Blood Cell-Based Drug Delivery Systems: From Biological and Mechanical Design to Clinical Applications
Abstract
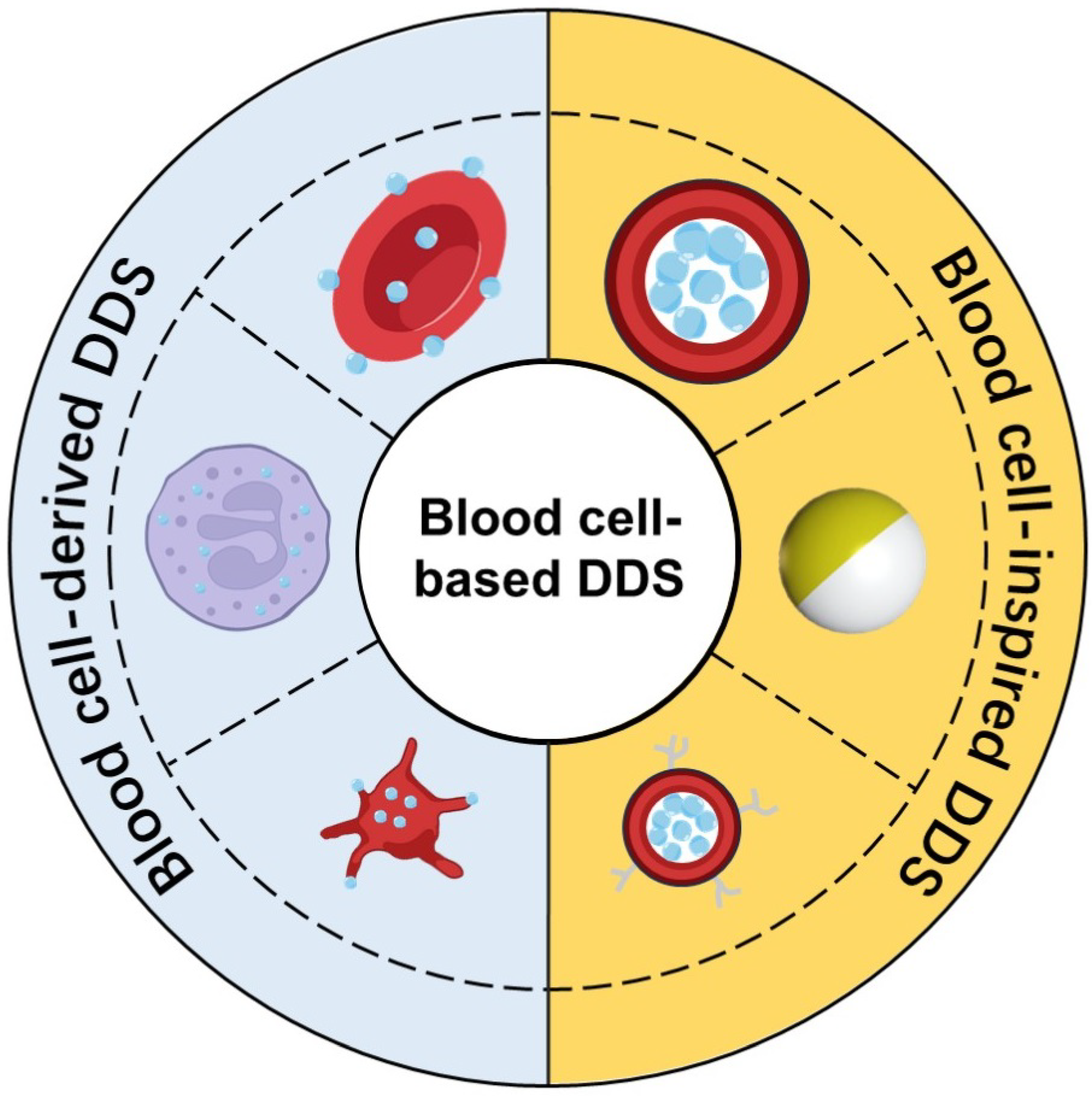
1. Introduction
2. Biological and Mechanical Design Principles
2.1. RBCs: Long-Circulating Carriers
2.2. WBCs: Actively Homing Vehicles
2.3. Platelets: Nature-Derived Carriers Targeting the Tumor Microenvironment
2.4. Blood Cell-Derived Membrane Vesicles: Biomimetic Nanocarriers
3. Engineering and Loading Strategies
3.1. RBC-Based DDSs: Encapsulation, Surface Engineering, and Controlled Release
3.2. WBC-Based DDSs: Leveraging Homing, Phagocytosis, and Bioinspired Designs
3.3. Platelet-Based DDSs: Granule Loading, Membrane Camouflage, and Stimuli- Responsive Design
3.4. Blood Cell-Derived Membrane Vesicle-Based DDSs: Fabrication and Cargo Loading
4. Therapeutic Applications in Oncology
4.1. RBC-Based DDSs in Tumor Therapy
4.2. WBC-Based DDSs in Tumor Therapy
4.3. Platelet-Based DDSs in Tumor Therapy
5. Emerging Applications Across Disease Settings
6. Current Challenges and Mitigation Strategies
6.1. Key Challenges
6.2. Strategies to Overcome Translational Barriers
7. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAV | Adeno-associated virus |
| ADP | Adenosine diphosphate |
| BBB | Blood–brain barrier |
| BDNF | Brain-derived neurotrophic factor |
| CARs | Chimeric antigen receptors |
| CTCs | Circulating tumor cells |
| CXCL12 | C-X-C motif chemokine ligand 12 |
| DDS | Drug delivery system |
| EGFR | Epidermal growth factor receptor |
| EPR | Enhanced permeability and retention |
| HER2 | Human epidermal growth factor receptor 2 |
| IL-6 | Interleukin-6 |
| MARVEL | Multiscale approach using RBC-mediated hitchhiking and vascular endothelium leakage |
| NK | Natural killer |
| PD-1 | Programmed cell death protein 1 |
| RBCs | Red blood cells |
| TNF- | Tumor necrosis factor alpha |
| VEGF | Vascular endothelial growth factor |
| vWF | von Willebrand factor |
| WBCs | White blood cells |
References
- Anselmo, A.C.; Mitragotri, S. An overview of clinical and commercial impact of drug delivery systems. J. Control. Release 2014, 190, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, J.; Wang, Y.; Gao, H.; Wei, G.; Huang, Y.; Yu, H.; Gan, Y.; Wang, Y.; Mei, L.; et al. Recent progress in drug delivery. Acta Pharm. Sin. B 2019, 9, 1145–1162. [Google Scholar] [CrossRef]
- Wang, L.L.W.; Gao, Y.; Feng, Z.; Mooney, D.J.; Mitragotri, S. Designing drug delivery systems for cell therapy. Nat. Rev. Bioeng. 2024, 2, 944–959. [Google Scholar] [CrossRef]
- Shaha, S.; Rodrigues, D.; Mitragotri, S. Locoregional drug delivery for cancer therapy: Preclinical progress and clinical translation. J. Control. Release 2024, 367, 737–767. [Google Scholar] [CrossRef] [PubMed]
- Fukuta, T.; Shaha, S.; da Silva-Candal, A.; Zhao, Z.; Mitragotri, S. Cell therapies against brain tumors: Clinical development and emerging prospects. Bioeng. Trans. Med. 2025, 10, e70018. [Google Scholar] [CrossRef] [PubMed]
- Gavas, S.; Quazi, S.; Karpiński, T.M. Nanoparticles for Cancer Therapy: Current Progress and Challenges. Nanoscale Res. Lett. 2021, 16, 173. [Google Scholar] [CrossRef]
- He, A.; Huang, Y.; Cao, C.; Li, X. Advances in drug delivery systems utilizing blood cells and their membrane-derived microvesicles. Drug Deliv. 2024, 31, 2425156. [Google Scholar] [CrossRef]
- Udofa, E.; Zhao, Z. In situ cellular hitchhiking of nanoparticles for drug delivery. Adv. Drug Deliv. Rev. 2024, 204, 115143. [Google Scholar] [CrossRef]
- Rossi, L.; Pierigè, F.; Antonelli, A.; Bigini, N.; Gabucci, C.; Peiretti, E.; Magnani, M. Engineering erythrocytes for the modulation of drugs’ and contrasting agents’ pharmacokinetics and biodistribution. Adv. Drug Deliv. Rev. 2016, 106, 73–87. [Google Scholar] [CrossRef]
- Zhang, S.; Fu, Q.; Zhang, Y.; Pan, J.; Zhang, L.; Zhang, Z.; Liu, Z. Surface loading of nanoparticles on engineered or natural erythrocytes for prolonged circulation time: Strategies and applications. Acta Pharmacol. Sin. 2021, 42, 1040–1054. [Google Scholar] [CrossRef]
- Nguyen, P.H.D.; Jayasinghe, M.K.; Le, A.H.; Peng, B.; Le, M.T.N. Advances in Drug Delivery Systems Based on Red Blood Cells and Their Membrane-Derived Nanoparticles. ACS Nano 2023, 17, 5187–5210. [Google Scholar] [CrossRef]
