Surface EMG-Validated Multi-DoF Wheelchair-Based Rehabilitation Device
Abstract
1. Introduction
2. System Model
3. Upper-Limb Exoskeleton Design
3.1. Wrist Design
3.2. Elbow Design
3.3. Shoulder Design
3.4. System Wiring
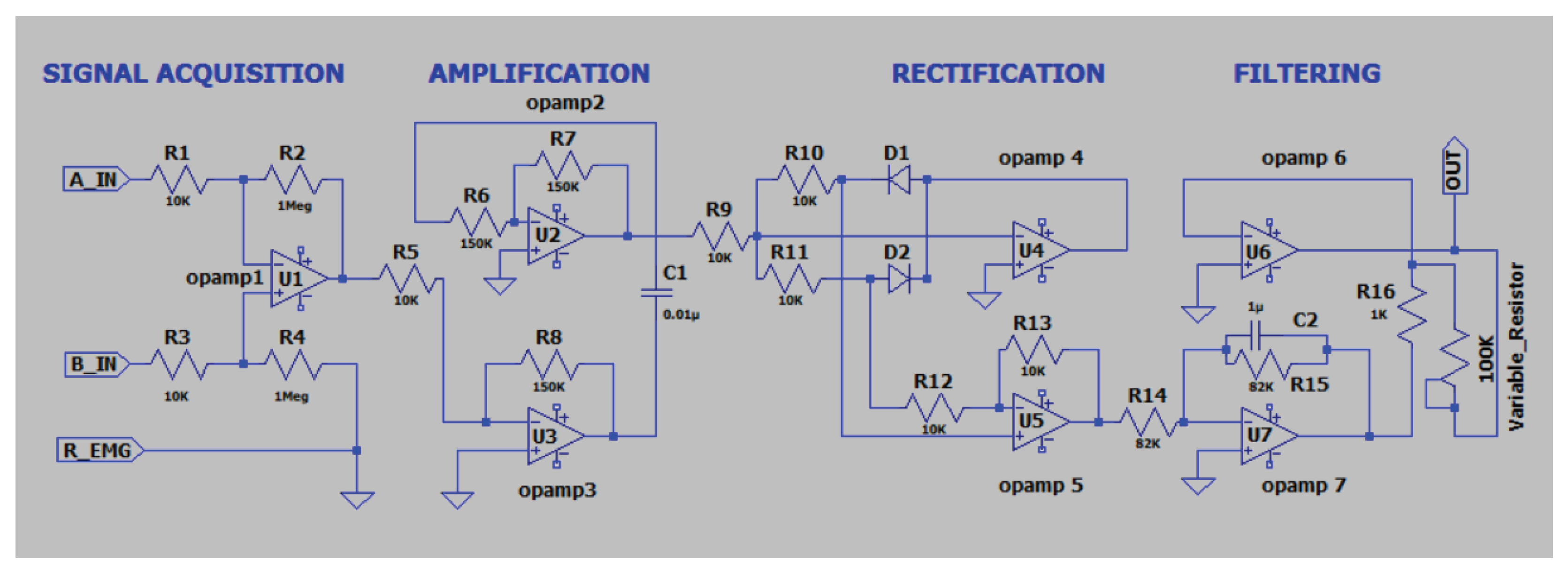
4. Surface EMG Signal
5. Experiments and Results
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Feigin, V.L.; Norrving, B.; Mensah, G.A. Global Burden of Stroke. Circ. Res. 2017, 120, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Rost, N.S.; Bottle, A.; Lee, J.; Randall, M.; Middleton, S.; Shaw, L.; Thijs, V.; Rinkel, G.J.E.; Hemmen, T.M. Stroke severity is a crucial predictor of outcome: An International Prospective Validation study. J. Am. Heart Assoc. 2016, 5, e002433. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.; Onuma, O.; Owolabi, M.; Sachdev, S. Stroke: A global response is needed. Bull. World Health Organ. 2016, 94, 634–634A. [Google Scholar] [CrossRef] [PubMed]
- Saini, V.; Guada, L.; Yavagal, D.R. Global Epidemiology of Stroke and Access to Acute Ischemic Stroke Interventions. Neurology 2021, 97, S6–S16. [Google Scholar] [CrossRef] [PubMed]
- McHugh, G.; Swain, I.D. A comparison between reported and ideal patient-to-therapist ratios for stroke rehabilitation. Health 2013, 5, 105–112. [Google Scholar] [CrossRef]
- Cram, J.R. The History of Surface Electromyography. Appl. Psychophysiol. Biofeedback 2003, 28, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Zieliński, G.; Gawda, P. Surface Electromyography in Dentistry—Past, Present and Future. J. Clin. Med. 2024, 13, 1328. [Google Scholar] [CrossRef] [PubMed]
- Banyai, A.D.; Brișan, C. Robotics in Physical Rehabilitation: Systematic Review. Healthcare 2024, 12, 1720. [Google Scholar] [CrossRef] [PubMed]
- Kapsalyamov, A.; Hussain, S.; Jamwal, P.K. State-of-the-Art assistive powered upper limb exoskeletons for elderly. IEEE Access 2020, 8, 178991–179001. [Google Scholar] [CrossRef]
- Lee, D.J.; Bae, S.J.; Jang, S.H.; Chang, P.H. Design of a clinically relevant upper-limb exoskeleton robot for stroke patients with spasticity. In Proceedings of the 2017 International Conference on Rehabilitation Robotics (ICORR), London, UK, 17–20 July 2017; pp. 622–627. [Google Scholar] [CrossRef]
- Gao, B.; Ma, H.; Guo, S.; Xu, H.; Yang, S. Design and evaluation of a 3-degree-of-freedom upper limb rehabilitation exoskeleton robot. In Proceedings of the 2017 IEEE International Conference on Mechatronics and Automation (ICMA), Takamatsu, Japan, 6–9 August 2017; pp. 938–942. [Google Scholar] [CrossRef]
- Lavado, D.M.; Vela, E.A. A Wearable Device based on IMU and EMG Sensors for Remote Monitoring of Elbow Rehabilitation. In Proceedings of the 2022 E-Health and Bioengineering Conference (EHB), Iasi, Romania, 17–18 November 2022; pp. 1–4. [Google Scholar] [CrossRef]
- Guo, S.; Ding, Y.; Guo, J. Control of A Lower Limb Exoskeleton Robot by Upper Limb sEMG Signal. In Proceedings of the 2021 IEEE International Conference on Mechatronics and Automation (ICMA), Takamatsu, Japan, 8–11 August 2021; pp. 1113–1118. [Google Scholar] [CrossRef]
