Echo Intensity Correction Method for Ultrasound Computed Tomography in Musculoskeletal Imaging
Abstract
1. Introduction
2. Materials and Methods
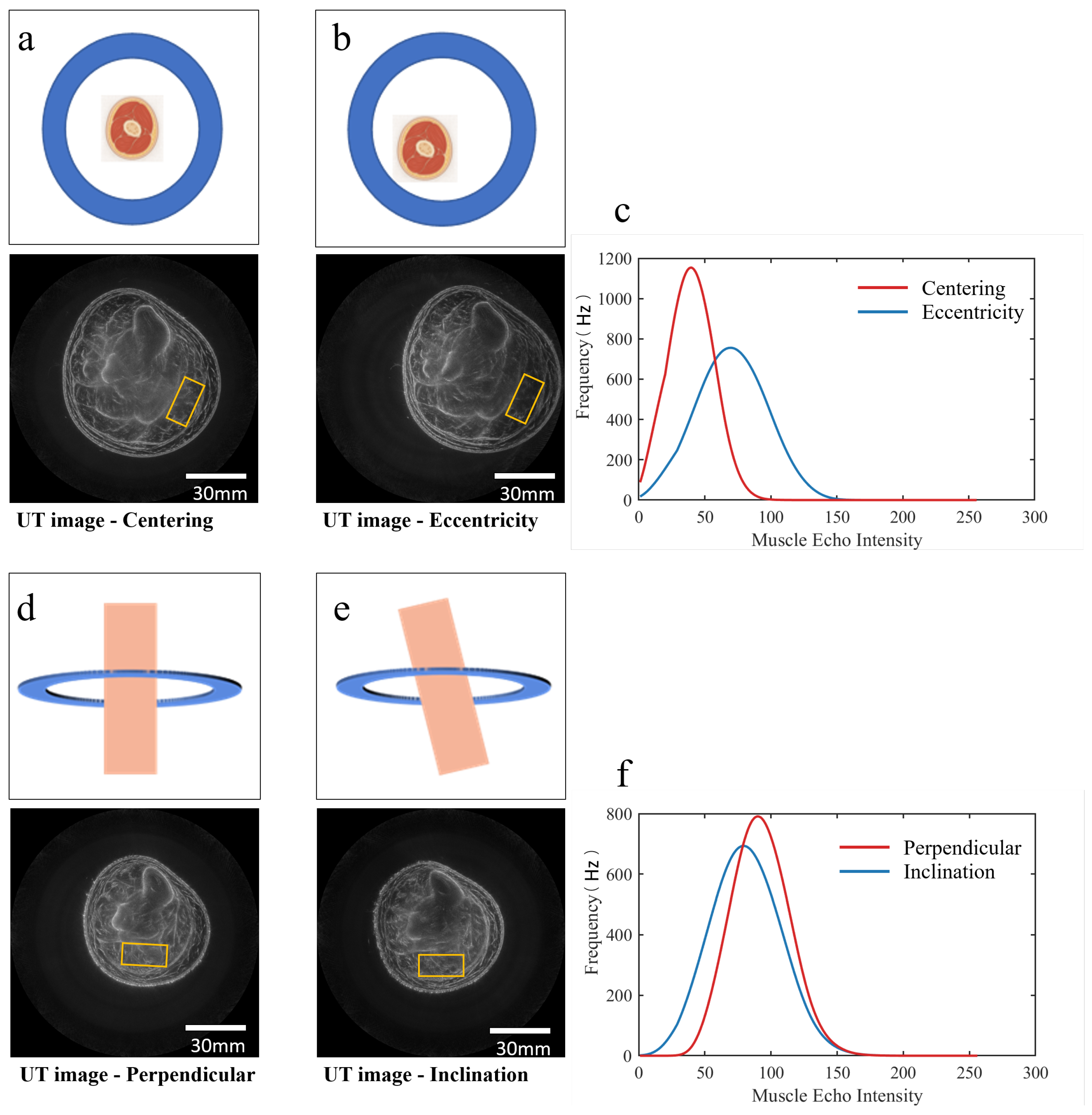
2.1. Identification of Influencing Factors
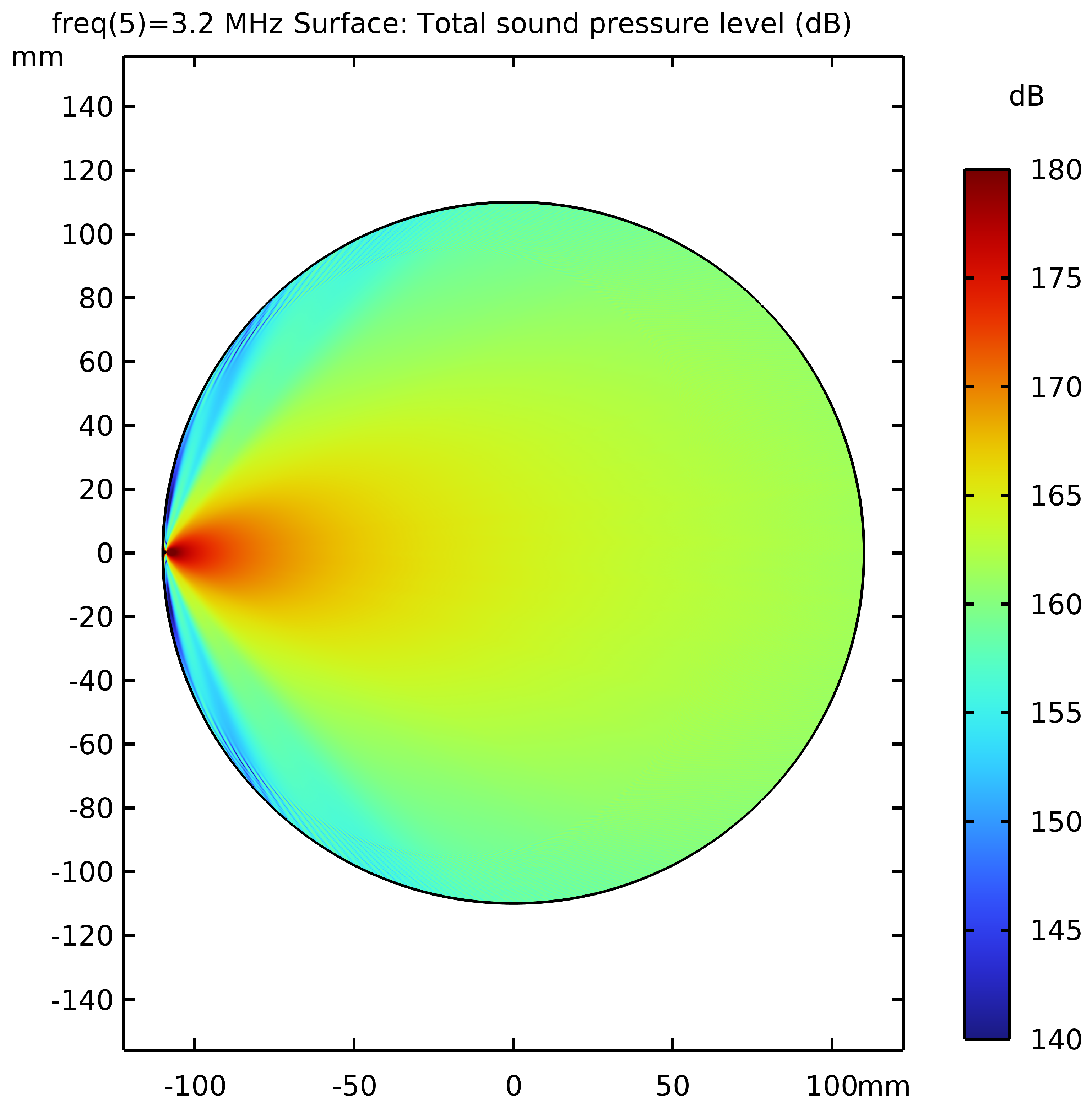
2.2. Numerical Simulations