- Quinn, D.; Pivkin, I.; Wong, S.; Chiam, K.; Dao, M.; Karniadakis, G.; Suresh, S. Combined simulation and experimental study of large deformation of red blood cells in microfluidic systems. Ann. Biomed. Eng. 2011, 39, 1041–1050. [Google Scholar] [CrossRef]
- Li, H.; Lu, L.; Li, X.; Buffet, P.A.; Dao, M.; Karniadakis, G.E.; Suresh, S. Mechanics of diseased red blood cells in human spleen and consequences for hereditary blood disorders. Proc. Natl. Acad. Sci. USA 2018, 115, 9574–9579. [Google Scholar] [CrossRef]
- Peng, Z.; Viallat, A.; Young, Y.N. Fluid Mechanics of Blood Cells and Vesicles Squeezing Through Narrow Constrictions. Annu. Rev. Fluid Mech. 2026, 58, 331–354. [Google Scholar] [CrossRef]
- Huang, Y.; Gao, X.; Chen, J. Leukocyte-derived biomimetic nanoparticle drug delivery systems for cancer therapy. Acta Pharm. Sin. B 2018, 8, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Chu, D.; Dong, X.; Shi, X.; Zhang, C.; Wang, Z. Neutrophil-based drug delivery systems. Adv. Mater. 2018, 30, 1706245. [Google Scholar] [CrossRef]
- Hu, Q.; Sun, W.; Qian, C.; Wang, C.; Bomba, H.; Gu, Z. Anticancer platelet-mimetic nanovehicles. Adv. Mater. 2015, 27, 7043–7050. [Google Scholar] [CrossRef]
- Wu, L.; Xie, W.; Zan, H.M.; Liu, Z.; Wang, G.; Wang, Y.; Liu, W.; Dong, W. Platelet membrane-coated nanoparticles for targeted drug delivery and local chemo-photothermal therapy of orthotopic hepatocellular carcinoma. J. Mater. Chem. B 2020, 8, 4648–4659. [Google Scholar] [CrossRef]
- Glassman, P.M.; Hood, E.D.; Ferguson, L.T.; Zhao, Z.; Siegel, D.L.; Mitragotri, S.; Brenner, J.S.; Muzykantov, V.R. Red blood cells: The metamorphosis of a neglected carrier into the natural mothership for artificial nanocarriers. Adv. Drug Deliv. Rev. 2021, 178, 113992. [Google Scholar] [CrossRef] [PubMed]
- Millán, C.; Marinero, M.; Castañeda, A.; Lanao, J. Drug, enzyme and peptide delivery using erythrocytes as carriers. J. Control. Release 2004, 95, 27–49. [Google Scholar] [CrossRef] [PubMed]
- Rarokar, N.; Yadav, S.; Saoji, S.; Bramhe, P.; Agade, R.; Gurav, S.; Khedekar, P.; Subramaniyan, V.; Wong, L.S.; Kumarasamy, V. Magnetic nanosystem a tool for targeted delivery and diagnostic application: Current challenges and recent advancement. Int. J. Pharm. X 2024, 7, 100231. [Google Scholar] [CrossRef]
- Park, K.S.; Chandran Suja, V.; Kim, J.; Rodrigues, D.; Mukherji, M.; Joshi, M.; Gao, Y.; Bibbey, M.G.; Choi, J.W.; Liao, R.; et al. Multiscale red blood cell hitchhiking for targeted deep tissue gene delivery in lungs. Nat. Commun. 2025, 16, 10280. [Google Scholar] [CrossRef] [PubMed]
- Oldenborg, P.A.; Zheleznyak, A.; Fang, Y.F.; Lagenaur, C.; Gresham, H.; Lindberg, F. Role of CD47 as a marker of self on red blood cells. Science 2000, 288, 2051–2054. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.; Harada, T.; Christian, D.; Pantano, D.; Tsai, R.; Discher, D. Minimal “Self” peptides that inhibit phagocytic clearance and enhance delivery of nanoparticles. Science 2013, 339, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Muzykantov, V. Drug delivery by red blood cells: Vascular carriers designed by mother nature. Expert Opin. Drug Deliv. 2010, 7, 403–427. [Google Scholar] [CrossRef]
- He, Y.; Cheng, C.; Liu, Y.; Chen, F.-M.; Chen, Y.; Yang, C.; Zhao, Z.; Dawulieti, J.; Shen, Z.; Zhang, Y.; et al. Intravenous Senescent Erythrocyte Vaccination Modulates Adaptive Immunity and Splenic Complement Production. ACS Nano 2024, 18, 470–482. [Google Scholar] [CrossRef]
- Li, Y.; Wu, J.; Liu, J.; Qiu, X.; Xu, W.; Tang, T.; Xiang, D. From blood to brain: Blood cell-based biomimetic drug delivery systems. Drug Deliv. 2021, 28, 1214–1225. [Google Scholar] [CrossRef]
- Klyachko, N.; Polak, R.; Haney, M.; Zhao, Y.; Neto, R.; Hill, M.; Kabanov, A.; Batrakova, E. Macrophages with cellular backpacks for targeted drug delivery to the brain. Biomaterials 2017, 140, 79–87. [Google Scholar] [CrossRef]
- Krishnamurthy, S.; Gnanasammandhan, M.; Xie, C.; Huang, K.; Cui, M.; Chan, J. Monocyte cell membrane-derived nanohosts for targeted cancer therapy. Nanoscale 2016, 8, 6981–6985. [Google Scholar] [CrossRef]
