Efficient Interleaved Multi-Band Outer Volume Suppression for Highly Accelerated Simultaneous Multi-Slice Imaging of the Heart
Abstract
1. Introduction
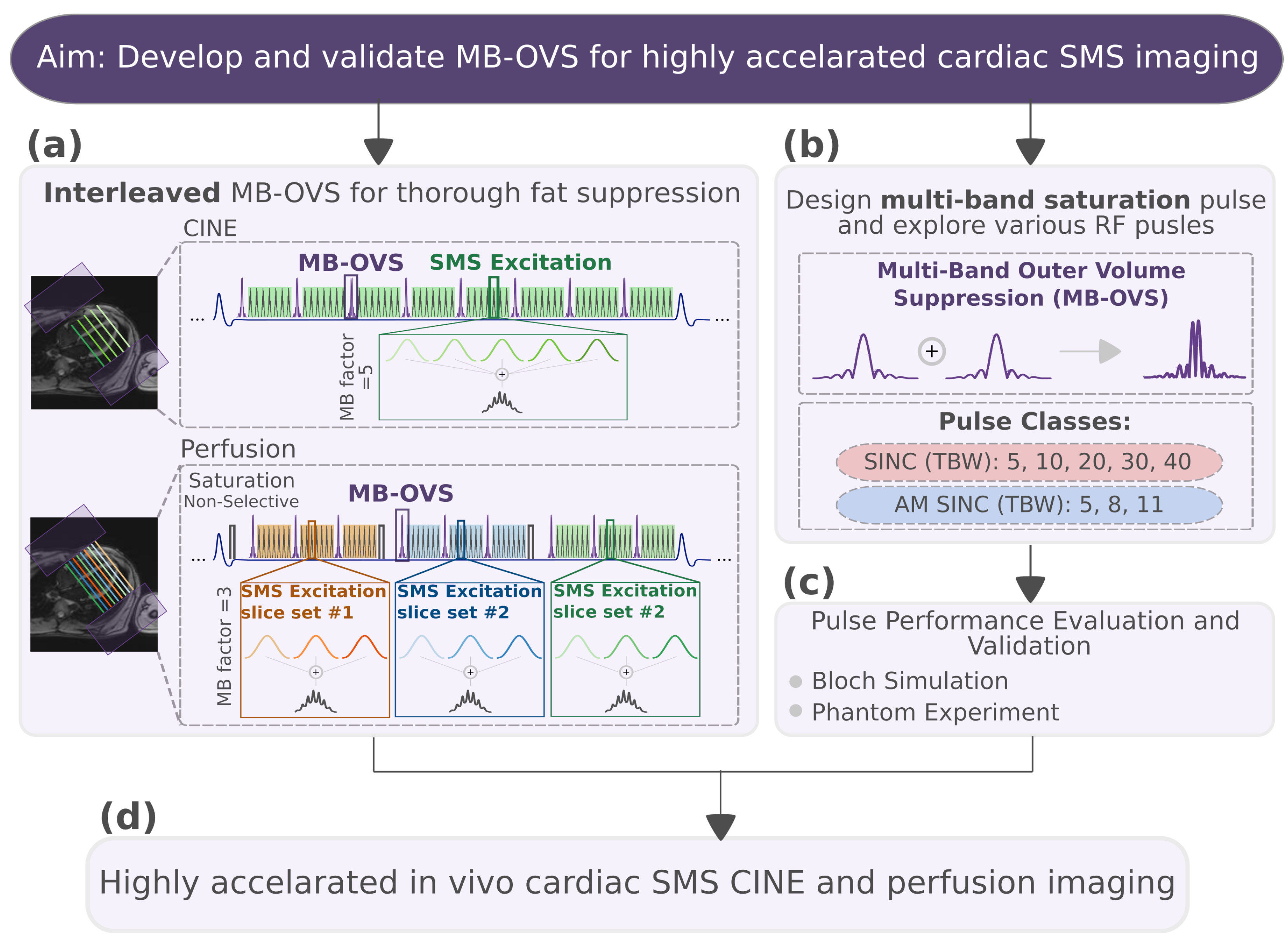
2. Materials and Methods
2.1. MB-OVS Design and Sequence
2.2. Bloch Simulation
- Residual signal analysis across a range of values using interleaved MB-OVS in the spGRE CINE sequence;
- Slice profile comparison for SINC and AM SINC pulses, including slice-selective gradient strength and requirements;
- scale and off-resonance effects on the chosen pulse to assess robustness in the presence of system imperfections.
