CFD Modelling Validated by PIV of Hydrodynamics in a Raceway Bioreactor: Dead Zone Detection and Flow Field Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Bioreactor
2.2. CFD Model
2.2.1. Multiphase Approach
2.2.2. Computational Mesh Resolution
2.3. Experimental Data of the Raceway Bioreactor
3. Results
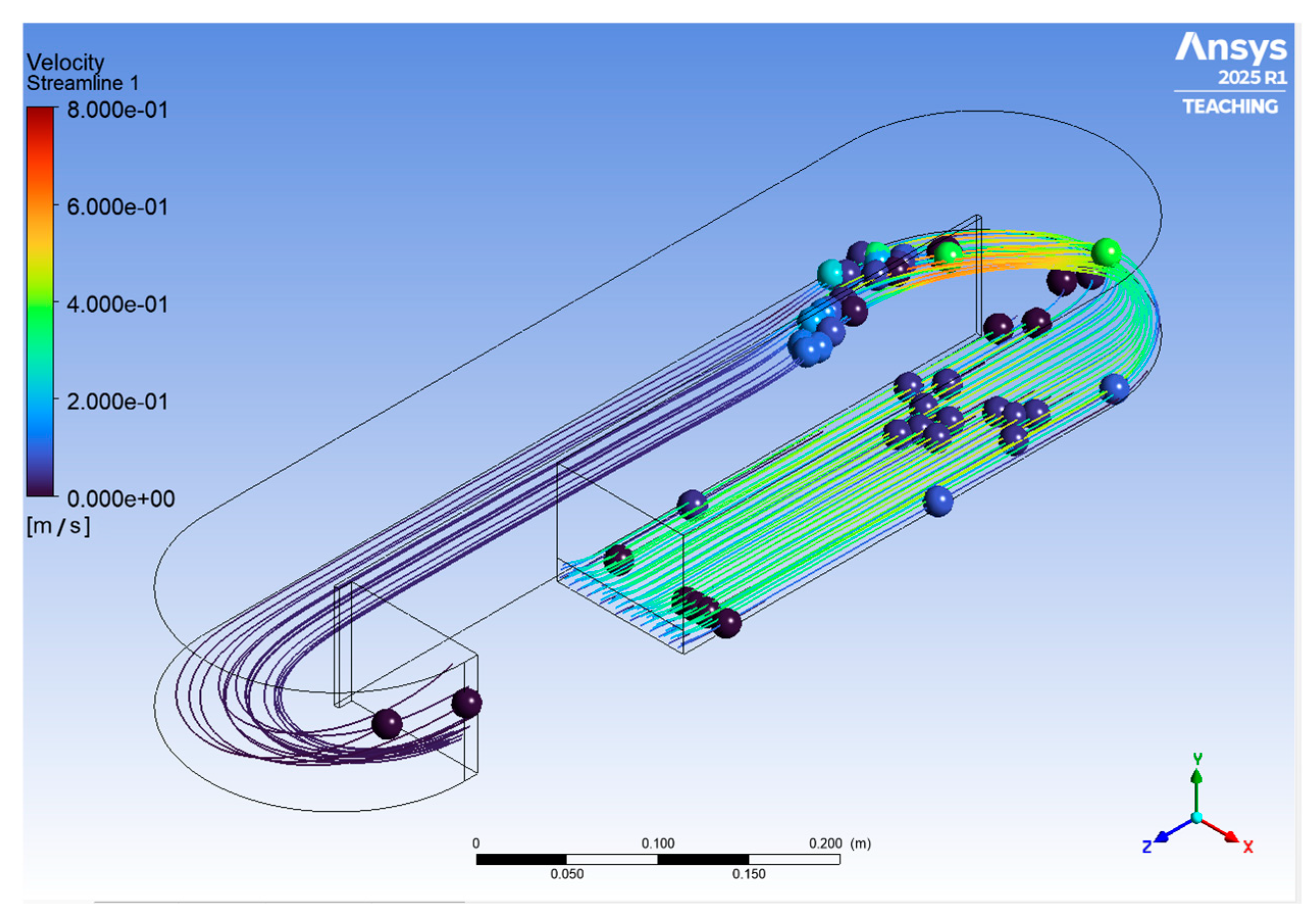
3.1. Raceway Bioreactor CFD
3.2. Validation Scope and Limitations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CFD | Computational Fluid Dynamics |
| fps | frames per second |
| FVM | Finite Volume Method |
| PIV | Particle Image Velocimetry |
| RANS | Reynolds-Averaged Navier–Stokes |
| RNG | Re-Normalization Group |
| RPM | Revolutions Per Minute |
References
- Acien, F.G.; Fernández, J.M.; Llamas, B.; Molina, E. Microalgae production systems. In Handbook of Microalgae-Based Processes and Products. Fundamentals and Advances in Energy, Food, Feed, Fertilizer, and Bioactive Compounds; Lopes, E.J., Manzoni, M., Queiroz, M.I., Queiroz, L., Eds.; Academic Press Elsevier Inc.: San Diego, CA, USA, 2020; pp. 127–163. [Google Scholar] [CrossRef]
- Park, S.; Li, Y. Integration of biological kinetics and computational fluid dynamics to model the growth of Nannochloropsis salina in an open channel raceway. Biotechnol. Bioeng. 2015, 112, 923–933. [Google Scholar] [CrossRef] [PubMed]
- Hreiz, R.; Sialve, B.; Morchain, J.; Escudié, R.; Steyer, J.; Guiraud, P. Experimental and numerical investigation of hydrodynamics in raceway reactors used for algaculture. Chem. Eng. J. 2014, 250, 230–239. [Google Scholar] [CrossRef]
- Jujjavarapu, S. Computational Fluid Dynamics: Fundamentals and Applications in the Design and Optimization of Various Bioreactors. In Computational Fluid Dynamics Applications in Bio and Biomedical Processes; Biotechnology Applications; Gupta, S., Kumar, T., Eds.; Springer Nature: Singapore, 2024; pp. 1–33. [Google Scholar] [CrossRef]
