2. Materials and Methods
2.1. Tissue Preparation of Porcine Pericardia
Porcine pericardia were isolated from the heart and were obtained from Taewoong Medical Inc. (Gimpo-si, Republic of Korea). For tissue preparation, porcine pericardium was rinsed in ice-cold phosphate normal saline with 1% antibiotic/antimycotic. The fat and connective tissue were trimmed. The tissues were immersed in 0.1% PAA and 4% ethanol in distilled water for 4 h, and then washed with buffered saline solution (PBS) under agitation at room temperature (RT). Some pericardia were analyzed as untreated specimens for DNA content after sectioning for histology.
2.2. Decellularization and Glutaraldehyde Fixation
The porcine pericardium was directly decellularized using 0.25% sodium dodecyl sulfate (SDS) (Sigma-Aldrich, St. Louis, MO, USA, 436143) in diH2O containing 0.5% (v/v) Triton X-100. For decellularization, each process using various solutions was carried out under an agitator (180 rpm) in an orbital shaker (SHO-2D, DAIHAN).
The porcine pericardial tissues were initially decellularized with a hypotonic solution with 0.25% SDS for 24 h at 4 °C and washed with distilled water for 1 h. Then they were treated with a hypotonic solution containing 0.5% Triton X-100 for 24 h at 4 °C, and then washed with distilled water for 12 h at 4 °C. After washing, these tissues were treated with an isotonic solution for 48 h at 4 °C. The tissues were treated in PBS with 30% polyethylene glycol 1000 (PEG-1000) and 1% antibiotic/antimycotic for 48 h at 4 °C. The tissues were finally treated with a hypertonic solution for 3 h at 4 °C, and then these tissues were washed in PBS. Decellularized pericardial tissues were obtained using our established protocol [
4].
For in vivo studies after recellularization, the porcine pericardial tissues were prepared into four groups: native, native + 0.5% glutaraldehyde (GA), decellularized, and decellularized + 0.5% GA. Tissues were fixed in PBS-buffered 0.5% GA solution (pH 7.4) at RT for 14 days (0.5% GA). Tissue was fixed in 0.5% GA for 72 h, and then fixed in 0.25% GA with 75% ethanol and 5% octanol for 48 h, and then the tissues were again treated in 0.25% GA for 7 days and then in 0.2 M glycine solution for 48 h (decellularized + 0.5% GA).
2.3. Enzyme Digestion
After decellularization, porcine pericardia were treated with PNGase-F (#P0704L, 1000U, NEB) in concentrations of 2000 unit/mL, whereas recombinant α-galactosidase (G7163, sigma) was treated with concentrations of 0.1 unit/mL. In addition, SDS + Tx-decellularized porcine pericardia were treated with α-galactosidase in combination with PNGase-F. Native pericardial tissues served as the untreated control or the negative control. Porcine pericardial tissues (1 × 1 cm size) with isotonic solution were applied for 24 h at 4 °C with agitation. Each enzymatic treatment with sliced tissues was performed at 37 °C for 24 h under agitation. After treatment, sliced tissues were washed in PBS under agitation (120 rpm; 10 min each; 3 times washes) at RT. Finally, the tissue samples were stored in sterile PBS supplemented with 1% (v/v) penicillin/streptomycin and 400 μL/L amphotericin B for 24 h at 4 °C.
2.4. Histology and Quantification of DNA Contents
Native and decellularized porcine pericardia were treated with different enzymatic treatments: SDS + Tx-decellularized tissues were treated with α-galactosidase, PNGase-F, and α-galactosidase in combination with PNGase-F or no enzyme. Dissected samples of native, decellularized, and both decellularized and enzymatically treated pericardia were fixed in 4% paraformaldehyde for 24 h and embedded in paraffin. Histological tissue sections (5 μm) of each sample were stained for hematoxylin and eosin (H&E) and immunofluorescence (IF) staining. Sliced samples of native and decellularized tissue for DNA quantification were homogenized, and then ground tissue samples (100 mg) were incubated in lysis buffer with proteinase K and RNase A solution for 24 h at 56 °C for 30 min. The QiAmp DNA Mini kit (Qiagen, Hilden, Germany) was used to measure DNA content according to the manufacturer’s instructions. Isolated DNA pellets were determined using a NanoDrop UV-VIS spectrophotometer (Thermo Fisher), and the absolute amount of DNA (ng) was quantified in relation to the total tissue mass (mg) used.
2.5. Cell Culture of Human Adipose Tissue-Derived Stem Cells (hADSCs)
Human adipose-derived stem cells (hADSCs) were purchased from Thermo Scientifics (R7788115). The hADSCs were cultured at 37 °C in a humidified atmosphere of 5% CO2 with Dulbecco’s Modified Eagle Medium (DMEM; LM001-05, Welgene, Gyeongsan, Republic of Korea) medium containing 10% fetal bovine serum (FBS), 1% penicillin/streptomycin (P/S). Rat bone marrow-derived mesenchymal stromal cells (BM-MSCs) were purchased from (Cell Biologics (RA-6043; Cell Biologics, Chicago, IL, USA). The BM-MSCs were cultured at 37 °C in a humidified atmosphere of 5% CO2 with Minimum Essential Medium Eagle, Alpha (α-MEM, Welgene, LM 008-01) medium containing 10% FBS, 1% P/S. The cells were grown in T-75 flasks for 5 to 7 days, and they were kept within 5 to 6 passages for the experiments. The cell culture medium of both cells was changed every 3 days.
After cell seeding on fabricated scaffolds, all pericardial scaffolds used in these experiments were cultured in DMEM or α-MEM, supplemented with 2% FBS, 1% P/S, transforming growth factor beta 1 (TGF-β1, 2.5 ng/mL, ab50036, Abcam), and bone morphogenetic protein 4 (BMP-4, 2.5 ng/mL, SRP6156, Sigma).
