The Emerging Role of Amino Acid PET in Neuro-Oncology
Abstract
1. Introduction
2. Role of Vascularity in Magnetic Resonance Imaging of CNS Tumors
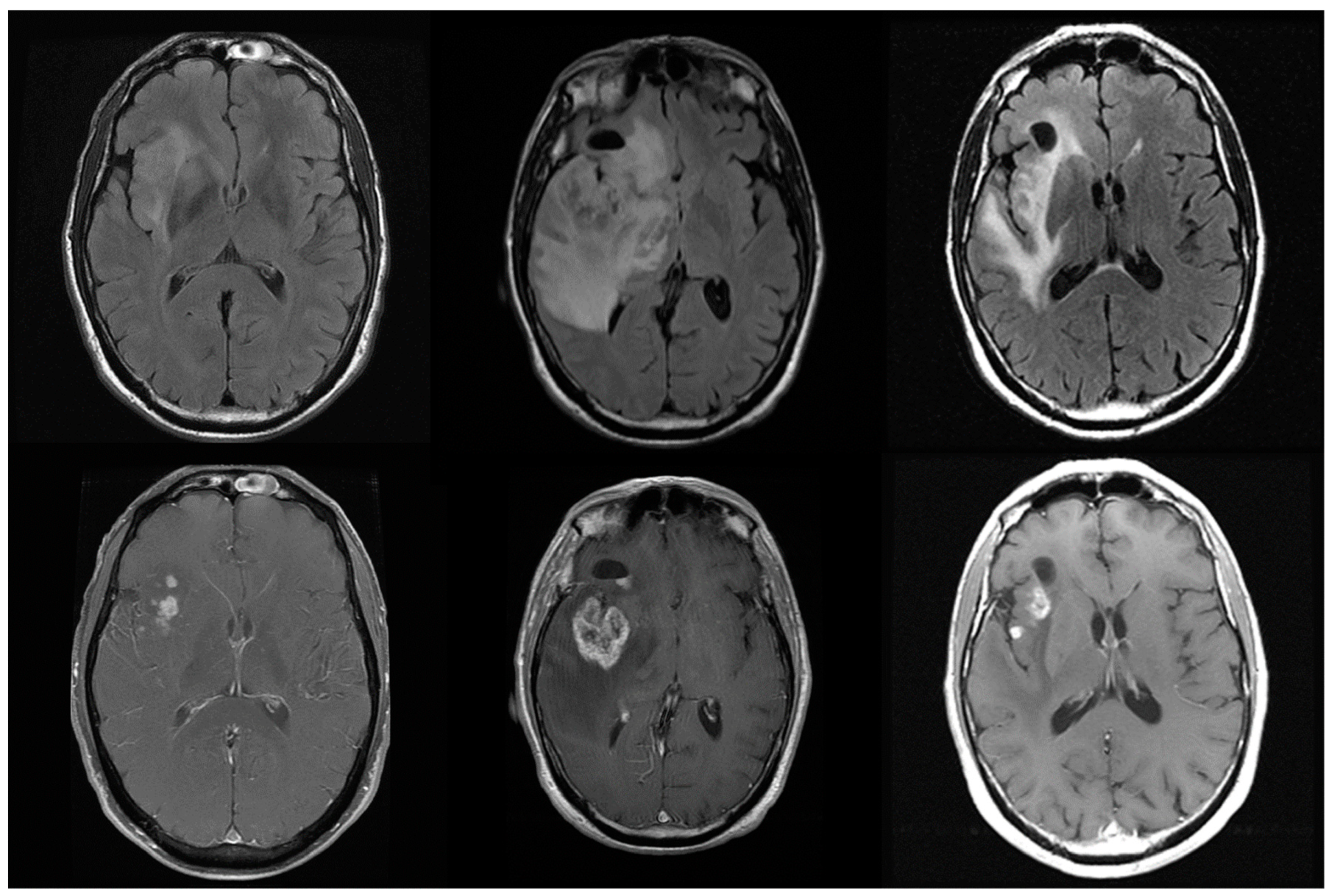
3. Limitations of Treatment Response Assessments by MRI
4. Positron Emission Tomography of CNS Tumors
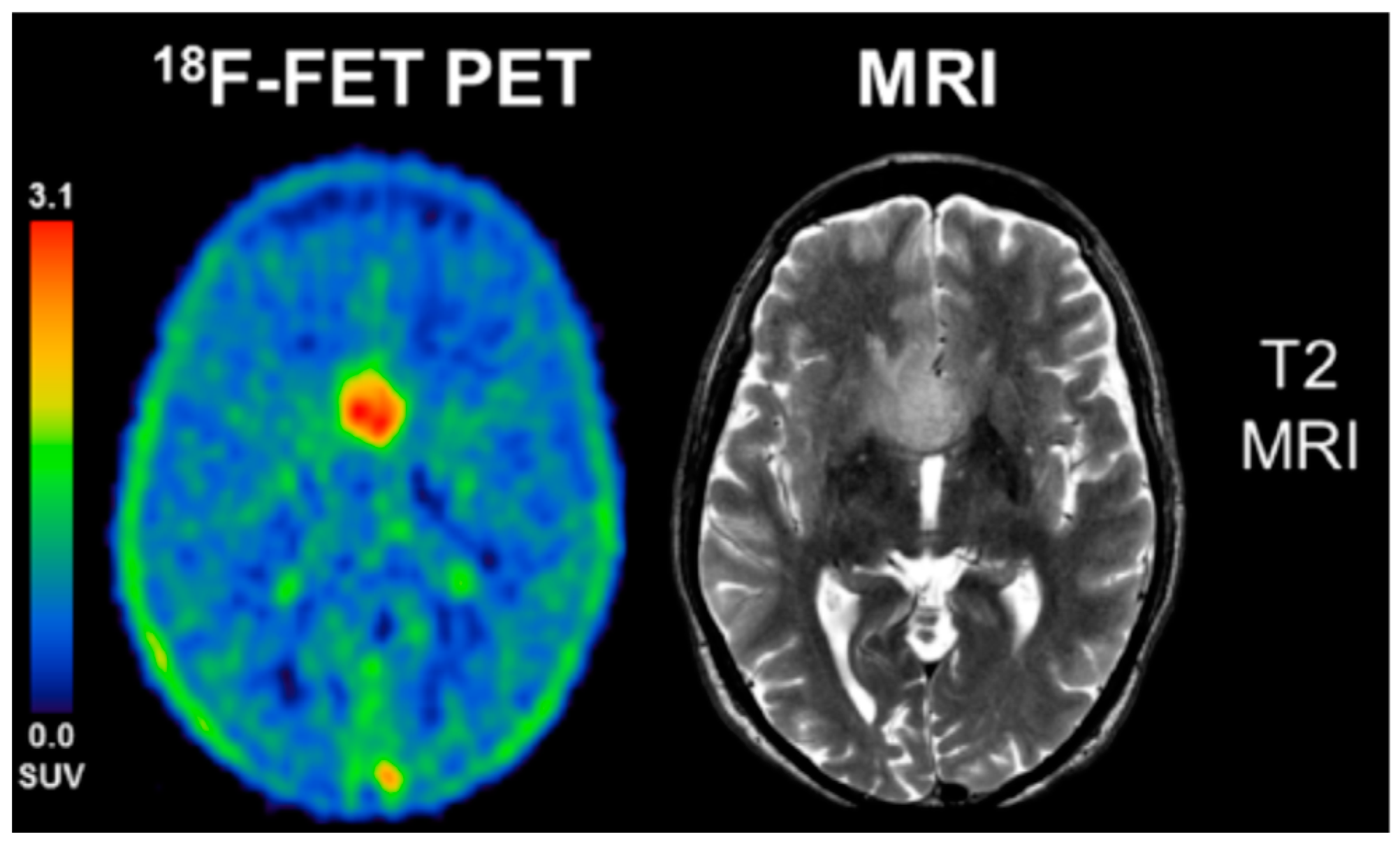
4.1. Amino Acid PET
4.2. Response to Therapy
4.3. Grade Differentiation
4.4. Prognostication
4.5. Biopsy Guidance
4.6. Case Studies Validating Amino Acid PET
5. Combined PET/MRI for the Management of CNS Tumors
6. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Cancer Facts & Figures 2018. Available online: https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2018.html (accessed on 10 September 2018).
- Ostrom, Q.T.; Gittleman, H.; Liao, P.; Vecchione-Koval, T.; Wolinsky, Y.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary brain and other central nervous system tumors diagnosed in the United States in 2010–2014. Neuro-Oncology 2017, 19 (Suppl. S5), v1–v88. [Google Scholar] [CrossRef] [PubMed]
- Nayak, L.; Lee, E.Q.; Wen, P.Y. Epidemiology of brain metastases. Curr. Oncol. Rep. 2012, 14, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Ohgaki, H.; Kleihues, P. Population-based studies on incidence, survival rates, and genetic alterations in astrocytic and oligodendroglial gliomas. J. Neuropathol. Exp. Neurol. 2005, 64, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J. Blood-brain barrier structure and function and the challenges for CNS drug delivery. J. Inherit. Metab. Dis. 2013, 36, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Da Fonseca, A.C.; Matias, D.; Garcia, C.; Amaral, R.; Geraldo, L.H.; Freitas, C.; Lima, F.R. The impact of microglial activation on blood-brain barrier in brain diseases. Front. Cell. Neurosci. 2014, 8, 362. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Ronnback, L.; Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.C. Blood-brain barrier breakdown in septic encephalopathy and brain tumours. J. Anat. 2002, 200, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, M.C.; Saadoun, S.; Davies, D.C.; Bell, B.A. Emerging molecular mechanisms of brain tumour oedema. Br. J. Neurosurg. 2001, 15, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Margolin, K.; Ernstoff, M.S.; Hamid, O.; Lawrence, D.; McDermott, D.; Puzanov, I.; Wolchok, J.D.; Clark, J.I.; Sznol, M.; Logan, T.F.; et al. Ipilimumab in patients with melanoma and brain metastases: An open-label, phase 2 trial. Lancet. Oncol. 2012, 13, 459–465. [Google Scholar] [CrossRef]
- Jain, R.; Griffith, B.; Alotaibi, F.; Zagzag, D.; Fine, H.; Golfinos, J.; Schultz, L. Glioma Angiogenesis and Perfusion Imaging: Understanding the Relationship between Tumor Blood Volume and Leakiness with Increasing Glioma Grade. AJNR Am. J. Neuroradiol. 2015, 36, 2030–2035. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.K.; di Tomaso, E.; Duda, D.G.; Loeffler, J.S.; Sorensen, A.G.; Batchelor, T.T. Angiogenesis in brain tumours. Nat. Rev. Neurosci. 2007, 8, 610–622. [Google Scholar] [CrossRef] [PubMed]
- Lyle, L.T.; Lockman, P.R.; Adkins, C.E.; Mohammad, A.S.; Sechrest, E.; Hua, E.; Palmieri, D.; Liewehr, D.J.; Steinberg, S.M.; Kloc, W.; et al. Alterations in Pericyte Subpopulations Are Associated with Elevated Blood-Tumor Barrier Permeability in Experimental Brain Metastasis of Breast Cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2016, 22, 5287–5299. [Google Scholar] [CrossRef] [PubMed]