2.3. Phantom Imaging
2.4. In Vivo MB-OVS Validation
2.4.1. CINE Imaging
2.4.2. Myocardial Perfusion Imaging
3. Results
3.1. Bloch Simulation
3.2. Phantom Imaging
3.3. In Vivo SMS Imaging
3.3.1. CINE Imaging
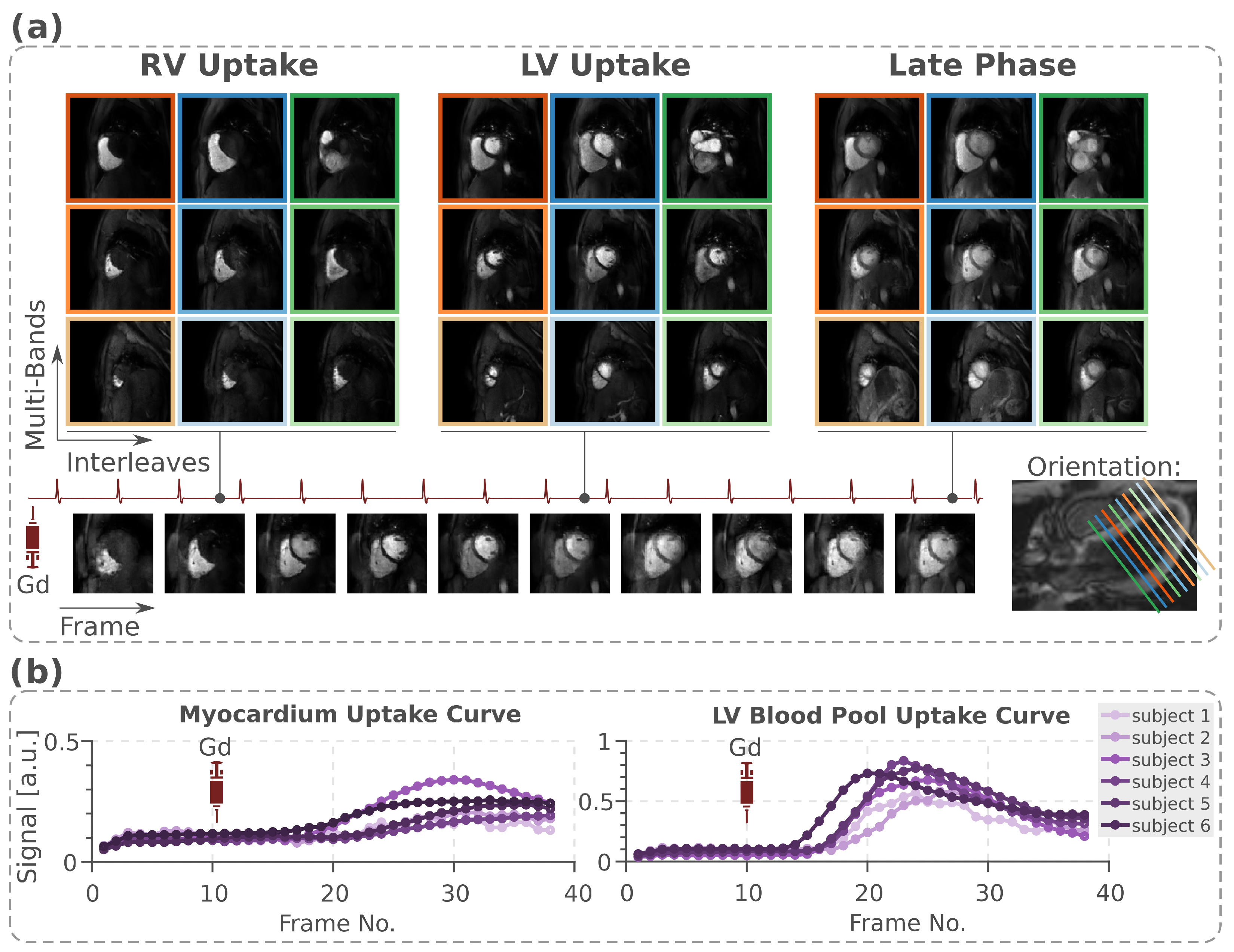
3.3.2. Myocardial Perfusion Imaging
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SMS | Simultaneous Multi-Slice |
| OVS | Outer Volume Suppression |
| RF | Radio Frequency |
| MB | Multi-Band |
| SAR | Specific Absorption Rate |
| PE | Phase-encoding |
| TBW | Time-Bandwidth product |
| spGRE | Spoiled Gradient Echo |
| ROI | Regions Of Interest |
| ACS | Auto Calibration Signal |
| LLR | Locally Low Rank |
| PD-DL | Physics-Driven Deep Learning |
| SIIM | Signal Intensity Informed Multi-coil |
| LV | Left Ventricle |
| SSDU | Self-Supervised Learning via Data Undersampling |
References
- Wieben, O.; Francois, C.; Reeder, S.B. Cardiac MRI of ischemic heart disease at 3 T: Potential and challenges. Eur. J. Radiol. 2008, 65, 15–28. [Google Scholar] [CrossRef]
- Guo, R.; Weingärtner, S.; Šiurytė, P.; Stoeck, C.T.; Füetterer, M.; E Campbell-Washburn, A.; Suinesiaputra, A.; Jerosch-Herold, M.; Nezafat, R. Emerging techniques in cardiac magnetic resonance imaging. J. Magn. Reson. Imaging 2022, 55, 1043–1059. [Google Scholar] [CrossRef]
- van der Geest, R.J.; Reiber, J.H. Quantification in cardiac MRI. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 1999, 10, 602–608. [Google Scholar] [CrossRef]
- Nandalur, K.R.; Dwamena, B.A.; Choudhri, A.F.; Nandalur, M.R.; Carlos, R.C. Diagnostic Performance of Stress Cardiac Magnetic Resonance Imaging in the Detection of Coronary Artery Disease. A Meta-Analysis. J. Am. Coll. Cardiol. 2007, 50, 1343–1353. [Google Scholar] [CrossRef]
- Curtis, A.D.; Cheng, H.L.M. Primer and historical review on rapid cardiac CINE MRI. J. Magn. Reson. Imaging 2022, 55, 373–388. [Google Scholar] [CrossRef] [PubMed]
- Rajiah, P.S.; François, C.J.; Leiner, T. Cardiac MRI: State of the art. Radiology 2023, 307, e223008. [Google Scholar] [CrossRef] [PubMed]
- Larkman, D.J.; Nunes, R.G. Parallel magnetic resonance imaging. Phys. Med. Biol. 2007, 52, R15. [Google Scholar] [CrossRef]
- Motwani, M.; Jogiya, R.; Kozerke, S.; Greenwood, J.P.; Plein, S. Advanced cardiovascular magnetic resonance myocardial perfusion imaging: High-spatial resolution versus 3-dimensional whole-heart coverage. Circ. Cardiovasc. Imaging 2013, 6, 339–348. [Google Scholar] [CrossRef]
- Nazir, M.S.; Neji, R.; Speier, P.; Reid, F.; Stäb, D.; Schmidt, M.; Forman, C.; Razavi, R.; Plein, S.; Ismail, T.F.; et al. Simultaneous multi slice (SMS) balanced steady state free precession first-pass myocardial perfusion cardiovascular magnetic resonance with iterative reconstruction at 1.5 T. J. Cardiovasc. Magn. Reson. 2018, 20, 84. [Google Scholar] [CrossRef]
