Advanced Biomaterials for Restorative Dentistry: From Biocompatibility to Bioactive and Smart Materials
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Literature Search Strategy
2.3. Inclusion and Exclusion Criteria
2.4. Data Extraction and Synthesis
3. Results
3.1. Resin-Based Composite Biomaterials
3.2. Bioactive Restorative Materials
3.3. Dental Ceramics and Hybrid Materials
3.4. CAD–CAM Materials in Digital Restorative Dentistry
3.5. Nanostructured and Smart Dental Biomaterials, Digital Technologies and Artificial Intelligence
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Woźniak-Budych, M.J.; Staszak, M.; Staszak, K. A Critical Review of Dental Biomaterials with an Emphasis on Biocompatibility. Dent. Med. Probl. 2023, 60, 709–739. [Google Scholar] [CrossRef]
- Abozaid, D.; Azab, A.; Bahnsawy, M.A.; Eldebawy, M.; Ayad, A.; Soomro, R.; Elwakeel, E.; Mohamed, M.A. Bioactive restorative materials in dentistry: A comprehensive review of mechanisms, clinical applications, and future directions. Odontology 2025. advance online publication. [Google Scholar] [CrossRef] [PubMed]
- Musani, A.; Sultana, M.; Ihsan, M.; Rizwanullah. Advances in Bioactive restorative materials for Dental Restorations: A Comprehensive Review of Mechanisms, Applications, and Future Directions. Am. J. Biomed. Sci. Res. 2025, 27, 376–382. [Google Scholar] [CrossRef]
- Balhaddad, A.A.; Melo, M.A.S.; Natto, Z.S. Editorial: Innovative Dental Biomaterials for Advancing Oral Health Care. Front. Dent. Med. 2025, 6, 1643992. [Google Scholar] [CrossRef]
- Sindi, R.A.; Alanozi, W.K.M.; Salloum, R.S.B.; Alqutub, S.M.; Alharthi, G.A.; Madkhali, A.M.; Althagafi, A.I.; Almosallam, A.A.; Banjar, A.T.; Dwead, R.M.A.; et al. Clinical Performance of Modern Dental Biomaterials: A Systematic Review of Aesthetic and Functional Restoration. Rev. Diabet. Stud. 2024, 20, 686–693. [Google Scholar] [CrossRef]
- Zoniou, A.K.; Antoniadou, M.; Saridou, S. Selection of Resin-Based Dental Restorative Materials: A Pilot Study on Professional Characteristics, Knowledge, and Selection Criteria. Appl. Sci. 2025, 15, 7987. [Google Scholar] [CrossRef]
- Kunert, M.; Piwonski, I.; Hardan, L.; Bourgi, R.; Sauro, S.; Inchingolo, F.; Lukomska-Szymanska, M. Dentine remineralisation induced by “bioactive” materials through mineral deposition: An in vitro study. Nanomaterials 2024, 14, 274. [Google Scholar] [CrossRef]
- Fatima, N.; Veg, E.; Khan, T. A Brief Review on the Exploration of Nanocomposites and Their Properties Through Computational Methods for Biological Activity Evaluation. Mater. Proc. 2025, 25, 1. [Google Scholar] [CrossRef]
- Selim, S.; Abdelghany, T.M.; Almuhayawi, M.S.; Nagshabandi, M.K.; Tarabulsi, M.K.; Elamir, M.Y.M.; Alharbi, A.A.; Al Jaouni, S.K. Biosynthesis and activity of Zn-MnO nanocomposite in vitro with molecular docking studies against multidrug resistance bacteria and inflammatory activators. Sci. Rep. 2025, 15, 2032. [Google Scholar] [CrossRef]
- Pathak, G.; Mangla, S.; Gupta, G.K.; Bhan, V.; Kapoor, R.K. Toxicological assessment and risk management of nanoparticles mediated composite materials—Critical review: State of the art. Discov. Polym. 2025, 2, 12. [Google Scholar] [CrossRef]