- Thøgersen, M.; Gull, M.A.; Kobbelgaard, F.V.; Mohammadi, M.; Bengtson, S.H.; Struijk, L.N.S.A. EXOTIC—A Discreet User-Based 5 DoF Upper-Limb Exoskeleton for Individuals with Tetraplegia. In Proceedings of the 2020 3rd International Conference on Mechatronics, Robotics and Automation (ICMRA), Shanghai, China, 16–18 October 2020; pp. 79–83. [Google Scholar] [CrossRef]
- Chandrasekhar, V.; Vazhayil, V.; Rao, M. Design of a real time portable low-cost multi-channel surface electromyography system to aid neuromuscular disorder and post stroke rehabilitation patients. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 4138–4142. [Google Scholar] [CrossRef]
- Laganà, F.; Pratticò, D.; Angiulli, G.; Oliva, G.; Pullano, S.A.; Versaci, M.; La Foresta, F. Development of an Integrated System of sEMG Signal Acquisition, Processing, and Analysis with AI Techniques. Signals 2024, 5, 476–493. [Google Scholar] [CrossRef]
- P, J.; GVK, S.; Rao, M.; Bapat, J.; Das, D. Advanced Wheelchair Rehabilitation: Surface EMG-augmented 9-DoF System for Upper and Lower Limb. In Proceedings of the 2024 46th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 15–19 July 2024; pp. 1–4. [Google Scholar] [CrossRef]
- Vinay, C.K.; Vazhiyal, V.; Rao, M. Design of a non-invasive pulse rate controlled deep vein thrombosis prophylaxis lower limb device. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; pp. 5407–5410. [Google Scholar] [CrossRef]
- Chandrasekhar, V.; Vazhayil, V.; Rao, M. Design of a portable anthropomimetic upper limb rehabilitation device for patients suffering from neuromuscular disability. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 4708–4712. [Google Scholar] [CrossRef]
- Bratanov, D.; Vitliemov, P. The Experience Behind The REHAROB Project—A New Robotic System For Simultaneous Rehabilitation Of Upper And Lower Human Limbs. In Proceedings of the 2020 International Conference on Assistive and Rehabilitation Technologies (iCareTech), Gaza, Palestine, 28–29 August 2020; pp. 19–23. [Google Scholar] [CrossRef]
- Laganà, F.; Pellicanò, D.; Arruzzo, M.; Pratticò, D.; Pullano, S.A.; Fiorillo, A.S. FEM-Based Modelling and AI-Enhanced Monitoring System for Upper Limb Rehabilitation. Electronics 2025, 14, 2268. [Google Scholar] [CrossRef]
- Qureshi, M.B.; Qureshi, M.S.; Tahir, S.; Anwar, A.; Hussain, S.; Uddin, M.; Chen, C.L. Encryption Techniques for Smart Systems Data Security Offloaded to the Cloud. Symmetry 2022, 14, 695. [Google Scholar] [CrossRef]
- Bateni, H.; Carruthers, J.; Mohan, R.; Pishva, S. Use of Virtual Reality in Physical Therapy as an Intervention and Diagnostic Tool. Rehabil. Res. Pract. 2024, 2024, 1–9. [Google Scholar] [CrossRef] [PubMed]
- P, J.; GVK, S.; Rao, M.; Bapat, J.; Das, D. XoRehab: IoT Enabled Wheelchair based Lower Limb Rehabilitation System. In Proceedings of the 2023 45th Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Sydney, Australia, 24–27 July 2023; pp. 1–5. [Google Scholar] [CrossRef]
- Garcia, G.F.; Gonçalves, R.S.; Carbone, G. A Review of Wrist Rehabilitation Robots and Highlights Needed for New Devices. Machines 2024, 12, 315. [Google Scholar] [CrossRef]
- Yang, H.; Wei, G.; Ren, L. Enhancing the Performance of a Biomimetic Robotic Elbow-and-Forearm System Through Bionics-Inspired Optimization. IEEE Trans. Robot. 2024, 40, 2692–2711. [Google Scholar] [CrossRef]
