Mastoid Obliteration After Canal Wall Down Mastoidectomy Using Tissue Engineering Approaches with Polymers, Mesenchymal Stem Cells, and Bioactive Molecules: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Reporting Standards
2.2. Eligibility Criteria (PICO Framework)
2.3. Information Sources and Search Strategy
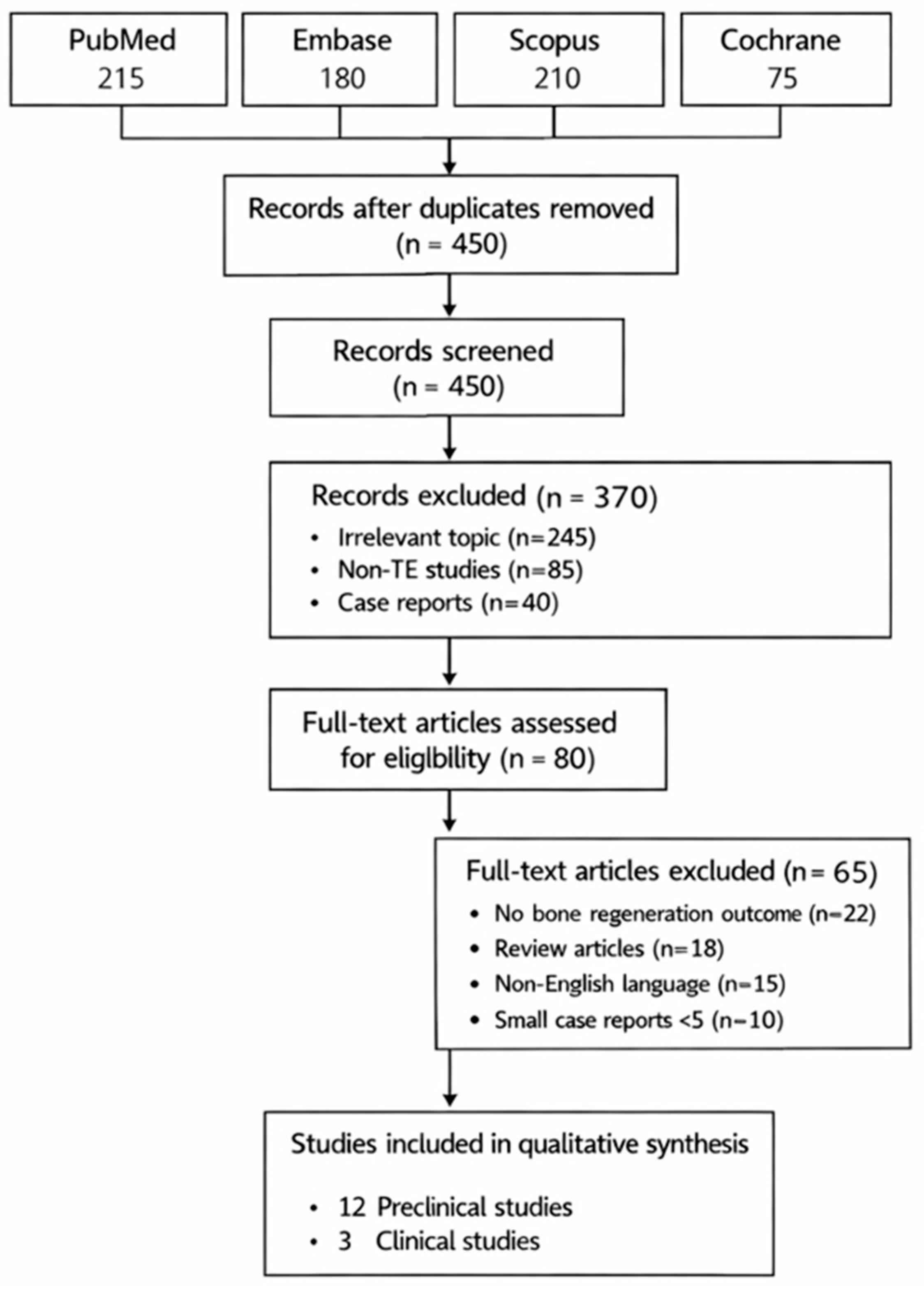
3. Results
3.1. Study Selection
3.2. Growth Factor-Augmented Mastoid Obliteration
3.3. Cell-Based Approaches
3.4. Polymer-Based Scaffolds and Carriers
3.5. 3D-Printed Architected Scaffolds
3.6. Critical Analysis of Heterogeneity
4. Discussion
4.1. Current Status of Mastoid Obliteration
4.2. Limitations of Current Tissue Engineering-Based Mastoid Obliteration
4.3. Future Directions: Toward Advanced Regenerative Mastoid Obliteration
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, M.; Zhang, L.; Zhang, Q.; Zeng, N.; Li, S.; Guo, S.; Zhao, Y.; Tang, M.; Yang, Q. Concentrated growth factors promote epithelization in the mastoid obliteration after canal wall down mastoidectomy. Braz. J. Otorhinolaryngol. 2025, 91, 101561. [Google Scholar] [CrossRef] [PubMed]
- Faita, A.; Giannì, E.; di Pasquale Fiasca, V.M. The effect of external auditory canal reconstruction with mastoid obliteration after canal wall down tympanomastoidectomy on hearing function: Narrative review and case series. Audiol. Foniatr. 2025, 10, 14–22. [Google Scholar]
- Elbary, M.E.-S.A.; Khaled, I.A.; El-Anwar, M.W.; Abdelhamid, H.I. Effect of platelet rich plasma on healing of mastoid cavity after canal wall down mastoidectomy. Eur. Arch. Oto-Rhino-Laryngol. 2025, 282, 6069–6072. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, M.; Zhang, Q.; Zeng, N.; Hu, J.; Cf, Z.; Yang, Q. Effect of hydroxyapatite combined with concentrated growth factor on mastoid cavity filling during endoscopic surgery for middle ear cholesteatoma. Zhonghua Yi Xue Za Zhi 2024, 104, 4391–4396. [Google Scholar]
- Jang, C.H.; Kim, M.S.; Cho, Y.B.; Jang, Y.S.; Kim, G.H. Mastoid obliteration using 3D PCL scaffold in combination with alginate and rhBMP-2. Int. J. Biol. Macromol. 2013, 62, 614–622. [Google Scholar] [CrossRef]
- Mangani, S.; Vetoulas, M.; Mineschou, K.; Spanopoulos, K.; Vivanco, M.d.; Piperigkou, Z.; Karamanos, N.K. Design and applications of extracellular matrix scaffolds in tissue engineering and regeneration. Cells 2025, 14, 1076. [Google Scholar] [CrossRef]
- Fan, J.; Ding, Z.; Cai, Y.; Lai, Y.; Huang, C.; Jiang, B.; Zhou, Z.; Luo, Z. Revolutionizing Bone Regeneration: Vascularized Bone Tissue Engineering with Advanced 3D Printing Technology. Aggregate 2025, 6, e731. [Google Scholar] [CrossRef]
- Abedi, N.; Sadeghian, A.; Kouhi, M.; Haugen, H.J.; Savabi, O.; Nejatidanesh, F. Immunomodulation in bone tissue engineering: Recent advancements in scaffold design and biological modifications for enhanced regeneration. ACS Biomater. Sci. Eng. 2025, 11, 1269–1290. [Google Scholar] [CrossRef]
- Chen, K.; Luo, L.; Tao, R.; Li, M.; Qu, S.; Wu, X.; Zhang, X.; Feng, H.; Zhu, Z.; Zhang, D. 3D-Printed PCL/SrHA@ DFO Bone Tissue Engineering Scaffold with Bone Regeneration and Vascularization Function. ACS Appl. Bio Mater. 2025, 8, 1684–1698. [Google Scholar] [CrossRef]