2.6. Fibrin Mesh Fabrication for Pericardial Scaffolds Modification
For recellularization of xenoantigen-free cardiac scaffolds, modification of porcine pericardia was performed in five experimental conditions: native, decellularized, decellularized + α-galactosidase (0.1 unit/mL), decellularized + PNGase-F (2000 unit/mL), and α-galactosidase in combination with PNGase-F. For in vivo studies after recellularization, the porcine pericardial tissues were modified into four groups: native, 0.5% GA, decellularized, and decellularized + 0.5% GA. The fibrin mesh was created by combining a solution containing fibrinogen, thrombin, and heparin, according to each process. The pericardial scaffolds were cut into approximately 1 × 1 cm (length × width, n = 3), and sliced samples were sterilized using 70% alcohol. Prior to cell seeding, the sterile scaffolds were washed using sterilized PBS, and the sliced pericardial scaffolds were fabricated with a fibrin mesh (P- or DP-Fb) in a 12-well plate. The scaffolds were washed using 0.05 M Tris-HCl buffer (TB) solution, which was modified by attachment of heparin (P- or DP-Fb + H) overnight at 4 °C. Upon overnight incubation, the fibrin mesh + heparin (P- or DP-Fb + H) scaffolds were washed with sterilized PBS, and were incubated with vascular endothelial growth factor (VEGF 165, 100 ng/mL in PBS, GeneScript USA Inc.) for 2 h at RT and washed with PBS, using an established protocol [
11].
2.7. Human Adipose-Derived Stem Cells Seeded onto Modified Pericardial Scaffolds
For cellular infiltration of hADSCs into the fibrin mesh-coated scaffolds, all pericardial scaffolds were spread on both sides and were seeded onto the central region. However, hADSCs were not seeded on some modified pericardial scaffolds, which were used as a negative control (n = 3). The hADSCs (passage 5) were seeded on the upper side of all modified pericardial scaffolds (P- or DP-Fb + H + VEGF) at a density of 5 × 104 cells (day 0) and per well in a 12-well plate. The