Looking for A Place for Dose-Dense TMZ Regimens in GBM Patients: An Experience with MGMT Exploratory Evaluation
Abstract
1. Introduction
2. Materials and Methods
2.1. Dose Finding Cohort
2.2. Patient Eligibility
2.3. Treatment Schedule
2.4. MGMT Analysis
2.5. Study Design and Statistical Analysis
3. Results
3.1. Patients’ Features
3.2. Activity and Efficacy
3.3. Dose-Intensity
3.4. Toxicity
3.5. Subsequent Treatments
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Stupp, R.; Mason, W.P.; Van Den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Gerson, S.L. MGMT: Its role in cancer aetiology and cancer therapeutics. Nat. Rev. Cancer 2004, 4, 296–307. [Google Scholar] [CrossRef]
- Esteller, M.; Garcia-Foncillas, J.; Andion, E.; Goodman, S.N.; Hidalgo, O.F.; Vanaclocha, V.; Baylin, S.B.; Herman, J.G. Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N. Engl. J. Med. 2000, 343, 1350–1354. [Google Scholar] [CrossRef] [PubMed]
- Hegi, M.E.; Diserens, A.C.; Gorlia, T.; Hamou, M.F.; De Tribolet, N.; Weller, M.; Kros, J.M.; Hainfellner, J.A.; Mason, W.; Mariani, L.; et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N. Engl. J. Med. 2005, 352, 997–1003. [Google Scholar] [CrossRef]
- Tolcher, A.W.; Gerson, S.L.; Denis, L.; Geyer, C.; Hammond, L.A.; Patnaik, A.; Goetz, A.D.; Schwartz, G.; Edwards, T.; Reyderman, L.; et al. Marked inactivation of O6-alkylguanine-DNA alkyltransferase activity with protracted temozolomide schedules. Br. J. Cancer 2003, 88, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Nagane, M. Dose-dense temozolomide: Is it still promising? Neurol. Med. Chir. 2015, 55, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Brandes, A.A.; Tosoni, A.; Cavallo, G.; Bertorelle, R.; Gioia, V.; Franceschi, E.; Biscuola, M.; Blatt, V.; Crino, L.; Ermani, M. Temozolomide 3 weeks on and 1 week off as first line therapy for recurrent glioblastoma: Phase II study from gruppo italiano cooperativo di neuro-oncologia (GICNO). Br. J. Cancer 2006, 95, 1155–1160. [Google Scholar] [CrossRef]
- Perry, J.R.; Bélanger, K.; Mason, W.P.; Fulton, D.; Kavan, P.; Easaw, J.; Shields, C.; Kirby, S.; Macdonald, D.R.; Eisenstat, D.D.; et al. Phase II trial of continous dose-intense temozolomide in recurrent malignant glioma: RESCUE study. J. Clin. Oncol. 2010, 28, 2051–2057. [Google Scholar] [CrossRef]
- Strik, H.M.; Buhk, J.H.; Wrede, A.; Hoffmann, A.L.; Bock, H.C.; Christmann, M.; Kaina, B. Rechallange with temozolomide with different scheduling is effective in recurrent malignant gliomas. Mol. Med. Rep. 2008, 1, 863–867. [Google Scholar]
