Three-Dimensional Culture Model of Skeletal Muscle Tissue with Atrophy Induced by Dexamethasone
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microdevice Design and Fabrication
2.2. Cell Culture
2.3. Construction of Skeletal Muscle Tissues
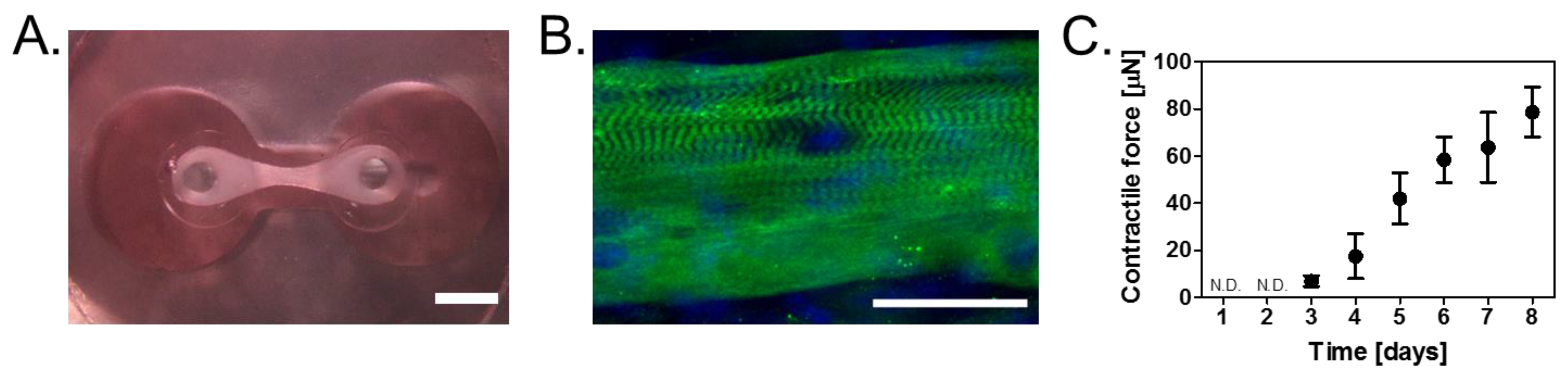
2.4. Contractile Force Measurement of the Muscle Tissues
2.5. Treatment with Dexamethasone and IGF-I
2.6. Real-Time PCR
2.7. Fluorescent Immunostaining
2.8. Statistical Analysis
3. Results and Discussion
3.1. Construction of Skeletal Muscle Tissues on Devices
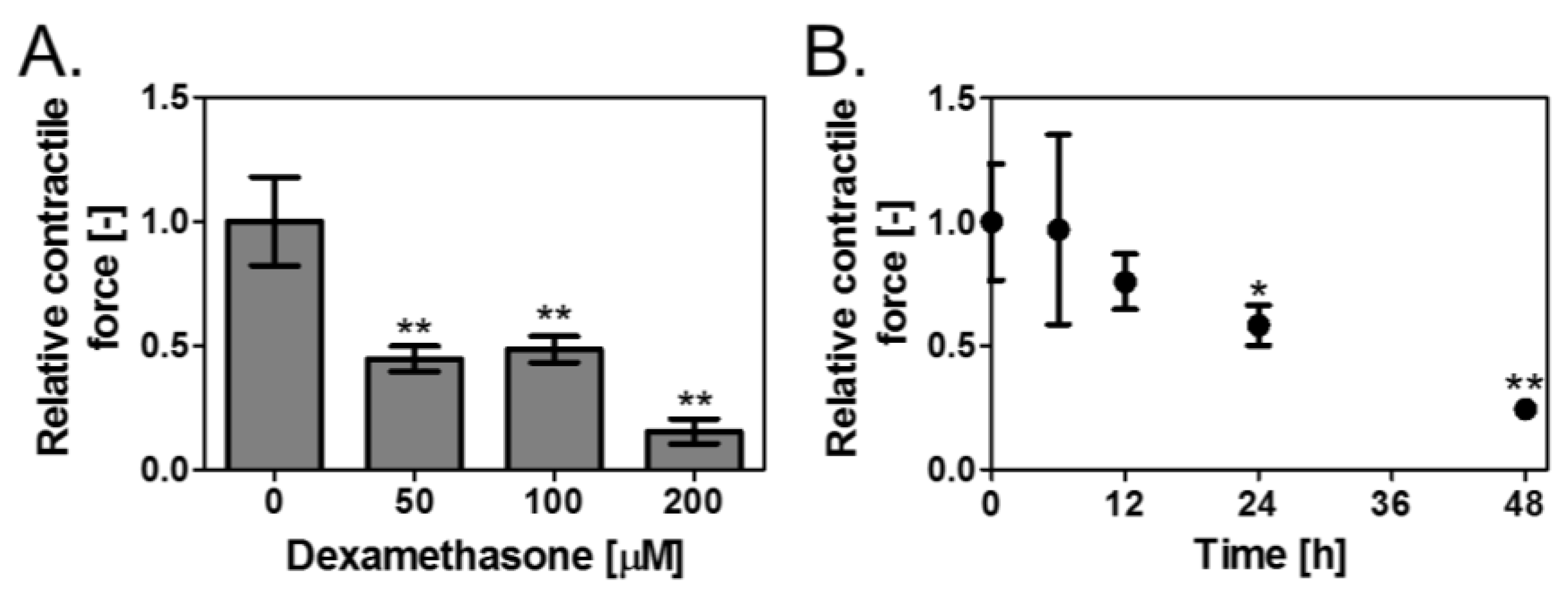
3.2. Effect of Dex on Contractile Force of the Skeletal Muscle Tissues
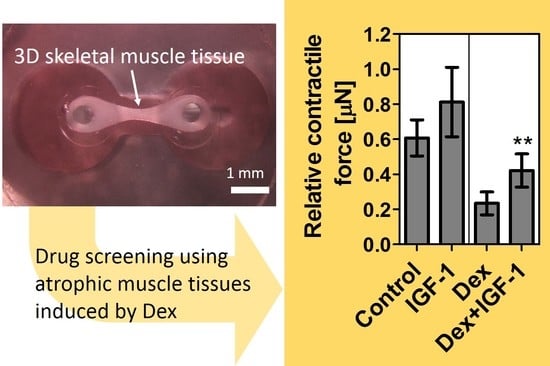
3.3. Evaluation of Muscle Tissue with Reduced Contractile Force Due to Addition of Dex
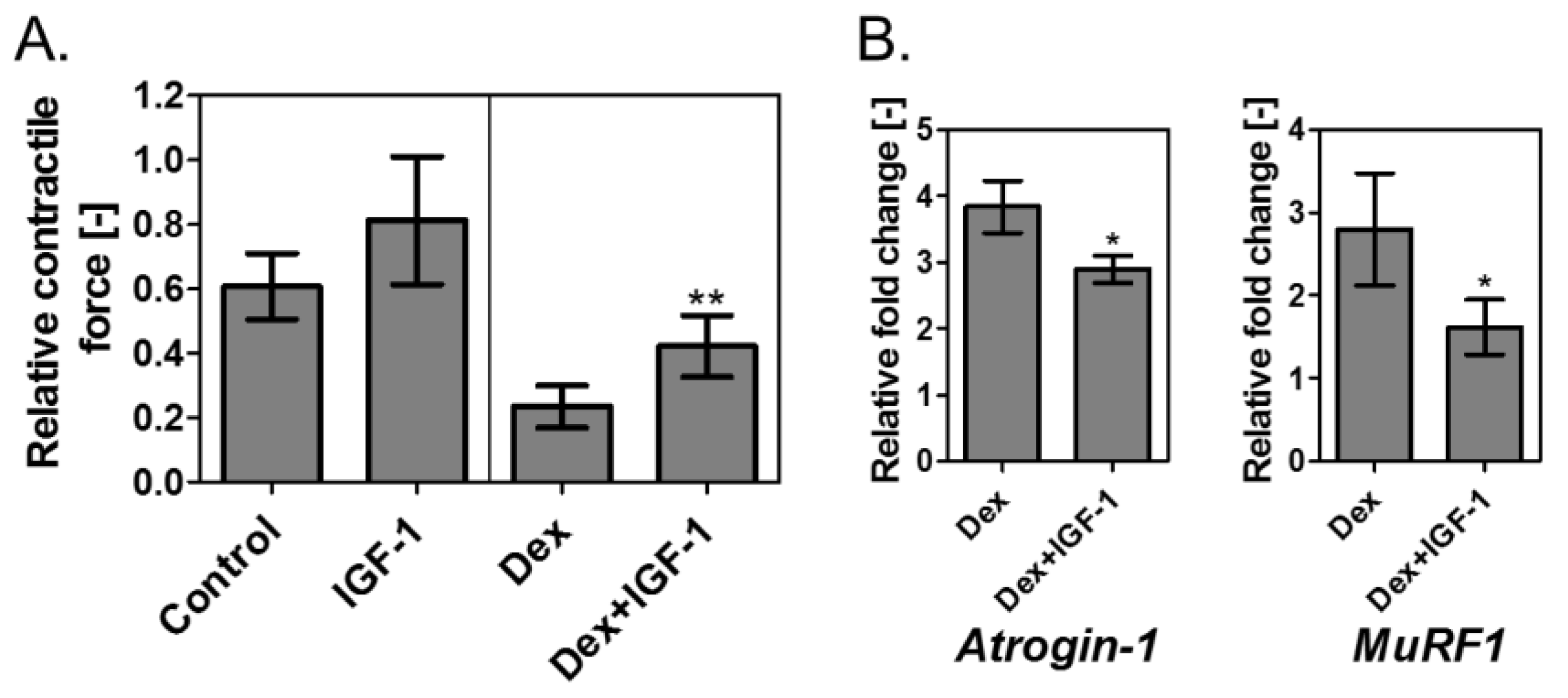
3.4. Verification of Contractile Force Recovery Effect by Addition of IGF-I to Muscle Atrophy Model
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bonaldo, P.; Sandri, M. Cellular and molecular mechanisms of muscle atrophy. Dis. Model. Mech. 2013, 6, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Schakman, O.; Kalista, S.; Barbe, C.; Loumaye, A.; Thissen, J.P. Glucocorticoid-induced skeletal muscle atrophy. Int. J. Biochem. Cell Biol. 2013, 45, 2163–2172. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.R. Loss of skeletal muscle mass in aging: Examining the relationship of starvation, sarcopenia and cachexia. Clin. Nutr. 2007, 26, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.H.; Churchward-Venne, T.A.; Mitchell, C.J.; Kolar, N.M.; Kassis, A.; Karagounis, L.G.; Burke, L.M.; Hawley, J.A.; Phillips, S.M. Hypoenergetic diet-induced reductions in myofibrillar protein synthesis are restored with resistance training and balanced daily protein ingestion in older men. Am. J. Physiol. Endocrinol. Metab. 2015, 308, E734–E743. [Google Scholar] [CrossRef] [PubMed]