- Cao, H.; Dan, Z.; He, X.; Zhang, Z.; Yu, H.; Yin, Q.; Li, Y. Liposomes coated with isolated macrophage membrane can target lung metastasis of breast cancer. ACS Nano 2016, 10, 7738–7748. [Google Scholar] [CrossRef]
- Zhu, S.; Yi, M.; Wu, Y.; Dong, B.; Wu, K. Roles of tumor-associated macrophages in tumor progression: Implications on therapeutic strategies. Exp. Hematol. Oncol. 2021, 10, 60. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Qin, J.; Lin, B.; Zhang, J.; Li, D.; Liu, M. CAR-T therapy in solid tumors. Cancer Cell 2025, 43, 665–679. [Google Scholar] [CrossRef] [PubMed]
- Mittelbrunn, M.; Gutierrez-Vazquez, C.; Villarroya-Beltri, C.; Gonzalez, S.; Sanchez-Cabo, F.; Gonzalez, M.; Bernad, A.; Sanchez-Madrid, F. Unidirectional transfer of microRNA-loaded exosomes from T cells to antigen-presenting cells. Nat. Commun. 2011, 2, 282. [Google Scholar] [CrossRef]
- Che, J.; Najer, A.; Blakney, A.; Edwards, C.; Yan, W.; Demello, A.; Stevens, M. Neutrophils enable local and non-invasive liposome delivery to inflamed skeletal muscle and ischemic heart. Adv. Mater. 2020, 32, 2003598. [Google Scholar] [CrossRef]
- Li, H.; Qiang, Y.; Li, X.; Brugnara, C.; Buffet, P.A.; Dao, M.; Karniadakis, G.E.; Suresh, S. Biomechanics of phagocytosis of red blood cells by macrophages in the human spleen. Proc. Natl. Acad. Sci. USA 2024, 121, e2414437121. [Google Scholar] [CrossRef]
- Semple, J.; Italiano, J.; Freedman, J. Platelets and the immune continuum. Nat. Rev. Immunol. 2011, 11, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Qin, J.; Plow, E. Platelet integrin αIIbβ3: Activation mechanisms. J. Thromb. Haemost. 2007, 5, 1345–1352. [Google Scholar] [CrossRef]
- Haemmerle, M.; Stone, R.; Menter, D.; Afshar-Khanghan, V.; Sood, A. The platelet lifeline to cancer: Challenges and opportunities. Cancer Cell 2018, 33, 965–983. [Google Scholar] [CrossRef]
- Wu, J.; Zhu, H.; Yang, G.; Wang, X.; Zhang, Y.; Liu, Y.; Chen, X.; Li, S.; Li, Y.; Zhang, Z.; et al. Design and synthesis of nanoscaled IQCA-TAVV as a delivery system capable of antiplatelet activation, targeting arterial thrombus and releasing IQCA. Int. J. Nanomedicine 2018, 13, 1139–1158. [Google Scholar] [CrossRef]
- Dehaini, D.; Wei, X.; Fang, R.; Masson, S.; Angsantikul, P.; Luk, B.; Zhang, Y.; Ying, M.; Jiang, Y.; Kroll, A.; et al. Erythrocyte-platelet hybrid membrane coating for enhanced nanoparticle functionalization. Adv. Mater. 2017, 29, 1606209. [Google Scholar] [CrossRef]
- Jiang, Q.; Wang, K.; Zhang, X.; Ouyang, B.; Liu, H.; Pang, Z.; Yang, W. Platelet membrane-camouflaged magnetic nanoparticles for ferroptosis-enhanced cancer immunotherapy. Small 2020, 16, 2001704. [Google Scholar] [CrossRef]
- Wang, S.; Duan, Y.; Zhang, Q.; Komarla, A.; Gong, H.; Gao, W.; Zhang, L. Drug targeting via platelet membrane-coated nanoparticles. Small Struct. 2020, 1, 2000018. [Google Scholar] [CrossRef]
- Zhang, G.; Huang, X.; Xiu, H.; Sun, Y.; Chen, J.; Cheng, G.; Song, Z.; Peng, Y.; Shen, Y.; Wang, J.; et al. Extracellular vesicles: Natural liver-accumulating drug delivery vehicles for the treatment of liver diseases. J. Extracell. Vesicles 2020, 10, e12030. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Badawi, M.; Pomeroy, S.; Sutaria, D.; Xie, Z.; Baek, A.; Jiang, J.; Elgamal, O.; Mo, X.; Perle, K.; et al. Comprehensive toxicity and immunogenicity studies reveal minimal effects in mice following sustained dosing of extracellular vesicles derived from HEK293T cells. J. Extracell. Vesicles 2017, 6, 1324730. [Google Scholar] [CrossRef] [PubMed]
- Secomb, T. Blood flow in the microcirculation. Annu. Rev. Fluid Mech. 2017, 49, 443–461. [Google Scholar] [CrossRef]
- Usman, W.; Pham, T.; Kwok, Y.; Vu, L.; Ma, V.; Peng, B.; Chan, Y.; Wei, L.; Chin, S.; Azad, A.; et al. Efficient RNA drug delivery using red blood cell extracellular vesicles. Nat. Commun. 2018, 9, 2186. [Google Scholar] [CrossRef]
- Anselmo, A.; Gupta, V.; Zern, B.; Pan, D.; Zakrewsky, M.; Muzykantov, V.; Mitragotri, S. Delivering nanoparticles to lungs while avoiding liver and spleen through adsorption on red blood cells. ACS Nano 2013, 7, 11129–11137. [Google Scholar] [CrossRef] [PubMed]