- McElroy, S.; Ferrazzi, G.; Nazir, M.S.; Kunze, K.P.; Neji, R.; Speier, P.; Stäb, D.; Forman, C.; Razavi, R.; Chiribiri, A.; et al. Combined simultaneous multislice bSSFP and compressed sensing for first-pass myocardial perfusion at 1.5 T with high spatial resolution and coverage. Magn. Reson. Med. 2020, 84, 3103–3116. [Google Scholar] [CrossRef] [PubMed]
- McElroy, S.; Ferrazzi, G.; Nazir, M.S.; Evans, C.; Ferreira, J.; Bosio, F.; Mughal, N.; Kunze, K.P.; Neji, R.; Speier, P.; et al. Simultaneous multislice steady-state free precession myocardial perfusion with full left ventricular coverage and high resolution at 1.5 T. Magn. Reson. Med. 2022, 88, 663–675. [Google Scholar] [CrossRef]
- Breuer, F.A.; Blaimer, M.; Heidemann, R.M.; Mueller, M.F.; Griswold, M.A.; Jakob, P.M. Controlled aliasing in parallel imaging results in higher acceleration (CAIPIRINHA) for multi-slice imaging. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 2005, 53, 684–691. [Google Scholar] [CrossRef]
- Larkman, D.J.; Hajnal, J.V.; Herlihy, A.H.; Coutts, G.A.; Young, I.R.; Ehnholm, G. Use of multicoil arrays for separation of signal from multiple slices simultaneously excited. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2001, 13, 313–317. [Google Scholar] [CrossRef]
- Todd, N.; Josephs, O.; Zeidman, P.; Flandin, G.; Moeller, S.; Weiskopf, N. Functional sensitivity of 2D simultaneous multi-slice echo-planar imaging: Effects of acceleration on g-factor and physiological noise. Front. Neurosci. 2017, 11, 158. [Google Scholar] [CrossRef]
- Hoch, M.; Bruno, M.; Pacione, D.; Lui, Y.; Fieremans, E.; Shepherd, T. Simultaneous multislice for accelerating diffusion MRI in clinical neuroradiology protocols. Am. J. Neuroradiol. 2021, 42, 1437–1443. [Google Scholar] [CrossRef]
- Chen, H.; Yin, J.; He, C.; Wu, Y.; Long, M.; Liu, G.; Ni, H.; Jin, H.; Liu, Y. Improved activation and hemodynamic response function of olfactory fMRI using simultaneous multislice with reduced TR acquisition. BioMed Res. Int. 2021, 2021, 9965756. [Google Scholar] [CrossRef]
- Moeller, S.; Yacoub, E.; Olman, C.A.; Auerbach, E.; Strupp, J.; Harel, N.; Uğurbil, K. Multiband multislice GE-EPI at 7 tesla, with 16-fold acceleration using partial parallel imaging with application to high spatial and temporal whole-brain fMRI. Magn. Reson. Med. 2010, 63, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.T.; Setsompop, K.; Ahveninen, J.; Belliveau, J.W.; Witzel, T.; Lin, F.H. Improving the spatial resolution of magnetic