- Matsui, N.; Maesako, M.; Alkhazaleh, A.; Irie, M.; Tsujimoto, A. Correlation Between Polymerization Shrinkage and Filler Content for Universal Shade Flowable Resin-Based Composites. J. Funct. Biomater. 2025, 16, 155. [Google Scholar] [CrossRef]
- Vouvoudi, E.C. Overviews on the Progress of Flowable Dental Polymeric Composites: Their Composition, Polymerization Process, Flowability and Radiopacity Aspects. Polymers 2022, 14, 4182. [Google Scholar] [CrossRef]
- Benelli, E.M.; Serra, M.C.; Rodrigues, A.L., Jr.; Cury, J.A. In situ anticariogenic potential of glass ionomer cement. Caries Res. 1993, 27, 280–284. [Google Scholar] [CrossRef]
- Hench, L.L. The Story of Bioglass®. J. Mater. Sci. Mater. Med. 2006, 17, 967–978. [Google Scholar] [CrossRef]
- Jones, J.R. Review of Bioactive Glass: From Hench to Hybrids. Acta Biomater. 2013, 9, 4457–4486. [Google Scholar] [CrossRef]
- Buzalaf, M.A.R.; Hannas, A.R.; Kato, M.T. Saliva and Dental Erosion. J. Appl. Oral Sci. 2012, 20, 493–502. [Google Scholar] [CrossRef]
- Camilleri, J. Mineral Trioxide Aggregate: Present and Future Developments. Endod. Top. 2015, 32, 31–46. [Google Scholar] [CrossRef]
- Gonçalves, F.; Silva, L.S.T.; Roschel, J.N.; de Souza, G.; Campos, L.P.M.; Varca, G.H.; Parra, D.; Perez, M.A.; Gordilho, A.C.; Brandt, W.C.; et al. Antibacterial Resin Composites with Sustained Chlorhexidine Release: One-Year In Vitro Study. Pharmaceutics 2025, 17, 1144. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Jiang, J.; Huang, Z.; Ma, X.; Shen, T.; Pan, J.; Bi, Z. Smart biomaterials in restorative dentistry: Recent advances and future perspectives. Mater. Today Bio 2025, 35, 102349. [Google Scholar] [CrossRef]
- Alhussein, A.; Alsahafi, R.; Balhaddad, A.A.; Mokeem, L.; Schneider, A.; Jabra-Rizk, M.-A.; Masri, R.; Hack, G.D.; Oates, T.W.; Sun, J.; et al. Novel bioactive nanocomposites containing calcium fluoride and calcium phosphate with antibacterial and low-shrinkage-stress capabilities to inhibit dental caries. Bioengineering 2023, 10, 991. [Google Scholar] [CrossRef] [PubMed]
- Ferracane, J.L. A Historical Perspective on Dental Composite Restorative Materials. J. Funct. Biomater. 2024, 15, 173. [Google Scholar] [CrossRef]
- Fugolin, A.P.; Pfeifer, C.S. Engineering a new generation of thermoset self-healing polymers based on intrinsic approaches. JADA Found. Sci. 2022, 1, 100014. [Google Scholar] [CrossRef]
- Chen, L.; Suh, B.I.; Yang, J. Antibacterial Dental Restorative Materials: A Review. Am. J. Dent. 2018, 31, 6B–12B. [Google Scholar] [PubMed]
- Varghese, E.J.; Sihivahanan, D.; Venkatesh, K.V. Development of novel antimicrobial dental composite resin with nano cerium oxide fillers. Int. J. Biomater. 2022, 2022, 3912290. [Google Scholar] [CrossRef]
- Zhang, Y.; Kelly, J.R. Dental Ceramics for Restoration and Metal Veneering. Dent. Clin. N. Am. 2017, 61, 797–819. [Google Scholar] [CrossRef] [PubMed]
- Sailer, I.; Makarov, N.A.; Thoma, D.S.; Zwahlen, M.; Pjetursson, B.E. All-Ceramic or Metal–Ceramic Tooth-Supported Fixed Dental Prostheses? A Systematic Review of the Survival and Complication Rates. Dent. Mater. 2015, 31, 603–623. [Google Scholar] [CrossRef]