- Rau, J.V.; Antoniac, I.; Cama, G.; Komlev, V.S.; Ravaglioli, A. Bioactive materials for bone tissue engineering. BioMed Res. Int. 2016, 2016, 3741428. [Google Scholar] [CrossRef]
- Swetha, S.; Lavanya, K.; Sruthi, R.; Selvamurugan, N. An insight into cell-laden 3D-printed constructs for bone tissue engineering. J. Mater. Chem. B 2020, 8, 9836–9862. [Google Scholar] [CrossRef]
- Picado-Tejero, D.; Mendoza-Cerezo, L.; Rodríguez-Rego, J.M.; Carrasco-Amador, J.P.; Marcos-Romero, A.C. Recent advances in 3D bioprinting of porous scaffolds for tissue engineering: A narrative and critical review. J. Funct. Biomater. 2025, 16, 328. [Google Scholar] [CrossRef]
- Arias-Peregrino, V.M.; Tenorio-Barajas, A.Y.; Mendoza-Barrera, C.O.; Román-Doval, J.; Lavariega-Sumano, E.F.; Torres-Arellanes, S.P.; Román-Doval, R. 3D printing for tissue engineering: Printing techniques, biomaterials, challenges, and the emerging role of 4D bioprinting. Bioengineering 2025, 12, 936. [Google Scholar] [CrossRef]
- Rama, M.; Suresh, A.S.; John, A.E.; Vijaylal, L.; Vijayalakshmi, U. Revolutionizing tissue engineering: Integrating 3D bioprinting and additive manufacturing for precision scaffold design and biomimetic tissue regeneration. Mater. Technol. 2025, 40, 2530639. [Google Scholar] [CrossRef]
- Askar, S.M.; Saber, I.M.; Omar, M. Mastoid reconstruction with platelet-rich plasma and bone pate after canal wall down mastoidectomy: A preliminary report. Ear Nose Throat J. 2021, 100, 485–489. [Google Scholar]
- Abd Elbary, M.E.-S.; Nasr, W.F.; Sorour, S.S. Platelet-rich plasma in reconstruction of posterior meatal wall after canal wall down mastoidectomy. Int. Arch. Otorhinolaryngol. 2018, 22, 103–107. [Google Scholar]
- Jang, C.H.; Cho, Y.B.; Choi, C.H.; Jang, Y.S.; Jung, W.K.; Lee, H.; Kim, G.H. Effect of umbilical cord serum coated 3D PCL/alginate scaffold for mastoid obliteration. Int. J. Pediatr. Otorhinolaryngol. 2014, 78, 1061–1065. [Google Scholar] [CrossRef]
- Jang, C.H.; Kim, W.; Kim, G. Effects of fibrous collagen/CDHA/hUCS biocomposites on bone tissue regeneration. Int. J. Biol. Macromol. 2021, 176, 479–489. [Google Scholar] [CrossRef]
- Jang, C.H.; Ahn, S.H.; Yang, G.-H.; Kim, G.H. A MSCs-laden polycaprolactone/collagen scaffold for bone tissue regeneration. RSC Adv. 2016, 6, 6259–6265. [Google Scholar] [CrossRef]
- Choi, S.W.; Kang, J.; Wang, C.; Lee, H.M.; Oh, S.J.; Pak, K.; Shin, N.; Lee, I.W.; Lee, J.; Kong, S.K. Human Tonsil-Derived Mesenchymal Stem Cells-Loaded Hydroxyapatite-Chitosan Patch for Mastoid Obliteration. ACS Appl. Bio Mater. 2020, 3, 1008–1017. [Google Scholar] [CrossRef]
- Skoloudik, L.; Chrobok, V.; Kalfert, D.; Koci, Z.; Sykova, E.; Chumak, T.; Popelar, J.; Syka, J.; Laco, J.; Dedková, J. Human multipotent mesenchymal stromal cells in the treatment of postoperative temporal bone defect: An animal model. Cell Transplant. 2016, 25, 1405–1414. [Google Scholar] [CrossRef]
- Park, S.-H.; Kim, H.; Lee, Y.Y.; Kim, Y.J.; Jang, J.H.; Choo, O.-S.; Choung, Y.-H. Development of intracorporeal differentiation of stem cells to induce one-Step mastoid bone reconstruction during otitis media surgeries. Polymers 2022, 14, 877. [Google Scholar] [CrossRef]