resonance inverse imaging via the blipped-CAIPI acquisition scheme. Neuroimage 2014, 91, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Schmitter, S.; Moeller, S.; Wu, X.; Auerbach, E.J.; Metzger, G.J.; Van de Moortele, P.F.; Uğurbil, K. Simultaneous multislice imaging in dynamic cardiac MRI at 7T using parallel transmission. Magn. Reson. Med. 2017, 77, 1010–1020. [Google Scholar] [CrossRef] [PubMed]
- Weingärtner, S.; Moeller, S.; Schmitter, S.; Auerbach, E.; Kellman, P.; Shenoy, C.; Akçakaya, M. Simultaneous multislice imaging for native myocardial T1 mapping: Improved spatial coverage in a single breath-hold. Magn. Reson. Med. 2017, 78, 462–471. [Google Scholar] [CrossRef]
- Yang, Y.; Meyer, C.H.; Epstein, F.H.; Kramer, C.M.; Salerno, M. Whole-heart spiral simultaneous multi-slice first-pass myocardial perfusion imaging. Magn. Reson. Med. 2019, 81, 852–862. [Google Scholar] [CrossRef]
- Bentatou, Z.; Troalen, T.; Bernard, M.; Guye, M.; Pini, L.; Bartoli, A.; Jacquier, A.; Kober, F.; Rapacchi, S. Simultaneous multi-slice T1 mapping using MOLLI with blipped CAIPIRINHA bSSFP. Magn. Reson. Imaging 2023, 95, 90–102. [Google Scholar] [CrossRef]
- Tian, Y.; Cui, S.X.; Lim, Y.; Lee, N.G.; Zhao, Z.; Nayak, K.S. Contrast-optimal simultaneous multi-slice bSSFP cine cardiac imaging at 0.55 T. Magn. Reson. Med. 2023, 89, 746–755. [Google Scholar] [CrossRef]
- Rapacchi, S.; Troalen, T.; Bentatou, Z.; Quemeneur, M.; Guye, M.; Bernard, M.; Jacquier, A.; Kober, F. Simultaneous multi-slice cardiac cine with Fourier-encoded self-calibration at 7 Tesla. Magn. Reson. Med. 2019, 81, 2576–2587. [Google Scholar] [CrossRef]
- Adluru, G.; Bieging, E.; Chen, L.; Kim, D.; Wilson, B.D.; DiBella, E.V. Radial simultaneous multi slice imaging for rapid cardiac imaging. J. Cardiovasc. Magn. Reson. 2016, 18, O111. [Google Scholar] [CrossRef]
- Price, A.N.; Cordero-Grande, L.; Malik, S.J.; Hajnal, J.V. Simultaneous multislice imaging of the heart using multiband balanced SSFP with blipped-CAIPI. Magn. Reson. Med. 2020, 83, 2185–2196. [Google Scholar] [CrossRef] [PubMed]
- Cauley, S.F.; Polimeni, J.R.; Bhat, H.; Wald, L.L.; Setsompop, K. Interslice leakage artifact reduction technique for simultaneous multislice acquisitions. Magn. Reson. Med. 2014, 72, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Lustig, M.; Pauly, J.M. SPIRiT: Iterative self-consistent parallel imaging reconstruction from arbitrary k-space. Magn. Reson. Med. 2010, 64, 457–471. [Google Scholar] [CrossRef] [PubMed]
- Blaimer, M.; Breuer, F.A.; Seiberlich, N.; Mueller, M.F.; Heidemann, R.M.; Jellus, V.; Wiggins, G.; Wald, L.L.; Griswold, M.A.; Jakob, P.M. Accelerated volumetric MRI with a SENSE/GRAPPA combination. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2006, 24, 444–450. [Google Scholar] [CrossRef]
