Robot-Assisted Placement of Thoracic Carbon-Fiber-Reinforced Polyetheretherketone (CFR-Peek) Pedicle Screws in the Cervical Spine for Giant Cell Tumor: Technical Note
Abstract
1. Introduction
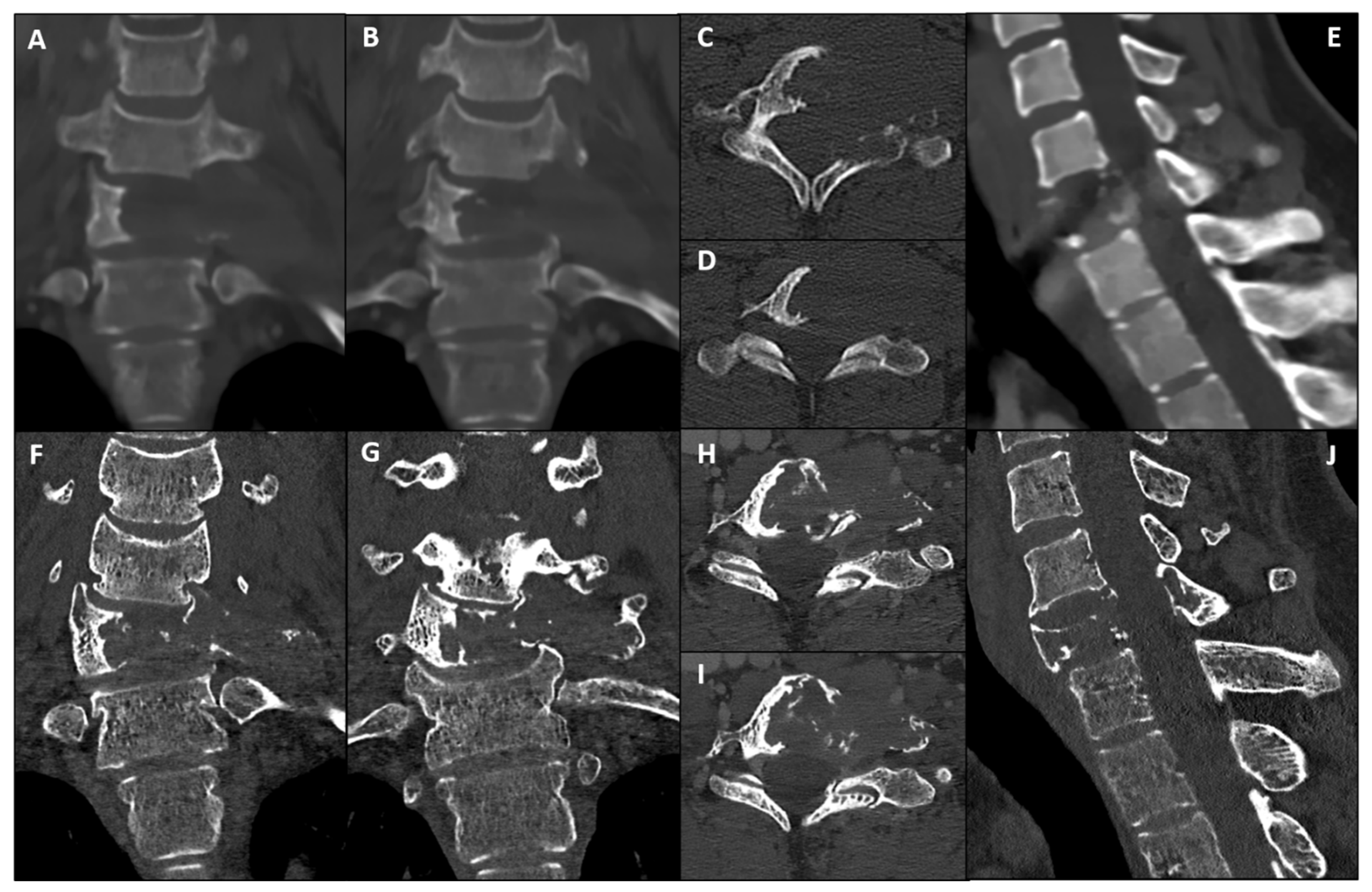
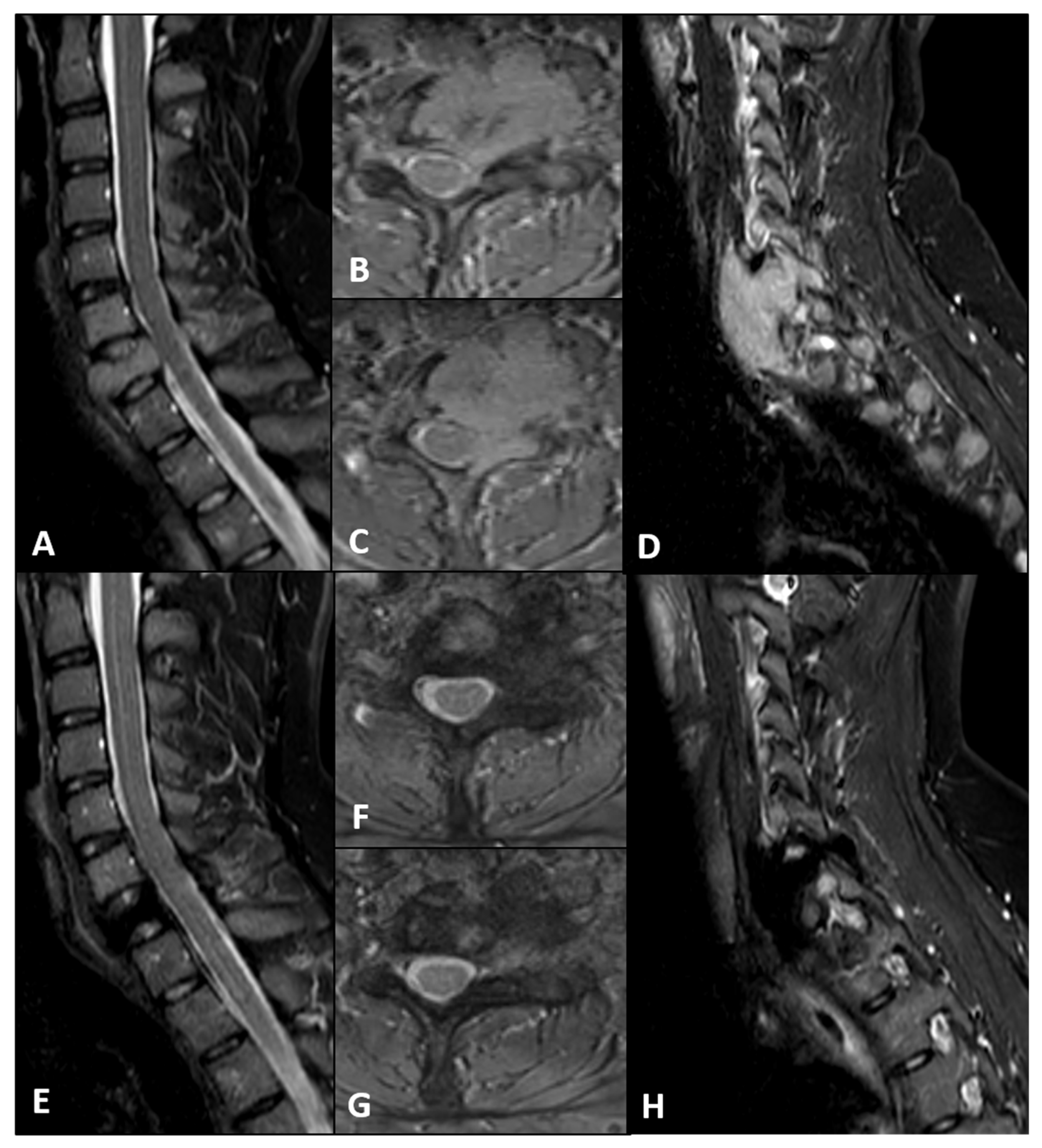
2. Case Presentation
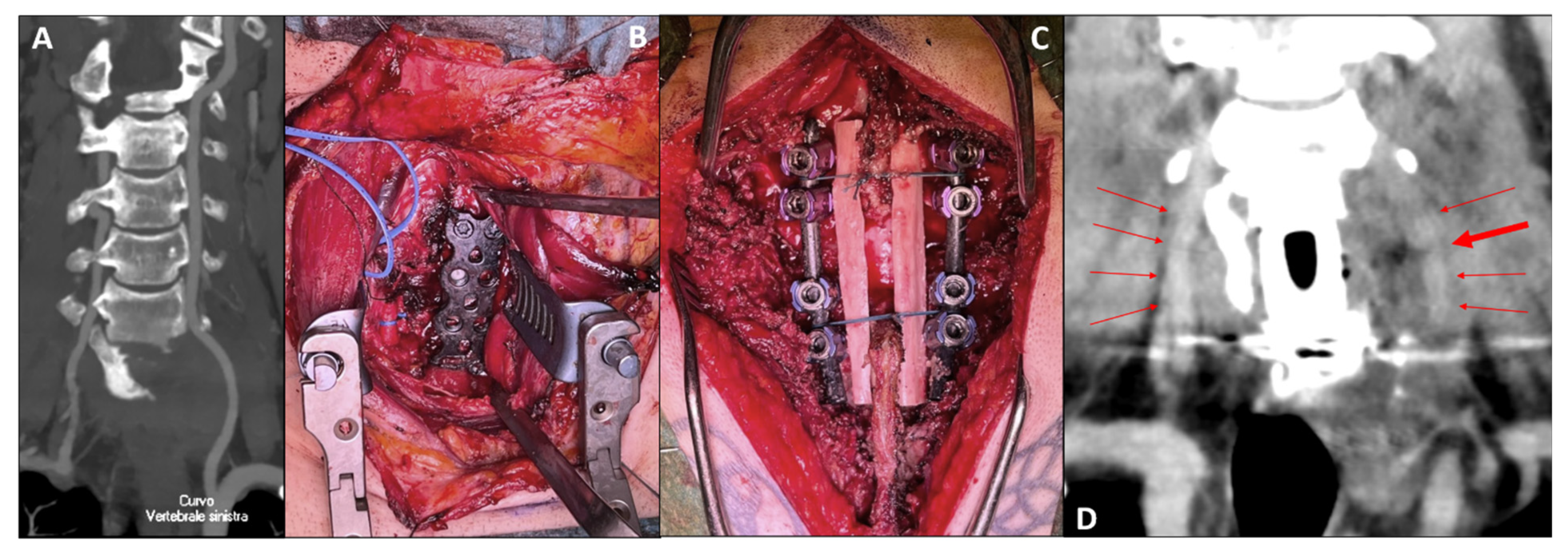
3. Surgical Technique
3.1. Anterior Step: Corpectomy and VA Repair
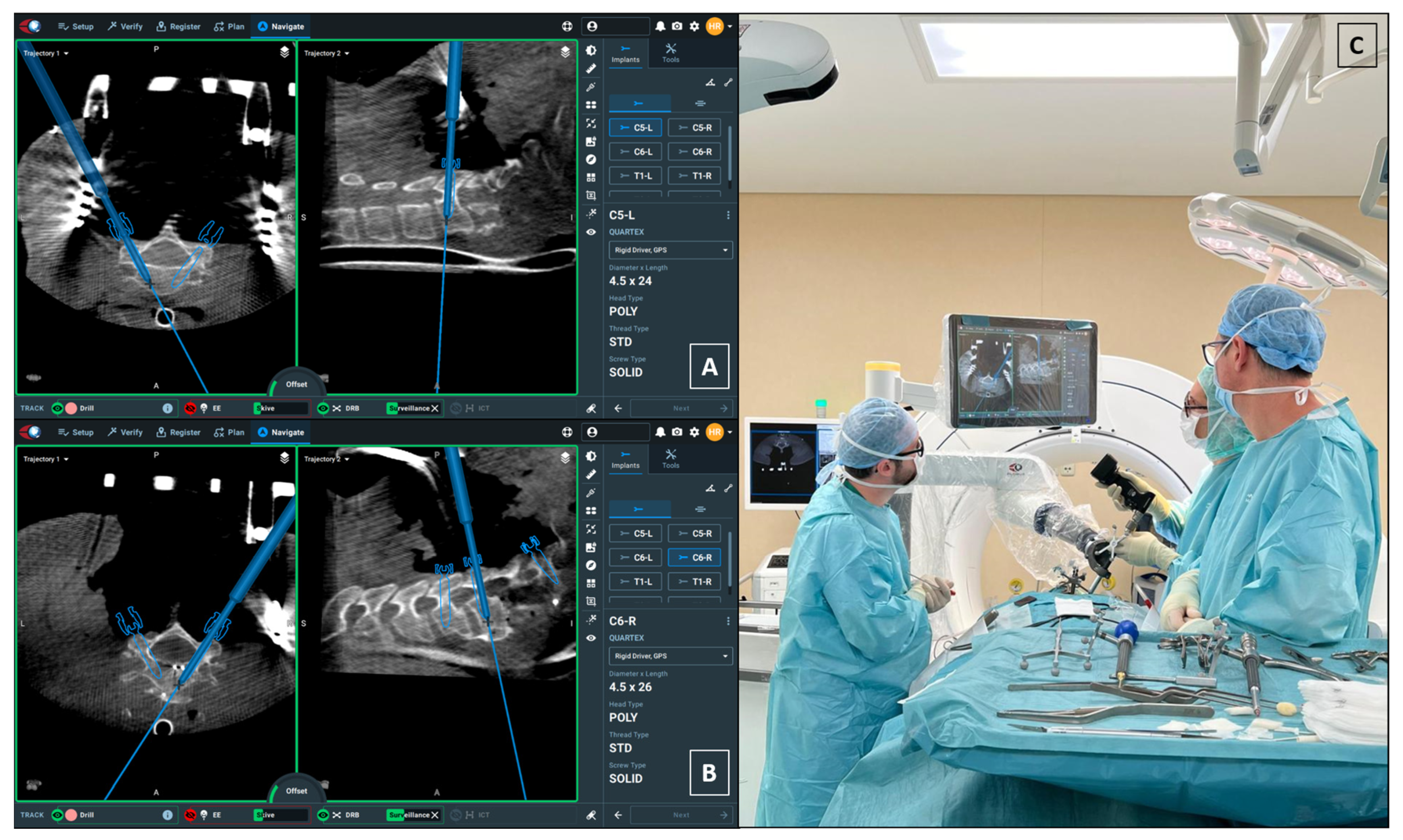
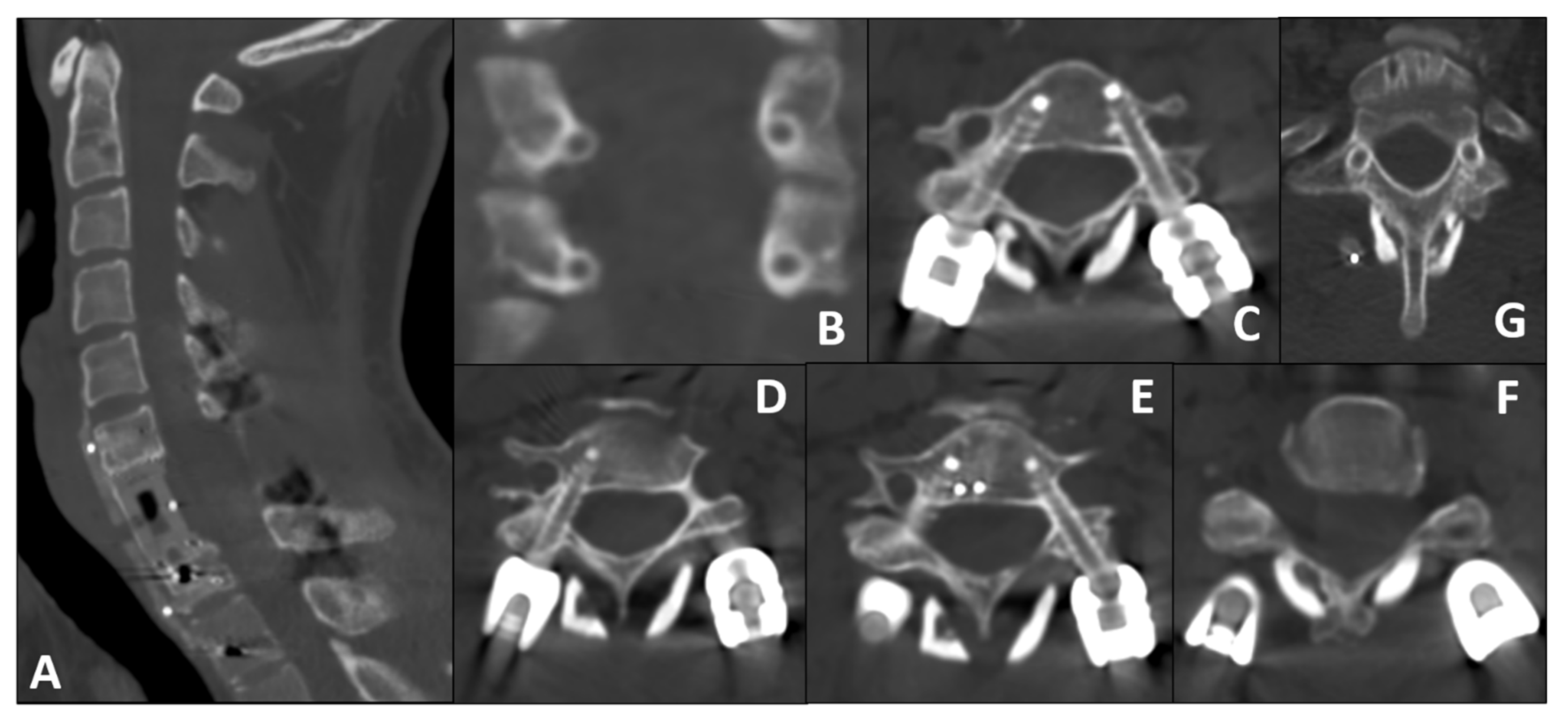
3.2. Posterior Step: Robot-Assisted Instrumentation
- Left Side (Repaired VA): The larger pedicle diameter allowed for a standard transpedicular trajectory.
- Right Side (Intact VA): Given the small pedicle diameter and the need to minimize lateral breach (risking injury to the only patent VA), we planned a small, controlled medial cortical violation, with limited encroachment into the canal, facilitated by robotic navigation. This choice is consistent with anatomical/clinical observations indicating higher vertebral artery risk with lateral breaches and short-term tolerance of limited medial violations [20,21,22].
4. Discussion
4.1. CFR-PEEK Instrumentation: Potential Advantages
4.2. Robotic Planning Under Hardware Constraints: Approach, Safety, and Alternatives
4.3. Multimodal and Complication Management
4.4. Limitations, Feasibility, and Applicability
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boriani, S.; Weinstein, J.N.; Biagini, R. Primary bone tumors of the spine: Terminology and surgical staging. Spine 1997, 22, 1036–1044. [Google Scholar] [CrossRef] [PubMed]