- Villa, C.; Anselmo, A.; Mitragotri, S.; Muzykantov, V. Red blood cells: Supercarriers for drugs, biologicals, and nanoparticles and inspiration for advanced delivery systems. Adv. Drug Deliv. Rev. 2016, 106, 88–103. [Google Scholar] [CrossRef]
- Zhou, X.; Cao, X.; Tu, H.; Zhang, Z.R.; Deng, L. Inflammation-targeted delivery of celastrol via neutrophil membrane-coated nanoparticles in the management of acute pancreatitis. Mol. Pharm. 2019, 16, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Wang, C. Platelet-derived extracellular vesicles for drug delivery. Biomater. Sci. 2023, 11, 5758–5768. [Google Scholar] [CrossRef]
- Ying, M.; Zhuang, J.; Wei, X.; Zhang, X.; Zhang, Y.; Jiang, Y.; Dehaini, D.; Chen, M.; Gu, S.; Gao, W.; et al. Remote-Loaded Platelet Vesicles for Disease-Targeted Delivery of Therapeutics. Adv. Funct. Mater. 2018, 28, 1801032. [Google Scholar] [CrossRef]
- Song, Y.; Huang, Z.; Liu, X.; Pang, Z.; Chen, J.; Yang, H.; Zhang, N.; Cao, Z.; Liu, M.; Cao, J.; et al. Platelet membrane-coated nanoparticle-mediated targeting delivery of Rapamycin blocks atherosclerotic plaque development and stabilizes plaque in apolipoprotein E-deficient mice. Nanomed. Nanotechnol. Biol. Med. 2019, 15, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Ben-Bassat, I.; Bensch, K.; Schrier, S. Drug-induced erythrocyte membrane internalization. J. Clin. Investig. 1972, 51, 1833–1844. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Koleva, L.; Bort, E.; Atuallakhanov, F.; Sinauridze, E. Erythrocytes as Carriers: From Drug Delivery to Biosensors. Pharmaceutics 2020, 12, 276. [Google Scholar] [CrossRef]
- Bourgeaux, V.; Lanao, J.; Bax, B.; Godfrin, Y. Drug-loaded erythrocytes: On the road toward marketing approval. Drug Des. Devel. Ther. 2016, 10, 665–676. [Google Scholar] [CrossRef] [PubMed]
- Ihler, G.; Glew, R.; Schnure, F. Enzyme loading of erythrocytes. Proc. Natl. Acad. Sci. USA 1973, 70, 2663–2666. [Google Scholar] [CrossRef]
- Tsai, R.; Discher, D. Inhibition of “self” engulfment through deactivation of myosin-II at the phagocytic synapse between human cells. J. Cell Biol. 2008, 180, 989–1003. [Google Scholar] [CrossRef]
- Villa, C.; Pan, D.; Johnston, I.; Greineder, C.; Walsh, L.; Hood, E.; Cines, D.; Poncz, M.; Siegel, D.; Muzykantov, V. Biocompatible coupling of therapeutic fusion proteins to human erythrocytes. Blood Adv. 2018, 2, 165–176. [Google Scholar] [CrossRef]
- Wang, S.; Guo, H.; Li, Y.; Li, X. Penetration of nanoparticles across a lipid bilayer: Effects of particle stiffness and surface hydrophobicity. Nanoscale 2019, 11, 4025–4034. [Google Scholar] [CrossRef]
- Dai, J.; Chen, Z.; Wang, S.; Xia, F.; Lou, X. Erythrocyte membrane-camouflaged nanoparticles as effective and biocompatible platform: Either autologous or allogeneic erythrocyte-derived. Mater. Today Bio 2022, 15, 100279. [Google Scholar] [CrossRef]
- Pan, Y.; He, Y.; Zhao, X.; Pan, Y.; Meng, X.; Lv, Z.; Hu, Z.; Mou, X.; Cai, Y. Engineered Red Blood Cell Membrane-Coating Salidroside/Indocyanine Green Nanovesicles for High-Efficiency Hypoxic Targeting Phototherapy of Triple-Negative Breast Cancer. Adv. Healthc. Mater. 2022, 11, e2200962. [Google Scholar] [CrossRef]
- Brenner, J.S.; Pan, D.C.; Myerson, J.W.; Marcos-Contreras, O.A.; Villa, C.H.; Patel, P.; Heikerski, H.; Chatterjee, S.; Tao, J.Q.; Parhiz, H.; et al. Red blood cell-hitchhiking boosts delivery of nanocarriers to chosen organs by orders of magnitude. Nat. Commun. 2018, 9, 2684. [Google Scholar] [CrossRef]
- Zhao, Z.; Ukidve, A.; Gao, Y.; Kim, J.; Mitragotri, S. Erythrocyte leveraged chemotherapy (ELecT): Nanoparticle assembly on erythrocyte surface to combat lung metastasis. Sci. Adv. 2019, 5, eaax9250. [Google Scholar] [CrossRef]
- Wang, S.; Ma, S.; Li, R.; Qi, X.; Han, K.; Guo, L.; Li, X. Probing the Interaction Between Supercarrier RBC Membrane and Nanoparticles for Optimal Drug Delivery. J. Mol. Biol. 2023, 435, 167539. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, Y.; Xin, J.; Zhang, Y.; Yang, K.; Luo, Y.; Wang, B. Red blood cells in biology and translational medicine: Natural vehicle inspires new biomedical applications. Theranostics 2024, 14, 220–248. [Google Scholar] [CrossRef]