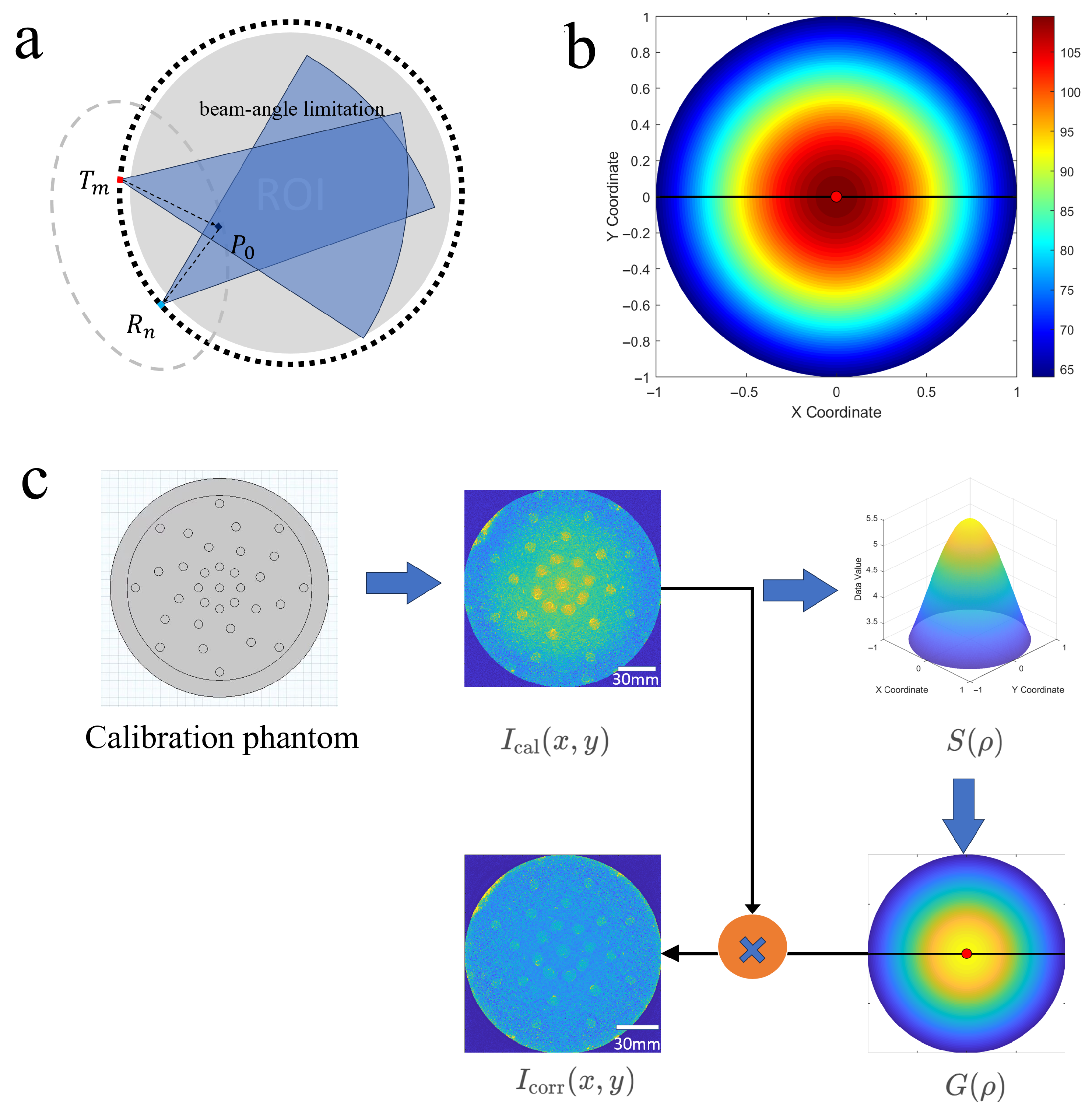
2.3. Theoretical Analysis and Compensation Method
2.3.1. Analysis and Compensation Method of Eccentricity
2.3.2. Analysis and Compensation Method of Inclination

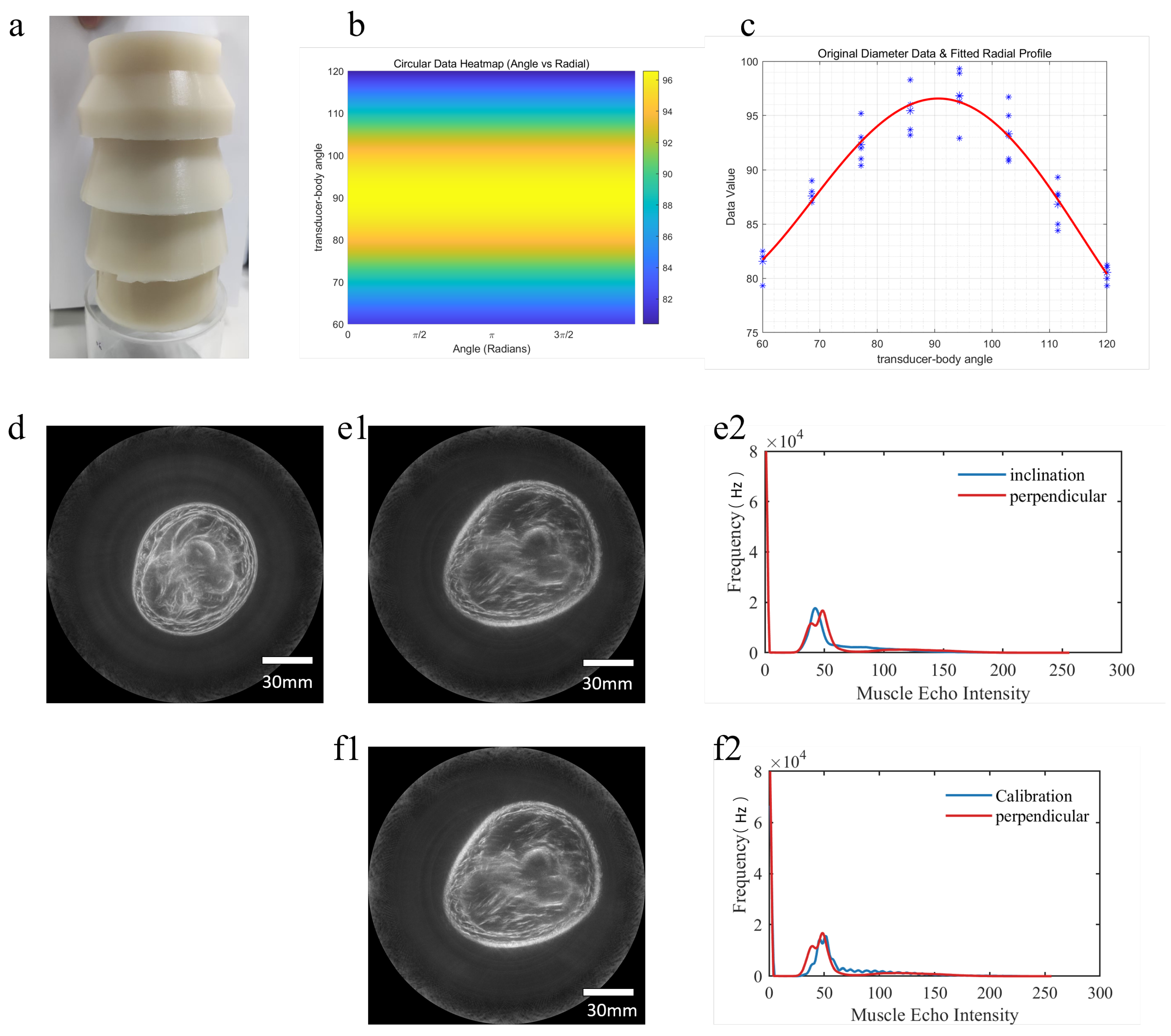
2.4. Phantom Experiment
3. Results
3.1. Validation of Eccentricity Effect
3.2. Validation of Inclination Effect
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vos, T.; Lim, S.S.; Abbafati, C.; Abbas, K.M.; Abbasi, M.; Abbasifard, M.; Abbasi-Kangevari, M.; Abbastabar, H.; Abd-Allah, F.; Abdelalim, A.; et al. Global Burden of 369 Diseases and Injuries in 204 Countries and Territories, 1990–2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Musculoskeletal Health. Available online: https://www.who.int/news-room/fact-sheets/detail/musculoskeletal-conditions (accessed on 14 July 2022).