- Yu, F.; Fan, X.; Wu, H.; Ou, Y.; Zhao, X.; Chen, T.; Qian, Y.; Kang, H. Mastoid obliteration and external auditory canal reconstruction using 3D printed bioactive glass S53P4/polycaprolactone scaffold loaded with bone morphogenetic protein-2: A simulation clinical study in rabbits. Regen. Ther. 2022, 21, 469–476. [Google Scholar] [PubMed]
- Jang, C.H.; Lee, J.; Kim, G. Synergistic effect of alginate/BMP-2/Umbilical cord serum-coated on 3D-printed PCL biocomposite for mastoid obliteration model. J. Ind. Eng. Chem. 2019, 72, 432–441. [Google Scholar] [CrossRef]
- Lee, J.; Kim, D.; Jang, C.H.; Kim, G.H. Highly elastic 3D-printed gelatin/HA/placental-extract scaffolds for bone tissue engineering. Theranostics 2022, 12, 4051–4066. [Google Scholar] [CrossRef]
- Kim, W.; Jang, C.H.; Kim, G. Bone tissue engineering supported by bioprinted cell constructs with endothelial cell spheroids. Theranostics 2022, 12, 5404–5417. [Google Scholar] [CrossRef] [PubMed]
- Jang, C.H.; Cho, Y.B.; Yeo, M.G.; Kim, G.H. Mastoid obliteration using three-dimensional composite scaffolds consisting of polycaprolactone/β-tricalcium phosphate/collagen nanofibers: An in vitro and in vivo study. Macromol. Biosci. 2013, 13, 660–668. [Google Scholar] [CrossRef]
- Jang, C.H.; Choi, C.H.; Cho, Y.B. Effect of BMP2–Platelet-rich plasma–biphasic calcium phosphate scaffold on accelerated osteogenesis in mastoid obliteration. In Vivo 2016, 30, 835–839. [Google Scholar] [CrossRef]
- Perri, B.; Cooper, M.; Lauryssen, C.; Anand, N. Adverse swelling associated with use of rh-BMP-2 in anterior cervical discectomy and fusion: A case study. Spine J. 2007, 7, 235–239. [Google Scholar] [CrossRef]
- Christensen, T.J.; Annis, P.; Hohl, J.B.; Patel, A.A. Neuroforaminal chondrocyte metaplasia and clustering associated with recombinant bone morphogenetic protein-2 usage in transforaminal lumbar interbody fusion. Spine J. 2014, 14, e23–e28. [Google Scholar] [CrossRef]
- Anderson, D.W.; Burton, D.C.; Jackson, R.S. Postoperative cervical myelopathy and cord compression associated with the use of recombinant bone morphogenetic protein-2 in posterior cervical decompression, instrumentation, and arthrodesis: A report of two cases. Spine 2011, 36, E682–E686. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, B.; Huang, R.; Guo, M.; Han, D.; Yin, L.; Zhang, X.; Huang, Y.; Li, X. Efforts to promote osteogenesis–angiogenesis coupling for bone tissue engineering. Biomater. Sci. 2024, 12, 2801–2830. [Google Scholar] [CrossRef]
- Bielecki, T.; Dohan Ehrenfest, D.M. Platelet-rich plasma (PRP) and Platelet-Rich Fibrin (PRF): Surgical adjuvants, preparations for in situ regenerative medicine and tools for tissue engineering. Curr. Pharm. Biotechnol. 2012, 13, 1121–1130. [Google Scholar] [CrossRef]
- Sharun, K.; Banu, S.A.; El-Husseiny, H.M.; Abualigah, L.; Pawde, A.M.; Dhama, K.; Amarpal. Exploring the applications of platelet-rich plasma in tissue engineering and regenerative medicine: Evidence from goat and sheep experimental research. Connect. Tissue Res. 2024, 65, 364–382. [Google Scholar] [CrossRef] [PubMed]