seeded scaffolds were turned over, and hADSCs were seeded on lower side of scaffolds at a density of 5 × 104 cells (day 0) in DMEM medium supplemented with 2% FBS, transforming growth factor beta 1 (TGF-β1, 2.5 ng/mL, ab50036, Abcam), and bone morphogenetic protein 4 (BMP-4, 2.5 ng/mL, SRP6156, Sigma), and these pericardial scaffolds were cultured until day 56. The rat BM-MSCs (passage 5) were seeded on top of each modified pericardial scaffold (P- or DP-Fb + H + VEGF) at a density of 1 × 105 cells (day 0) in 50 μL culture medium with 10% FBS for 3 h at 37 °C and 5% CO2. The seeded scaffolds were turned over, and the cells were seeded on the bottom of the scaffolds at a density of 1 × 105 cells (day 0) using 50 μL α-MEM medium in a 12-well plate. After cell attachment to the scaffolds, 2 mL culture media was added to each cell-seeded scaffold, and then the scaffolds were cultured for 3 days.
2.8. Immuno-Fluorescence Staining of Recellularized Pericardial Scaffolds
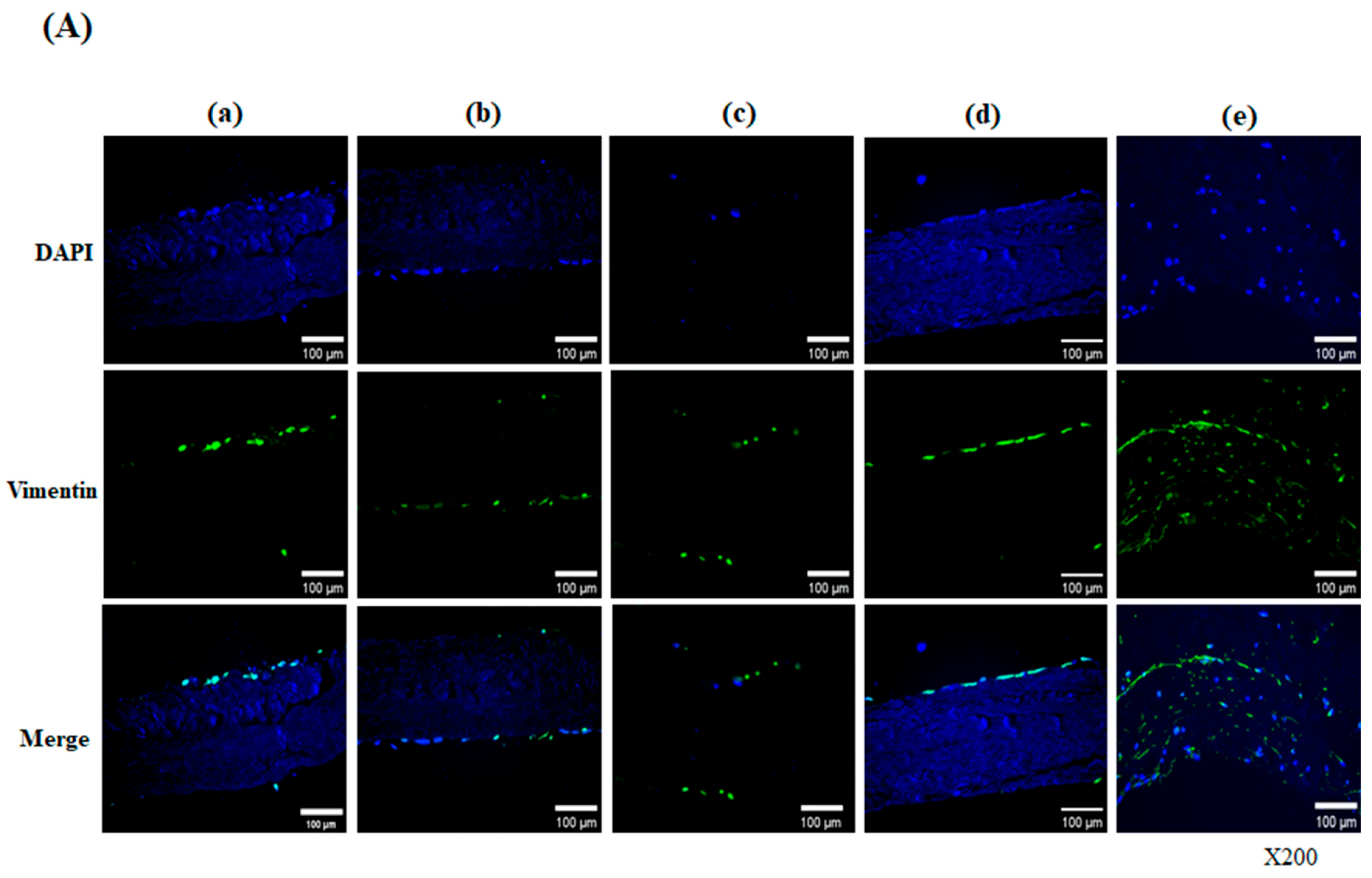
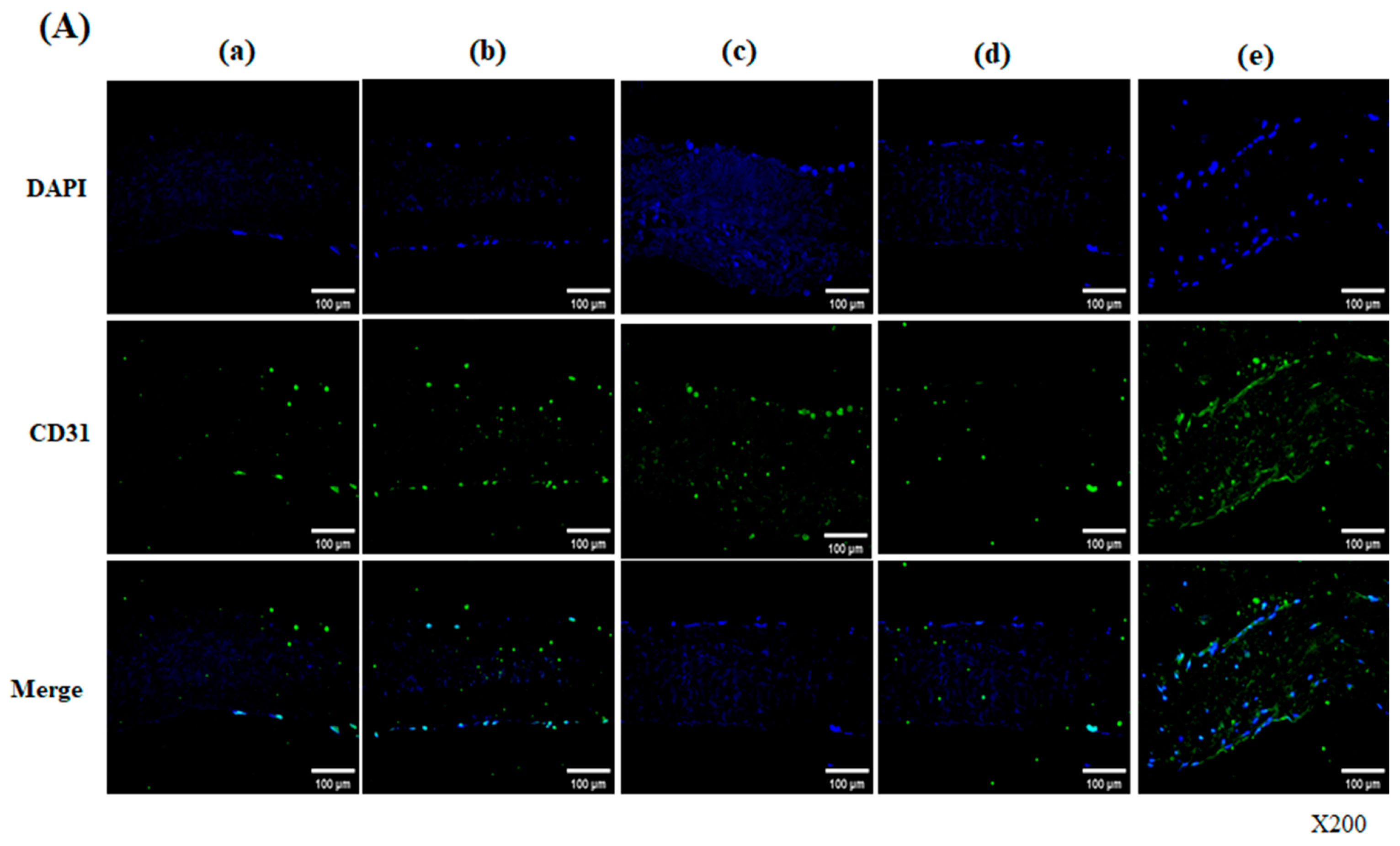
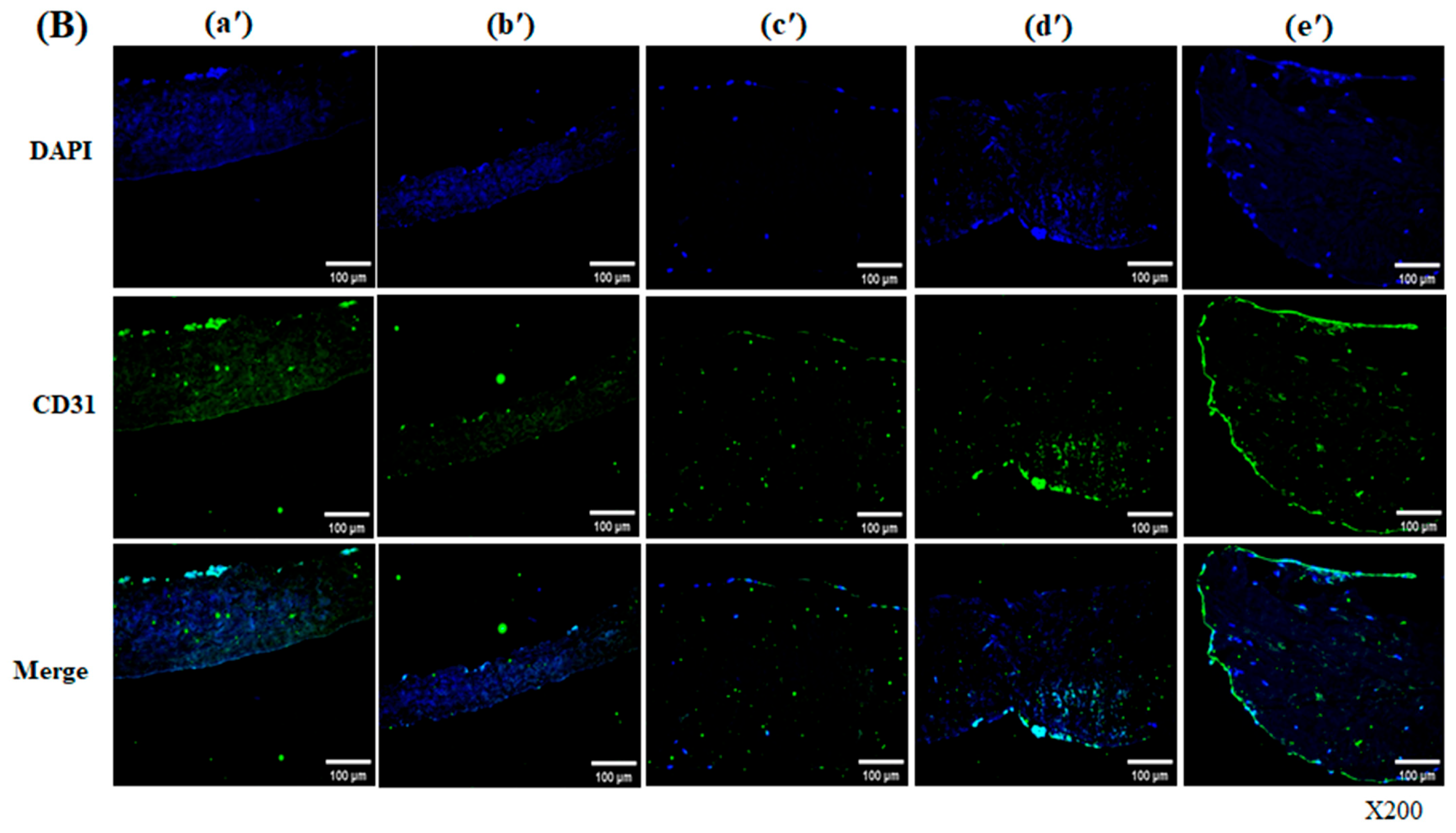
After 56 days of in vitro culturing, the seeded scaffolds were cut in half, fixed in 4% para-formaldehyde solution, and processed for H&E and IF staining. IF staining was performed using embedded sections and deparaffinized sections. For the IF staining, all scaffolds were incubated in antigen retrieval buffer (10 mM citrate buffer, pH 6.0) at 95 °C for 10 min, followed by 3 washes in PBS for 5 min each. The samples were then incubated in 5% blocking serum for 60 min at RT. The seeded and unseeded scaffold samples were then incubated with primary antibodies for vimentin (ab16700, Abcam, dilution 1:200), calponin (ab227661, Abcam, dilution 1:100), fibronectin (F0916, sigma, dilution 1:100), and CD31 (ab182981, Abcam, dilution 1:2000) at 4 °C overnight. Upon overnight incubation, all scaffolds were then washed 3 times for 10 min each in PBS-T, and incubated for 2 h at RT with anti-rabbit secondary