- Demirel, O.B.; Weingärtner, S.; Moeller, S.; Akçakaya, M. Improved simultaneous multislice cardiac MRI using readout concatenated k-space SPIRiT (ROCK-SPIRiT). Magn. Reson. Med. 2021, 85, 3036–3048. [Google Scholar] [CrossRef]
- Knoll, F.; Hammernik, K.; Zhang, C.; Moeller, S.; Pock, T.; Sodickson, D.K.; Akcakaya, M. Deep-learning methods for parallel magnetic resonance imaging reconstruction: A survey of the current approaches, trends, and issues. IEEE Signal Process. Mag. 2020, 37, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Demirel, O.B.; Yaman, B.; Shenoy, C.; Moeller, S.; Weingärtner, S.; Akçakaya, M. Signal intensity informed multi-coil encoding operator for physics-guided deep learning reconstruction of highly accelerated myocardial perfusion CMR. Magn. Reson. Med. 2023, 89, 308–321. [Google Scholar] [CrossRef] [PubMed]
- Menon, R.G.; Miller, G.W.; Jeudy, J.; Rajagopalan, S.; Shin, T. Free breathing three-dimensional late gadolinium enhancement cardiovascular magnetic resonance using outer volume suppressed projection navigators. Magn. Reson. Med. 2017, 77, 1533–1543. [Google Scholar] [CrossRef] [PubMed]
- Henningsson, M.; Abou Zahr, R.; Dyer, A.; Greil, G.F.; Burkhardt, B.; Tandon, A.; Hussain, T. Feasibility of 3D black-blood variable refocusing angle fast spin echo cardiovascular magnetic resonance for visualization of the whole heart and great vessels in congenital heart disease. J. Cardiovasc. Magn. Reson. 2018, 20, 76. [Google Scholar] [CrossRef]
- Jang, M.; Gupta, A.; Kovanlikaya, A.; Scholl, J.E.; Zun, Z. High-resolution anatomical imaging of the fetal brain with a reduced field of view using outer volume suppression. Magn. Reson. Med. 2024, 92, 1556–1567. [Google Scholar] [CrossRef]
- Felmlee, J.P.; Ehman, R.L. Spatial presaturation: A method for suppressing flow artifacts and improving depiction of vascular anatomy in MR imaging. Radiology 1987, 164, 559–564. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, L.; Chen, X.; Shaw, P.W.; Gonzalez, J.A.; Epstein, F.H.; Meyer, C.H.; Kramer, C.M.; Salerno, M. Reduced field of view single-shot spiral perfusion imaging. Magn. Reson. Med. 2018, 79, 208–216. [Google Scholar] [CrossRef]
- Wang, J.; Yang, Y.; Weller, D.S.; Zhou, R.; Van Houten, M.; Sun, C.; Epstein, F.H.; Meyer, C.H.; Kramer, C.M.; Salerno, M. High spatial resolution spiral first-pass myocardial perfusion imaging with whole-heart coverage at 3 T. Magn. Reson. Med. 2021, 86, 648–662. [Google Scholar] [CrossRef]
