Merging Heterocyclic Chemistry and Biocatalysis in One-Pot Processes through Compartmentalization of the Reaction Steps
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the PDMS Thimbles for Compartmentalization of the Reaction Steps
2.2. Construction and Expression of Whole-Cell Catalyst
2.3. GC Analytics for the Determination of the Conversion
- Temperature program: 40 °C–200 °C, 10 °C·min−1.
- Retention time for 2,2,3-trimethyl-1-thia-4-azaspiro[4.4]non-3-ene (3a): 8.52 min.
- Retention time for 2,2,3-Trimethyl-1-thia-4-azaspiro[4.4]nonane (4a): 8.96 min.
2.4. Derivatization of 3-Thiazolidine (4a) with Phenylisocyanate
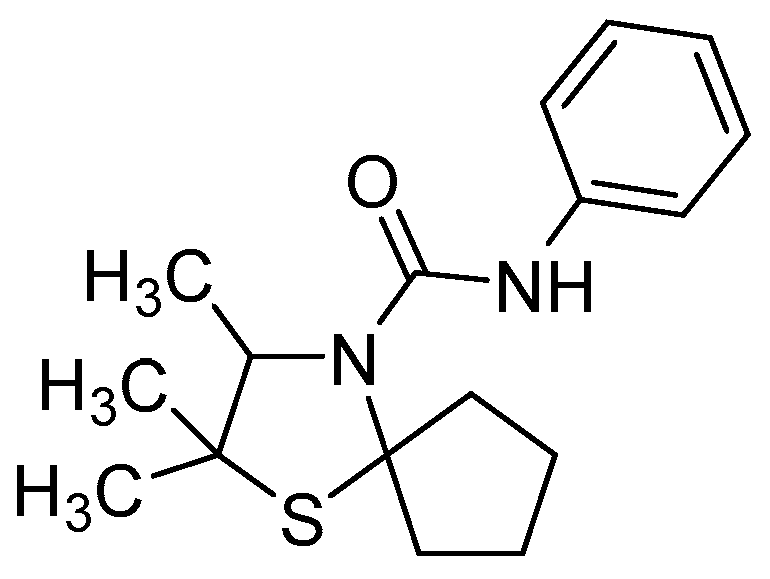
2.5. HPLC Analytics for the Determination of the Enantiomeric Excess
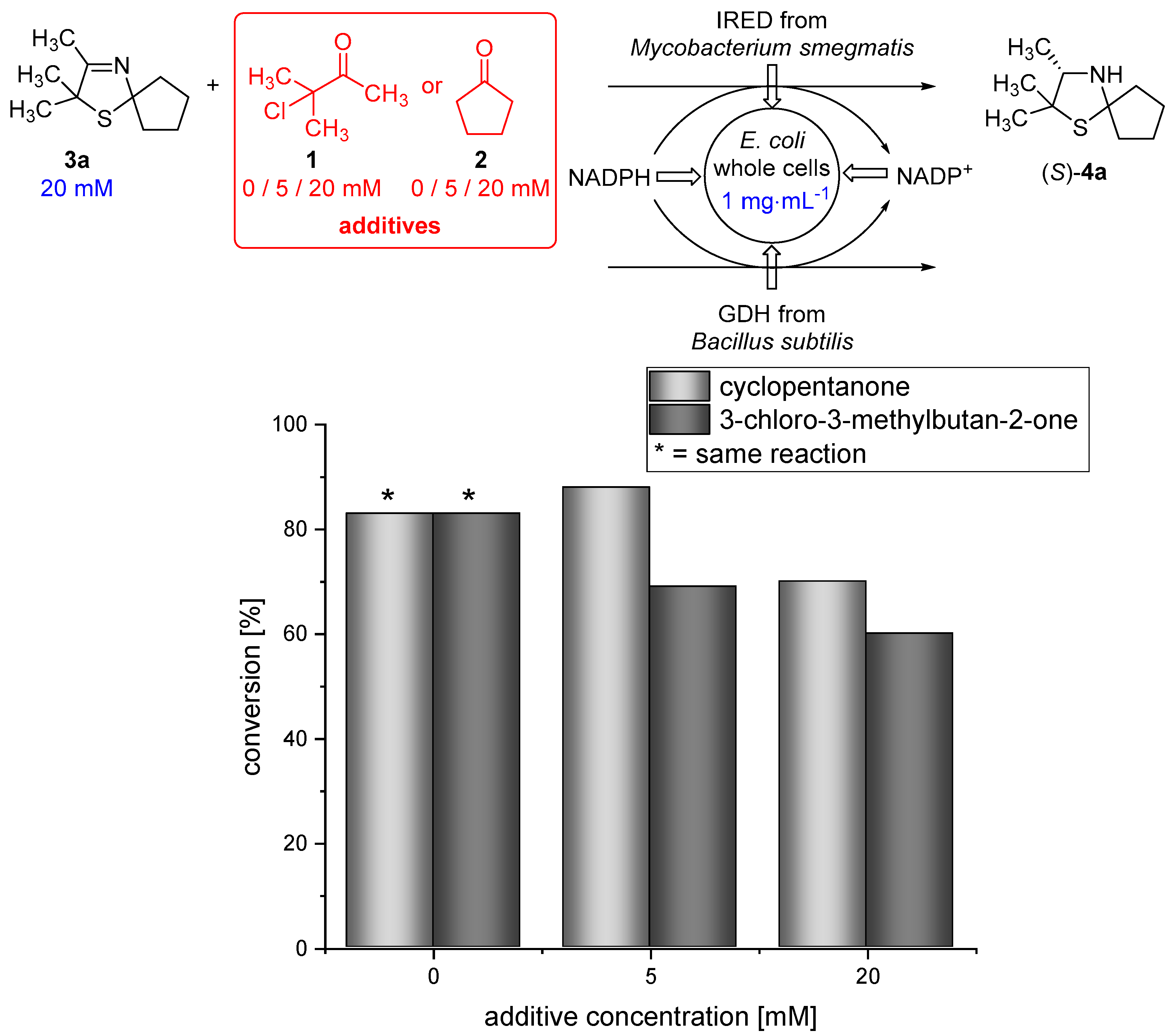
2.6. Biotransformation of 3-Thiazoline (3a) in the Presence of Different Amounts of Dimethyl Sulfoxide (DMSO)/3-Chloro-3-methylbutan-2-one (1)/Cyclopentanone (2)
2.7. Studies on Diffusion of 3a through the PDMS Membrane
2.8. Synthesis of 3-Thiazoline (3a) via Asinger-Reaction in Different Reactor Types
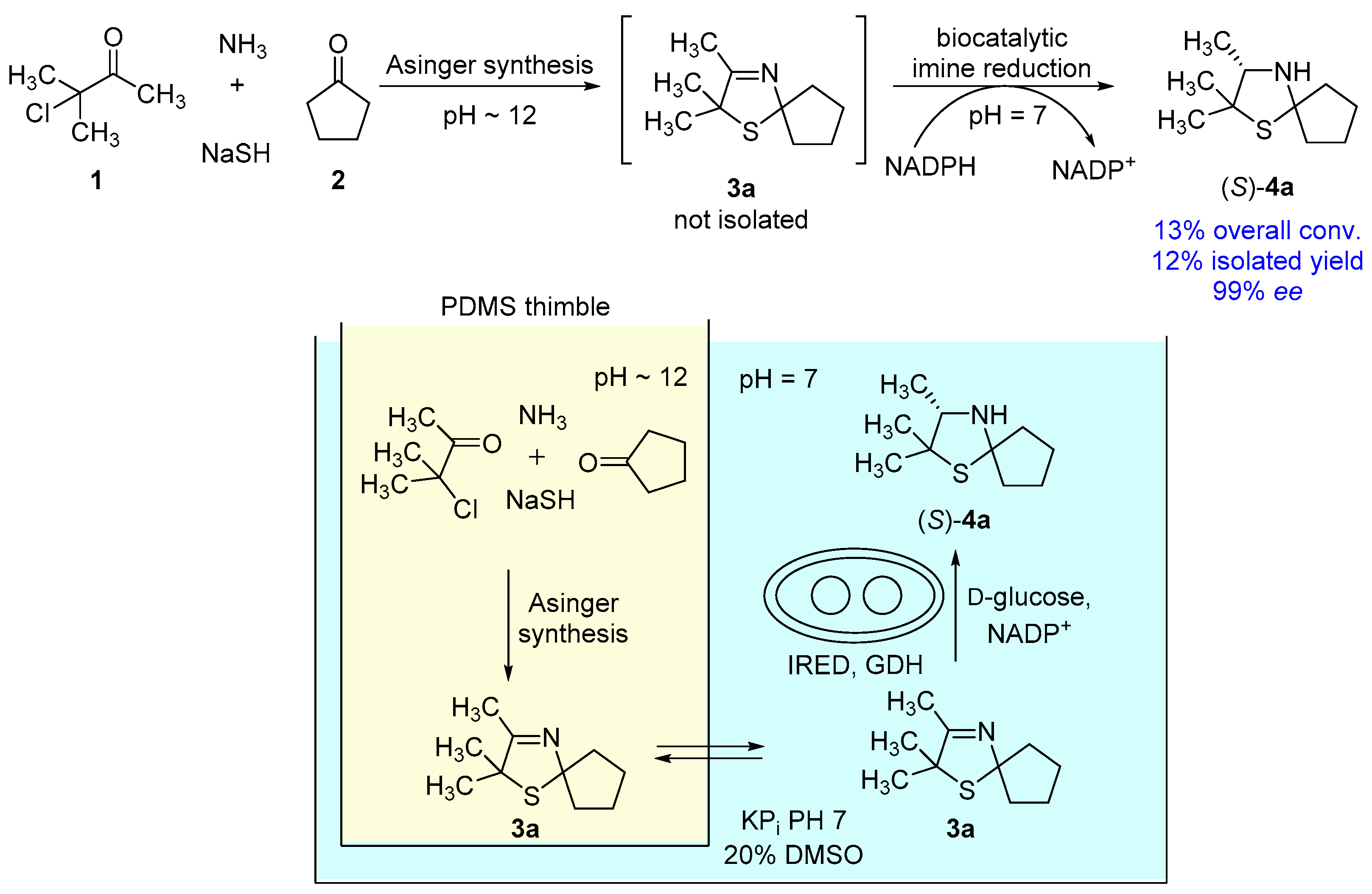
2.9. One-Pot Process Combining the Asinger-Synthesis of 3-Thiazoline (3a) with an Enzymatic Reduction via Compartmentalization of the Reaction Steps
3. Results and Discussion
3.1. Compartmentation Approach
3.2. Studies on Asinger-Type Multi-Component Reaction
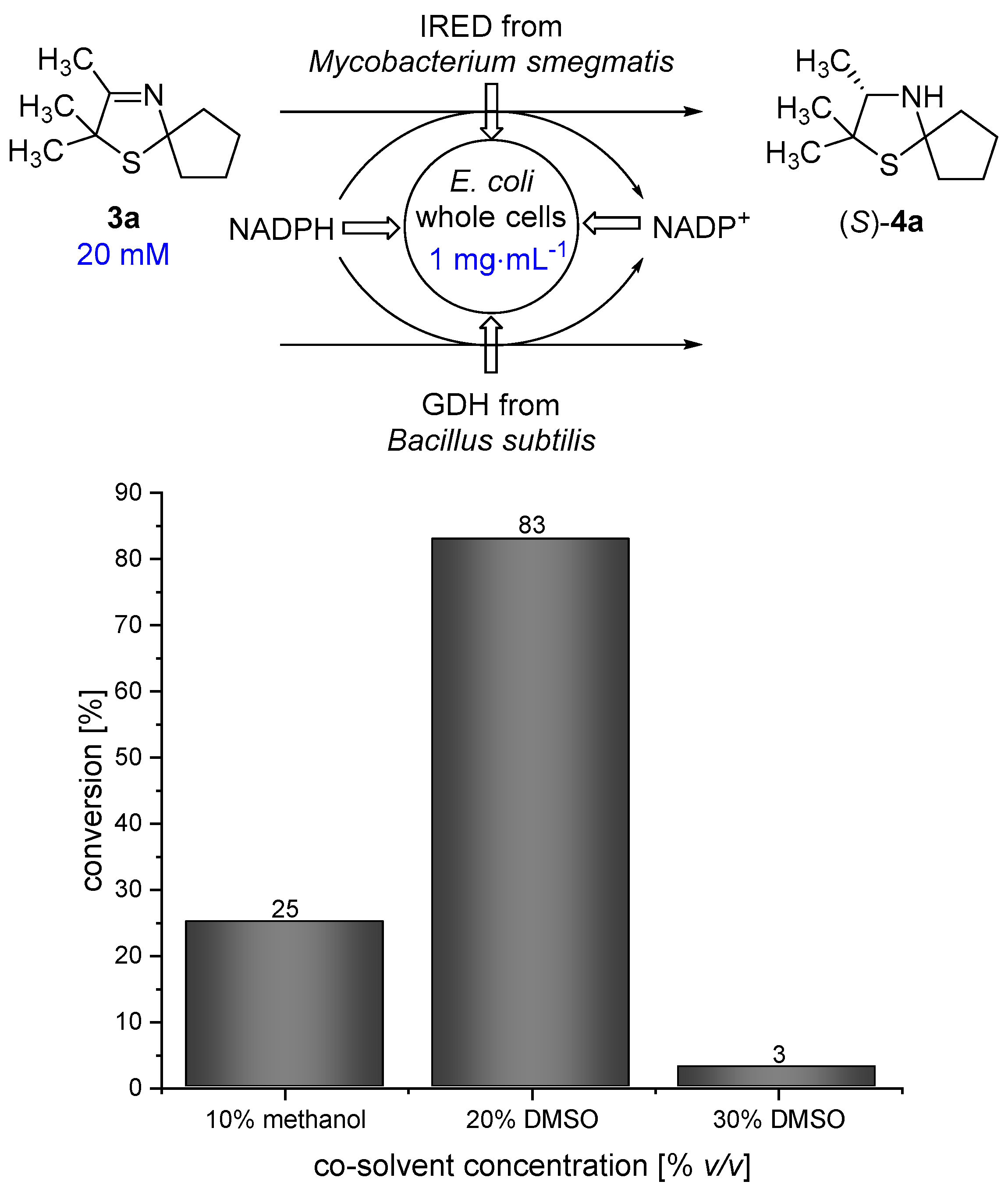
3.3. Diffusion Efficiency and Biocatalytic Reduction Using Imine Reductases