- Turcotte, R.E. Giant cell tumor of bone. Orthop. Clin. N. Am. 2006, 37, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Boriani, S.; Bandiera, S.; Casadei, R.; Boriani, L.; Donthineni, R.; Gasbarrini, A.; Pignotti, E.; Biagini, R.; Schwab, J.H. Giant cell tumor of the mobile spine: A review of 49 cases. Spine 2012, 37, E37–E45. [Google Scholar] [CrossRef]
- Goldschlager, T.; Dea, N.; Boyd, M.; Reynolds, J.; Patel, S.; Rhines, L.D.; Mendel, E.; Pacheco, M.; Ramos, E.; Mattei, T.A.; et al. Giant cell tumors of the spine: Has denosumab changed the treatment paradigm? J. Neurosurg. Spine 2015, 22, 526–533. [Google Scholar] [CrossRef]
- Chawla, S.; Henshaw, R.; Seeger, L.; Choy, E.; Blay, J.; Ferrari, S.; Kroep, J.; Grimer, R.; Reichardt, P.; Rutkowski, P.; et al. Safety and efficacy of denosumab for adults and skeletally mature adolescents with giant cell tumour of bone: Interim analysis of an open-label, parallel-group, phase 2 study. Lancet Oncol. 2013, 14, 901–908. [Google Scholar] [CrossRef]
- Rutkowski, P.; Ferrari, S.; Grimer, R.J.; Stalley, P.D.; Dijkstra, S.P.D.; Pienkowski, A.; Vaz, G.; Wunder, J.S.; Seeger, L.L.; Feng, A.; et al. Surgical downstaging in an open-label phase II trial of denosumab in patients with giant cell tumor of bone. Ann. Surg. Oncol. 2015, 22, 2860–2868. [Google Scholar] [CrossRef]
- Kumar, N.; Kumar, P.; Zihui, G.L.; Seow, L.; Liang, S.; Hui, S.J.; Parihar, R.; Hallinan, J.; Vellayappan, B.; Tan, J.H.J. Nonmetallic Carbon Fiber-Reinforced Polyetheretherketone Implants Vs Titanium Implants: Analysis of Clinical Outcomes and Influence on Postoperative Radiotherapy Planning in Metastatic Spine Tumor Surgery. Int. J. Spine Surg. 2024, 18, 603–610. [Google Scholar] [CrossRef]