- Russell, S.M.; Elliott, R.; Forshaw, D.; Golfinos, J.G.; Nelson, P.K.; Kelly, P.J. Glioma vascularity correlates with reduced patient survival and increased malignancy. Surg. Neurol. 2009, 72, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Leon, S.P.; Folkerth, R.D.; Black, P.M. Microvessel density is a prognostic indicator for patients with astroglial brain tumors. Cancer 1996, 77, 362–372. [Google Scholar] [CrossRef]
- Wesseling, P.; van der Laak, J.A.; Link, M.; Teepen, H.L.; Ruiter, D.J. Quantitative analysis of microvascular changes in diffuse astrocytic neoplasms with increasing grade of malignancy. Hum. Pathol. 1998, 29, 352–358. [Google Scholar] [CrossRef]
- Chinot, O.L.; Wick, W.; Mason, W.; Henriksson, R.; Saran, F.; Nishikawa, R.; Carpentier, A.F.; Hoang-Xuan, K.; Kavan, P.; Cernea, D.; et al. Bevacizumab plus radiotherapy-temozolomide for newly diagnosed glioblastoma. N. Engl. J. Med. 2014, 370, 709–722. [Google Scholar] [CrossRef] [PubMed]
- Lamborn, K.R.; Yung, W.K.; Chang, S.M.; Wen, P.Y.; Cloughesy, T.F.; DeAngelis, L.M.; Robins, H.I.; Lieberman, F.S.; Fine, H.A.; Fink, K.L.; et al. Progression-free survival: An important end point in evaluating therapy for recurrent high-grade gliomas. Neuro-Oncology 2008, 10, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, D.R.; Cascino, T.L.; Schold, S.C., Jr.; Cairncross, J.G. Response criteria for phase II studies of supratentorial malignant glioma. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 1990, 8, 1277–1280. [Google Scholar] [CrossRef] [PubMed]
- Chinot, O.L.; Macdonald, D.R.; Abrey, L.E.; Zahlmann, G.; Kerloeguen, Y.; Cloughesy, T.F. Response assessment criteria for glioblastoma: Practical adaptation and implementation in clinical trials of antiangiogenic therapy. Curr. Neurol. Neurosci. Rep. 2013, 13, 347. [Google Scholar] [CrossRef] [PubMed]
- Chiou, V.L.; Burotto, M. Pseudoprogression and Immune-Related Response in Solid Tumors. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2015, 33, 3541–3543. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.K. Antiangiogenic therapy for cancer: Current and emerging concepts. Oncology 2005, 19, 7–16. [Google Scholar] [PubMed]
- Hygino da Cruz, L.C., Jr.; Rodriguez, I.; Domingues, R.C.; Gasparetto, E.L.; Sorensen, A.G. Pseudoprogression and pseudoresponse: Imaging challenges in the assessment of posttreatment glioma. AJNR Am. J. Neuroradiol. 2011, 32, 1978–1985. [Google Scholar] [CrossRef] [PubMed]
- Herbel, C.; Patsoukis, N.; Bardhan, K.; Seth, P.; Weaver, J.D.; Boussiotis, V.A. Clinical significance of T cell metabolic reprogramming in cancer. Clin. Transl. Med. 2016, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Bollineni, V.R.; Kramer, G.M.; Jansma, E.P.; Liu, Y.; Oyen, W.J. A systematic review on [(18)F]FLT-PET uptake as a measure of treatment response in cancer patients. Eur. J. Cancer 2016, 55, 81–97. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Delaloye, S.; Silverman, D.H.; Geist, C.; Czernin, J.; Sayre, J.; Satyamurthy, N.; Pope, W.; Lai, A.; Phelps, M.E.; et al. Predicting treatment response of malignant gliomas to bevacizumab and irinotecan by imaging proliferation with [18F] fluorothymidine positron emission tomography: A pilot study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2007, 25, 4714–4721. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenberg, J.; Czernin, J.; Cloughesy, T.F.; Ellingson, B.M.; Pope, W.B.; Geist, C.; Dahlbom, M.; Silverman, D.H.; Satyamurthy, N.; Phelps, M.E.; et al. 3′-deoxy-3′-18F-fluorothymidine PET and MRI for early survival predictions in patients with recurrent malignant glioma treated with bevacizumab. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2012, 53, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Wardak, M.; Schiepers, C.; Cloughesy, T.F.; Dahlbom, M.; Phelps, M.E.; Huang, S.C. (1)(8)F-FLT and (1)(8)F-FDOPA PET kinetics in recurrent brain tumors. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Nowosielski, M.; DiFranco, M.D.; Putzer, D.; Seiz, M.; Recheis, W.; Jacobs, A.H.; Stockhammer, G.; Hutterer, M. An intra-individual comparison of MRI, [18F]-FET and [18F]-FLT PET in patients with high-grade gliomas. PLoS ONE 2014, 9, e95830. [Google Scholar] [CrossRef] [PubMed]