antibody conjugated with Alexa Fluor 488 (A11008, Invitrogen, Carlsbad, CA, USA; 1:500) and anti-mouse secondary antibody conjugated with Alexa Fluor 488 (AB150113, Invitrogen, 1:500). The samples were then washed with PBS-T for 10 min. Nuclei were counterstained with 4ʹ,6-diamidino-2-phenylindole (DAPI) (Invitrogen, 1:1000) for 1 min. The sections were washed 3 times with PBS-T and mounted using ShandonTM Immuno-Mount (Thermo Fischer Scientific). The stained sections were imaged using an inverted fluorescence microscope (DMI4000B, Leica Microsystems, Wetzlar, Germany). Merging of the stained images and DAPI was processed through Application Suite X Image Viewer software version 3.5.7 (Leica), and cell counting was performed using the ImageJ/FIJI software (NIH, Bethesda, MD, USA), version 1.54f (FIJI distribution).
2.9. Immuno-Fluorescence Using Confocal Microscopy
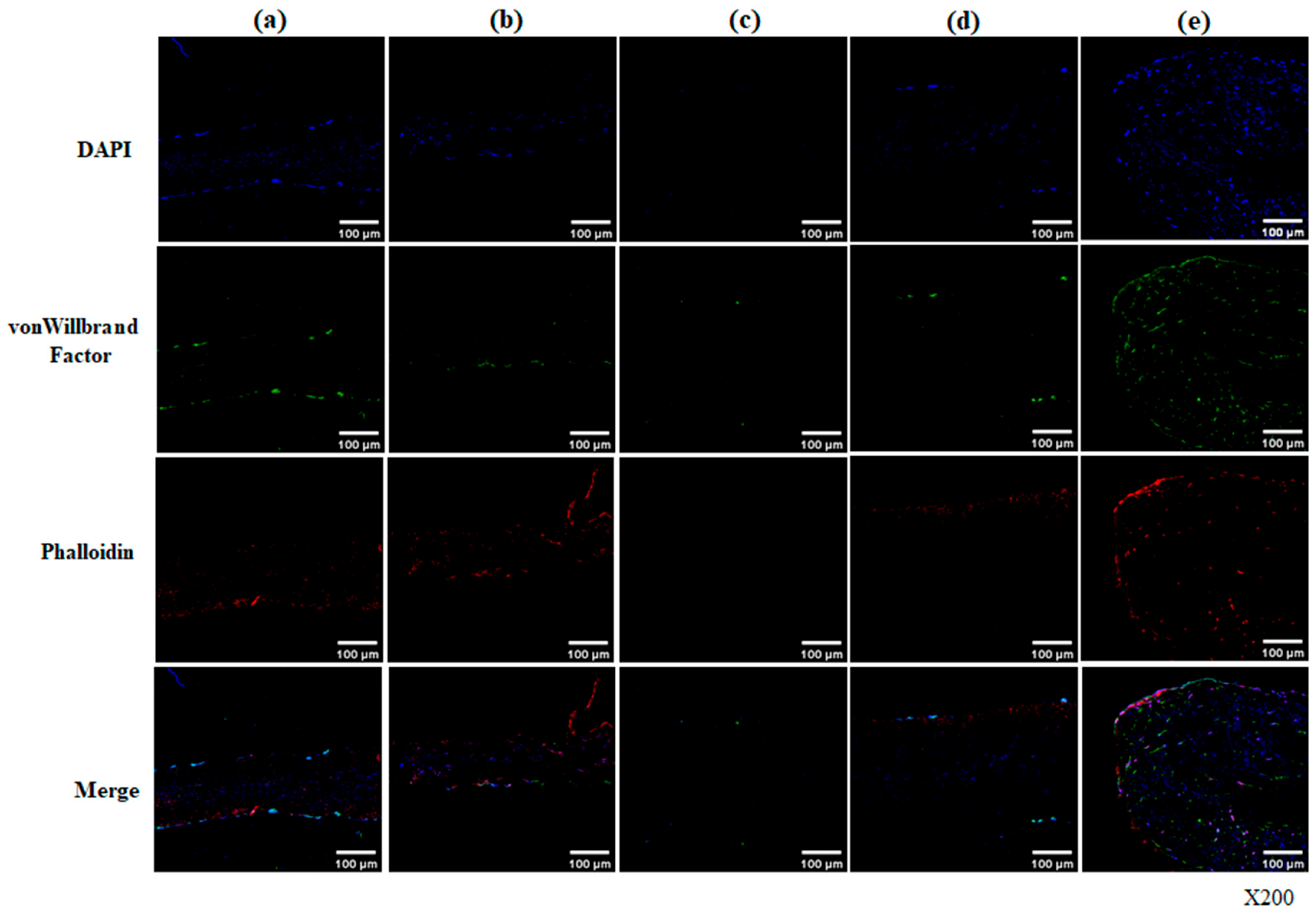
All scaffolds were fixed in 4% paraformaldehyde solution and processed for confocal microscopy. Paraffin-embedded sections were incubated in 0.2% Triton-X for 15 min, and then the sections were washed 3 times with PBS-T. After washing in PBS-T, the samples were then incubated in 5% blocking serum for 60 min at RT. The sections were incubated overnight at 4 °C with primary antibodies directed against von Willebrand factor (F8/86) (MA5-14029, Thermo Fischer Scientific, dilution 1:50). After incubation, the sections were further incubated with goat anti-rabbit secondary antibody conjugated with Alexa Fluor 488 (A11008, Invitrogen, 1:500) for 1 h at RT, and then phalloidin-647 (ab176759, Abcam, dilution 1:1000) for 30 min at RT. After the animal experiment, confocal analysis of harvested tissue sections was conducted. BM-MSC seeded sections were incubated with vimentin-647 (ab176759, Abcam, dilution 1:1000) for 30 min at RT. Nuclei were counterstained with DAPI (Invitrogen, 1:1000) for 1 min. The stained sections were analyzed by confocal microscopy (STELLARIS5, Leica).