- Krätzig, T.; Mende, K.C.; Mohme, M.; Kniep, H.; Dreimann, M.; Stangenberg, M.; Westphal, M.; Gauer, T.; Eicker, S.O. Carbon fiber-reinforced PEEK versus titanium implants: An in vitro comparison of susceptibility artifacts in CT and MR imaging. Neurosurg. Rev. 2021, 44, 2163–2170. [Google Scholar] [CrossRef]
- Ringel, F.; Ryang, Y.M.; Kirschke, J.S.; Müller, B.S.; Wilkens, J.J.; Brodard, J.; Combs, S.E.; Meyer, B. Radiolucent Carbon Fiber-Reinforced Pedicle Screws for Treatment of Spinal Tumors: Advantages for Radiation Planning and Follow-Up Imaging. World Neurosurg. 2017, 105, 294–301. [Google Scholar] [CrossRef]
- Chhabra, A.M.; Snider, J.W.; Kole, A.J.; Stock, M.; Holtzman, A.L.; Press, R.; Wang, C.J.; Li, H.; Lin, H.; Shi, C.; et al. Proton Therapy for Spinal Tumors: A Consensus Statement From the Particle Therapy Cooperative Group. Int. J. Radiat. Oncol. Biol. Phys. 2024, 120, 1135–1148. [Google Scholar] [CrossRef] [PubMed]
- Boriani, S.; Tedesco, G.; Ming, L.; Ghermandi, R.; Amichetti, M.; Fossati, P.; Krengli, M.; Mavilla, L.; Gasbarrini, A. Carbon-fiber-reinforced PEEK fixation system in the treatment of spine tumors: A preliminary report. Eur. Spine J. 2018, 27, 874–881. [Google Scholar] [CrossRef]
- Tedesco, G.; Gasbarrini, A.; Bandiera, S.; Ghermandi, R.; Boriani, S. Composite PEEK/Carbon fiber implants can increase the effectiveness of radiotherapy in the management of spine tumors. J. Spine Surg. 2017, 3, 323–329, Erratum in J. Spine Surg. 2018, 4, 167. https://doi.org/10.21037/jss.2018.01.01.. [Google Scholar] [CrossRef] [PubMed]