- Rapp, M.; Floeth, F.W.; Felsberg, J.; Steiger, H.J.; Sabel, M.; Langen, K.J.; Galldiks, N. Clinical value of O-(2-[(18)F]-fluoroethyl)-l-tyrosine positron emission tomography in patients with low-grade glioma. Neurosurg. Focus 2013, 34, E3. [Google Scholar] [CrossRef] [PubMed]
- Unterrainer, M.; Schweisthal, F.; Suchorska, B.; Wenter, V.; Schmid-Tannwald, C.; Fendler, W.P.; Schuller, U.; Bartenstein, P.; Tonn, J.C.; Albert, N.L. Serial 18F-FET PET Imaging of Primarily 18F-FET-Negative Glioma: Does It Make Sense? J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2016, 57, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Okubo, S.; Zhen, H.N.; Kawai, N.; Nishiyama, Y.; Haba, R.; Tamiya, T. Correlation of l-methyl-11C-methionine (MET) uptake with L-type amino acid transporter 1 in human gliomas. J. Neuro-Oncol. 2010, 99, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Youland, R.S.; Kitange, G.J.; Peterson, T.E.; Pafundi, D.H.; Ramiscal, J.A.; Pokorny, J.L.; Giannini, C.; Laack, N.N.; Parney, I.F.; Lowe, V.J.; et al. The role of LAT1 in (18)F-DOPA uptake in malignant gliomas. J. Neuro-Oncol. 2013, 111, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Habermeier, A.; Graf, J.; Sandhofer, B.F.; Boissel, J.P.; Roesch, F.; Closs, E.I. System L amino acid transporter LAT1 accumulates O-(2-fluoroethyl)-l-tyrosine (FET). Amino Acids 2015, 47, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Dadone-Montaudie, B.; Ambrosetti, D.; Dufour, M.; Darcourt, J.; Almairac, F.; Coyne, J.; Virolle, T.; Humbert, O.; Burel-Vandenbos, F. [18F] FDOPA standardized uptake values of brain tumors are not exclusively dependent on LAT1 expression. PLoS ONE 2017, 12, e0184625. [Google Scholar] [CrossRef] [PubMed]
- Stegmayr, C.; Bandelow, U.; Oliveira, D.; Lohmann, P.; Willuweit, A.; Filss, C.; Galldiks, N.; Lubke, J.H.; Shah, N.J.; Ermert, J.; et al. Influence of blood-brain barrier permeability on O-(2-(18)F-fluoroethyl)-l-tyrosine uptake in rat gliomas. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Stegmayr, C.; Oliveira, D.; Niemietz, N.; Willuweit, A.; Lohmann, P.; Galldiks, N.; Shah, N.J.; Ermert, J.; Langen, K.J. Influence of Bevacizumab on Blood-Brain Barrier Permeability and O-(2-(18)F-Fluoroethyl)-l-Tyrosine Uptake in Rat Gliomas. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2017, 58, 700–705. [Google Scholar]
- Carideo, L.; Minniti, G.; Mamede, M.; Scaringi, C.; Russo, I.; Scopinaro, F.; Cicone, F. (18)F-DOPA uptake parameters in glioma: Effects of patients’ characteristics and prior treatment history. Br. J. Radiol. 2018, 91, 20170847. [Google Scholar] [CrossRef] [PubMed]
- O’Tuama, L.A.; Phillips, P.C.; Strauss, L.C.; Carson, B.C.; Uno, Y.; Smith, Q.R.; Dannals, R.F.; Wilson, A.A.; Ravert, H.T.; Loats, S.; et al. Two-phase [11C]l-methionine PET in childhood brain tumors. Pediatric Neurol. 1990, 6, 163–170. [Google Scholar] [CrossRef]
- Kits, A.; Martin, H.; Sanchez-Crespo, A.; Delgado, A.F. Diagnostic accuracy of (11)C-methionine PET in detecting neuropathologically confirmed recurrent brain tumor after radiation therapy. Ann. Nucl. Med. 2018, 32, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Gao, L.; Shao, A.; Zheng, J.; Zhang, J. The performance of 11C-Methionine PET in the differential diagnosis of glioma recurrence. Oncotarget 2017, 8, 91030–91039. [Google Scholar] [CrossRef] [PubMed]