- Luo, J.; Addy, N.O.; Ingle, R.R.; Hargreaves, B.A.; Hu, B.S.; Nishimura, D.G.; Shin, T. Combined outer volume suppression and T2 preparation sequence for coronary angiography. Magn. Reson. Med. 2015, 74, 1632–1639. [Google Scholar] [CrossRef]
- Coristine, A.J.; Yerly, J.; Stuber, M. A cylindrical, inner volume selecting 2D-T2-Prep improves GRAPPA-accelerated image quality in MRA of the right coronary artery. PLoS ONE 2016, 11, e0163618. [Google Scholar] [CrossRef]
- Raman, S.V.; Markl, M.; Patel, A.R.; Bryant, J.; Allen, B.D.; Plein, S.; Seiberlich, N. 30-minute CMR for common clinical indications:  a Society for Cardiovascular Magnetic Resonance white paper. J. Cardiovasc. Magn. Reson. 2022, 24, 13. [Google Scholar] [CrossRef]
- Isensee, F.; Jaeger, P.F.; Kohl, S.A.; Petersen, J.; Maier-Hein, K.H. nnU-Net: A self-configuring method for deep learning-based biomedical image segmentation. Nat. Methods 2021, 18, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yang, C.; Schweidtmann, A.; Tao, Q. Efficient Bayesian uncertainty estimation for nnU-Net. In Proceedings of the International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer Nature: Cham, Switzerland, 2022; pp. 535–544. [Google Scholar]
- Zhao, Y.; Tourais, J.; Pierce, I.; Nitsche, C.; Treibel, T.A.; Weingärtner, S.; Schweidtmann, A.M.; Tao, Q. Bayesian Uncertainty Estimation by Hamiltonian Monte Carlo: Applications to Cardiac MRI Segmentation. Mach. Learn. Biomed. Imaging 2024, 2, 856–887. [Google Scholar] [CrossRef]
- Ogg, R.J.; Kingsley, R.; Taylor, J.S. WET, a T1-and B1-insensitive water-suppression method for in vivo localized 1H NMR spectroscopy. J. Magn. Reson. Ser. B 1994, 104, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yaman, B.; Hosseini, S.A.H.; Moeller, S.; Ellermann, J.; Uğurbil, K.; Akçakaya, M. Self-supervised learning of physics-guided reconstruction neural networks without fully sampled reference data. Magn. Reson. Med. 2020, 84, 3172–3191. [Google Scholar] [CrossRef] [PubMed]
- Yaman, B.; Gu, H.; Hosseini, S.A.H.; Demirel, O.B.; Moeller, S.; Ellermann, J.; Uğurbil, K.; Akçakaya, M. Multi-mask self-supervised learning for physics-guided neural networks in highly accelerated magnetic resonance imaging. NMR Biomed. 2022, 35, e4798. [Google Scholar] [CrossRef]
- Tao, Q.; van der Tol, P.; Berendsen, F.F.; Paiman, E.H.; Lamb, H.J.; van der Geest, R.J. Robust motion correction for myocardial T1 and extracellular volume mapping by principle component analysis-based groupwise image registration. J. Magn. Reson. Imaging 2018, 47, 1397–1405. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Y.; Huang, L.; Xia, L.; Tao, Q. Deep-learning-based groupwise registration for motion correction of cardiac T1 mapping. In Proceedings of the International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer Nature: Cham, Switzerland, 2024. [Google Scholar]