- Yoo, J.; Irvine, D.; Discher, D.; Mitragotri, S. Bio-inspired, bioengineered and biomimetic drug delivery carriers. Nat. Rev. Drug Discov. 2011, 10, 521–535. [Google Scholar] [CrossRef]
- Kang, T.; Zhu, Q.; Wei, D.; Feng, J.; Yao, J.; Jiang, T.; Song, Q.; Wei, X.; Chen, H.; Gao, X.; et al. Nanoparticles coated with neutrophil membranes can effectively treat cancer metastasis. ACS Nano 2017, 11, 1397–1411. [Google Scholar] [CrossRef] [PubMed]
- Murciano, J.; Al-Roof Higaai, A.; Cines, D.; Muzykantov, V. Soluble urokinase receptor conjugated to carrier red blood cells binds latent pro-urokinase and alters its functional profile. J. Control. Release 2009, 139, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Muzykantov, V.; Sakharov, D.; Smirnov, M.; Domogatsky, S.; Samokhin, G. Targeting of enzyme immobilized on erythrocyte membrane to collagen-coated surface. FEBS Lett. 1985, 182, 62–66. [Google Scholar] [CrossRef]
- Alapan, Y.; Bozuyuk, U.; Erkoc, P.; Karacakol, A.; Sitti, M. Multifunctional surface microrollers for targeted cargo delivery in physiological blood flow. Sci. Robot. 2020, 5, eaba5726. [Google Scholar] [CrossRef] [PubMed]
- Bozuyuk, U.; Alapan, Y.; Aghakhani, A.; Yunusa, M.; Sitti, M. Shape anisotropy-governed locomotion of surface microrollers on vessel-like microtopographies against physiological flows. Proc. Natl. Acad. Sci. USA 2021, 118, e2022090118. [Google Scholar] [CrossRef] [PubMed]
- Aybar Tural, G.; Bozuyuk, U.; Dogan, N.O.; Alapan, Y.; Akolpoglu, M.B.; Aghakhani, A.; Lazovic, J.; Ozer, O.; Sitti, M. Magnetic Mesoporous Janus Microrollers for Combined Chemo- and Photothermal Ablation Therapy. Adv. Ther. 2024, 7, 2300319. [Google Scholar] [CrossRef]
- Jiang, Y.; Wei, Z.Y.; Song, Z.F.; Qian, H.Y. Platelet-inspired targeting delivery for coronary heart disease. Heliyou 2024, 10, e27166. [Google Scholar] [CrossRef]
- Hammel, P.; Fabienne, P.; Mineur, L.; Metges, J.; Andre, T.; De La Fouchardiere, C.; Louvet, C.; El Hajbi, F.; Faroux, R.; Guimbaud, R.; et al. Erythrocyte-encapsulated asparaginase (eryaspace) combined with chemotherapy in second-line treatment of advanced pancreatic cancer: An open-label, randomized Phase IIb trial. Eur. J. Cancer 2020, 124, 91–101. [Google Scholar] [CrossRef]
- Huang, Y.; Yu, L.; Ren, J.; Wang, L.; Yang, W.; Zhang, L.; Zhang, L.; Chen, X.; Ma, J.; Li, Y.; et al. An activated-platelet-sensitive nano-carrier enables targeted delivery of tissue plasminogen activator for effective thrombolytic therapy. J. Control. Release 2019, 300, 1–12. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Q.; Li, C.; Zhou, Z.; Lei, H.; Liu, M.; Zhang, D. Advances in cell membrane-based biomimetic nanodelivery systems for natural products. Drug Deliv. 2024, 31, 2361169. [Google Scholar] [CrossRef]
- Zhang, R.; Wu, S.; Ding, Q.; Fan, Q.; Dai, Y.; Guo, S.; Ye, Y.; Li, C.; Zhou, M. Recent advances in cell membrane-camouflaged nanoparticles for inflammation therapy. Drug Deliv. 2021, 28, 1109–1119. [Google Scholar] [CrossRef]
- Li, Y.; Raza, F.; Liu, Y.; Wei, Y.; Rong, R.; Zheng, M.; Yuan, W.; Su, J.; Qiu, M.; Li, Y.; et al. Clinical progress and advanced research of red blood cells based drug delivery system. Biomaterials 2021, 279, 121202. [Google Scholar] [CrossRef]
- Wibroe, P.; Anselmo, A.; Nilsson, P.; Sarode, A.; Gupta, V.; Urbanics, R.; Szebeni, J.; Hunter, A.; Mitragotri, S.; Mollnes, T.; et al. Bypassing adverse injection reactions to nanoparticles through shape modification and attachment to erythrocytes. Nat. Nanotechnol. 2017, 12, 589–594. [Google Scholar] [CrossRef]
- Ukidve, A.; Zhao, Z.; Fehnel, A.; Krishnan, V.; Pan, D.; Gao, Y.; Mandal, A.; Muzykantov, V.; Mitragotri, S. Erythrocyte-driven immunization via biomimicry of their natural antigen-presenting function. Proc. Natl. Acad. Sci. USA 2020, 117, 17727–17736. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, C.; Wang, Y.; Chen, H.; Zhang, X.; Luo, C.; Zhou, W.; Li, L.; Teng, L.; Yu, H.; et al. Smart drug delivery systems for precise cancer therapy. Acta Pharm. Sin. B 2022, 12, 4098–4121. [Google Scholar] [CrossRef]