- Chen, L.K.; Liu, L.K.; Woo, J.; Assantachai, P.; Auyeung, T.W.; Bahyah, K.S.; Chou, M.; Chen, L.; Hsu, P.; Krairit, O.; et al. Sarcopenia in Asia: Consensus Report of the Asian Working Group for Sarcopenia. J. Am. Med. Dir. Assoc. 2014, 15, 95–101. [Google Scholar] [CrossRef]
- Xu, W.; Chen, T.; Cai, Y.; Hu, Y.; Fan, L.; Wu, C. Sarcopenia in Community-Dwelling Oldest Old Is Associated with Disability and Poor Physical Function. J. Nutr. Health Aging 2020, 24, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Petermann-Rocha, F.; Balntzi, V.; Gray, S.R.; Lara, J.; Ho, F.K.; Pell, J.P.; Celis-Morales, C. Global Prevalence of Sarcopenia and Severe Sarcopenia: A Systematic Review and Meta-Analysis. J. Cachexia Sarcopenia Muscle 2022, 13, 86–99. [Google Scholar] [CrossRef]
- Shin, Y.; Yang, J.; Lee, Y.H.; Kim, S. Artificial Intelligence in Musculoskeletal Ultrasound Imaging. Ultrasonography 2021, 40, 30–44. [Google Scholar] [CrossRef]
- Shinohara, I.; Inui, A.; Mifune, Y.; Nishimoto, H.; Yamaura, K.; Mukohara, S.; Yoshikawa, T.; Kato, T.; Furukawa, T.; Hoshino, Y.; et al. Motion Analysis of Triangular Fibrocartilage Complex by Using Ultrasonography Images: Preliminary Analysis. Sensors 2022, 22, 345. [Google Scholar] [CrossRef]
- Özçakar, L.; Ata, A.M.; Kaymak, B.; Kara, M.; Kumbhare, D. Ultrasound Imaging for Sarcopenia, Spasticity and Painful Muscle Syndromes. Curr. Opin. Support. Palliat. Care 2018, 12, 373–381. [Google Scholar] [CrossRef]
- Wu, J.; Luo, H.; Ren, S.; Shen, L.; Cheng, D.; Wang, N. Enhanced Echo Intensity of Skeletal Muscle Is Associated with Poor Physical Function in Hemodialysis Patients: A Cross-Sectional Study. BMC Nephrol. 2022, 23, 186. [Google Scholar] [CrossRef]
- Mikołajowski, G.; Pałac, M.; Wolny, T.; Linek, P. Lateral Abdominal Muscles Shear Modulus and Thickness Measurements under Controlled Ultrasound Probe Compression by External Force Sensor: A Comparison and Reliability Study. Sensors 2021, 21, 4036. [Google Scholar] [CrossRef]
- Iqbal, I.; Younus, M.; Walayat, K.; Kakar, M.U.; Ma, J. Automated multi-class classification of skin lesions through deep convolutional neural network with dermoscopic images. Comput. Med. Imaging Graph. 2021, 88, 101843. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, I. Deep learning-based automated detection of human knee joint’s synovial fluid from magnetic resonance images with transfer learning. IET Image Process. 2020, 14, 2884–2892. [Google Scholar] [CrossRef]
- Ali, R.; Mitcham, T.M.; Brevett, T.; Agudo, O.C.; Martinez, C.D.; Li, C.; Doyley, M.M.; Duric, N. 2-D Slicewise Waveform Inversion of Sound Speed and Acoustic Attenuation for Ring Array Ultrasound Tomography Based on a Block LU Solver. IEEE Trans. Med. Imaging 2024, 43, 2988–3000. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Choi, J.; Moon, J.; Sung, K.R.; Choi, J. A Sarcopenia Detection System Using an RGB-D Camera and an Ultrasound Probe: Eye-in-Hand Approach. Biosensors 2021, 11, 243. [Google Scholar]