- Yomo, S.; Oguchi, K. Prospective study of (11)C-methionine PET for distinguishing between recurrent brain metastases and radiation necrosis: Limitations of diagnostic accuracy and long-term results of salvage treatment. BMC Cancer 2017, 17, 713. [Google Scholar] [CrossRef] [PubMed]
- Tomura, N.; Kokubun, M.; Saginoya, T.; Mizuno, Y.; Kikuchi, Y. Differentiation between Treatment-Induced Necrosis and Recurrent Tumors in Patients with Metastatic Brain Tumors: Comparison among (11)C-Methionine-PET, FDG-PET, MR Permeability Imaging, and MRI-ADC-Preliminary Results. AJNR Am. J. Neuroradiol. 2017, 38, 1520–1527. [Google Scholar] [CrossRef] [PubMed]
- Tomura, N.; Saginoya, T.; Goto, H. 11C-Methionine Positron Emission Tomography/Computed Tomography Versus 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography in Evaluation of Residual or Recurrent World Health Organization Grades II and III Meningioma After Treatment. J. Comput. Assist. Tomogr. 2018, 42, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Kratochwil, C.; Combs, S.E.; Leotta, K.; Afshar-Oromieh, A.; Rieken, S.; Debus, J.; Haberkorn, U.; Giesel, F.L. Intra-individual comparison of (1)(8)F-FET and (1)(8)F-DOPA in PET imaging of recurrent brain tumors. Neuro-Oncology 2014, 16, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Calcagni, M.L.; Galli, G.; Giordano, A.; Taralli, S.; Anile, C.; Niesen, A.; Baum, R.P. Dynamic O-(2-[18F]fluoroethyl)-l-tyrosine (F-18 FET) PET for glioma grading: Assessment of individual probability of malignancy. Clin. Nucl. Med. 2011, 36, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Popperl, G.; Kreth, F.W.; Mehrkens, J.H.; Herms, J.; Seelos, K.; Koch, W.; Gildehaus, F.J.; Kretzschmar, H.A.; Tonn, J.C.; Tatsch, K. FET PET for the evaluation of untreated gliomas: Correlation of FET uptake and uptake kinetics with tumour grading. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 1933–1942. [Google Scholar] [CrossRef] [PubMed]
- Weckesser, M.; Langen, K.J.; Rickert, C.H.; Kloska, S.; Straeter, R.; Hamacher, K.; Kurlemann, G.; Wassmann, H.; Coenen, H.H.; Schober, O. O-(2-[18F]fluorethyl)-l-tyrosine PET in the clinical evaluation of primary brain tumours. Eur. J. Nucl. Med. Mol. Imaging 2005, 32, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Moulin-Romsee, G.; D’Hondt, E.; de Groot, T.; Goffin, J.; Sciot, R.; Mortelmans, L.; Menten, J.; Bormans, G.; Van Laere, K. Non-invasive grading of brain tumours using dynamic amino acid PET imaging: Does it work for 11C-methionine? Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 2082–2087. [Google Scholar] [CrossRef] [PubMed]
- Unterrainer, M.; Winkelmann, I.; Suchorska, B.; Giese, A.; Wenter, V.; Kreth, F.W.; Herms, J.; Bartenstein, P.; Tonn, J.C.; Albert, N.L. Correction to: Biological tumour volumes of gliomas in early and standard 20-40 min (18)F-FET PET images differ according to IDH mutation status. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1078. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Rapp, M.; Stoffels, G.; Dunkl, V.; Sabel, M.; Langen, K.J. Earlier diagnosis of progressive disease during bevacizumab treatment using O-(2-18F-fluorethyl)-l-tyrosine positron emission tomography in comparison with magnetic resonance imaging. Mol. Imaging 2013, 12, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Tsuyuguchi, N.; Terakawa, Y.; Uda, T.; Nakajo, K.; Kanemura, Y. Diagnosis of Brain Tumors Using Amino Acid Transport PET Imaging with (18)F-fluciclovine: A Comparative Study with l-methyl-(11)C-methionine PET Imaging. Asia Ocean. J. Nucl. Med. Boil. 2017, 5, 85–94. [Google Scholar]
