded cornstarch | PHBHV (about 11 mol.% 3HV) | 5 L bioreactor; pH-stat feeding strategy; 77.8 g/L PHA | [50] |
| ´´ | 200 g/L NaCl; Rice-based stillage T: n.r, | PHBHV (17.9 mol.% 3HV) | Unsterile 50 L plug-flow PMMA bioreactor; techno-economic assessment 13 g/L PHA, 63 wt.% PHA in CDM, 0.14 g/(L·h) | [52] | |
| ´´ | ´´ | 156 g/L NaCl; Mixes of butyric & valeric acid; Tween80 T = 37 °C | PHBHV (43 mol.% 3HV at butyric/valeric acid = 56/44) | Fed-batch bioreactor cultivation 4.01 g/L PHA, 59 wt.% PHA in CDM; 0.01 g/(L·h) | [65] |
| ´´ (EPS-negative mutant; strain “ES1”) | ´´ | 140 g/L total salts (110 g/L NaCl) Glucose and valerate T = 37 °C | b-PHBHV (up to 50 mol.% 3HV at end of fermentation) | 7 L fed-batch bioreactor cultivation Results only reported for shaking flask experiments: max. ca. 5 g/L PHA, 50 wt.% PHA in CDM; 0.17 g/(L·h) | [64] |
| Hgr. amylolyticum | Tainan marine solar saltern near Lianyungang, PR China | 200 g/L NaCl; T = 37 °C Glucose | PHBHV (>20 mol.% 3HV) | 7.5 L bioreactor; fed-batch feeding strategy; 0.074 g/(L·h), 14 g/L PHBHV, 48 wt.% PHA in CDM | [89] |
| Hpg. Aswanensis (“strain 56”) | Samples collected from surface of hypersaline soil collected in Aswan, Egypt | 250 g/L NaCl; T = 40 °C Sodium acetate and butyric acid | PHB | Corrosion-resistant 8 L composite bioreactor; batch feeding; 0.0045 g/(L·h), 53 wt.% PHB in CDM, 4.6 g/L PHB, 0.018 g(/L·h) | [80] |
| Htg. hispanica | Saltern crystallizer pond at Fuente de Piedra saline lake, Malaga, Spain | 200 g/L NaCl; T = 37 °C Complex medium Carrot waste | PHB (complex medium) P(3HB-co-3HV-co-4HB) (carrot waste) | Bioreactor; batch setups and bioreactor equipped with ultrafiltration unit 0.135 wt.% PHA in CDM (complex medium); 0.125 wt.% PHA in CDM (carrot waste) | [91] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koller, M. Polyhydroxyalkanoate Biosynthesis at the Edge of Water Activity-Haloarchaea as Biopolyester Factories. Bioengineering 2019, 6, 34. https://doi.org/10.3390/bioengineering6020034
Koller M. Polyhydroxyalkanoate Biosynthesis at the Edge of Water Activity-Haloarchaea as Biopolyester Factories. Bioengineering. 2019; 6(2):34. https://doi.org/10.3390/bioengineering6020034
Chicago/Turabian StyleKoller, Martin. 2019. "Polyhydroxyalkanoate Biosynthesis at the Edge of Water Activity-Haloarchaea as Biopolyester Factories" Bioengineering 6, no. 2: 34. https://doi.org/10.3390/bioengineering6020034
APA StyleKoller, M. (2019). Polyhydroxyalkanoate Biosynthesis at the Edge of Water Activity-Haloarchaea as Biopolyester Factories. Bioengineering, 6(2), 34. https://doi.org/10.3390/bioengineering6020034