- Song, J.; Zhang, Q.; Zhou, L.; Quan, Z.; Wang, S.; Liu, Z. Design and implementation of a modular and scalable research platform for ultrasound computed tomography. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2021, 69, 62–72. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, X.; Yang, H.; Zhang, Q.; Zhou, L.; Wu, Y.; Liu, Q.; Yan, W.; Song, J.; Ding, M.; et al. Reconstruction of reflection ultrasound computed tomography with sparse transmissions using conditional generative adversarial network. Ultrasonics 2025, 145, 107486. [Google Scholar] [CrossRef]
- Savitzky, A.; Golay, M.J.E. Smoothing and differentiation of data by simplified least squares procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- du Toit, C.; Dima, R.; Papernick, S.; Jonnalagadda, M.; Tessier, D.; Fenster, A.; Lalone, E. Three-dimensional ultrasound to investigate synovitis in first carpometacarpal osteoarthritis: A feasibility study. Med. Phys. 2024, 51, 1092–1104. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.N.; Park, M.S.; Yang, S.J.; Yoo, H.J.; Kang, H.J.; Song, W.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Baik, S.H.; et al. Prevalence and Determinant Factors of Sarcopenia in Patients with Type 2 Diabetes: The Korean Sarcopenic Obesity Study (KSOS). Diabetes Care 2010, 33, 1497–1499. [Google Scholar] [CrossRef]
- Wolfson, L.; Judge, J.; Whipple, R.; King, M. Strength Is a Major Factor in Balance, Gait, and the Occurrence of Falls. J. Gerontol. A Biol. Sci. Med. Sci. 1995, 50, 64–67. [Google Scholar] [CrossRef]
- Wilhelm, E.N.; Rech, A.; Minozzo, F.; Radaelli, R.; Botton, C.E.; Pinto, R.S. Relationship between Quadriceps Femoris Echo Intensity, Muscle Power, and Functional Capacity of Older Men. Age 2014, 36, 9625. [Google Scholar] [CrossRef]
- Watanabe, Y.; Ikenaga, M.; Yoshimura, E.; Yamada, Y.; Kimura, M. Association between Echo Intensity and Attenuation of Skeletal Muscle in Young and Older Adults: A Comparison between Ultrasonography and Computed Tomography. Clin. Interv. Aging 2018, 13, 1871–1878. [Google Scholar] [CrossRef]
- Isaka, M.; Sugimoto, K.; Yasunobe, Y.; Akasaka, H.; Fujimoto, T.; Kurinami, H.; Takeya, Y.; Yamamoto, K.; Rakugi, H. The Usefulness of an Alternative Diagnostic Method for Sarcopenia Using Thickness and Echo Intensity of Lower Leg Muscles in Older Males. J. Am. Med. Dir. Assoc. 2019, 20, 1185.e1–1185.e8. [Google Scholar] [CrossRef] [PubMed]