- Zavras, A.G.; Schoenfeld, A.J.; Patt, J.C.; Munim, M.A.; Goodwin, C.R.; Goodwin, M.L.; Lo, S.L.; Redmond, K.J.; Tobert, D.G.; Shin, J.H.; et al. Attitudes and trends in the use of radiolucent spinal implants: A survey of the North American Spine Society section of spinal oncology. N. Am. Spine Soc. J. 2022, 10, 100105. [Google Scholar] [CrossRef]
- Ghermandi, R.; Tosini, G.; Lorenzi, A.; Griffoni, C.; La Barbera, L.; Girolami, M.; Pipola, V.; Barbanti Brodano, G.; Bandiera, S.; Terzi, S.; et al. Carbon Fiber-Reinforced PolyEtherEtherKetone (CFR-PEEK) Instrumentation in Degenerative Disease of Lumbar Spine: A Pilot Study. Bioengineering 2023, 10, 872. [Google Scholar] [CrossRef] [PubMed]
- Damante, M.; Wilson, S.B.; Ward, J.; Mensah-Arhin, K.; Gandhi, S.; Bardeesi, A.; Xu, D.; Palmer, J.; Elder, J.B.; Chakravarthy, V.B. Durability of carbon fiber-reinforced PEEK spinal instrumentation in spine oncology: A single-institution longitudinal experience. Neurosurg. Focus 2025, 58, E10. [Google Scholar] [CrossRef] [PubMed]
- Saluja, S.; Patil, S.; Vasudeva, N. Morphometric Analysis of Sub-axial Cervical Vertebrae and Its Surgical Implications. J. Clin. Diagn. Res. 2015, 9, AC01-4. [Google Scholar] [CrossRef]
- Abumi, K.; Shono, Y.; Ito, M.; Taneichi, H.; Kotani, Y.; Kaneda, K. Complications of pedicle screw fixation in reconstructive surgery of the cervical spine. Spine 2000, 25, 962–969. [Google Scholar] [CrossRef]