- Wick, A.; Felsberg, J.; Steinbach, J.P.; Herrlinger, U.; Platten, M.; Blaschke, B.; Meyermann, R.; Reifenberger, G.; Weller, M.; Wick, W. Efficacy and tolerability of temozolomide in an alternating weekly regimen in patients with recurrent glioma. J. Clin. Oncol. 2007, 25, 3357–3361. [Google Scholar] [CrossRef]
- Brada, M.; Stenning, S.; Gabe, R.; Thompson, L.C.; Levy, D.; Rampling, R.; Erridge, S.; Saran, F.; Gattamaneni, R.; Hopkins, K.; et al. Temozolomide versus procarbazine, lomustine and vincristine in recurrent high-grade glioma. J. Clin. Oncol. 2010, 28, 4601–4608. [Google Scholar] [CrossRef] [PubMed]
- Han, S.J.; Rolston, J.D.; Molinaro, A.M.; Clarke, J.L.; Prados, M.D.; Chang, S.M.; Berger, M.S.; DeSilva, A.; Butowski, N.A. Phase II trial of 7 days on/7 days off temozolomide for recurrent high grade glioma. Neuro-Oncology 2014, 16, 1255–1262. [Google Scholar] [CrossRef]
- Taal, W.; Segers-van Rijn, J.M.; Kros, J.M.; van Heuvel, I.; van der Rijt, C.C.; Bromberg, J.E.; Smitt, P.A.; van den Bent, M.J. Dose dense 1 week on/1 week off temozolomide in recurrent glioma: A retrospective study. J. Neur-Oncol. 2012, 108, 195–200. [Google Scholar] [CrossRef]
- Berrocal, A.; Segura, P.P.; Gil, M.; Balana, C.; Lopez, J.G.; Yaya, R.; Rodríguez, J.; Reynes, G.; Gallego, O.; Iglesias, L. Extended-schedule dose-dense temozolomide in refractory gliomas. J. Neuro-Oncol. 2010, 96, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.R.; Wang, M.; Aldape, K.D.; Stupp, R.; Hegi, M.E.; Jaeckle, K.A.; Armstrong, T.S.; Wefel, J.S.; Won, M.; Blumenthal, D.T.; et al. Dose-Dense Temozolomide for Newly Diagnosed Glioblastoma: A Randomized Phase III Clinical Trial. J. Clin. Oncol. 2013, 31, 4085–4091. [Google Scholar] [CrossRef]
- Napoleoni, L.; Bruera, G.; Adinolfi, M.; Catalucci, A.; Bonfili, P.; Di Staso, M.; Ricci, A.; Di Cesare, E.; Galzio, R.; Ricevuto, E. Temozolomide dose-dense regimen in high grade gliomas: Dose-finding/phase II study. Ann. Oncol. 2015, 26, vi. [Google Scholar] [CrossRef]
- Storer, B.E. Design and analysis of phase I clinical trials. Biometrics 1989, 45, 925–937. [Google Scholar] [CrossRef] [PubMed]
- Simon, R.; Rubinstein, L.; Arbuck, S.G.; Christian, M.C.; Freidlin, B.; Collins, J. Accelereted titration designs for phase I clinical trials in oncology. J. Natl. Cancer Inst. 1997, 89, 1138–1147. [Google Scholar] [CrossRef]