- Lewelt, A.; Krosschell, K.J.; Stoddard, G.J.; Weng, C.D.; Xue, M.; Marcus, R.L.; Gappmaier, E.; Viollet, L.; Johnson, B.A.; White, A.T.; et al. Resistance strength training exercise in children with spinal muscular atrophy. Muscle Nerve 2015, 52, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Hemdan, D.I.; Hirasaka, K.; Nakao, R.; Kohno, S.; Kagawa, S.; Abe, T.; Harada-Sukeno, A.; Okumura, Y.; Nakaya, Y.; Terao, J.; et al. Polyphenols prevent clinorotation-induced expression of atrogenes in mouse C2C12 skeletal myotubes. J. Med. Investig. 2009, 56, 26–32. [Google Scholar] [CrossRef]
- Kawai, N.; Hirasaka, K.; Maeda, T.; Haruna, M.; Shiota, C.; Ochi, A.; Abe, T.; Kohno, S.; Ohno, A.; Teshima-Kondo, S.; et al. Prevention of skeletal muscle atrophy in vitro using anti-ubiquitination oligopeptide carried by atelocollagen. Biochim. Biophys. Acta (BBA) Mol. Cell. Res. 2015, 1853, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Alamdari, N.; Aversa, Z.; Castillero, E.; Gurav, A.; Petkova, V.; Tizio, S.; Hasselgren, P.O. Resveratrol prevents dexamethasone-induced expression of the muscle atrophy-related ubiquitin ligases atrogin-1 and murf1 in cultured myotubes through a sirt1-dependent mechanism. Biochem. Biophys. Res. Commun. 2012, 417, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.T.; Yin, Y.; Yang, Y.J.; Lv, P.J.; Shi, Y.; Lu, L.; Wei, L.B. Resveratrol prevents tnf-alpha-induced muscle atrophy via regulation of akt/mtor/foxo1 signaling in C2C12 myotubes. Int. Immunopharmacol. 2014, 19, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Sasaki, H.; Hida, H.; Fujita, H.; Obinata, K.; Shikida, M.; Nagamori, E. Assembly of skeletal muscle cells on a si-mems device and their generative force measurement. Biomed. Microdevices 2010, 12, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Fujita, H.; Nagamori, E. Evaluation systems of generated forces of skeletal muscle cell-based bio-actuators. J. Biosci. Bioeng. 2013, 115, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Araki, H.; Sakata, K.; Tonomura, W.; Hashida, M.; Konishi, S. Microfluidic devices for construction of contractile skeletal muscle microtissues. J. Biosci. Bioeng. 