2.10. Animal Experiments
Four-week-old male Sprague-Dawley rats (n = 20) were purchased from the KOATECH Co., Ltd. All procedures were carried out in accordance with the Guiding Principles on Care and Use of Animals of Seoul National University Hospital (IACUC No.24-00181-S1A0). For recellularized in vivo experiments, anesthesia was performed by intraperitoneal (i.p) injection with a mixture of Zoletil 50 25 mg/kg and xylazine 10 mg/kg. After the rats were anesthetized and shaved, six subcutaneous pouches were created in the dorsal area of each animal. Each group had its sliced scaffold samples (6 × 6 mm each) implanted into each pouch, and the wounds were closed with 6/0 nylon sutures. Native and decellularized porcine pericardial scaffolds with or without 0.5% GA, in the absence or in the presence of seeded BM-MSC, were implanted intradermally in a rat dorsal skin and explanted after 7 and 28 days. The animals were divided into 4 groups: native (n = 12), 0.5% glutaraldehyde (n = 12), decellularized (n = 12), and 0.5% GA-decellularized (n = 12). After each implantation, a papule was observed at the implantation site in the dorsal area of each animal. Cell-seeded scaffold-implanted rats were kept in a light/dark cycle until the next experiment. Four weeks after implantation, the scaffold samples were harvested, and finally, the implanted scaffolds were harvested on day 28. Upon harvest, examination of histological features and calcification using the harvested scaffolds was conducted.
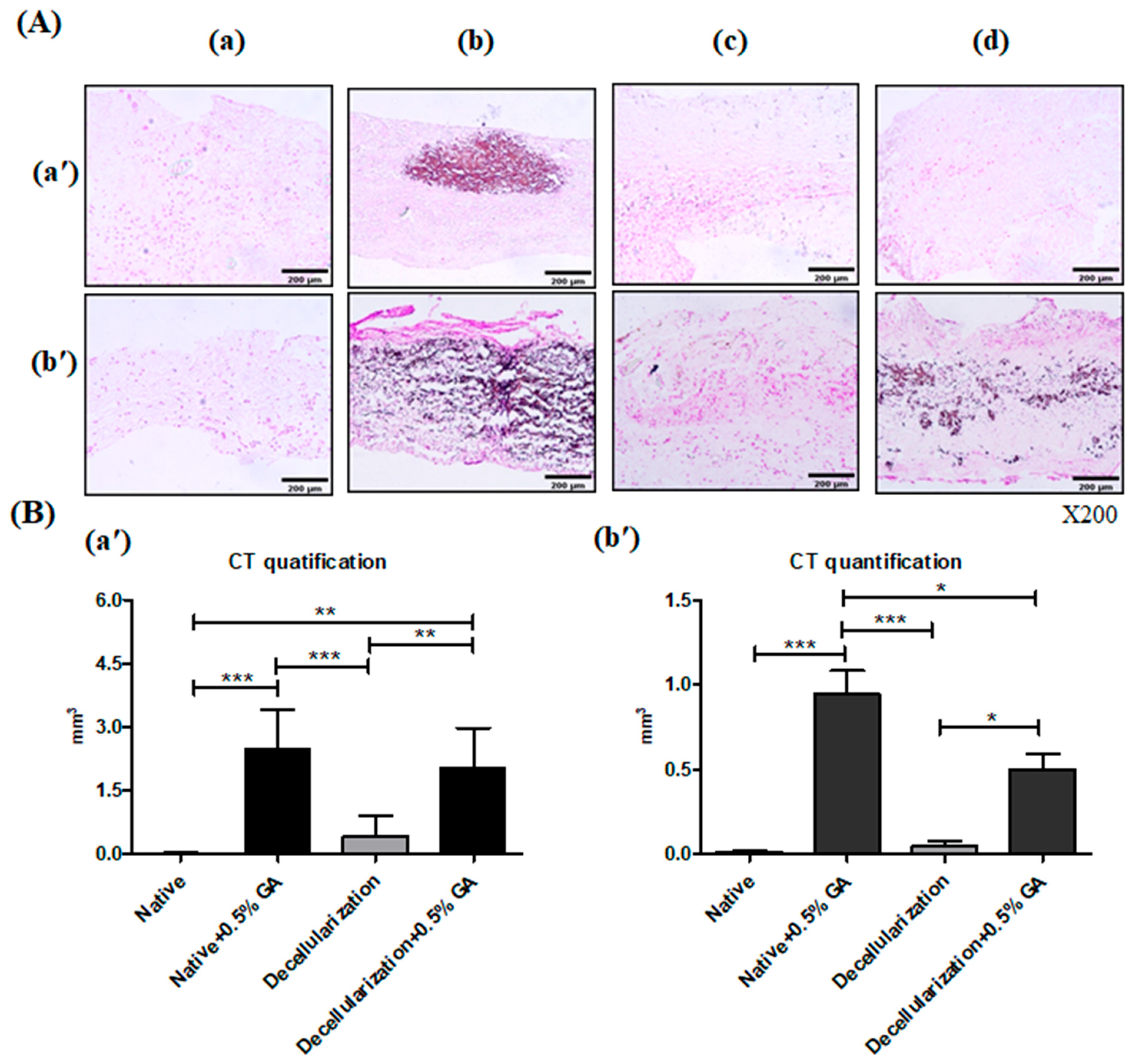
2.11. Von Kossa Calcium Staining of Scaffolds After Implantation
Histological paraffin sections (5 μm) of each sample were stained for hematoxylin and eosin (H&E) and Von Kossa staining. Von Kossa staining was used to examine the calcium deposition in the implanted samples. The tissue sections were stained for optical microscope analysis (Nikon ECLipse Ci-L, Version 22.5 x64, Tokyo). Stained pictures were obtained using iSolution Lite (IMT i-Solution Inc., Vancouver, BC, Canada).