- Kleihues, P.; Louis, D.N.; Scheithauer, B.W.; Rorke, L.B.; Reifenberger, G.; Burger, P.C.; Cavenee, W.K. The WHO Classification of Tumors of the Nervous System. J. Neuropathol. Exp. Neurol. 2002, 61, 215–225. [Google Scholar] [CrossRef]
- Louis, D.N.; Ohgaki, H.; Wiestler, O.D.; Cavenee, W.K.; Burger, P.C.; Jouvet, A.; Scheithauer, B.W.; Kleihues, P. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007, 114, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Therasse, P.; Arbuck, S.G.; Eisenhauer, E.A.; Wanders, J.; Kaplan, R.S.; Rubinstein, L.; Verweij, J.; Van Glabbeke, M.; van Oosterom, A.T.; Christian, M.C.; et al. New guidelines to evaluate the response to treatment in solid tumors: European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J. Natl. Cancer Inst. 2000, 92, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, E.L.; Meier, P. Nonparametric estimation of incomplete observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- Sanai, N.; Berger, M.S. Glioma extent of resection and its impact on patient outcome. Neurosurgery 2008, 62, 753–764, discussion 264–266. [Google Scholar] [CrossRef] [PubMed]
- Morgan, E.R.; Norman, A.; Laing, K.; Seal, M.D. Treatment and outcomes for glioblastoma in elderly compared with non-elderly patients: A population-based study. Curr. Oncol. 2017, 24, e92–e98. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.A. On the interpretation of χ2 from contingency tables, and the calculation of P. J. R. Stat. Soc. 1922, 85, 87–94. [Google Scholar] [CrossRef]
- Cox, D.R. Regression models and life tables (with discussion). J. R. Stat. Soc. (Ser. B) 1972, 74, 187–200. [Google Scholar]
- Marill, K.A. Advanced statistics: Linear regression, part II: Multiple linear regression. Acad. Emerg. Med. 2004, 11, 94–102. [Google Scholar] [CrossRef]
- Gallego, O. Nonsurgical treatment of recurrent glioblastoma. Curr. Oncol. 2015, 22, e273–e281. [Google Scholar] [CrossRef]
- Donson, A.M.; Addo-Yobo, S.O.; Handler, M.H.; Gore, L.; Foreman, N.K. MGMT promoter methylation correlates with survival benefit and sensitivity to temozolomide in pediatric glioblastoma. Pediatr. Blood Cancer 2007, 48, 403–407. [Google Scholar] [CrossRef]