- Cicone, F.; Filss, C.P.; Minniti, G.; Rossi-Espagnet, C.; Papa, A.; Scaringi, C.; Galldiks, N.; Bozzao, A.; Shah, N.J.; Scopinaro, F.; et al. Volumetric assessment of recurrent or progressive gliomas: Comparison between F-DOPA PET and perfusion-weighted MRI. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 905–915. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenberg, J.; Czernin, J.; Cloughesy, T.F.; Ellingson, B.M.; Pope, W.B.; Grogan, T.; Elashoff, D.; Geist, C.; Silverman, D.H.; Phelps, M.E.; et al. Treatment response evaluation using 18F-FDOPA PET in patients with recurrent malignant glioma on bevacizumab therapy. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2014, 20, 3550–3559. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Kracht, L.W.; Burghaus, L.; Thomas, A.; Jacobs, A.H.; Heiss, W.D.; Herholz, K. Use of 11C-methionine PET to monitor the effects of temozolomide chemotherapy in malignant gliomas. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Kracht, L.W.; Burghaus, L.; Ullrich, R.T.; Backes, H.; Brunn, A.; Heiss, W.D.; Jacobs, A.H. Patient-tailored, imaging-guided, long-term temozolomide chemotherapy in patients with glioblastoma. Mol. Imaging 2010, 9, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Dunkl, V.; Ceccon, G.; Tscherpel, C.; Stoffels, G.; Law, I.; Henriksen, O.M.; Muhic, A.; Poulsen, H.S.; Steger, J.; et al. Early treatment response evaluation using FET PET compared to MRI in glioblastoma patients at first progression treated with bevacizumab plus lomustine. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 2377–2386. [Google Scholar] [CrossRef] [PubMed]
- Ono, T.; Sasajima, T.; Doi, Y.; Oka, S.; Ono, M.; Kanagawa, M.; Baden, A.; Mizoi, K.; Shimizu, H. Amino acid PET tracers are reliable markers of treatment responses to single-agent or combination therapies including temozolomide, interferon-beta, and/or bevacizumab for glioblastoma. Nucl. Med. Boil. 2015, 42, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Heinzel, A.; Muller, D.; Langen, K.J.; Blaum, M.; Verburg, F.A.; Mottaghy, F.M.; Galldiks, N. The use of O-(2-18F-fluoroethyl)-l-tyrosine PET for treatment management of bevacizumab and irinotecan in patients with recurrent high-grade glioma: a cost-effectiveness analysis. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2013, 54, 1217–1222. [Google Scholar] [CrossRef] [PubMed]
- Jansen, N.L.; Graute, V.; Armbruster, L.; Suchorska, B.; Lutz, J.; Eigenbrod, S.; Cumming, P.; Bartenstein, P.; Tonn, J.C.; Kreth, F.W.; et al. MRI-suspected low-grade glioma: Is there a need to perform dynamic FET PET? Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Lopez, W.O.; Cordeiro, J.G.; Albicker, U.; Doostkam, S.; Nikkhah, G.; Kirch, R.D.; Trippel, M.; Reithmeier, T. Correlation of (18)F-fluoroethyl tyrosine positron-emission tomography uptake values and histomorphological findings by stereotactic serial biopsy in newly diagnosed brain tumors using a refined software tool. OncoTargets Ther. 2015, 8, 3803–3815. [Google Scholar] [CrossRef] [PubMed]
- Cicuendez, M.; Lorenzo-Bosquet, C.; Cuberas-Borros, G.; Martinez-Ricarte, F.; Cordero, E.; Martinez-Saez, E.; Castell-Conesa, J.; Sahuquillo, J. Role of [(11)C] methionine positron emission tomography in the diagnosis and prediction of survival in brain tumours. Clin. Neurol. Neurosurg. 2015, 139, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Nishii, R.; Higashi, T.; Kagawa, S.; Arimoto, M.; Kishibe, Y.; Takahashi, M.; Yamada, S.; Saiki, M.; Arakawa, Y.; Yamauchi, H.; et al. Differential Diagnosis between Low-Grade and High-Grade Astrocytoma Using System A Amino Acid Transport PET Imaging with C-11-MeAIB: A Comparison Study with C-11-Methionine PET Imaging. Contrast Media Mol. Imaging 2018, 2018, 1292746. [Google Scholar] [CrossRef] [PubMed]