- Di Bella, E.; Wu, Y.; Alexander, A.; Parker, D.; Green, D.; McGann, C. Comparison of temporal filtering methods for dynamic contrast MRI myocardial perfusion studies. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 2003, 49, 895–902. [Google Scholar] [CrossRef]
- Bojorquez, J.Z.; Bricq, S.; Acquitter, C.; Brunotte, F.; Walker, P.M.; Lalande, A. What are normal relaxation times of tissues at 3 T? Magn. Reson. Imaging 2017, 35, 69–80. [Google Scholar] [CrossRef]
- Auerbach, E.J.; Xu, J.; Yacoub, E.; Moeller, S.; Uğurbil, K. Multiband accelerated spin-echo echo planar imaging with reduced peak RF power using time-shifted RF pulses. Magn. Reson. Med. 2013, 69, 1261–1267. [Google Scholar] [CrossRef]
- Pauly, J.; Le Roux, P.; Nishimura, D.; Macovski, A. Parameter relations for the Shinnar-Le Roux selective excitation pulse design algorithm (NMR imaging). IEEE Trans. Med Imaging 1991, 10, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Schulte, R.; Henning, A.; Tsao, J.; Boesiger, P.; Pruessmann, K.P. Design of broadband RF pulses with polynomial-phase response. J. Magn. Reson. 2007, 186, 167–175. [Google Scholar] [CrossRef]
- Luo, Y.; de Graaf, R.A.; DelaBarre, L.; Tannús, A.; Garwood, M. BISTRO: An outer-volume suppression method that tolerates RF field inhomogeneity. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 2001, 45, 1095–1102. [Google Scholar] [CrossRef]
- Smith, T.B.; Nayak, K.S. Reduced field of view MRI with rapid, B1-robust outer volume suppression. Magn. Reson. Med. 2012, 67, 1316–1323. [Google Scholar] [CrossRef] [PubMed]
- Feinberg, D.A.; Hoenninger, J.; Crooks, L.; Kaufman, L.; Watts, J.; Arakawa, M. Inner volume MR imaging: Technical concepts and their application. Radiology 1985, 156, 743–747. [Google Scholar] [CrossRef]
- Gibiino, F.; Lechner-Greite, S.; Schirmer, T.; Positano, V.; Brau, A.; Stainsby, J.A.; Slavin, G.S.; Landini, L.; Janich, M.A. Effects of inner volume field-of-view reduction on myocardial T2 mapping. J. Magn. Reson. Imaging 2015, 42, 175–179. [Google Scholar] [CrossRef]
- Rieseberg, S.; Frahm, J.; Finsterbusch, J. Two-dimensional spatially-selective RF excitation pulses in echo-planar imaging. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 2002, 47, 1186–1193. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elmoniem, K.Z.; Barmet, C.; Stuber, M. Free-breathing inner-volume black-blood imaging of the human heart using two-dimensionally selective local excitation at 3 T. Magn. Reson. Med. 2012, 68, 822–829. [Google Scholar] [CrossRef][Green Version]