3.4. One-Pot Process Combining the Asinger-Synthesis of 3-Thiazoline (3a) with an Enzymatic Reduction via Compartmentalization of the Reaction Steps
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rudroff, F.; Mihovilovic, M.D.; Gröger, H.; Snajdrova, R.; Iding, H.; Bornscheuer, U.T. Opportunities and challenges for combining chemo- and biocatalysis. Nat. Catal. 2018, 1, 12–22. [Google Scholar] [CrossRef]
- Gröger, H. Emerging Fields in One-Pot Multi-step Synthesis with Combined Chemo- and Bio-catalysts: Sequential- and Domino-Type Process Concepts as well as Compartmentation Strategies. In Modern Biocatalysis: Advances Towards Synthetic Biological Systems; Williams, G., Hall, M., Eds.; Royal Society of Chemistry: Cambridge, UK, 2018; pp. 439–472. [Google Scholar]
- Denard, C.A.; Huang, H.; Bartlett, M.J.; Lu, L.; Tan, Y.; Zhao, H.; Hartwig, J.F. Cooperative tandem catalysis by an organometallic complex and a metalloenzyme. Angew. Chem. Int. Ed. 2014, 53, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Heidlindemann, M.; Rulli, G.; Berkessel, A.; Hummel, W.; Gröger, H. Combination of asymmetric organo- and biocatalytic reactions in organic media using immobilized catalysts in different compartments. ACS Catal. 2014, 4, 1099–1103. [Google Scholar] [CrossRef]
- Wang, Z.J.; Clary, K.N.; Bergman, R.G.; Raymond, K.N.; Toste, F.D. A supramolecular approach to combining enzymatic and transition metal catalysis. Nat. Chem. 2013, 5, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Köhler, V.; Wilson, Y.M.; Dürrenberger, M.; Ghislieri, D.; Churakova, E.; Quinto, T.; Knörr, L.; Häussinger, D.; Hollmann, F.; Turner, N.J.; et