- Möllemann, M.; Wolter, M.; Felsberg, J.; Collins, V.P.; Reifenberger, G. Frequent promoter hypermethylation and low expression of the MGMT gene in oligodendroglial tumors. Int. J. Cancer 2005, 113, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Brell, M.; Tortosa, A.; Verger, E.; Gil, J.M.; Viñolas, N.; Villá, S.; Acebes, J.J.; Caral, L.; Pujol, T.; Ferrer, I.; et al. Prognostic significance of O6-methylguanine-DNA methyltransferase determined by promoter hypermethylation and immunohistochemical expression in anaplastic gliomas. Clin. Cancer Res. 2005, 11, 5167–5174. [Google Scholar] [CrossRef] [PubMed]
- Pieper, R.O.; Patel, S.; Ting, S.A.; Futscher, B.W.; Costello, J.F. Methylation of CpG island transcription factor binding sites is unnecessary for aberrant silencing of the human MGMT gene. J. Biol. Chem. 1996, 271, 13916–13924. [Google Scholar] [CrossRef] [PubMed]
- Bearzatto, A.; Szadkowski, M.; Macpherson, P.; Jiricny, J.; Karran, P. Epigenetic regulation of the MGMT and hMSH6 DNA repair genes in cells resistant to methylating agents. Cancer Res. 2000, 60, 3262–3270. [Google Scholar]
- Bhakat, K.K.; Mitra, S. CpG methylation-dependent repression of the human O6-methylguanine-DNA methyltransferase gene linked to chromatin structure alteration. Carcinogenesis 2003, 24, 1337–1345. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.W.; Schiff, D. Treatment considerations for MGMT-unmethylated glioblastoma. Curr. Neurol. Neurosci. Rep. 2015, 15, 507. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Plutynski, A.; Ward, S.; Rubin, J.B. An integrative view on sex differences in brain tumors. Cell. Mol. Life Sci. 2015, 72, 3323–3342. [Google Scholar] [CrossRef] [PubMed]
- Vogelbaum, M.A. The benefit of surgical resection in recurrent glioblastoma. Neuro-Oncology 2016, 18, 462–463. [Google Scholar] [CrossRef] [PubMed]
- McGranahan, T.; Li, G.; Nagpal, S. History and current state of immunotherapy in glioma and brain metastasis. Ther. Adv. Med. Oncol. 2017, 9, 347–368. [Google Scholar] [CrossRef]
- Winograd, E.K.; Ciesielski, M.J.; Fenstermaker, R.A. Novel vaccines for glioblastoma: Clinical update and perspective. Immunotherapy 2016, 8, 1293–1308. [Google Scholar] [CrossRef]
| No. of Patients (%), Total 36 | |
|---|---|
| Gender | |
| Male | 23 (63.2) |
| Female | 13 (36.1) |
| Age (Years) | |
| Median | 61 |
| Range | (24–75) |
| Elderly (≥65) | 13 (36.1) |
| ECOG-PS | |
| 0–1 | 28 (77.8) |
| 2 | 8 (22.2) |
| Type of Surgery | |
| Extensive resection | 19 (52.8) |
| Non–extensive resection | 17 (47.2) |
| MGMT (IHC Expression) | |
| Positive | 29 (80.6) |
| Negative | 7 (19.4) |
| Overall | ||
|---|---|---|
| N° | % | |
| Evaluable Patients | 36 | 100 |