- Neo, M.; Sakamoto, T.; Fujibayashi, S.; Nakamura, T. The clinical risk of vertebral artery injury from cervical pedicle screws inserted in degenerative vertebrae. Spine 2005, 30, 2800–2805. [Google Scholar] [CrossRef]
- Cofano, F.; Marengo, N.; Colonna, S.; Rizzo, F.; Lacatena, F.; Garbossa, D. Cervical carbon fiber–reinforced PEEK pedicle screw fixation with 3D custom-made template guides for spinal oncology surgery: Case report. Front. Surg. 2026, 12, 1725931. [Google Scholar] [CrossRef]
- Gertzbein, S.D.; Robbins, S.E. Accuracy of pedicular screw placement in vivo. Spine 1990, 15, 11–14. [Google Scholar] [CrossRef]
- Boriani, S.; Cecchinato, R.; Cuzzocrea, F.; Bandiera, S.; Gambarotti, M.; Gasbarrini, A. Denosumab in the treatment of giant cell tumor of the spine. Preliminary report, review of the literature and protocol proposal. Eur. Spine J. 2020, 29, 257–271. [Google Scholar] [CrossRef] [PubMed]
- Lieberman, I.H.; Hardenbrook, M.A.; Wang, J.C.; Guyer, R.D. Assessment of pedicle screw placement accuracy, procedure time, and radiation exposure using a miniature robotic guidance system. J. Spinal Disord. Tech. 2012, 25, 241–248. [Google Scholar] [CrossRef]
- McKenzie, D.M.; Westrup, A.M.; O’Neal, C.M.; Lee, B.J.; Shi, H.H.; Dunn, I.F.; Snyder, L.A.; Smith, Z.A. Robotics in spine surgery: A systematic review. J. Clin. Neurosci. 2021, 89, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Karaikovic, E.E.; Daubs, M.D.; Madsen, R.W.; Gaines, R.W., Jr. Morphologic characteristics of human cervical pedicles. Spine 1997, 22, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Sundaresan, N.; Rosen, G.; Boriani, S. Surgical treatment of spinal tumors. Neurosurgery 1986, 19, 753–759. [Google Scholar]