- Coldea, A.; Swain, M.V.; Thiel, N. Mechanical Properties of Polymer-Infiltrated-Ceramic-Network Materials. Dent. Mater. 2013, 29, 419–426. [Google Scholar] [CrossRef]
- Conti, G.; Veneri, F.; Amadori, F.; Garzoni, A.; Majorana, A.; Bardellini, E. Evaluation of Antibacterial Activity of a Bioactive Restorative Material Versus a Glass-Ionomer Cement on Streptococcus mutans: In Vitro Study. Dent. J. 2023, 11, 149. [Google Scholar] [CrossRef]
- Bourgi, R.; Doumandji, Z.; Cuevas-Suárez, C.E.; Ben Ammar, T.; Laporte, C.; Kharouf, N.; Haikel, Y. Exploring the Role of Nanoparticles in Dental Materials: A Comprehensive Review. Coatings 2025, 15, 33. [Google Scholar] [CrossRef]
- Ferracane, J.L. Resin composite—State of the art. Dent. Mater. 2011, 27, 29–38. [Google Scholar] [CrossRef]
- Chen, M.-H. Update on Dental Nanocomposites. J. Dent. Res. 2010, 89, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Moreau, J.L.; Sun, L.; Chow, L.C. Nanocomposite Containing Amorphous Calcium Phosphate Nanoparticles for Caries Inhibition. Dent. Mater. 2011, 27, 762–769. [Google Scholar] [CrossRef]
- Ruse, N.D.; Sadoun, M.J. Resin-composite blocks for dental CAD/CAM applications. J. Dent. Res. 2014, 93, 1232–1234. [Google Scholar] [CrossRef]
- Abuljadayel, R.; Aljadani, N.; Almutairi, H.; Turkistani, A. Effect of Antibacterial Agents on Dentin Bond Strength of Bioactive Restorative Materials. Polymers 2023, 15, 2612. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, C.; Shan, Z. Application of Artificial Intelligence in Orthodontics: Current State and Future Perspectives. Healthcare 2023, 11, 2760. [Google Scholar] [CrossRef]
- Ilie, N.; Hickel, R. Resin Composite Restorative Materials. Aust. Dent. J. 2011, 56, 59–66. [Google Scholar] [CrossRef]
- Mitwalli, H.; Balhaddad, A.A.; AlSahafi, R.; Oates, T.W.; Melo, M.A.S.; Xu, H.H.K.; Weir, M.D. Novel CaF2 nanocomposites with antibacterial function and fluoride and calcium ion release to inhibit oral biofilm and protect teeth. J. Funct. Biomater. 2020, 11, 56. [Google Scholar] [CrossRef]
- Zhang, Y.; Lawn, B.R. Novel Zirconia Materials in Dentistry. J. Dent. Res. 2018, 97, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Mainjot, A.; Dupont, N.; Oudkerk, J.; Dewael, T.; Sadoun, M. From Artisanal to CAD–CAM Blocks. J. Dent. Res. 2016, 95, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Almutairi, N.; Alhussein, A.; Alenizy, M.; Ba-Armah, I.; Alqarni, H.; Oates, T.W.; Masri, R.; Hack, G.D.; Sun, J.; Weir, M.D.; et al. Novel Bioactive Resin Coating with Calcium Phosphate Nanoparticles for Antibacterial and Remineralization Abilities to Combat Tooth Root Caries. Int. J. Mol. Sci. 2025, 26, 2490. [Google Scholar] [CrossRef]
- Ramburrun, P.; Pringle, N.A.; Dube, A.; Adam, R.Z.; D’Souza, S.; Aucamp, M. Recent advances in the development of antimicrobial and antifouling biocompatible materials for dental applications. Materials 2021, 14, 3167. [Google Scholar] [CrossRef] [PubMed]
- Marsh, P.D. Contemporary Perspective on Plaque Control. Br. Dent. J. 2012, 212, 601–606. [Google Scholar] [CrossRef] [PubMed]