- Li, J.; Ai, Y.; Wang, L.; Bu, P.; Sharkey, C.; Wu, Q.; Wun, B.; Roy, S.; Shen, X.; King, M. Targeted drug delivery to circulating tumor cells via platelet membrane-functionalized particles. Biomaterials 2016, 76, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, H.; Wang, J.; Hu, X.; Zhang, L.; Chen, B. Platelet-Vesicles-Encapsulated RSL-3 Enable Anti-Angiogenesis and Induce Ferroptosis to Inhibit Pancreatic Cancer Progress. Front. Endocrinol. 2022, 13, 841461. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, Q.; Liu, L.; Wang, Y.; Wang, L.; Zhang, L.; Zhang, L.; Li, Y.; Li, Z.; Zhang, Y.; et al. Double-sided effect of tumor microenvironment on platelets targeting nanoparticles. Biomaterials 2018, 183, 258–267. [Google Scholar] [CrossRef]
- She, S.; Li, Q.; Shan, B.; Tong, W.; Gao, C. Fabrication of red-blood-cell-like polyelectrolyte microcapsules and their deformation and recovery behavior through a microcapillary. Adv. Mater. 2013, 25, 5814–5818. [Google Scholar] [CrossRef]
- Zhang, N.; Li, M.; Yang, Y.; Wang, Z.; Chen, X.; Liu, J. Application of therapeutical nanoparticles with neutrophil membrane camouflaging for inflammatory plaques targeting against atherosclerosis. Mater. Today Bio 2024, 29, 101397. [Google Scholar] [CrossRef] [PubMed]
- Biagiotti, S.; Rossi, L.; Bianchi, M.; Giacomini, E.; Pierigè, F.; Serafini, G.; Conaldi, P.; Magnani, M. Immunophilin-loaded erythrocytes as a new delivery strategy for immunosuppressive drugs. J. Control. Release 2011, 154, 306–313. [Google Scholar] [CrossRef]
- Bustamante Lopez, S.C.; Meissner, K.E. Characterization of carrier erythrocytes for biosensing applications. J. Biomed. Opt. 2017, 22, 091510. [Google Scholar] [CrossRef]
- Chang, M.; Hsiao, J.; Yao, M.; Chien, L.; Hsu, S.; Ko, B.; Chen, S.; Liu, H.; Chen, Y.; Yang, C.; et al. Homologous RBC-derived vesicles as ultrasmall carriers of iron oxide for magnetic resonance imaging of stem cells. Nanotechnology 2010, 21, 235103. [Google Scholar] [CrossRef]
- Cremel, M.; Guerin, N.; Campello, G.; Barthe, Q.; Berlier, W.; Horand, F.; Godfrin, Y. Innovative approach in Pompe disease therapy: Induction of immune tolerance by antigen-encapsulated red blood cells. Int. J. Pharm. 2015, 491, 69–77. [Google Scholar] [CrossRef]
- DeLoach, J.; Barton, C. Glutaraldehyde-treated carrier erythrocytes for organ targeting of methotrexate in dogs. Am. J. Vet. Res. 1981, 42, 1971–1974. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Agola, J.; Serda, R.; Franco, S.; Qi, L.; Wang, L.; Minster, J.; Croissant, J.; Butler, K.; Zhu, W.; et al. Biomimetic rebuilding of multifunctional red blood cells: Modular design using functional components. ACS Nano 2020, 14, 7847–7859. [Google Scholar] [CrossRef]
- Ho-Tin-Noé, B.; Demers, M.; Wagner, D. How platelets safeguard vascular integrity. J. Thromb. Haemost. 2011, 9, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Hunault-Berger, M.; Leguay, T.; Huguet, F.; Lepretre, S.; Deconinck, E.; Ojeda-Uribe, M.; Bonmati, C.; Escoffre-Barbe, M.; Bories, P.; Himberlin, C.; et al. A Phase 2 study of L-asparaginase encapsulated in erythrocytes in elderly patients with Philadelphia chromosome negative acute lymphoblastic leukemia: The GRASPALL/GRAALL-SA2-2008 study. Am. J. Hematol. 2015, 90, 811–818. [Google Scholar] [CrossRef]
- Ju, X.; Wang, X.; Liu, Z.; Xie, R.; Wang, W.; Chu, L. Red-blood-cell-shaped chitosan microparticles prepared by electrospraying. Particuology 2017, 30, 151–157. [Google Scholar] [CrossRef]
- Nie, X.; Liu, Y.; Yao, X.; Zhang, Q.; Huang, Y.; Mattursun, K.; Feng, Y.; Liang, T.; Yang, L.; Gao, X. Erythrocyte–anti-PD1 conjugates in persons with advanced solid tumors resistant to anti-PD1/PDL1: Preclinical characterization and results of a phase 1 trial. Nat. Cancer, 2026; in press. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, D.; Fang, G.; Wang, S. Erythrocyte membrane biosinspired near-infrared persistent luminescence nanocarriers for in vivo long-circulating bioimaging and drug delivery. Biomaterials 2018, 165, 39–47. [Google Scholar] [CrossRef]
- Rossi, L.; Serafini, S.; Pierigé, F.; Antonelli, A.; Cerasi, A.; Fraternale, A.; Chiarantini, L.; Magnani, M. Erythrocyte-based drug delivery. Expert Opin. Drug Deliv. 2005, 18, 965–982. [Google Scholar] [CrossRef]