2015, 119, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Vandenburgh, H.; Shansky, J.; Benesch-Lee, F.; Barbata, V.; Reid, J.; Thorrez, L.; Valentini, R.; Crawford, G. Drug-screening platform based on the contractility of tissue-engineered muscle. Muscle Nerve 2008, 37, 438–447. [Google Scholar] [CrossRef] [PubMed]
- Sakar, M.S.; Neal, D.; Boudou, T.; Borochin, M.A.; Li, Y.Q.; Weiss, R.; Kamm, R.D.; Chen, C.S.; Asada, H.H. Formation and optogenetic control of engineered 3D skeletal muscle bioactuators. Lab Chip 2012, 12, 4976–4985. [Google Scholar] [CrossRef] [PubMed]
- Fujita, H.; Shimizu, K.; Nagamori, E. Novel method for fabrication of skeletal muscle construct from the C2C12 myoblast cell line using serum-free medium AIM-V. Biotechnol. Bioeng. 2009, 103, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
- Fujita, H.; Shimizu, K.; Nagamori, E. Novel method for measuring active tension generation by C2C12 myotube using UV-crosslinked collagen film. Biotechnol. Bioeng. 2010, 106, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Madden, L.; Juhas, M.; Kraus, W.E.; Truskey, G.A.; Bursac, N. Bioengineered human myobundles mimic clinical responses of skeletal muscle to drugs. eLIFE 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Dennis, R.G.; Kosnik, P.E., 2nd. Excitability and isometric contractile properties of mammalian skeletal muscle constructs engineered in vitro. In Vitro Cell. Dev. Biol. Anim. 2000, 36, 327–335. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Ito, A.; Fujita, H.; Nagamori, E.; Kawabe, Y.; Kamihira, M. Functional evaluation of artificial skeletal muscle tissue constructs fabricated by a magnetic force-based tissue engineering technique. Tissue Eng. Part A 2011, 17, 107–114. [Google Scholar] [CrossRef] [PubMed]