- Salazar, A.; Anderson, N.; Stansbury, J. Formulating Mechanically Robust Composite Restorative Materials for High Performance. J. Funct. Biomater. 2025, 16, 101. [Google Scholar] [CrossRef] [PubMed]
- Rokaya, D.; Al Jaghsi, A.; Jagtap, R.; Srinameepong, V. Artificial Intelligence in Dentistry and Dental Biomaterials. Front. Dent. Med. 2024, 5, 1525505. [Google Scholar] [CrossRef]


| Material Type | Main Composition | Key Advantages | Limitations | Typical Clinical Applications | Clinical Relevance | References |
|---|---|---|---|---|---|---|
| Amalgam | Silver, tin, copper alloy with mercury | High durability, good compressive strength, long clinical history | Poor aesthetics, environmental concerns, limited adhesion | Posterior restorations (historically common) | Suitable for posterior restorations where durability is prioritized over aesthetics, although its use has declined due to aesthetic and environmental concerns | [1,5] |
| Glass Ionomer Cement (GIC) | Fluoroaluminosilicate glass + polyacrylic acid | Fluoride release, chemical adhesion to tooth structure, biocompatibility | Lower mechanical strength, wear susceptibility | Cervical lesions, pediatric dentistry, temporary restorations | Recommended for cervical lesions and pediatric applications due to fluoride release and chemical adhesion, particularly in low-stress areas | [1,13] |
| Resin-Based Composites | Organic resin matrix (Bis-GMA, UDMA) + inorganic fillers | Excellent aesthetics, strong adhesion, minimally invasive preparation | Polymerization shrinkage, technique sensitivity | Direct restorations in anterior and posterior teeth | Preferred for aesthetic and minimally invasive restorations; widely used in both anterior and posterior regions depending on load conditions | [6,11] |
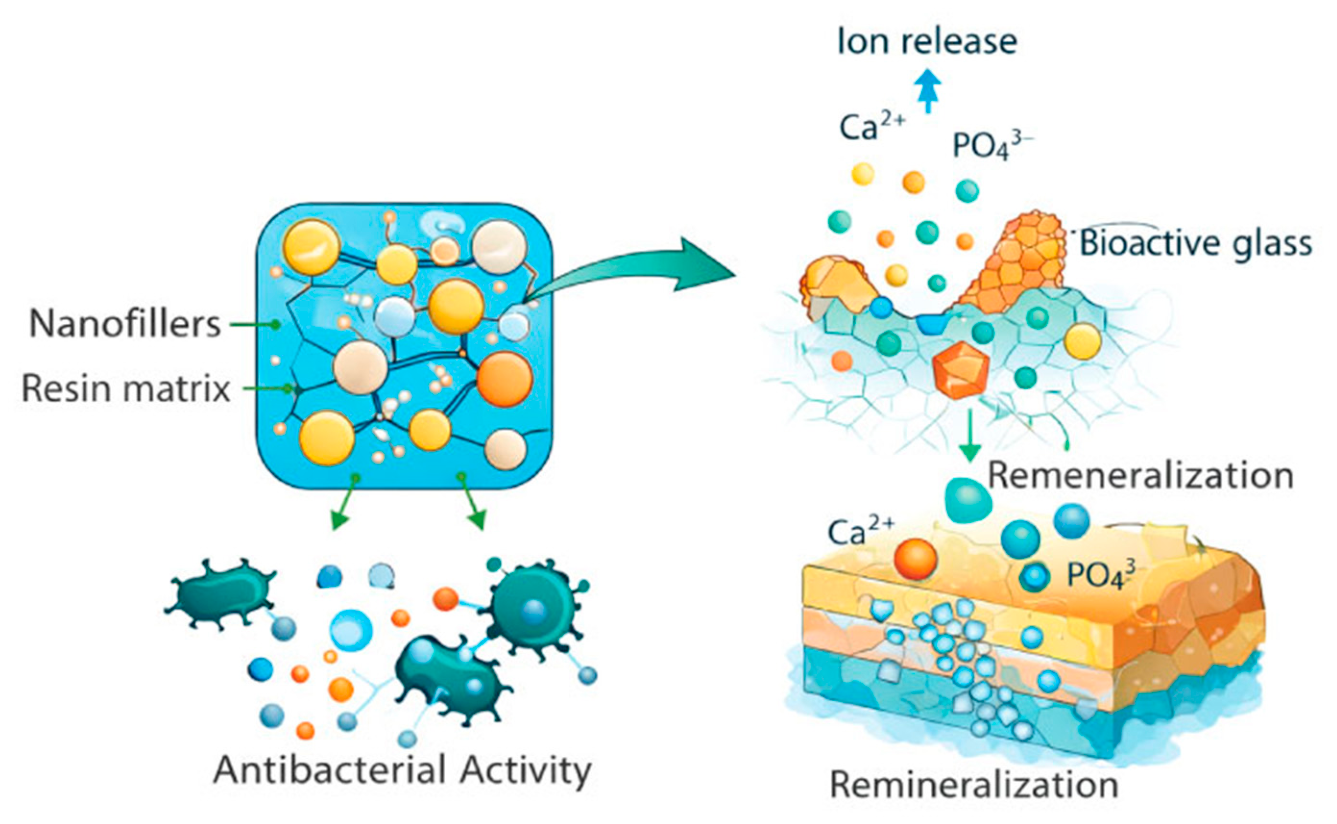
| Nanocomposites | Resin matrix with nanoscale filler particles | Improved polishability, enhanced mechanical strength, better wear resistance | Technique sensitive, higher cost | Highly aesthetic restorations | Indicated for restorations requiring superior aesthetics and improved surface properties, particularly in visible areas | [8,13] |
| Bioactive restorative materials | Calcium silicates, bioactive glass, ion-releasing components | Promote remineralization, antimicrobial potential, improved tissue interaction | Limited long-term clinical data | Regenerative and minimally invasive dentistry | Promising for preventive and remineralizing strategies, especially in patients with high caries risk, although long-term clinical validation is still needed | [2,3,14] |
| Dental Ceramics | Feldspathic porcelain, lithium disilicate, zirconia | Excellent aesthetics, high strength, biocompatibility | Brittle behavior, higher cost, laboratory procedures required | Crowns, veneers, inlays/onlays | Ideal for indirect restorations requiring high aesthetics and mechanical strength, particularly in long-term rehabilitations | [19,20] |
| CAD–CAM Materials | Hybrid ceramics, resin nanoceramics, zirconia blocks | High precision, digital workflow compatibility, reproducibility | Equipment cost, learning curve | Indirect restorations fabricated digitally | Recommended for digitally fabricated restorations with high precision and reproducibility, especially in modern digital workflows | [6,8] |