- Wang, Q.; Ma, Y.; Lu, Z.; Yu, H.; Li, Z. Co-delivery of chemotherapeutic drugs and immune checkpoint inhibitors by engineered erythrocytes for synergistic cancer therapy. ACS Appl. Nano Mater. 2022, 5, 101–106. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Z.; Zhang, Y.; Guan, Y.; Liu, Z.; Ren, J.; Qu, X. Artificial metalloenzyme-based enzyme replacement therapy for the treatment of hyperuricemia. Adv. Funct. Mater. 2016, 26, 7921–7928. [Google Scholar] [CrossRef]
- Yamagata, K.; Kawasaki, E.; Kawarai, H.; Iino, M. Encapsulation of Concentrated Protein Into Erythrocyte Ported by Continuous-Wave Ultrasound. Ultrasound Med. Biol. 2008, 34, 1924–1933. [Google Scholar] [CrossRef]
- Wang, J.; Tang, W.; Yang, M.; Yin, Y.; Li, H.; Hu, F.; Tang, L.; Ma, X.; Zhang, Y.; Wang, Y. Inflammatory tumor microenvironment responsive neutrophil exosomes-based drug delivery system for targeted glioma therapy. Biomaterials 2021, 273, 120784. [Google Scholar] [CrossRef] [PubMed]
- Gomes, F.L.; Jeong, S.H.; Shin, S.R.; Leijten, J.; Jonkheijm, P. Engineering Synthetic Erythrocytes as Next-Generation Blood Substitutes. Adv. Funct. Mater. 2024, 34, 2315879. [Google Scholar] [CrossRef] [PubMed]
- Biagiotti, S.; Canonico, B.; Tiboni, M.; Abbas, F.; Perla, E.; Montanari, M.; Battistelli, M.; Papa, S.; Casettari, L.; Rossi, L.; et al. Efficient and highly reproducible production of red blood cell-derived extracellular vesicle mimetics for the loading and delivery of RNA molecules. Sci. Rep. 2024, 14, 14610. [Google Scholar] [CrossRef]
- Kweon, S.; Kim, S.; Baek, E.J. Current status of red blood cell manufacturing in 3D culture and bioreactors. Blood Res. 2023, 58, S46–S51. [Google Scholar] [CrossRef] [PubMed]
- Magnani, M.; Chiarantini, L.; Mancini, U. Preparation and characterization of biotinylated red blood cells. Biotechnol. Appl. Biochem. 1994, 20, 335–345. [Google Scholar] [CrossRef]
- Park, J.; Jiang, Y.; Zhou, J.; Gong, H.; Mohanty, A.; Goh, C.; Tang, J.; Zhang, B.; Liu, W.; Ruan, J.; et al. Genetically engineered cell membrane-coated nanoparticles for targeted delivery of dexamethasone to inflamed lungs. Sci. Adv. 2021, 7, eabf7820. [Google Scholar] [CrossRef]
- Zhu, K.; Huang, Y.; Li, K.; Zhang, K. Biomimetic erythrocyte-based drug delivery systems for organ-targeted therapy. Front. Bioeng. Biotechnol. 2025, 13, 1663092. [Google Scholar] [CrossRef]
- Jangid, A.K.; Kim, S.; Kim, K. Polymeric biomaterial-inspired cell surface modulation for the development of novel anticancer therapeutics. Biomater. Res. 2023, 27, 59. [Google Scholar] [CrossRef]
- Dai, Z.; Zhao, T.; Song, N.; Pan, K.; Yang, Y.; Zhu, X.; Chen, P.; Zhang, J.; Xia, C. Platelets and platelet extracellular vesicles in drug delivery therapy: A review of the current status and future prospects. Front. Pharmacol. 2022, 13, 1026386. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, R.; Tian, X.; Zhang, Y.; Liu, X.; Li, Z. A novel bispecific antibody fusion protein co-targeting EGFR and CD47 with enhanced therapeutic index. Biotechnol. Lett. 2018, 40, 789–795. [Google Scholar] [CrossRef]
- Heemskerk, N.; Gruijs, M.; Temming, A.R.; Heineke, M.H.; Gout, D.Y.; Hellingman, T.; Tuk, C.W.; Winter, P.J.; Lissenberg-Thunnissen, S.; Bentlage, A.E.H.; et al. Augmented antibody-based anticancer therapeutics boost neutrophil cytotoxicity. J. Clin. Investig. 2021, 131, e134680. [Google Scholar] [CrossRef]
- Chapanian, R.; Constantinescu, I.; Brooks, D.E.; Scott, M.D.; Kizhakkedathu, J.N. Antigens protected functional red blood cells by the membrane grafting of compact hyperbranched polyglycerols. J. Vis. Exp. 2013, 71, e50075. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, R.; Meng, N.; Guo, H.; Zhang, S.; Ren, J.; Liu, F.; Lu, W.; Liu, M. Platelet membrane-functionalized nanoparticles with improved targeting ability and lower hemorrhagic risk for thrombolysis therapy. J. Control. Release 2020, 328, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, R.; Meng, N.; Lu, L.; Wang, J.; Zhou, J.; Lu, J.; Xu, Q.; Xie, C.; Zhan, C.; et al. Engineered platelets-based drug delivery platform for targeted thrombolysis. Acta Pharm. Sin. B 2022, 12, 2000–2013. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Li, X.; Wang, Y.; Liu, C.; Ling, G.; Zhang, P. Biomimetic platelet-camouflaged drug-loaded polypyrrole for the precise targeted antithrombotic therapy. J. Nanobiotechnology 2023, 21, 439. [Google Scholar] [CrossRef]