- Steiner, N.; Vozel, D.; Bozanic Urbancic, N.; Troha, K.; Lazar, A.; Kralj-Iglic, V.; Battelino, S. Enhancing the Outcomes of Temporalis Fascia Tympanoplasty Using Autologous Platelet-Rich Plasma and Gel: A Randomized Controlled Trial. J. Pers. Med. 2025, 15, 233. [Google Scholar] [CrossRef]
- Fouad, Y.A.; Abdelhady, M.; El-Anwar, M.; Merwad, E. Topical platelet rich plasma versus hyaluronic acid during fat graft myringoplasty. Am. J. Otolaryngol. 2018, 39, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Shi, Y.; Wu, L.; Lv, C.; Hu, Y.; Shen, Y. Comparative efficacy of platelet-rich plasma applied in myringoplasty: A systematic review and meta-analysis. PLoS ONE 2021, 16, e0245968. [Google Scholar] [CrossRef]
- Sharma, P.; Parida, P.K.; Preetam, C.; Mukherjee, S.; Nayak, A.; Pradhan, P. Outcome of Temporalis Fascia Myringoplasty With and Without use of Platelet Rich Plasma: A Randomized Control Trial. Indian. J. Otolaryngol. Head. Neck Surg. 2022, 74, 3832–3840. [Google Scholar] [CrossRef] [PubMed]
- Shiomi, Y.; Shiomi, Y. Surgical outcomes of myringoplasty using platelet-rich plasma and evaluation of the outcome-associated factors. Auris Nasus Larynx 2020, 47, 191–197. [Google Scholar] [CrossRef]
- Rodríguez Calvo-de-Mora, M.; Domínguez-Ruiz, C.; Barrero-Sojo, F.; Rodríguez-Moreno, G.; Antúnez Rodríguez, C.; Ponce Verdugo, L.; Hernández Lamas, M.D.C.; Hernández-Guijarro, L.; Villalvilla Castillo, J.; Fernández-Baca Casares, I.; et al. Autologous versus allogeneic versus umbilical cord sera for the treatment of severe dry eye disease: A double-blind randomized clinical trial. Acta Ophthalmol. 2022, 100, e396–e408. [Google Scholar] [CrossRef]
- Orlando, N.; Pellegrino, C.; Valentini, C.G.; Bianchi, M.; Barbagallo, O.; Sparnacci, S.; Forni, F.; Fontana, T.M.; Teofili, L. Umbilical cord blood: Current uses for transfusion and regenerative medicine. Transfus. Apher. Sci. 2020, 59, 102952. [Google Scholar] [CrossRef]
- Manivasagam, V.K.; Popat, K.C. Hydrothermally treated titanium surfaces for enhanced osteogenic differentiation of adipose derived stem cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 128, 112315. [Google Scholar] [CrossRef]
- Tanideh, N.; Sarikhani, M.; Emami, M.; Alipanah, M.; Mohammadi, Y.; Mokhtarzadegan, M.; Jamshidzadeh, A.; Zare, S.; Daneshi, S.; Feiz, A. Fabrication of porous collagen-stem cells–dexamethasone scaffold as a novel approach for regeneration of mandibular bone defect. Oral Maxillofac. Surg. 2025, 29, 65. [Google Scholar]
- Lee, S.-H.; Lee, J.; Kang, N.-U.; Cho, Y.S.; Heo, S.C.; Park, Y.; Cho, Y.-S. PCL scaffold with well-defined hierarchical pores effectively controls cell migration and alignment of human mesenchymal stem cells. Sci. Rep. 2025, 15, 11542. [Google Scholar] [CrossRef]
- Olza, S.; Bouzidi, N.M.H.; Rubatat, L.; Pellerin, V.; Montejo, U.; Alonso-Varona, A.; Fernandes, S.C. Mineralized chitin nanocrystals enhance osteoinductive ability of chitosan 3D porous biohybrid scaffolds for bone tissue regeneration. Carbohydr. Polym. 2025, 366, 123911. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, E.M.; Yamamoto, M.; Park, H.; Shin, H. Engineering Multi-Cellular Spheroids for Tissue Engineering and Regenerative Medicine. Adv. Health Mater. 2020, 9, e2000608. [Google Scholar] [CrossRef]