| Material Type | Main Components | Bioactive Mechanism | Clinical Applications | Key References |
|---|---|---|---|---|
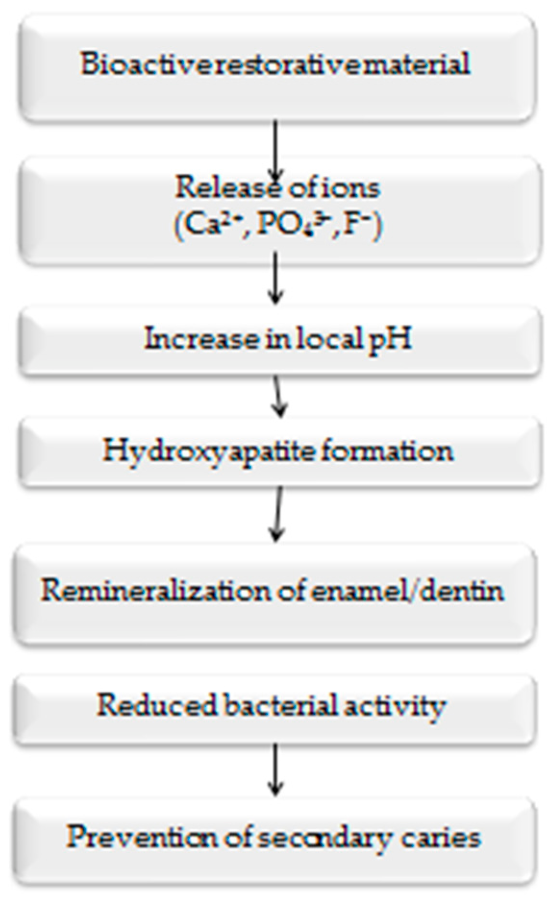
| Bioactive Glass-Based Materials | SiO2–CaO–Na2O–P2O5 glass particles | Release of Ca2+ and PO43− ions leading to hydroxyapatite formation | Remineralizing restorative materials, dentin repair | [11,12] |
| Calcium Silicate Cements (e.g., MTA) | Tricalcium silicate, dicalcium silicate, calcium aluminate | Induce mineralized tissue formation and dentin bridge development | Pulp capping, root-end filling, endodontic repair | [14,15,16,17] |
| Fluoride-Releasing Restorative Materials | Glass ionomer matrix or resin-modified composites with fluoride fillers | Continuous fluoride ion release enhancing enamel remineralization | Cervical lesions, pediatric dentistry | [13] |
| Ion-Releasing Composite Resins | Resin matrix with calcium phosphate or bioactive glass fillers | Release of Ca2+ and PO43− ions to promote remineralization | Direct restorations with preventive effect | [15,16,17] |
| Smart Bioactive restorative materials | pH-responsive nanoparticles and ion-releasing fillers | Stimulus-responsive ion release under acidic conditions | Prevention of secondary caries, minimally invasive dentistry | [16,17,18] |
| Material Type | Main Composition | Mechanical Properties | Advantages | Clinical Applications | References |
|---|---|---|---|---|---|
| Feldspathic Ceramics | Silica-based glass ceramics | Moderate strength, high translucency | Excellent aesthetics and optical properties | Veneers, anterior restorations | [20] |
| Lithium Disilicate Ceramics | Lithium disilicate glass-ceramic (Li2Si2O5) | Flexural strength ~360–400 MPa | High aesthetics, good durability | Crowns, veneers, inlays/onlays | [20,23] |
| Zirconia Ceramics (Y-TZP) | Yttria-stabilized zirconium dioxide | Very high strength (>900 MPa) | Exceptional fracture resistance | Posterior crowns, bridges, implant prostheses | [19,21] |
| Hybrid Ceramics (PICN) | Polymer-infiltrated ceramic network | Improved elasticity and fracture resistance | Reduced brittleness, better shock absorption | CAD–CAM restorations | [22] |
| Material | Flexural Strength (MPa) | Elastic Modulus (GPa) | Fracture Toughness (MPa·m½) | References |
|---|---|---|---|---|
| Feldspathic ceramic | 60–120 | 60–70 | 0.7–1.0 | [20] |
| Lithium disilicate | 360–400 | 90–100 | 2.5–3.0 | [20,21] |
| Zirconia (Y-TZP) | 900–1200 | 200–210 | 5–10 | [19,21] |
| Hybrid ceramics (PICN) | 150–200 | 25–35 | 1.5–2.0 | [22] |
| Resin nanoceramic | 150–250 | 12–15 | 1.2–1.8 | [23] |
| Technology/Material Type | Main Characteristics | Advantages | Clinical Longevity Evidence | Clinical Relevance | References |
|---|---|---|---|---|---|
| Nanostructured Resin Composites | Resin matrix with nanoscale fillers (silica, zirconia nanoparticles) | Improved polishability, wear resistance, aesthetic integration | Long-term clinical studies report survival rates comparable to conventional composites, with improved surface stability | Suitable for aesthetic restorations requiring enhanced surface quality and wear resistance, particularly in anterior and moderate-load posterior regions | [24,31] |