al. Synthetic cascades are enabled by combining biocatalysts with artificial metalloenzymes. Nat. Chem. 2013, 5, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Köhler, V.; Ward, T.R. An NAD(P)H-Dependent artificial transfer hydrogenase for multienzymatic cascades. J. Am. Chem. Soc. 2016, 138, 5781–5784. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Köhler, V.; Paul, C.E.; Hollmann, F.; Ward, T.R. Efficient in situ regeneration of NADH mimics by an artificial metalloenzyme. ACS Catal. 2016, 6, 3553–3557. [Google Scholar] [CrossRef]
- Brahma, A.; Musio, B.; Ismayilova, U.; Nikbin, N.; Kamptmann, S.; Siegert, P.; Jeromin, G.; Ley, S.; Pohl, M. An orthogonal biocatalytic approach for the safe generation and use of HCN in a multistep continuous preparation of chiral O-acetylcyanohydrins. Synlett 2016, 27, 262–266. [Google Scholar] [CrossRef]
- Miller, A.L.; Bowden, N.B. Site-isolation and recycling of PdCl(2) using PDMS thimbles. J. Org. Chem. 2009, 74, 4834–4840. [Google Scholar] [CrossRef] [PubMed]
- Mwangi, M.T.; Runge, M.B.; Hoak, K.M.; Schulz, M.D.; Bowden, N.B. A materials approach to site-isolation of Grubbs catalysts from incompatible solvents and m-chloroperoxybenzoic acid. Chem. Eur. J. 2008, 14, 6780–6788. [Google Scholar] [CrossRef] [PubMed]
- Runge, M.B.; Mwangi, M.T.; Miller, A.L.; Perring, M.; Bowden, N.B. Cascade reactions using LiAlH4 and grignard reagents in the presence of water. Angew. Chem. Int. Ed. 2008, 47, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Hummel, W.; Gröger, H. Cooperative catalysis of noncompatible catalysts through compartmentalization: Wacker oxidation and enzymatic reduction in a one-pot process in aqueous media. Angew. Chem. Int. Ed. 2015, 54, 4488–4492. [Google Scholar] [CrossRef] [PubMed]