- Fang, Y.; Ji, M.; Wu, B.; Xu, X.; Wang, G.; Zhang, Y.; Xia, Y.; Li, Z.; Zhang, T.; Sun, W.; et al. Engineering Highly Vascularized Bone Tissues by 3D Bioprinting of Granular Prevascularized Spheroids. ACS Appl. Mater. Interfaces 2023, 15, 43492–43502. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, N.; Hamlet, S.; Love, R.M.; Nguyen, N.-T. Porous scaffolds for bone regeneration. J. Sci.: Adv. Mater. Devices 2020, 5, 1–9. [Google Scholar]
- Hasani, M.; Abdouss, M.; Shojaei, S.; Khonakdar, H.A. Controlled release in sodium alendronate/halloysite/hydroxyapatite/gelatin nanocomposite scaffolds: A new insight into bone tissue engineering. Mater. Chem. Phys. 2024, 327, 129821. [Google Scholar]
- Jessy, R.S.; Ibrahim, M.H. Biodegradability and biocompatibility of polymers with emphasis on bone scaffolding: A brief review. Int. J. Sci. Res. Publ. 2014, 4, 1–3. [Google Scholar]
- Yang, X.; Wang, Y.; Zhou, Y.; Chen, J.; Wan, Q. The application of polycaprolactone in three-dimensional printing scaffolds for bone tissue engineering. Polymers 2021, 13, 2754. [Google Scholar] [CrossRef]
- Ge, Z.; Tian, X.; Heng, B.C.; Fan, V.; Yeo, J.F.; Cao, T. Histological evaluation of osteogenesis of 3D-printed poly-lactic-co-glycolic acid (PLGA) scaffolds in a rabbit model. Biomed. Mater. 2009, 4, 021001. [Google Scholar] [CrossRef]
- Li, C.; Zhang, W.; Nie, Y.; Du, X.; Huang, C.; Li, L.; Long, J.; Wang, X.; Tong, W.; Qin, L. Time-sequential and multi-functional 3D printed MgO2/PLGA scaffold developed as a novel biodegradable and bioactive bone substitute for challenging postsurgical osteosarcoma treatment. Adv. Mater. 2024, 36, 2308875. [Google Scholar] [CrossRef]
- Keogh, M.B.; O’Brien, F.J.; Daly, J.S. A novel collagen scaffold supports human osteogenesis—Applications for bone tissue engineering. Cell Tissue Res. 2010, 340, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Paiva, S.S.; Ferreira, A.; Pakenham, E.; Kaur, K.; Cavanagh, B.; O’Brien, F.J.; Murphy, C.M. Magnesium Ion-Mediated Regulation of Osteogenesis and Osteoclastogenesis in 2D Culture and 3D Collagen/Nano-Hydroxyapatite Scaffolds for Enhanced Bone Repair. J. Funct. Biomater. 2025, 16, 363. [Google Scholar] [CrossRef] [PubMed]
- Baniameri, S.; Aminianfar, H.; Gharehdaghi, N.; Yousefi-Koma, A.A.; Mohaghegh, S.; Nokhbatolfoghahaei, H.; Khojasteh, A. Tissue Engineering 3D-Printed Scaffold Using Allograft/Alginate/Gelatin Hydrogels Coated with Platelet-Rich Fibrin or Adipose Stromal Vascular Fraction Induces Osteogenesis In Vitro. J. Cell. Physiol. 2025, 240, e31497. [Google Scholar] [CrossRef]
- Zhou, E.; He, P.; Yang, Z.; Li, C.; Fang, G.; Wu, J.; Zhuang, W.; Sang, H. 3D-printed GelMA-Alginate microsphere scaffold with staged dual-growth factor release for enhanced bone regeneration. Mater. Today Bio 2025, 35, 102422. [Google Scholar] [PubMed]
- Ferjaoui, Z.; López-Muñoz, R.; Akbari, S.; Mantovani, D.; Fanganiello, R. Design of Alginate/Gelatin Hydrogels for Craniofacial Bone Tissue Engineering: Optimizing Osteogenesis in Dental Pulp Stem Cells Without Compromising Other Cellular Functions. In Proceedings of the 1st International Online Conference on Gels; MDPI: Basel, Switzerland, 2025. [Google Scholar]