| Smart Bioactive Restorative Materials | pH-responsive materials releasing Ca2+, PO43− or F− ions | Promote remineralization, reduce secondary caries risk, dynamic response to oral environment | Limited long-term clinical data; promising in vitro and short-term clinical studies | Recommended for patients with high caries risk or in minimally invasive approaches, where preventive and therapeutic effects are desired | [26,32] |
| Antimicrobial Nanocomposite Materials | Incorporation of antibacterial nanoparticles (Ag, ZnO) or antimicrobial monomers | Inhibition of bacterial adhesion and biofilm formation, prevention of recurrent caries | Mostly experimental or short-term clinical studies | Promising for reducing bacterial colonization and secondary caries, although clinical validation remains limited | [27,33] |
| CAD–CAM Restorative Materials | Digitally fabricated ceramics, hybrid ceramics and resin nanoceramics | High precision, reproducibility, improved marginal adaptation | Long-term clinical data available for zirconia and lithium disilicate restorations | Ideal for indirect restorations requiring high precision and durability, especially in digitally guided workflows | [23,34] |
| Artificial Intelligence in Biomaterial Design | Machine learning algorithms for predicting material performance and optimizing composition | Accelerated biomaterial development, predictive modeling of mechanical and biological properties | Emerging research field; limited direct clinical evidence | Useful for future personalized dentistry and predictive material selection, but currently limited to research and early clinical applications | [35,36] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Albu, M.C.; Ștefănescu, C.L.; Murineanu, R.M.; Grigorian, M.; Sachelarie, L.; Zaharia, A.; Hurjui, L.L.; Caraiane, A. Advanced Biomaterials for Restorative Dentistry: From Biocompatibility to Bioactive and Smart Materials. Bioengineering 2026, 13, 542. https://doi.org/10.3390/bioengineering13050542
Albu MC, Ștefănescu CL, Murineanu RM, Grigorian M, Sachelarie L, Zaharia A, Hurjui LL, Caraiane A. Advanced Biomaterials for Restorative Dentistry: From Biocompatibility to Bioactive and Smart Materials. Bioengineering. 2026; 13(5):542. https://doi.org/10.3390/bioengineering13050542
Chicago/Turabian StyleAlbu, Maria Claudia, Corina Laura Ștefănescu, Rodica Maria Murineanu, Mircea Grigorian, Liliana Sachelarie, Agripina Zaharia, Loredana Liliana Hurjui, and Aureliana Caraiane. 2026. "Advanced Biomaterials for Restorative Dentistry: From Biocompatibility to Bioactive and Smart Materials" Bioengineering 13, no. 5: 542. https://doi.org/10.3390/bioengineering13050542
APA StyleAlbu, M. C., Ștefănescu, C. L., Murineanu, R. M., Grigorian, M., Sachelarie, L., Zaharia, A., Hurjui, L. L., & Caraiane, A. (2026). Advanced Biomaterials for Restorative Dentistry: From Biocompatibility to Bioactive and Smart Materials. Bioengineering, 13(5), 542. https://doi.org/10.3390/bioengineering13050542