- Roser, F.; Tatagiba, M.; Maier, G. Spinal robotics: Current applications and accuracy. Neurosurgery 2013, 72, 113–120. [Google Scholar] [CrossRef]
- Kantelhardt, S.R.; Martinez, R.; Baerwinkel, S.; Burger, R.; Giese, A.; Rohde, V. Perioperative course and accuracy of screw positioning in conventional, open robotic-guided and percutaneous robotic-guided, pedicle screw placement. Eur. Spine J. 2011, 20, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Lunardini, D.J.; Eskander, M.S.; Even, J.L.; Dunlap, J.T.; Chen, A.F.; Lee, J.Y.; Ward, T.W.; Kang, J.D.; Donaldson, W.F. Vertebral artery injuries in cervical spine surgery. Spine J. 2014, 14, 1520–1525. [Google Scholar] [CrossRef]
- Neo, M.; Fujibayashi, S.; Miyata, M.; Takemoto, M.; Nakamura, T. Vertebral artery injury during cervical spine surgery: A survey of more than 5600 operations. Spine 2008, 33, 779–785. [Google Scholar] [CrossRef]
- Kumar, N.; Hui, S.J.; Kumar, P.; Gabriel, L.Z.; Parihar, R.; Tan, J.H.; Yang, Y.O.; Ryang, Y.M. Carbon-fiber-reinforced polyetheretherketone instrumentation in metastatic spine tumor surgery: Technical considerations and potential pitfalls to avoid. Asian Spine J. 2025, 19, 836–846. [Google Scholar] [CrossRef]