- Venkata Prathyusha, E.; Gomte, S.S.; Ahmed, H.; Prabakaran, A.; Agrawal, M.; Chella, N.; Alexander, A. Nanostructured polymer composites for bone and tissue regeneration. Int. J. Biol. Macromol. 2025, 284, 137834. [Google Scholar] [CrossRef]
- Lesko, L.; Jungova, P.; Culenova, M.; Thurzo, A.; Danisovic, L. Polymer-Based Scaffolds as an Implantable Material in Regenerative Dentistry: A Review. J. Funct. Biomater. 2025, 16, 80. [Google Scholar] [CrossRef]
- Zheng, Y.; Fu, L.; Zhang, Z.; Wu, J.; Yuan, X.; Ding, Z.; Ning, C.; Sui, X.; Liu, S.; Guo, Q. Three-dimensional bioprinting of growth differentiation factor 5-preconditioned mesenchymal stem cell-derived exosomes facilitates articular cartilage endogenous regeneration. ACS Nano 2025, 19, 15281–15301. [Google Scholar]
- Noh, J.H.; Lee, Y.; Kim, M.S. 3D-printed scaffolds: Incorporating dexamethasone microspheres and BMP2 for enhanced osteogenic differentiation of human mesenchymal stem cells. Colloids Surf. B Biointerfaces 2025, 253, 114705. [Google Scholar] [CrossRef]
- Marin-Tapia, H.A.; Romero-Salazar, L.; Mayorga-Rojas, M.; Arteaga-Arcos, J.C. Scaffold-Free Extrusion-Based 3D Bioprinting of Cornea Constructs Using a Decellularized Corneal Extracellular Matrix Based Bioink and Human Placenta-Derived Mesenchymal Stem Cells. Macromol. Biosci. 2025, 25, e00276. [Google Scholar]
- Domingues, M.F.; Carvalho, M.S.; Sanjuan-Alberte, P.; Silva, J.C. Synthetic scaffolds functionalized with mesenchymal stem/stromal cells-derived extracellular matrix for bone tissue engineering: A review. RSC Adv. 2025, 15, 31812–31829. [Google Scholar] [CrossRef]
- Klabukov, I.; Shestakova, V.; Garifullin, A.; Yakimova, A.; Baranovskii, D.; Yatsenko, E.; Ignatyuk, M.; Atiakshin, D.; Shegay, P.; Kaprin, A.D. Critical Systematic Review of 3D Bioprinting in Biomedicine. Int. J. Mol. Sci. 2025, 26, 11882. [Google Scholar] [CrossRef]
- de Carvalho, A.B.G.; Rahimnejad, M.; Oliveira, R.; Sikder, P.; Saavedra, G.; Bhaduri, S.B.; Gawlitta, D.; Malda, J.; Kaigler, D.; Trichês, E.S.; et al. Personalized bioceramic grafts for craniomaxillofacial bone regeneration. Int. J. Oral Sci. 2024, 16, 62. [Google Scholar] [CrossRef] [PubMed]
- Latimer, J.M.; Maekawa, S.; Yao, Y.; Wu, D.T.; Chen, M.; Giannobile, W.V. Regenerative Medicine Technologies to Treat Dental, Oral, and Craniofacial Defects. Front. Bioeng. Biotechnol. 2021, 9, 704048. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Li, J.; Lei, X.; Cheng, P.; Song, Y.; Gao, Y.; Hu, J.; Wang, C.; Zhang, S.; Li, D.; et al. 3D-bioprinted functional and biomimetic hydrogel scaffolds incorporated with nanosilicates to promote bone healing in rat calvarial defect model. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 112, 110905. [Google Scholar] [CrossRef]
- Mosher, H.P. A method of filling the excavated mastoid with a flap from the back of the auricle. Laryngoscope 1911, 21, 1158–1163. [Google Scholar] [CrossRef]
- Zwierz, A.; Staszak, M.; Scheich, M.; Domagalski, K.; Hackenberg, S.; Burduk, P. A Comparison of the Sticky Bone Obliteration Technique and Obliteration Using S53P4 Bioactive Glass After Canal Wall Down Ear Surgery: A Preliminary Study. J. Clin. Med. 2025, 14, 1681. [Google Scholar] [CrossRef]