- Bolsterlee, B.; Veeger, D.H.E.J.; Chadwick, E.K. Clinical applications of musculoskeletal modelling for the shoulder and upper limb. Med Biol. Eng. Comput. 2013, 51, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Hermens, H.J.; Freriks, B.; Disselhorst-Klug, C.; Rau, G. Development of recommendations for SEMG sensors and sensor placement procedures. J. Electromyogr. Kinesiol. 2000, 10, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Ashcroft, R.E. The Declaration of Helsinki. In The Oxford Textbook of Clinical Research Ethics; Oxford University Press: Oxford, UK, 2008. [Google Scholar] [CrossRef]
- Kubo, A.; Onoda, K.; Nishida, Y.; Kurosawa, K.; Morita, M. Effects of gender and grade on differences in physiotherapy students’ interest and reasons for participating in overseas volunteer programs. J. Phys. Ther. Sci. 2020, 32, 597–600. [Google Scholar] [CrossRef] [PubMed]







| Paper | IoT- Enabled | Exoskeleton Mechanism for Targeted Area | EMG Enhanced | Edge Communication | Cloud and Client Application | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Upper Limb | Lower Limb | Hip Flexion–Extension | |||||||||
| 3DoF Wrist | Elbow Flexion–Extension | Shoulder Flexion–Extension | Shoulder Abduction–Adduction | Knee Flexion–Extension | Ankle Flexion–Extension | ||||||
| [10] | No | Yes | Yes | No | No | No | No | No | No | No | No |
| [11] | No | Yes | Yes | No | No | No | No | No | No | No | No |
| [12] | Yes | No | Yes | No | No | No | No | No | Yes | Yes | Yes |
| [13] | No | No | No | No | No | Yes | Yes | No | Yes | No | No |
| [14] | No | 1 DoF | Yes | Yes | Yes | No | No | No | No | No | No |
| [18] | No | No | No | No | No | Yes | Yes | No | No | No | No |
| [20] | No | No | Yes | Yes | Yes | Yes | Yes | No | No | No | No |
| [24] | Yes | No | No | No | No | Yes | Yes | Yes | No | Yes | Yes |
| Proposed | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Raspberry Pi | Yes |
| Exercise Mode | Total Expected ROM | Total Device ROM | Each ROM in Degrees | Difference |
|---|---|---|---|---|
| Wrist flexion and extension | 78° | 82° | 0–40 flexion 40–82 extension | +4° (5%) |
| Wrist pronation and supination | 180° | 125° | 0–60 pronation 60–125 supination | −55° (33%) |
| Wrist radial and ulnar deviation | 66° | 70° | 0–30 radial 30–70 ulnar | +4° (6%) |
| Elbow flexion and extension | 142° | 55° | 0–55 flexion 55–0 extension | −87° (53%) |
| Shoulder flexion and extension | 180° | 100° | 0–100 flexion 100–0 extension | −80° (44%) |
| Shoulder abduction and adduction | 150° | 140° | 0–140 abduction 140–0 adduction | +10° (6.6%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
P, J.; Rao, M. Surface EMG-Validated Multi-DoF Wheelchair-Based Rehabilitation Device. Bioengineering 2026, 13, 350. https://doi.org/10.3390/bioengineering13030350
P J, Rao M. Surface EMG-Validated Multi-DoF Wheelchair-Based Rehabilitation Device. Bioengineering. 2026; 13(3):350. https://doi.org/10.3390/bioengineering13030350
Chicago/Turabian StyleP, Jagan, and Madhav Rao. 2026. "Surface EMG-Validated Multi-DoF Wheelchair-Based Rehabilitation Device" Bioengineering 13, no. 3: 350. https://doi.org/10.3390/bioengineering13030350
APA StyleP, J., & Rao, M. (2026). Surface EMG-Validated Multi-DoF Wheelchair-Based Rehabilitation Device. Bioengineering, 13(3), 350. https://doi.org/10.3390/bioengineering13030350