- Kumari, R.; Das, B.; Devi, K.; Khuntia, J.; Mohanty, M. Hydrodynamic performance of Raceway pond using k-ω and LES turbulence models. J. Renew. Sustain. Energy 2024, 16, 063101. [Google Scholar] [CrossRef]
- Pandey, R.; Premalatha, M. Design and analysis of flow velocity distribution inside a Raceway pond using computational fluid dynamics. Bioprocess Biosyst. Eng. 2016, 40, 439–450. [Google Scholar] [CrossRef]
- Oliveira, C.; Pace, Z.; Thomas, J.; DeVincentis, B.; Sirasitthichoke, C.; Egan, S.; Lee, J. CFD-based bioreactor model with proportional–integral–derivative controller functionality for dissolved oxygen and pH. Biotechnol. Bioeng. 2023, 121, 655–669. [Google Scholar] [CrossRef]
- Inostroza, C.; Solimeno, A.; García, J.; Fernandez-Sevilla, J.; Acién, F. Improvement of real-scale Raceway bioreactors for microalgae production using Computational Fluid Dynamics (CFD). Algal Res. 2021, 54, 102207. [Google Scholar] [CrossRef]
- Rajput, S.; Das, B.; Sharma, A. Review on modelling approaches of Raceway pond: Experimental, numerical and machine learning approaches. ISH J. Hydraul. Eng. 2025, 31, 382–397. [Google Scholar] [CrossRef]
- Campos, G.; Renato, N.; Oliveira, V.; Rodrigues, P.; Martins, M.A. Influence of paddlewheel geometry on hydrodynamic performance and energy consumption in microalgae cultivations in open raceway ponds. Bioprocess Biosyst. Eng. 2025, 48, 1171–1188. [Google Scholar] [CrossRef]
- Musgrove, E. Investigating the Performance of Paddlewheels Used in Microalgae Raceways for the Production of Biomass. Doctoral Dissertation, University of Southampton, Southampton, UK, 2017. Available online: https://eprints.soton.ac.uk/420751/ (accessed on 30 July 2023).