- Faita, A.; Volpato, G.M.; Trojan, D.; Montagner, G.; Di Pasquale Fiasca, V.M. Mastoid Obliteration with the “Cupeta Technique” After Canal Wall Down Tympanoplasty in Chronic Otitis Media with Cholesteatoma: Preliminary Results. Biomedicines 2025, 13, 2391. [Google Scholar] [CrossRef] [PubMed]
- Kemper, M.; Kluge, A.; Zeidler-Rentzsch, I.; Günther, S.I.; Neudert, M. Improvement of the Obliteration of Non-Critical Size Defects by Using a Mixture of Bone Dust and Bone Replacement Material (Bioactive Glass S53P4). Osteology 2025, 5, 15. [Google Scholar] [CrossRef]
- Hashmi, S.; Hussain, S.Z.M.; Matto, O.; Dewhurst, S.; Qayyum, A. To evaluate the results of mastoid obliteration and reconstruction of posterior meatal wall after canal wall down mastoidectomy using ready-to-use, self-setting hydroxyapatite bone cement. J. Laryngol. Otol. 2025, 139, 458–463. [Google Scholar] [CrossRef]
- Pozharskaya, D.D.; Vakhrushev, S.G.; Kashirtseva, I.A. Mastoid obliteration using platelet-rich plasma. Folia Otorhinolaryngol. Pathol. Respir. 2025, 31, 40–45. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Vahabi, H.; Rabiee, N.; Rabiee, M.; Bagherzadeh, M.; Saeb, M.R. Green composites in bone tissue engineering. Emergent Mater. 2022, 5, 603–620. [Google Scholar] [CrossRef]
- Miguez, P.A.; de Paiva Gonçalves, V.; Musskopf, M.L.; Rivera-Concepcion, A.; McGaughey, S.; Yu, C.; Lee, D.J.; Tuin, S.A.; Ali, A. Mitigation of BMP-induced inflammation in craniofacial bone regeneration and improvement of bone parameters by dietary hesperidin. Sci. Rep. 2024, 14, 2602. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.M.; Smyth, M.D.; Woo, A.S. Adverse facial edema associated with off-label use of recombinant human bone morphogenetic protein–2 in cranial reconstruction for craniosynostosis: Case report. J. Neurosurg. Pediatr. 2008, 1, 255–257. [Google Scholar] [PubMed]

| Author | Type | Material | Result | Reference |
|---|---|---|---|---|
| MESA EIbary et al. [3] | Clinical | PRP only | Enhanced epithelization | Eur Arch Otorhinolaryngol 2025; 282: 6069–6072 |
| Askar SM et al. [15] | Clinical | PRP with bone pate | Achieved complete epithelialization | Ear Nose Throat J 2021; 100: 485–489 |
| MES bd EIbary et al. [16] | Clinical | PRP with bone pate Titanium mesh | Surface of the neocanal wall appeared smooth | Int Arch Otorhinolaryngol 2018; 22: 103–107 |
| Jang CH et al. [17] | Preclinical | Umbilical cord serum PCL/alginate scaffold | GFs in umbilical cord serum enhanced osteogenesis | Int J Pediatr Otorhinolaryngol 2014; 78: 1061–1065 |
| Jang CH et al. [18] | Preclinical | Fibrous collagen, calcium dehiscent HA, umbilical cord serum | Rapid osteogenesis | Int J Biol Macromol 2021; 176: 479–489 |
| Jang CH et al. [19] | Preclinical | MSCs laden PCL/ Collagen scaffold | MSCs enhanced more rapid osteogenesis | RSC Advances 2016; 6: 6259–6265 |
| Choi SW et al. [20] | Preclinical | Tonsil derived MSCs/HA/chitosan patch | MSCs enhanced efficient osteogenesis | ACS Appl Bio Mater 2020; 3: 1008–1017 |