2.12. Calcification Analysis of the Scaffold After Implantation
For calcium quantification analysis of the harvested scaffolds, Micro-Computed Tomography (CT) analysis was conducted. Harvested scaffold samples from each group were rinsed with normal saline and then dried at RT for 5 min. CT images of each harvested sample were measured using a Micro-CT analyzer (Quantum GX II in vivo micro-CT, BR70000032-01, PerkinElmer), and the degree (volume of interest (VOI), mm3) of calcification in the images was analyzed by the Caliper Analyze 12.0 software program.
4. Discussion
In this study, we successfully developed a clinically applicable recellularization strategy for decellularized and enzymatically xenoantigen-removed porcine pericardial scaffolds using hADSCs. Dual enzymatic removal of xenoantigens using α-Gal and PNGase-F significantly improved scaffold biocompatibility and promoted effective recellularization. Human ADSCs demonstrated infiltration, survival, and differentiation into valvular interstitial and endothelial-like phenotypes, as evidenced by expression of vimentin, fibronectin, calponin, CD31, and vWF. The functional integration of endothelial-like cells and ECM remodeling was further supported by phalloidin co-localization and progressive fibronectin expression. Furthermore, in vivo experiments confirmed that recellularization occurred within the biocompatible scaffolds after implantation, and a marked reduction in calcification was observed.
Importantly, the novelty of this study lies in several key aspects that distinguish it from prior work. While previous studies on xenogeneic valve recellularization have been limited by incomplete cell engraftment, reliance on non-human or mixed cell populations, and persistent xenoantigen-related immune responses, our approach addresses these challenges in an integrated manner. First, we established a xenoantigen-free scaffold through dual enzymatic removal of both α-Gal and non-α-Gal epitopes, thereby creating a biologically permissive environment for human cell engraftment. Second, we demonstrate, to the best of our knowledge, the first successful recellularization of porcine-derived cardiovascular tissue using hADSCs as a single-cell source without co-culture or pre-differentiation. Third, our strategy was directly applied to a clinically relevant xenograft platform, PULSTA, significantly enhancing its translational potential. Finally, by combining recellularization with a multi-step anti-calcification treatment, we extend the impact of recellularization beyond biocompatibility to improved resistance against structural degeneration, which remains a major limitation of current bioprosthetic valves.
To address the shortage of human-derived organs, the development of artificial or animal-derived tissues with excellent biocompatibility through tissue engineering has emerged as a particularly important issue, especially for patients with congenital heart disease. In our previous studies, we optimized the decellularization process to preserve the ECM structure and applied it to tissue valve construction [
7]. However, to overcome the limitations of decellularized xenograft valves—particularly their deficiency in essential biological functions like regeneration, endothelialization, and immunomodulation—we shifted our focus toward strategies involving recellularization following decellularization [
12,
13]. We demonstrated that porcine xenograft tissue, after appropriate decellularization, could be recellularized in vitro by co-culturing hADSCs with HUVECs to accelerate the process [
10]. To streamline the process for greater clinical feasibility, we explored a monoculture recellularization approach using adipose-derived stem cells alone.
Initially, our goal was to enhance the efficiency of recellularization using mesenchymal stem cells (MSCs) by modifying the surface of decellularized scaffolds, thereby addressing their functional limitations. Earlier studies have shown that integrating decellularized porcine aortic valves with porous matrix metalloproteinase (MMP) and degradable polyethylene glycol (PEG) hydrogels [
14], or applying gelatin or fibronectin to the surface, resulted in improved cell attachment and proliferation [
15]. As demonstrated in our previous study [
10], we sought to improve MSC attachment and proliferation by incorporating heparin, fibrin mesh, and VEGF [
11]. Fibrin-based modification can bind various growth factors and fibronectin present in the serum of the culture medium [
16]. Additionally, fibronectin binding can be further enhanced through collagen-binding domains of the decellularized pericardium and through heparin conjugation [
17], thereby promoting cell adhesion, proliferation, and migration.
Even if cells adhere to the surface of the scaffold, they may fail to engraft due to inflammatory immune responses, and calcification or thrombosis may occur first, leading to inflammation. Therefore, it is essential to sufficiently remove residual antigens from the xenogeneic tissue that can trigger inflammation. Notably, scaffolds treated with both α-galactosidase and PNGase-F showed superior recellularization outcomes compared to single enzyme or untreated controls, in addition to the decellularization strategy we have developed [
2]. These findings highlight the synergistic role of dual enzyme pretreatment in reducing xeno-antigens and immunogenicity, thereby enhancing scaffold biocompatibility and enhancing cell adhesion and migration.
In addition, we used adipose-derived MSCs, which are relatively easy to obtain, for recellularization. Adipose-derived MSCs (AD-MSCs) and bone marrow-derived MSCs (BM-MSCs) are typically the most frequently utilized cell types for recellularization. Prior studies have indicated that these two MSC sources exhibit comparable levels of invasiveness, adhesion, and proliferation [
18]. Moreover, MSCs exhibit distinct immunomodulatory properties, and research has demonstrated their ability to survive even in an allogenic environment [
19]. If allogeneic MSCs can engraft and differentiate within xenogeneic tissues, they may retain their biological activity in a host environment. This potential has led to extensive research utilizing MSCs in tissue engineering.