- Perez-Roman, R.J.; Boddu, J.V.; Bashti, M.; Bryant, J.P.; Amadasu, E.; Gyedu, J.S.; Wang, M.Y. The Use of Carbon Fiber-Reinforced Instrumentation in Patients with Spinal Oncologic Tumors: A Systematic Review of Literature and Future Directions. World Neurosurg. 2023, 173, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Xu, G.; Zhou, X.; Qiu, J.; Lan, T.; Zhang, S.; Li, W. Combined preoperative denosumab and adjuvant microwave ablation for high-risk giant cell tumor of bone: A retrospective study in a single center. J. Orthop. Surg. Res. 2024, 19, 488. [Google Scholar] [CrossRef] [PubMed]
- Osterne, R.-L.-V.; Araújo, P.-M.; de Souza-Carvalho, A.-C.-G.; Cavalcante, R.-B.; Sant’Ana, E.; Nongueira, R.-L.-M. Intralesional corticosteroid injection as a treatment for central giant cell lesions of the jaws. Med. Oral Patol. Oral Cir. Bucal 2013, 18, e226–e232. [Google Scholar] [CrossRef]
- Thangaraj, R.; Grimer, R.J.; Carter, S.R.; Stirling, A.J.; Spilsbury, J.; Spooner, D. Giant cell tumour of the sacrum: A suggested algorithm for treatment. Eur. Spine J. 2010, 19, 1189–1194. [Google Scholar] [CrossRef] [PubMed]

| Level | Side | Pedicle Width (mm) | Screw Diameter (mm) | Mismatch (mm) |
|---|---|---|---|---|
| C5 | Right | 3.2–3.7 | 4.5 | +0.8–1.3 |
| C5 | Left | 3.7–4.0 | 4.5 | +0.5–0.8 |
| C6 | Right | 3.2–3.7 | 4.5 | +0.8–1.3 |
| C6 | Left | 3.7–4.0 | 4.5 | +0.5–0.8 |
| Level | Side | Intended Medial Encroachment | GR Grade | Revision Required |
|---|---|---|---|---|
| C5 | Right | Yes | C | No |
| C5 | Left | No | A | No |
| C6 | Right | Yes | C | No |
| C6 | Left | No | A | No |
| T1 | Right | No | A | No |
| T1 | Left | No | A | No |
| T2 | Right | No | A | No |