- Juhasz, C.; Muzik, O.; Chugani, D.C.; Chugani, H.T.; Sood, S.; Chakraborty, P.K.; Barger, G.R.; Mittal, S. Differential kinetics of alpha-[(1)(1)C]methyl-l-tryptophan on PET in low-grade brain tumors. J. Neuro-Oncol. 2011, 102, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Naslund, O.; Smits, A.; Forander, P.; Laesser, M.; Bartek, J., Jr.; Gempt, J.; Liljegren, A.; Daxberg, E.L.; Jakola, A.S. Amino acid tracers in PET imaging of diffuse low-grade gliomas: A systematic review of preoperative applications. Acta Neurochir. 2018, 160, 1451–1460. [Google Scholar] [CrossRef] [PubMed]
- Pauleit, D.; Stoffels, G.; Bachofner, A.; Floeth, F.W.; Sabel, M.; Herzog, H.; Tellmann, L.; Jansen, P.; Reifenberger, G.; Hamacher, K.; et al. Comparison of (18)F-FET and (18)F-FDG PET in brain tumors. Nucl. Med. Boil. 2009, 36, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, P.; Herzog, H.; Rota Kops, E.; Stoffels, G.; Judov, N.; Filss, C.; Galldiks, N.; Tellmann, L.; Weiss, C.; Sabel, M.; et al. dual-time-point O-(2-[(18)F]fluoroethyl)-l-tyrosine PET for grading of cerebral gliomas. Eur. Radiol. 2015, 25, 3017–3024. [Google Scholar] [CrossRef] [PubMed]
- Albert, N.L.; Winkelmann, I.; Suchorska, B.; Wenter, V.; Schmid-Tannwald, C.; Mille, E.; Todica, A.; Brendel, M.; Tonn, J.C.; Bartenstein, P.; et al. Early static (18)F-FET-PET scans have a higher accuracy for glioma grading than the standard 20–40 min scans. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Jansen, N.L.; Suchorska, B.; Wenter, V.; Eigenbrod, S.; Schmid-Tannwald, C.; Zwergal, A.; Niyazi, M.; Drexler, M.; Bartenstein, P.; Schnell, O.; et al. Dynamic 18F-FET PET in newly diagnosed astrocytic low-grade glioma identifies high-risk patients. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2014, 55, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Piroth, M.D.; Holy, R.; Pinkawa, M.; Stoffels, G.; Kaiser, H.J.; Galldiks, N.; Herzog, H.; Coenen, H.H.; Eble, M.J.; Langen, K.J. Prognostic impact of postoperative, pre-irradiation (18)F-fluoroethyl-l-tyrosine uptake in glioblastoma patients treated with radiochemotherapy. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 2011, 99, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Langen, K.J.; Holy, R.; Pinkawa, M.; Stoffels, G.; Nolte, K.W.; Kaiser, H.J.; Filss, C.P.; Fink, G.R.; Coenen, H.H.; et al. Assessment of treatment response in patients with glioblastoma using O-(2-18F-fluoroethyl)-l-tyrosine PET in comparison to MRI. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2012, 53, 1048–1057. [Google Scholar] [CrossRef] [PubMed]
- Beppu, T.; Terasaki, K.; Sasaki, T.; Sato, Y.; Tomabechi, M.; Kato, K.; Sasaki, M.; Ogasawara, K. MRI and 11C-methyl-l-methionine PET Differentiate Bevacizumab True Responders After Initiating Therapy for Recurrent Glioblastoma. Clin. Nucl. Med. 2016, 41, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Yoo, M.Y.; Paeng, J.C.; Cheon, G.J.; Lee, D.S.; Chung, J.K.; Kim, E.E.; Kang, K.W. Prognostic Value of Metabolic Tumor Volume on (11)C-Methionine PET in Predicting Progression-Free Survival in High-Grade Glioma. Nucl. Med. Mol. Imaging 2015, 49, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.Y.; Min, J.J.; Bom, H.S.; Jung, S.; Kim, I.Y.; Lim, S.H.; Kim, D.Y.; Kwon, S.Y. Prognostic value of post-treatment metabolic tumor volume from (11)C-methionine PET/CT in recurrent malignant glioma. Neurosurg. Rev. 2017, 40, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Floeth, F.W.; Sabel, M.; Stoffels, G.; Pauleit, D.; Hamacher, K.; Steiger, H.J.; Langen, K.J. Prognostic value of 18F-fluoroethyl-l-tyrosine PET and MRI in small nonspecific incidental brain lesions. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2008, 49, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Stoffels, G.; Ruge, M.I.; Rapp, M.; Sabel, M.; Reifenberger, G.; Erdem, Z.; Shah, N.J.; Fink, G.R.; Coenen, H.H.; et al. Role of O-(2-18F-fluoroethyl)-l-tyrosine PET as a diagnostic tool for detection of malignant progression in patients with low-grade glioma. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2013, 54, 2046–2054. [Google Scholar] [CrossRef] [PubMed]
- Morana, G.; Piccardo, A.; Garre, M.L.; Nozza, P.; Consales, A.; Rossi, A. Multimodal magnetic resonance imaging and 18F-l-dihydroxyphenylalanine positron emission tomography in early characterization of pseudoresponse and nonenhancing tumor progression in a pediatric patient with malignant transformation of ganglioglioma treated with bevacizumab. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2013, 31, e1–e5. [Google Scholar]
- Pirotte, B.; Goldman, S.; David, P.; Wikler, D.; Damhaut, P.; Vandesteene, A.; Salmon, L.; Brotchi, J.; Levivier, M. Stereotactic brain biopsy guided by positron emission tomography (PET) with [F-18]fluorodeoxyglucose and [C-11]methionine. Acta Neurochir. Suppl. 1997, 68, 133–138. [Google Scholar] [PubMed]
- Pirotte, B.; Goldman, S.; Massager, N.; David, P.; Wikler, D.; Vandesteene, A.; Salmon, I.; Brotchi, J.; Levivier, M. Comparison of 18F-FDG and 11C-methionine for PET-guided stereotactic brain biopsy of gliomas. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2004, 45, 1293–1298. [Google Scholar]
- Pafundi, D.H.; Laack, N.N.; Youland, R.S.; Parney, I.F.; Lowe, V.J.; Giannini, C.; Kemp, B.J.; Grams, M.P.; Morris, J.M.; Hoover, J.M.; et al. Biopsy validation of 18F-DOPA PET and biodistribution in gliomas for neurosurgical planning and radiotherapy target delineation: Results of a prospective pilot study. Neuro-Oncology 2013, 15, 1058–1067. [Google Scholar] [CrossRef] [PubMed]
- Pirotte, B.J.; Lubansu, A.; Massager, N.; Wikler, D.; Goldman, S.; Levivier, M. Results of positron emission tomography guidance and reassessment of the utility of and indications for stereotactic biopsy in children with infiltrative brainstem tumors. J. Neurosurg. 2007, 107 (Suppl. S5), 392–399. [Google Scholar] [CrossRef]
- Galldiks, N.; Schroeter, M.; Fink, G.R.; Kracht, L.W. Interesting image. PET imaging of a butterfly glioblastoma. Clin. Nucl. Med. 2010, 35, 49–50. [Google Scholar] [CrossRef] [PubMed]
- Calabria, F.; Schillaci, O. Recurrent glioma and crossed cerebellar diaschisis in a patient examined with 18F-DOPA and 18F-FDG PET/CT. Clin. Nucl. Med. 2012, 37, 878–879. [Google Scholar] [CrossRef] [PubMed]
- Kebir, S.; Rauschenbach, L.; Gielen, G.H.; Schafer, N.; Tzaridis, T.; Scheffler, B.; Giordano, F.A.; Lazaridis, L.; Herrlinger, U.; Glas, M. Recurrent pseudoprogression in isocitrate dehydrogenase 1 mutant glioblastoma. J. Clin. Neurosci. Off. J. Neurosurg. Soc. Australas 2018, 53, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Juhasz, C.; Bhambhani, K.; Wu, D.; Chugani, D.C.; Chugani, H.T. Assessment of progression and treatment response of optic pathway glioma with positron emission tomography using alpha-[(11)C]methyl-l-tryptophan. Mol. Imaging Boil. MIB Off. Publ. Acad. Mol. Imaging 2007, 9, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Viader, F.; Derlon, J.M.; Petit-Taboue, M.C.; Shishido, F.; Hubert, P.; Houtteville, J.P.; Courtheoux, P.; Chapon, F. Recurrent oligodendroglioma diagnosed with 11C-l-methionine and pet: A case report. Eur. Neurol. 1993, 33, 248–251. [Google Scholar] [CrossRef] [PubMed]