Interestingly, stem cell proliferation and differentiation are regulated by interactions with the extracellular matrix and the surrounding culture environment. Thus, decellularized tissue scaffolds can serve as a key signaling platform guiding cell fate decisions. A previous study investigated the chondrogenic differentiation of bovine-derived MSCs when cultured in a collagen type II-rich environment [
20]. Given that porcine pericardial tissue was used in our study, we anticipated that the seeded stem cells would interact with the residual ECM components and differentiate into cell types characteristic of cardiac or valvular tissue. It has been shown that decellularized rat-derived cardiac ECM can promote the recellularization of human embryonic stem cell-derived cardiomyocytes [
21]. In another study of Fernanda et al., atrial-derived ECM was found to direct human iPSC-derived cardiomyocytes toward an atrial-specific phenotype [
22]. These results indicate that residual ECM components within the scaffold play a critical role in guiding the acquisition of mature phenotypes in multipotent cells. Although this study did not determine the specific ECM components that remained or the signaling pathways driving stem cell differentiation, immunohistochemical staining revealed the potential for differentiation into valvular interstitial cells or valvular endothelial cells. Therefore, it is crucial to preserve the structural and biochemical integrity of the ECM during the cell removal process, as well as to remove it effectively.
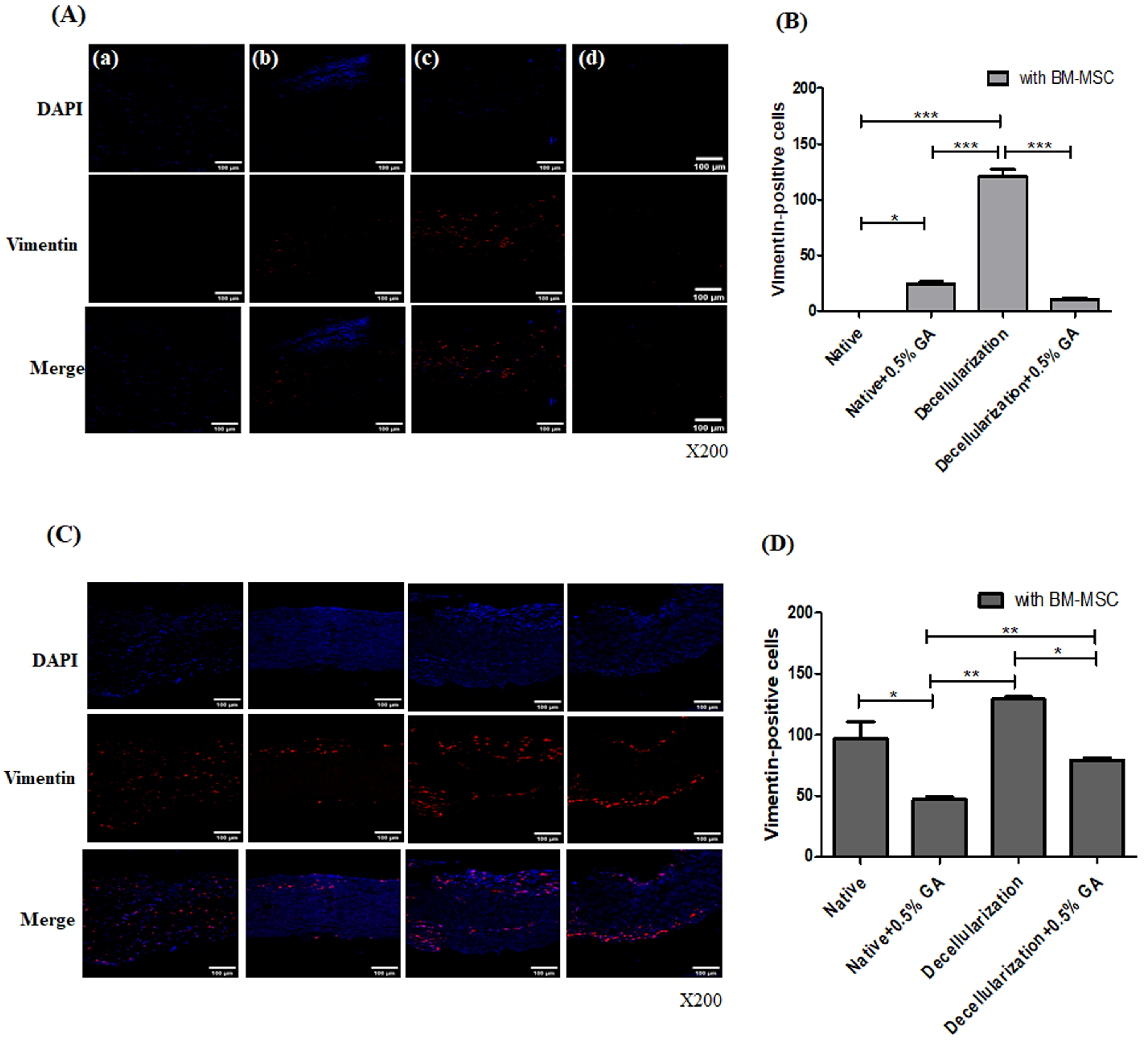
In vitro culture of hADSC-seeded decellularized scaffolds demonstrated progressive recellularization and phenotypic differentiation. Vimentin-positive cells infiltrated the scaffold by day 28, with widespread distribution by day 56. Particularly, α-galactosidase/PNGase F-treated scaffolds showed enhanced vimentin-positive cell penetration, indicating effective recellularization. Fibronectin, a key ECM protein involved in cell adhesion, was initially weakly expressed at day 28 but became prominently localized on the surface and within the matrix by day 56. This temporal increase suggests that MSCs not only adhered and survived on the scaffold but also actively participated in ECM remodeling. While some fibronectin may have originated from residual native ECM, the increased expression over time implies new synthesis by hADSCs. This aligns with previous findings by Lee et al., who reported that fibronectin-derived peptides promote stem cell adhesion, proliferation, and differentiation via integrin-mediated signaling [
23]. Calponin expression within the scaffold matrix suggests early differentiation of hADSCs toward smooth muscle-like cells, potentially supporting vascular remodeling. Although markers such as α-SMA, actin/myosin filaments, and collagen types I and III were not assessed, the positive expression of calponin and vimentin indicates that the ADSCs likely differentiated into myofibroblast-like cells, reflecting the contractile property and ECM-producing capability required in heart valve tissue [
24].
Endothelial differentiation was evidenced by the presence of CD31- and von Willebrand factor (vWF)-positive cells forming a monolayer across the scaffold surface. These cells mimic native endothelial function, acting as a barrier to blood components and contributing to immunomodulation and thromboresistance. Endothelial cells additionally release a range of growth factors and signaling molecules that support the regeneration of surrounding tissue and the remodeling of the ECM, forming a basis for self-remodeling [
25]. Importantly, vWF and phalloidin co-expression indicated that the differentiated cells not only acquired endothelial identity but also developed a well-organized cytoskeletal structure, suggesting a stable, functionally active state [
26,
27]. This co-expression was observed not only on the surface but also within the scaffold, demonstrating integration and migration of endothelial-like cells throughout the construct. This implies both functional endothelial differentiation and structural stability, both of which are crucial factors in tissue engineering applications.