- Kim, D.; Cauley, S.F.; Nayak, K.S.; Leahy, R.M.; Haldar, J.P. Region-optimized virtual (ROVir) coils: Localization and/or suppression of spatial regions using sensor-domain beamforming. Magn. Reson. Med. 2021, 86, 197–212. [Google Scholar] [CrossRef]
- Kim, D.; Coll-Font, J.; Lobos, R.A.; Stäb, D.; Pang, J.; Foster, A.; Garrett, T.; Bi, X.; Speier, P.; Haldar, J.P.; et al. Single breath-hold CINE imaging with combined simultaneous multislice and region-optimized virtual coils. Magn. Reson. Med. 2023, 90, 222–230. [Google Scholar] [CrossRef]




| AM SINC X | SINC X | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameters | No OVS | RF-0 | AM SINC 5 | AM SINC 8 | AM SINC 11 | SINC 5 | SINC 10 | SINC 20 | SINC 30 | SINC 40 |
| Duration [ms] | 3.84 | 3.84 | 3.84 | 1.92 | 3.84 | 7.68 | 11.52 | 15.36 | ||
| TBW [-] | 5 | 8 | 11 | 5 | 10 | 20 | 30 | 40 | ||
| SAR [W/kg] | 0.09 | 0.007 | 0.81 | 1.26 | 1.60 | 1.40 | 1.46 | 1.35 | 1.27 | 1.16 |
| Residual Signal [%] | 100 | 110.8 | 32.69 | 23.72 | 24.14 | 27.34 | 24.95 | 24.02 | 24.08 | 22.21 |
| Gr [T/m] | 203 | 326 | 448 | 407 | 407 | 407 | 407 | 407 | ||
| [T] | 8.44 | 12.8 | 16.4 | 15.4 | 14.7 | 15.6 | 15.1 | 15.4 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Arami, A.; Demirel, O.B.; Kilic, T.; Moeller, S.; Zhao, Y.; Zhang, Y.; Tao, Q.; Lamb, H.J.; Akçakaya, M.; Weingärtner, S. Efficient Interleaved Multi-Band Outer Volume Suppression for Highly Accelerated Simultaneous Multi-Slice Imaging of the Heart. Bioengineering 2026, 13, 286. https://doi.org/10.3390/bioengineering13030286
Arami A, Demirel OB, Kilic T, Moeller S, Zhao Y, Zhang Y, Tao Q, Lamb HJ, Akçakaya M, Weingärtner S. Efficient Interleaved Multi-Band Outer Volume Suppression for Highly Accelerated Simultaneous Multi-Slice Imaging of the Heart. Bioengineering. 2026; 13(3):286. https://doi.org/10.3390/bioengineering13030286
Chicago/Turabian StyleArami, Ayda, Omer Burak Demirel, Toygan Kilic, Steen Moeller, Yidong Zhao, Yi Zhang, Qian Tao, Hildo J. Lamb, Mehmet Akçakaya, and Sebastian Weingärtner. 2026. "Efficient Interleaved Multi-Band Outer Volume Suppression for Highly Accelerated Simultaneous Multi-Slice Imaging of the Heart" Bioengineering 13, no. 3: 286. https://doi.org/10.3390/bioengineering13030286
APA StyleArami, A., Demirel, O. B., Kilic, T., Moeller, S., Zhao, Y., Zhang, Y., Tao, Q., Lamb, H. J., Akçakaya, M., & Weingärtner, S. (2026). Efficient Interleaved Multi-Band Outer Volume Suppression for Highly Accelerated Simultaneous Multi-Slice Imaging of the Heart. Bioengineering, 13(3), 286. https://doi.org/10.3390/bioengineering13030286