- Uthoff, F.; Sato, H.; Gröger, H. Formal enantioselective hydroamination of non-activated alkenes: Transformation of styrenes into enantiomerically pure 1-phenylethylamines in chemoenzymatic one-pot synthesis. Chem. Cat. Chem. 2017, 9, 555–558. [Google Scholar] [CrossRef]
- Latham, J.; Henry, J.M.; Sharif, H.H.; Menon, B.R.K.; Shepherd, S.A.; Greaney, M.F.; Micklefield, J. Integrated catalysis opens new arylation pathways via regiodivergent enzymatic C-H activation. Nat. Commun. 2016, 7, 11873. [Google Scholar] [CrossRef] [PubMed]
- Uthoff, F.; Gröger, H. Asymmetric synthesis of 1-phenylethylamine from styrene via combined wacker-oxidation and enzymatic reductive amination. J. Org. Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zumbrägel, N.; Merten, C.; Huber, S.M.; Gröger, H. Enantioselective reduction of sulfur-containing cyclic imines through biocatalysis. Nat. Commun. 2018, 9, 1949. [Google Scholar] [CrossRef] [PubMed]
- Wetzl, D.; Berrera, M.; Sandon, N.; Fishlock, D.; Ebeling, M.; Müller, M.; Hanlon, S.; Wirz, B.; Iding, H. Expanding the imine reductase toolbox by exploring the bacterial protein-sequence space. Chem. Biol. Chem. 2015, 16, 1749–1756. [Google Scholar] [CrossRef] [PubMed]
- Zumbrägel, N.; Wetzl, D.; Iding, H.; Gröger, H. Asymmetric biocatalytic reduction of cyclic imines: Design and application of a tailor-made whole-cell catalyst. Heterocycles 2017, 95, 1261–1271. [Google Scholar] [CrossRef]