- McAleer, C.W.; Smith, A.S.T.; Najjar, S.; Pirozzi, K.; Long, C.J.; Hickman, J.J. Mechanistic investigation of adult myotube response to exercise and drug treatment in vitro using a multiplexed functional assay system. J. Appl. Physiol. 2014, 117, 1398–1405. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Duffy, R.; Lee, A.; Feinberg, A.W. Optimizing the structure and contractility of engineered skeletal muscle thin films. Acta Biomater. 2013, 9, 7885–7894. [Google Scholar] [CrossRef] [PubMed]
- Vandenburgh, H.; Shansky, J.; Benesch-Lee, F.; Skelly, K.; Spinazzola, J.M.; Saponjian, Y.; Tseng, B.S. Automated drug screening with contractile muscle tissue engineered from dystrophic myoblasts. FASEB J. 2009, 23, 3325–3334. [Google Scholar] [CrossRef] [PubMed]
- Massaccesi, L.; Goi, G.; Tringali, C.; Barassi, A.; Venerando, B.; Papini, N. Dexamethasone-induced skeletal muscle atrophy increases o-glcnacylation in C2C12 cells. J. Cell. Biochem 2016, 117, 1833–1842. [Google Scholar] [CrossRef] [PubMed]
- Castillero, E.; Alamdari, N.; Lecker, S.H.; Hasselgren, P.O. Suppression of atrogin-1 and murf1 prevents dexamethasone-induced atrophy of cultured myotubes. Metabolism 2013, 62, 1495–1502. [Google Scholar] [CrossRef] [PubMed]
- Bian, W.; Liau, B.; Badie, N.; Bursac, N. Mesoscopic hydrogel molding to control the 3D geometry of bioartificial muscle tissues. Nat. Protoc. 2009, 4, 1522–1534. [Google Scholar] [CrossRef] [PubMed]
- Latres, E.; Amini, A.R.; Amini, A.A.; Griffiths, J.; Martin, F.J.; Wei, Y.; Lin, H.C.; Yancopoulos, G.D.; Glass, D.J. Insulin-like growth factor-1 (IGF-1) inversely regulates atrophy-induced genes via the phosphatidylinositol 3-kinase/akt/mammalian target of rapamycin (PI3K/Akt/mTOR) pathway. J. Biol. Chem. 2005, 280, 2737–2744. [Google Scholar] [CrossRef] [PubMed]
- Fujita, H.; Hirano, M.; Shimizu, K.; Nagamori, E. Rapid decrease in active tension generated by C2C12 myotubes after termination of artificial exercise. J. Muscle Res. Cell Motil. 2010, 31, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Fujita, H.; Nedachi, T.; Kanzaki, M. Accelerated de novo sarcomere assembly by electric pulse stimulation in C2C12 myotubes. Exp. Cell. Res. 2007, 313, 1853–1865. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K.; Ito, A.; Sato, M.; Kawabe, Y.; Kamihira, M. Improved contractile force generation of tissue-engineered skeletal muscle constructs by IGF-I and Bcl-2 gene transfer with electrical pulse stimulation. Regen. Ther. 2016, 3, 38–44. [Google Scholar] [CrossRef]
- Ito, A.; Yamamoto, Y.; Sato, M.; Ikeda, K.; Yamamoto, M.; Fujita, H.; Nagamori, E.; Kawabe, Y.; Kamihira, M. Induction of functional tissue-engineered skeletal muscle constructs by defined electrical stimulation. Sci. Rep. 2014, 4, 4781. [Google Scholar] [CrossRef] [PubMed]
- Cohick, W.S.; Clemmons, D.R. The insulin-like growth-factors. Annu. Rev. Physiol. 1993, 55, 131–153. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Ito, A.; Kawabe, Y.; Nagamori, E.; Kamihira, M. Enhanced contractile force generation by artificial skeletal muscle tissues using IGF-I gene-engineered myoblast cells. J. Biosci. Bioeng. 2011, 112, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.C.; Dennis, R.G.; Larkin, L.; Baar, K. Rapid formation of functional muscle in vitro using fibrin gels. J. Appl. Physiol. 2005, 98, 706–713. [Google Scholar] [CrossRef] [PubMed]
- Sacheck, J.M.; Ohtsuka, A.; McLary, S.C.; Goldberg, A.L. IGF-I stimulates muscle growth by suppressing protein breakdown and expression of atrophy-related ubiquitin ligases, atrogin-1 and murf1. Am. J. Physiol. -Endocrinol. Metab. 2004, 287, E591–E601. [Google Scholar] [CrossRef] [PubMed]
- Rommel, C.; Bodine, S.C.; Clarke, B.A.; Rossman, R.; Nunez, L.; Stitt, T.N.; Yancopoulos, G.D.; Glass, D.J. Mediation of IGF-1-induced skeletal myotube hypertrophy by PI(3)K/Akt/mTOR and PI(3)K/Akt/GSK3 pathways. Nat. Cell. Biol. 2001, 3, 1009–1013. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Woltjen, K.; Miyake, K.; Hotta, A.; Ikeya, M.; Yamamoto, T.; Nishino, T.; Shoji, E.; Sehara-Fujisawa, A.; Manabe, Y.; et al. Efficient and reproducible myogenic differentiation from human iPS cells: Prospects for modeling miyoshi myopathy in vitro. PLoS ONE 2013, 8, e61540. [Google Scholar] [CrossRef]