- O’Halloran, P.J.; Viel, T.; Murray, D.W.; Wachsmuth, L.; Schwegmann, K.; Wagner, S.; Kopka, K.; Jarzabek, M.A.; Dicker, P.; Hermann, S.; et al. Mechanistic interrogation of combination bevacizumab/dual PI3K/mTOR inhibitor response in glioblastoma implementing novel MR and PET imaging biomarkers. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1673–1683. [Google Scholar] [CrossRef] [PubMed]
- Deuschl, C.; Moenninghoff, C.; Goericke, S.; Kirchner, J.; Koppen, S.; Binse, I.; Poeppel, T.D.; Quick, H.H.; Forsting, M.; Umutlu, L.; et al. Response assessment of bevacizumab therapy in GBM with integrated 11C-MET-PET/MRI: A feasibility study. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1285–1295. [Google Scholar] [CrossRef] [PubMed]
- Deuschl, C.; Goericke, S.; Grueneisen, J.; Sawicki, L.M.; GOEBEL, J.; El Hindy, N.; Wrede, K.; Binse, I.; Poeppel, T.; Quick, H.; et al. Simultaneous 11C-Methionine Positron Emission Tomography/Magnetic Resonance Imaging of Suspected Primary Brain Tumors. PLoS ONE 2016, 11, e0167596. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.R.; Cozar, M.; Baquero, M.; Fernandez Barrionuevo, J.M.; Jaramillo, A.; Rubio, J.; Maida, G.; Soler, M.; Riera, E. The value of (11)C-methionine PET in the early differentiation between tumour recurrence and radionecrosis in patients treated for a high-grade glioma and indeterminate MRI. Rev. Esp. Med. Nucl. Imagen Mol. 2017, 36, 85–90. [Google Scholar] [PubMed]
- Gauvain, K.; Ponisio, M.R.; Barone, A.; Grimaldi, M.; Parent, E.; Leeds, H.; Goyal, M.; Rubin, J.; McConathy, J. (18)F-FDOPA PET/MRI for monitoring early response to bevacizumab in children with recurrent brain tumors. Neuro-Oncol. Pract. 2018, 5, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Filss, C.P.; Cicone, F.; Shah, N.J.; Galldiks, N.; Langen, K.J. Amino acid PET and MR perfusion imaging in brain tumours. Clin. Transl. Imaging 2017, 5, 209–223. [Google Scholar] [CrossRef] [PubMed]
- Muoio, B.; Giovanella, L.; Treglia, G. Recent Developments of 18F-FET PET in Neuro-oncology. Curr. Med. Chem. 2018, 25, 3061–3073. [Google Scholar] [CrossRef] [PubMed]
- Albert, N.L.; Weller, M.; Suchorska, B.; Galldiks, N.; Soffietti, R.; Kim, M.M.; la Fougere, C.; Pope, W.; Law, I.; Arbizu, J.; et al. Response Assessment in Neuro-Oncology working group and European Association for Neuro-Oncology recommendations for the clinical use of PET imaging in gliomas. Neuro-Oncology 2016, 18, 1199–1208. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Najjar, A.M.; Johnson, J.M.; Schellingerhout, D. The Emerging Role of Amino Acid PET in Neuro-Oncology. Bioengineering 2018, 5, 104. https://doi.org/10.3390/bioengineering5040104
Najjar AM, Johnson JM, Schellingerhout D. The Emerging Role of Amino Acid PET in Neuro-Oncology. Bioengineering. 2018; 5(4):104. https://doi.org/10.3390/bioengineering5040104
Chicago/Turabian StyleNajjar, Amer M., Jason M. Johnson, and Dawid Schellingerhout. 2018. "The Emerging Role of Amino Acid PET in Neuro-Oncology" Bioengineering 5, no. 4: 104. https://doi.org/10.3390/bioengineering5040104
APA StyleNajjar, A. M., Johnson, J. M., & Schellingerhout, D. (2018). The Emerging Role of Amino Acid PET in Neuro-Oncology. Bioengineering, 5(4), 104. https://doi.org/10.3390/bioengineering5040104