To investigate the functional relevance of recellularization in vivo, we implanted BM-MSC–seeded scaffolds into subcutaneous pockets in rats. Histological analysis revealed increased vimentin expression and greater cell infiltration in the decellularized groups, particularly in scaffolds without GA fixation. Furthermore, von Kossa staining and Micro-CT analysis demonstrated reduced calcification in recellularized biocompatible scaffolds compared to GA-treated scaffolds. These results indicate that recellularization promotes cell integration and ECM remodeling while also reducing the risk of calcification- a critical drawback of GA-fixed bioprosthetic valves. We have recognized that xenogeneic tissues tend to lose durability due to calcification after implantation in the human body, and we have been studying methods to reduce this issue. We developed a four-step anti-calcification protocol—decellularization [
4], space filling, organic solvent treatment, and detoxification—to reduce immunogenicity, prevent calcification, and improve the durability of xenograft tissues for cardiovascular use [
28]. GA treatment has been widely used to enhance the durability of decellularized heart valve tissues. However, residual aldehyde groups left after GA fixation can induce cytotoxicity, inhibit endothelialization, and accelerate valve dysfunction by promoting calcification, thrombosis, and chronic inflammation [
29,
30]. In this study, even when MSCs were seeded onto GA-fixed tissues, in vivo analysis showed that recellularization of the scaffold occurred. Notably, the recellularized tissues exhibited the lowest levels of calcification, which was quantitatively confirmed. Although further studies are needed to determine whether the recellularized tissue can maintain durability and support ongoing cell survival and remodeling in vivo, it is encouraging that recellularization was achievable even after GA fixation, leading to successful cell engraftment and reduced calcification.
This study strongly supports that stem cell-based recellularization can produce functionally viable tissue grafts, particularly when combined with effective xenoantigen removal, and also demonstrates the potential of recellularization to enhance anti-calcification properties. However, we were limited by the inability to quantitatively assess the extent of immunostaining and did not include gene expression data as additional indicators [
24]. While our findings provide that recellularization can reduce the degree of calcification, further research is required to clarify how the recellularized tissue responds in vivo, especially in terms of its immunogenic profile and the potential for sustained histological remodeling. Furthermore, it remains unclear to what extent the recellularized valve may induce inflammation upon in vivo implantation, and whether this inflammatory response could ultimately promote an M2-mediated immune reaction leading to long-term tissue healing and regeneration [
13]. According to previous studies, heart valves recellularized with MSCs showed increased expression of CD163, a marker of M2 macrophages [
31], which is associated with an anti-inflammatory and pro-regenerative environment. Recellularized xenogeneic valves using autologous mesenchymal stem cells showed a marked reduction in xenoreactive immune response, with immune cell infiltration and immunoglobulin production levels comparable to those of autologous tissue [
32]. In our next study, we plan to further investigate the immune response in vivo. It has been demonstrated that applying biomimetic flow conditions to tissue-engineered constructs in vitro promotes tissue maturation and enhances mechanical properties. This study underscores the need for further investigation to assess whether the recellularized xenogeneic tissue can preserve its structural integrity and facilitate tissue development and cellular differentiation under biomimetic conditions.
This study has several limitations that should be considered in light of its defined scope. First, the analysis was primarily focused on early-stage recellularization and endothelial-like differentiation based on structural and phenotypic markers. Accordingly, detailed characterization of immune responses, macrophage involvement, and endothelial activation was not included. Even at early stages of differentiation, endothelial-like cell development is inherently associated with inflammatory and immune processes, particularly involving macrophage activity and smooth muscle cell (SMC) behavior. In particular, macrophage polarization—especially toward an M2 phenotype—plays a critical role in inducing the contractile phenotype of SMCs. These contractile SMCs provide a necessary substrate for endothelial cell adhesion, as endothelial-like cells are unlikely to directly attach to decellularized valve matrices in the absence of an underlying SMC layer. Furthermore, M2 macrophages and contractile SMCs are known to act synergistically to promote the formation of a stable endothelial monolayer, which is essential for sustained antithrombogenic function. In this context, markers such as CD86/CD206 for macrophage polarization, SM22α and osteopontin for SMC phenotypic switching, and E-selectin for endothelial activation, as well as functional assays including nitric oxide production, would provide a more comprehensive evaluation of these coordinated processes.
Second, endothelialization in this study was assessed mainly through established marker expression without comprehensive functional validation. While additional functional assays and advanced experimental models—such as biomimetic flow systems, long-term culture, and in vivo functional studies—would allow for a more detailed investigation of immune–mechanical interactions and endothelial functionality, these aspects were beyond the primary objectives of the present work. Importantly, although these mechanistic pathways were not directly evaluated, the in vivo biocompatibility and calcification findings provide supportive, albeit indirect, evidence of a favorable host response and tissue integration. The observed reduction in calcification and enhanced cellular incorporation suggest the establishment of a microenvironment that may be permissive to macrophage-mediated modulation and subsequent SMC and endothelial organization.
Finally, given that vascular remodeling and endothelialization are temporally regulated, multicellular processes involving coordinated interactions among macrophages, smooth muscle cells, and endothelial cells, the present findings should be interpreted as reflecting an early stage of tissue development rather than a fully matured or functionally complete tissue. Future studies incorporating targeted analyses of macrophage polarization, SMC contractility, and endothelial functionality under physiologically relevant conditions will be essential to further elucidate these mechanisms.
In summary, our dual enzyme treatment significantly enhanced scaffold biocompatibility and hADSC-mediated recellularization. Vimentin and fibronectin expression confirmed interstitial-like cell phenotypes and ECM remodeling, while CD31 and vWF expression with phalloidin co-localization indicated successful endothelial differentiation and cytoskeletal organization. In vivo, recellularized biocompatible scaffolds exhibited increased cell infiltration and markedly reduced calcification compared to GA-fixed controls. Our hADSC-based monoculture recellularization, combined with optimized xenoantigen removal, effectively supports functional repopulation and reduces calcification in porcine pericardial scaffolds. This approach offers a simplified and scalable path toward developing next-generation, recellularized tissue-engineered heart valves.