- Shan, L.; Wang, J.; Tu, H.; Zhang, W.; Li, H.; Slezak, P.; Lu, F.; Lee, D.; Hu, E.; Geng, Z.; et al. Drug delivery under cover of erythrocytes extends drug half-life: A thrombolytic targeting therapy utilizing microenvironment-responsive artificial polysaccharide microvesicles. Carbohydrate Polymers 2024, 343, 122505. [Google Scholar] [CrossRef]
- Friedman, S.H. The Photoactivated Depot (PAD): Light Triggered Control of Therapeutic Protein Solubility and Release. Acc. Chem. Res. 2025, 58, 461–472. [Google Scholar] [CrossRef]
- Pei, W.; Li, X.; Bi, R.; Zhang, X.; Zhong, M.; Yang, H.; Zhang, Y.; Lv, K. Exosome membrane-modified M2 macrophages targeted nanomedicine: Treatment for allergic asthma. J. Control. Release 2021, 338, 253–267. [Google Scholar] [CrossRef]
- Mura, S.; Nicolas, J.; Couvreur, P. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 2013, 12, 991–1003. [Google Scholar] [CrossRef]
- Karimi, M.; Ghasemi, A.; Sahandi Zangabad, P.; Rahighi, R.; Moosavi Basri, S.; Mirshekari, H.; Amiri, M.; Pishabad, Z.; Aslani, A.; Bozorgomid, M.; et al. Smart micro/nanoparticles in stimulus-responsive drug/gene delivery systems. Chem. Soc. Rev. 2016, 45, 1457–1501. [Google Scholar] [CrossRef] [PubMed]
- Nomikou, N.; Masood, H.; Patel, S. Stimulus-Responsive Nanoparticles for Drug Delivery. In Fundamentals of Pharmaceutical Nanoscience; Uchegbu, I.F., Schätzlein, A.G., Lalatsa, A., Lopez, D.R.S., Eds.; Springer Nature: Cham, Switzerland, 2024; pp. 389–422. [Google Scholar] [CrossRef]



| Carrier Type | Biocompatibility | Stability | Drug-Loading Capacity | Circulation Duration |
|---|---|---|---|---|
| Nanoparticle-based DDS | Variable (material-dependent) | High (but degradation-sensitive) | High | Short (rapid RES clearance) |
| Liposome-based DDS | High (biomimetic membrane) | Moderate (oxidation-sensitive) | Moderate to High | Moderate (improved by PEGylation) |
| Blood Cell-based DDS | High (autologous, ‘self’ markers) | High (but sensitive to handling) | Moderate (cell volume limited) | Long (comparable to natural cell lifespan) |
| Carrier Type | Core Advantages | Key Challenges | Main Applications |
|---|---|---|---|
| RBCs | Long circulation time, high biocompatibility, large loading capacity | Limited targeting capability, modifications may affect lifespan | Chronic diseases, enzyme replacement therapy, palliative care for solid tumors |
| WBCs | Active inflammation/tumor homing, deep tissue penetration | Complex isolation and modification, potential immunosuppression | Autoimmune diseases, infections, solid tumors (esp. hypoxic regions) |
| Platelets | Natural injury/thrombus targeting, ability to cross biological barriers | Relatively short lifespan, demanding storage requirements | Thrombotic diseases, tumors (esp. pancreatic cancer, glioblastoma) |
| Blood Cell-derived Vesicles | Low immunogenicity, retains source cell targeting, easy to modify | Heterogeneity, difficulties in isolation and purification | Neurological disorders, cancer, inflammatory diseases |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Xu, G.; Li, X. Blood Cell-Based Drug Delivery Systems: From Biological and Mechanical Design to Clinical Applications. Bioengineering 2026, 13, 340. https://doi.org/10.3390/bioengineering13030340
Xu G, Li X. Blood Cell-Based Drug Delivery Systems: From Biological and Mechanical Design to Clinical Applications. Bioengineering. 2026; 13(3):340. https://doi.org/10.3390/bioengineering13030340
Chicago/Turabian StyleXu, Gang, and Xuejin Li. 2026. "Blood Cell-Based Drug Delivery Systems: From Biological and Mechanical Design to Clinical Applications" Bioengineering 13, no. 3: 340. https://doi.org/10.3390/bioengineering13030340
APA StyleXu, G., & Li, X. (2026). Blood Cell-Based Drug Delivery Systems: From Biological and Mechanical Design to Clinical Applications. Bioengineering, 13(3), 340. https://doi.org/10.3390/bioengineering13030340