- Hosoyama, T.; McGivern, J.V.; Van Dyke, J.M.; Ebert, A.D.; Suzuki, M. Derivation of myogenic progenitors directly from human pluripotent stem cells using a sphere-based culture. Stem Cells Transl. Med. 2014, 3, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Chal, J.; Al Tanoury, Z.; Hestin, M.; Gobert, B.; Aivio, S.; Hick, A.; Cherrier, T.; Nesmith, A.P.; Parker, K.K.; Pourquie, O. Generation of human muscle fibers and satellite-like cells from human pluripotent stem cells in vitro. Nat. Protoc. 2016, 11, 1833–1850. [Google Scholar] [CrossRef] [PubMed]
- Biedasek, K.; Andres, J.; Mai, K.; Adams, S.; Spuler, S.; Fielitz, J.; Spranger, J. Skeletal muscle 11beta-HSD1 controls glucocorticoid-induced proteolysis and expression of E3 ubiquitin ligases atrogin-1 and murf-1. PLoS ONE 2011, 6, e16674. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shimizu, K.; Genma, R.; Gotou, Y.; Nagasaka, S.; Honda, H. Three-Dimensional Culture Model of Skeletal Muscle Tissue with Atrophy Induced by Dexamethasone. Bioengineering 2017, 4, 56. https://doi.org/10.3390/bioengineering4020056
Shimizu K, Genma R, Gotou Y, Nagasaka S, Honda H. Three-Dimensional Culture Model of Skeletal Muscle Tissue with Atrophy Induced by Dexamethasone. Bioengineering. 2017; 4(2):56. https://doi.org/10.3390/bioengineering4020056
Chicago/Turabian StyleShimizu, Kazunori, Riho Genma, Yuuki Gotou, Sumire Nagasaka, and Hiroyuki Honda. 2017. "Three-Dimensional Culture Model of Skeletal Muscle Tissue with Atrophy Induced by Dexamethasone" Bioengineering 4, no. 2: 56. https://doi.org/10.3390/bioengineering4020056
APA StyleShimizu, K., Genma, R., Gotou, Y., Nagasaka, S., & Honda, H. (2017). Three-Dimensional Culture Model of Skeletal Muscle Tissue with Atrophy Induced by Dexamethasone. Bioengineering, 4(2), 56. https://doi.org/10.3390/bioengineering4020056