| T2 | Left | No | A | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Stucchi, E.; De Robertis, M.; Capo, G.; Baram, A.; De Gennaro Aquino, G.; Creatura, D.; Anselmi, L.; Fornari, M.; Pessina, F.; Brembilla, C. Robot-Assisted Placement of Thoracic Carbon-Fiber-Reinforced Polyetheretherketone (CFR-Peek) Pedicle Screws in the Cervical Spine for Giant Cell Tumor: Technical Note. Bioengineering 2026, 13, 361. https://doi.org/10.3390/bioengineering13030361
Stucchi E, De Robertis M, Capo G, Baram A, De Gennaro Aquino G, Creatura D, Anselmi L, Fornari M, Pessina F, Brembilla C. Robot-Assisted Placement of Thoracic Carbon-Fiber-Reinforced Polyetheretherketone (CFR-Peek) Pedicle Screws in the Cervical Spine for Giant Cell Tumor: Technical Note. Bioengineering. 2026; 13(3):361. https://doi.org/10.3390/bioengineering13030361
Chicago/Turabian StyleStucchi, Emanuele, Mario De Robertis, Gabriele Capo, Ali Baram, Giuseppe De Gennaro Aquino, Donato Creatura, Leonardo Anselmi, Maurizio Fornari, Federico Pessina, and Carlo Brembilla. 2026. "Robot-Assisted Placement of Thoracic Carbon-Fiber-Reinforced Polyetheretherketone (CFR-Peek) Pedicle Screws in the Cervical Spine for Giant Cell Tumor: Technical Note" Bioengineering 13, no. 3: 361. https://doi.org/10.3390/bioengineering13030361
APA StyleStucchi, E., De Robertis, M., Capo, G., Baram, A., De Gennaro Aquino, G., Creatura, D., Anselmi, L., Fornari, M., Pessina, F., & Brembilla, C. (2026). Robot-Assisted Placement of Thoracic Carbon-Fiber-Reinforced Polyetheretherketone (CFR-Peek) Pedicle Screws in the Cervical Spine for Giant Cell Tumor: Technical Note. Bioengineering, 13(3), 361. https://doi.org/10.3390/bioengineering13030361