| Objective Response Rate | 19.4% (95% CI: 7.8–40.1) | |
| Partial Response | 6 | |
| Complete Response | 1 | |
| Disease Control Rate | 55.5% (95% CI: 33.9–85.8) | |
| Stable Disease | 13 | |
| Progression Disease | 16 | |
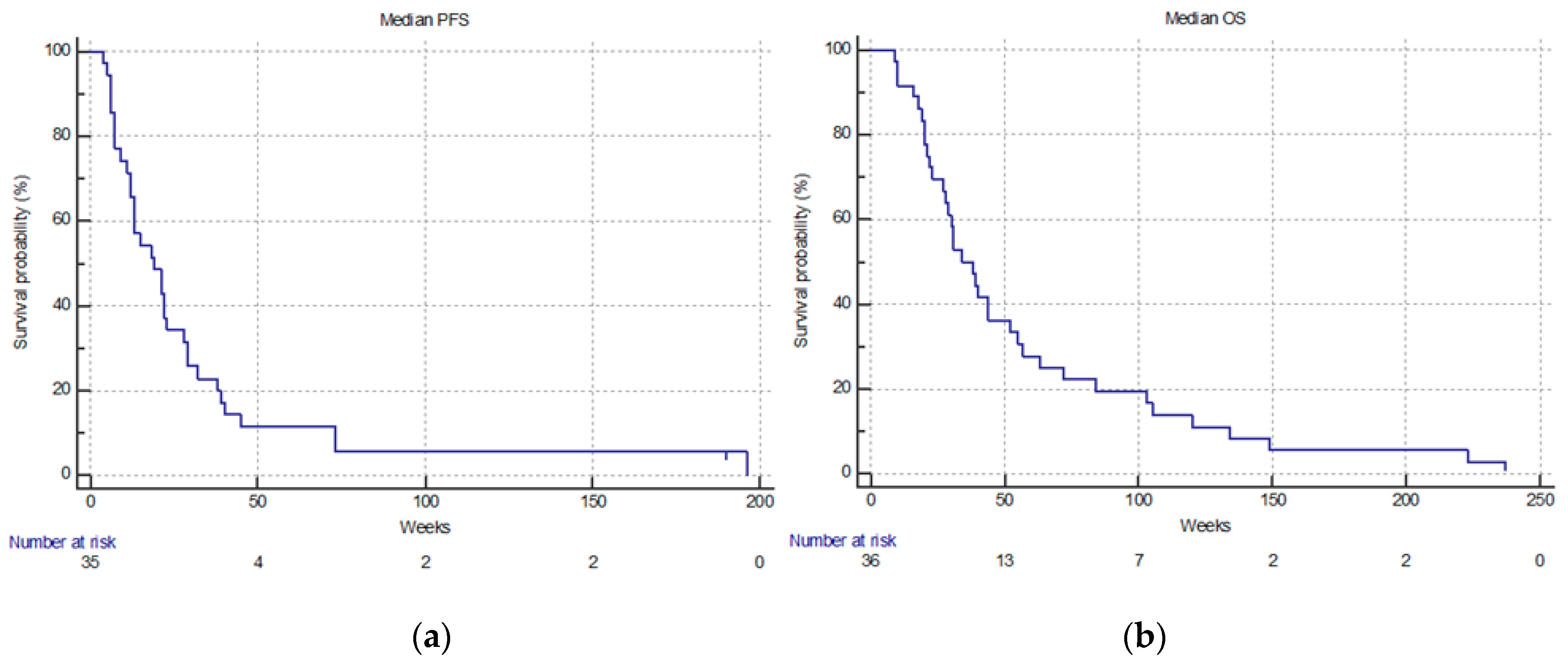
| Median PFS (weeks) | 19.0 | |
| Range | 4–196 | |
| Progression events | 35 | |
| Median OS (weeks) | 34 | |
| Range | 9–237 | |
| Deaths | 35 | |
| Univariate Analysis | |||
|---|---|---|---|
| Variable (N°) | DCR (95% CI) | p-value | |
| Age | |||
| Non Elderly (23) | 56.5% (34.4–76.8) | 1.0000 | |
| Elderly (13) | 53.8 % (25.1–80.7) | ||
| ECOG-PS | |||
| 0–1 (28) | 67.9% (47.6–84.1) | 0.0121 * | |
| 2 (8) | 12.5% (0.3–52.6) | ||
| Sex | |||
| Male (23) | 39.1% (19.7–61.4) | 0.0139 * | |
| Female (13) | 84.6% (54.5–98.1) | ||
| Type of resection | |||
| Extensive (19) | 68.4 % (43.4–87.4) | 0.1786 | |
| Non-extensive (17) | 41.2% (18.4–67.1) | ||
| MGMT expression | |||
| Positive (28) | 51.7% (32.5–70.5) | 0.4264 | |
| Negative (7) | 71.4% (29.1–96.3) | ||
| MGMT expression | |||
| ≥50% (12) | 41.7% (15.1–72.3) | 0.2983 | |
| <50% (24) | 62.5% (40.5–81.2) | ||
| MGMT expression | |||
| ≥70% (7) | 14.3% (0.3–57.8) | 0.0298 * | |
| <70% (29) | 65.5% (45.6–82.1) | ||
| Multivariate Analysis | |||
| Variable | Coefficient | Std. Error | p-value |
| ECOG-PS 0–1 vs 2 | 0.3482 | 0.1712 | 0.0503 |
| Sex male vs female | 0.3630 | 0.1481 | 0.0199 * |
| MGMT (70% cut-off) | 0.5153 | 0.1650 | 0.0038 * |
| Coefficient of Determination R2: 0.4567 | |||
| Univariate Analysis | ||||
|---|---|---|---|---|
| Variable (N°) | PFS | OS | ||
| HR (95% CI) | p-value | HR (95% CI) | p-value | |
| Age | ||||
| Non Elderly (23) Elderly (13) | 1.3 (0.6–2.7) | 0.4068 | 1.7 (0.8–3.5) | 0.1526 |
| ECOG-PS | ||||
| 0–1 (28) 2 (8) | 26.3 (5.2–132.4) | 0.0001 * | 5.1 (2.1–12.3) | 0.0003 * |
| Sex | ||||
| Male (23) Female (13) | 2.2 (1.1–4.5) | 0.0328 * | 1.2 (0.6–2.4) | 0.5828 |
| Type of resection | ||||
| Extensive (19) Non-extensive (17) | 3.2 (1.5–7.0) | 0.0028 * | 2.7 (1.3–5.6) | 0.0058 * |
| MGMT expression | ||||
| Positive (28) Negative (7) | 1.6 (0.6–4.3) | 0.3251 | 1.2 (0.5–2.7) | 0.7209 |
| MGMT expression | ||||
| ≥ 50% (12) < 50% (24) | 1.2 (0.6–2.4) | 0.6838 | 0.9 (0.4–1.9) | 0.8945 |
| MGMT expression | ||||
| ≥ 70% (7) < 70% (29) | 2.7 (1.1–6.9) | 0.0379 * | 2.1 (0.9–5.2) | 0.0803 |
| Multivariate Analysis | ||||
| ECOG-PS | 11.5 (1.9–66.9) | 0.0064 * | 3.3 (1.2–9.1) | 0.0179 * |