- Yamada, M.; Kimura, Y.; Ishiyama, D.; Nishio, N.; Abe, Y.; Kakehi, T.; Fujimoto, J.; Tanaka, T.; Ohji, S.; Otobe, Y.; et al. Differential Characteristics of Skeletal Muscle in Community-Dwelling Older Adults. J. Am. Med. Dir. Assoc. 2017, 18, 807.e9–807.e16. [Google Scholar] [CrossRef] [PubMed]
- Formenti, P.; Umbrello, M.; Coppola, S.; Froio, S.; Chiumello, D. Clinical Review: Peripheral Muscular Ultrasound in the ICU. Ann. Intensive Care 2019, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Scafoglieri, A.; Van den Broeck, J.; Bartocci, P.; Cattrysse, E.; Jager-Wittenaar, H.; Gonzalez, M.C. Skeletal Muscle Echo Intensity Values Differ Significantly across Ultrasound Parameter Settings. Life 2024, 14, 291. [Google Scholar] [CrossRef]




| Ingredient | Target Tissues Material | Background Tissues Material |
|---|---|---|
| agar power | 3.5 g | 3.5 g |
| cellulose | 1.5 g | - |
| glycerol | 8 mL | - |
| Metric | Before (Mean ± SD) | After (Mean ± SD) | p-Value |
|---|---|---|---|
| Radial Intensity Slope (RIS) | 0.05612 ± 0.00023 | 0.000818 ± 0.000012 | <0.05 |
| Center-to-Periphery Ratio (CPR) | 1.660 ± 0.018 | 0.985 ± 0.004 | <0.05 |
| Radial Non-Uniformity (RNU) | 0.702 ± 0.005 | 0.127 ± 0.003 | <0.05 |
| Coefficient of Variation (CV) | 0.267 ± 0.002 | 0.173 ± 0.002 | <0.05 |
| Metric | Before (Mean ± SD) | After (Mean ± SD) | Reduction | p-Value |
|---|---|---|---|---|
| RIS | 0.106 ± 0.010 | 0.0296 ± 0.0001 | 72% ↓ | <0.05 |
| CPR | 2.776 ± 0.063 | 1.313 ± 0.017 | 53% ↓ | <0.05 |
| RNU | 1.583 ± 0.057 | 1.044 ± 0.008 | 34% ↓ | <0.05 |
| CV | 0.629 ± 0.011 | 0.491 ± 0.007 | 22% ↓ | <0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zeng, J.; Lou, D.; Zhang, Q.; Zhang, H.; Zhu, H.; Cheng, X.; Wang, T.; Xu, S.; Ling, Y.; Ding, M. Echo Intensity Correction Method for Ultrasound Computed Tomography in Musculoskeletal Imaging. Bioengineering 2026, 13, 352. https://doi.org/10.3390/bioengineering13030352
Zeng J, Lou D, Zhang Q, Zhang H, Zhu H, Cheng X, Wang T, Xu S, Ling Y, Ding M. Echo Intensity Correction Method for Ultrasound Computed Tomography in Musculoskeletal Imaging. Bioengineering. 2026; 13(3):352. https://doi.org/10.3390/bioengineering13030352
Chicago/Turabian StyleZeng, Junchao, Ding Lou, Qin Zhang, Hui Zhang, Hongyi Zhu, Xing Cheng, Tengfei Wang, Sanping Xu, Yan Ling, and Mingyue Ding. 2026. "Echo Intensity Correction Method for Ultrasound Computed Tomography in Musculoskeletal Imaging" Bioengineering 13, no. 3: 352. https://doi.org/10.3390/bioengineering13030352
APA StyleZeng, J., Lou, D., Zhang, Q., Zhang, H., Zhu, H., Cheng, X., Wang, T., Xu, S., Ling, Y., & Ding, M. (2026). Echo Intensity Correction Method for Ultrasound Computed Tomography in Musculoskeletal Imaging. Bioengineering, 13(3), 352. https://doi.org/10.3390/bioengineering13030352