| Skoloudik L et al. [21] | Preclinical | Human MSCs/HA | hMSCs showed a significantly higher ratio of new bone formation | Cell Transplantation 2016; 25: 1405–1414 |
| Park SH et al. [22] | Preclinical | PCL/stromal vascular fraction cells | Autologous SVF cells with PCL are promising | Polymers 2022; 14: 877 |
| Yu F et al. [23] | Preclinical | 3D-printed BAG/PCL/BMP-2 | Enhanced osteogenesis | Regenerative Therapy 2022; 21: 469–476 |
| Jang CH et al. [5] | Preclinical | 3D PCL/alginate/BMP-2 | Enhanced osteogenesis | Int J Biol Macromol 2013; 62: 614–622 |
| Jang CH et al. [24] | Preclinical | 3D PCL/alginate/BMP-2/UCS | Enhanced osteogenesis | J Industrial Eng Chem 2019; 72: 432–441 |
| Lee J et al. [25] | Preclinical | Highly elastic 3D-printed gelatin/HA/placental extract | GFs from placental extract enhanced osteogenesis | Theranostics 2022; 12: 4051–4066 |
| Kim W et al. [26] | Preclinical | Bioprinted cell constructs with endothelial cell spheroids | Early angiogenesis prominently enhanced | Theranostics 2022; 12: 5404–5417 |
| Jang CH et al. [27] | Preclinical | 3D PCL/beta TCP/collagen nanofiber | 3D porosity and collagen nanofiber stimulated osteogenesis | Macromol Biosci 2013; 13: 660–668 |
| Aspect | Current Limitations | Future Directions |
|---|---|---|
| Biological adjunct | Predominant use of PRP with short-lived effects | MSCs, MSC-secretome, extracellular vesicles |
| Growth factor delivery | Non-specific, variable release | Controlled, sustained release of BMPs, VEGF |
| Scaffold design | Heterogeneous polymers, non-personalized | Patient-specific 3D-bioprinted scaffolds |
| Regenerative control | Limited spatial and temporal control | Bioprinting with spatial cell/molecule distribution |
| Personalization | One-size-fits-all approach | AI-driven personalized scaffold optimization |
| Evidence level | Small retrospective studies | Prospective trials with long-term follow-up |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sun, K.H.; Choi, C.H.; Kim, M.; Jang, C.H. Mastoid Obliteration After Canal Wall Down Mastoidectomy Using Tissue Engineering Approaches with Polymers, Mesenchymal Stem Cells, and Bioactive Molecules: A Systematic Review. Bioengineering 2026, 13, 305. https://doi.org/10.3390/bioengineering13030305
Sun KH, Choi CH, Kim M, Jang CH. Mastoid Obliteration After Canal Wall Down Mastoidectomy Using Tissue Engineering Approaches with Polymers, Mesenchymal Stem Cells, and Bioactive Molecules: A Systematic Review. Bioengineering. 2026; 13(3):305. https://doi.org/10.3390/bioengineering13030305
Chicago/Turabian StyleSun, Kyung Hoon, Cheol Hee Choi, Minseong Kim, and Chul Ho Jang. 2026. "Mastoid Obliteration After Canal Wall Down Mastoidectomy Using Tissue Engineering Approaches with Polymers, Mesenchymal Stem Cells, and Bioactive Molecules: A Systematic Review" Bioengineering 13, no. 3: 305. https://doi.org/10.3390/bioengineering13030305
APA StyleSun, K. H., Choi, C. H., Kim, M., & Jang, C. H. (2026). Mastoid Obliteration After Canal Wall Down Mastoidectomy Using Tissue Engineering Approaches with Polymers, Mesenchymal Stem Cells, and Bioactive Molecules: A Systematic Review. Bioengineering, 13(3), 305. https://doi.org/10.3390/bioengineering13030305