| Type of resection | 2.1 (0.8–5.3) | 0.0898 | 1.9 (0.8–4.5) | 0.1228 |
| MGMT (cut-off 70%) | 2.5 (1.01–6.38) | 0.0592 | - | - |
| Sex | 2.54 (0.9–6.6) | 0.1507 | - | - |
| Number | Overall Patients | |||
|---|---|---|---|---|
| NCI-CTC Grade | 1 | 2 | 3 | 4 |
| Asthenia (%) | 17 (47.2) | 5 (13.9) | - | - |
| Anorexia (%) | 4 (11.1) | - | - | - |
| Nausea (%) | 7 (19.4) | 1 (2.8) | - | - |
| Vomiting (%) | 3 (8.3) | 1 (2.8) | - | - |
| Dysgeusia (%) | 3 (8.3) | 1 (2.8) | - | - |
| Mucositis (%) | 1 (2.8) | - | - | - |
| Constipation (%) | 12 (33.3) | 4 (11.1) | - | - |
| Rash (%) | 1 (2.8) | 1 (2.8) | - | - |
| Increased transaminases (%) | 17 (47.2) | 6 (16.7) | - | - |
| Edema (%) | 7 (19.4) | 2 (5.6) | - | - |
| Anemia (%) | 5 (13.9) | 3 (8.3) | 1 (2.8) | - |
| Leukopenia (%) | 12 (33.3) | 9 (25) | 4 (11.1) | 1 (2.8) |
| Neutropenia (%) | 7 (19.4) | 4 (11.1) | 3 (8.3) | 1 (2.8) |
| Thrombocytopenia (%) | 9 925) | 4 (11.1) | 2 (5.6) | 2 (5.6) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Napoleoni, L.; Cortellini, A.; Cannita, K.; Parisi, A.; Dal Mas, A.; Calvisi, G.; Venditti, O.; Lanfiuti Baldi, P.; Cocciolone, V.; Ricci, A.; et al. Looking for A Place for Dose-Dense TMZ Regimens in GBM Patients: An Experience with MGMT Exploratory Evaluation. Bioengineering 2019, 6, 11. https://doi.org/10.3390/bioengineering6010011
Napoleoni L, Cortellini A, Cannita K, Parisi A, Dal Mas A, Calvisi G, Venditti O, Lanfiuti Baldi P, Cocciolone V, Ricci A, et al. Looking for A Place for Dose-Dense TMZ Regimens in GBM Patients: An Experience with MGMT Exploratory Evaluation. Bioengineering. 2019; 6(1):11. https://doi.org/10.3390/bioengineering6010011
Chicago/Turabian StyleNapoleoni, Luca, Alessio Cortellini, Katia Cannita, Alessandro Parisi, Antonella Dal Mas, Giuseppe Calvisi, Olga Venditti, Paola Lanfiuti Baldi, Valentina Cocciolone, Alessandro Ricci, and et al. 2019. "Looking for A Place for Dose-Dense TMZ Regimens in GBM Patients: An Experience with MGMT Exploratory Evaluation" Bioengineering 6, no. 1: 11. https://doi.org/10.3390/bioengineering6010011
APA StyleNapoleoni, L., Cortellini, A., Cannita, K., Parisi, A., Dal Mas, A., Calvisi, G., Venditti, O., Lanfiuti Baldi, P., Cocciolone, V., Ricci, A., & Ficorella, C. (2019). Looking for A Place for Dose-Dense TMZ Regimens in GBM Patients: An Experience with MGMT Exploratory Evaluation. Bioengineering, 6(1), 11. https://doi.org/10.3390/bioengineering6010011