- Biermann, M.; Bakonyi, D.; Hummel, W.; Gröger, H. Design of recombinant whole-cell catalysts for double reduction of C=C and C=O bonds in enals and application in the synthesis of Guerbet alcohols as industrial bulk chemicals for lubricants. Green Chem. 2017, 19, 405–410. [Google Scholar] [CrossRef]
- Reiners, I.; Gröger, H.; Martens, J.A. New enantioselective synthetic approach to β-aminothio-compoundsvia enantioselective reduction of N,S-heterocyclic imines. J. Prakt. Chem. 1997, 339, 541–546. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zumbrägel, N.; Gröger, H. Merging Heterocyclic Chemistry and Biocatalysis in One-Pot Processes through Compartmentalization of the Reaction Steps. Bioengineering 2018, 5, 60. https://doi.org/10.3390/bioengineering5030060
Zumbrägel N, Gröger H. Merging Heterocyclic Chemistry and Biocatalysis in One-Pot Processes through Compartmentalization of the Reaction Steps. Bioengineering. 2018; 5(3):60. https://doi.org/10.3390/bioengineering5030060
Chicago/Turabian StyleZumbrägel, Nadine, and Harald Gröger. 2018. "Merging Heterocyclic Chemistry and Biocatalysis in One-Pot Processes through Compartmentalization of the Reaction Steps" Bioengineering 5, no. 3: 60. https://doi.org/10.3390/bioengineering5030060
APA StyleZumbrägel, N., & Gröger, H. (2018). Merging Heterocyclic Chemistry and Biocatalysis in One-Pot Processes through Compartmentalization of the Reaction Steps. Bioengineering, 5(3), 60. https://doi.org/10.3390/bioengineering5030060