- Bautista-Monroy, S.; Salgado-Ramírez, J.; Téllez-Jurado, A.; Ramírez-Vargas, M.; Gómez-Aldapa, C.; Pérez-Viveros, K.; Cadena-Ramírez, A. Hydrodynamic characterization in a raceway bioreactor with different stirrers. Rev. Mex. Ing. Química 2019, 18, 605–619. [Google Scholar] [CrossRef]
- Sawant, S.; Khadamkar, H.; Mathpati, C.; Pandit, R.; Lali, A. Computational and experimental studies of high depth algal Raceway pond photobioreactor. Renew. Energy 2018, 118, 152–159. [Google Scholar] [CrossRef]
- Zeng, F.; Huang, J.; Meng, C.; Zhu, F.; Chen, J.; Li, Y. Investigation on novel Raceway pond with inclined paddle wheels through simulation and microalgae culture experiments. Bioprocess Biosyst. Eng. 2016, 39, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Lee, W.; Lee, Y.; Ali, H.; Kwak, M.; Park, C. Particle Image Velocimetry Measurement of Hydrodynamic Properties of Raceway Pond with the Effect of Central Wall. Sens. Mater. 2016, 28, 957–966. [Google Scholar] [CrossRef][Green Version]
- Quiroz-Arita, C.; Blaylock, M.; Gharagozloo, P.; Bark, D.; Prasad, D.; Bradley, T. Pilot-scale open-channel raceways and flat-panel photobioreactors maintain well-mixed conditions under a wide range of mixing energy inputs. Biotechnol. Bioeng. 2020, 117, 959–969. [Google Scholar] [CrossRef]
- Yang, Z.; Cheng, J.; Ye, Q.; Liu, J.; Zhou, J.; Cen, K. Decrease in light/dark cycle of microalgal cells with computational fluid dynamics simulation to improve microalgal growth in a raceway pond. Bioresour. Technol. 2016, 220, 352–359. [Google Scholar] [CrossRef]
- Del Olmo, P.; Acién, F.; Fernández-Sevilla, J. Analysis of productivity in raceway photobioreactor using computational fluid dynamics particle tracking coupled to a dynamic photosynthesis model. Bioresour. Technol. 2021, 334, 125226. [Google Scholar] [CrossRef]
- Shen, C.; Dandy, D. Application of passive vortex generators to enhance vertical mixing in an open raceway pond. Algal Res. 2024, 79, 103434. [Google Scholar] [CrossRef]
- Konozsy, L. Introduction to Classical Analytical Solutions for Wall-Bounded Turbulence. In A New Hypothesis on the Anisotropic Reynolds Stress Tensor for Turbulent Flows; Thess, A., Moreau, R., Eds.; Springer Nature: Cham, Switzerland, 2021; pp. 1–114. [Google Scholar] [CrossRef]
- ANSYS. Turbulence. In ANSYS Fluent Theory Guide; ANSYS Inc.: Canonsburg, PA, USA, 2021; pp. 41–154. [Google Scholar]
- Chen, X. Three-Dimensional Numerical Investigation on Velocity Field and Mixing Characteristics at Channel Junctions. Master’s Thesis, University of Alberta, Edmonton, AB, Canada, 2014. [Google Scholar] [CrossRef]
- Pineda, S.; Blanco, A.; Rojas-Solórzano, L. CFD Software Applications for Transcritical Free Surface Flow. In Proceedings of the FEDSM2009 ASME 2009 Fluids Engineering Division Summer Meeting, Vail, CO, USA, 2–6 August 2009; pp. 1987–1994. [Google Scholar] [CrossRef]
- Horritt, M. Development of physically based meshes for two-dimensional models of meandering channel flow. Int. J. Numer. Methods Eng. 2000, 47, 2019–2037. [Google Scholar] [CrossRef]
- Chila, R.; Kaminski, D. Automated grid independence via unstructured adaptive refinement. In Proceedings of the Insert Conference Abbreviation in: 36th AIAA Fluid Dynamics Conference and Exhibit, San Francisco, CA, USA, 5–8 June 2006; AIAA: Reston, VA, USA, 2012; pp. 2006–3062. [Google Scholar] [CrossRef]
- Bautista-Monroy, S.; Salgado-Ramirez, J.; Gómez-Aldapa, C.; Téllez-Jurado, A.; Ortega-Palacios, R.; Cadena-Ramirez, A. Image segmentation with fast distance transform (FDT) and morphological skeleton in microalgae Raceway culture systems applications. Rev. Mex. Ing. Quimica 2021, 20, 885–897. [Google Scholar] [CrossRef]
- Bautista-Monroy, S.; Chávez-Urbiola, E.A.; Ortega-Palacios, R.; González-Sánchez, A.; Gómez-Aldapa, C.A.; Rodríguez-Nava, O.; Cadena-Ramírez, A. Insights of raceway bioreactor scale-up: Effect of agitation on microalgae culture and reduction of the liquid medium speed. Appl. Sci. 2022, 12, 1513. [Google Scholar] [CrossRef]
- Skiba, Y.N. Ecuaciones Básicas de Movimiento. In Introducción a la Dinámica de Fluidos Centro de Ciencias de la Atmósfera; Universidad Nacional Autónoma de México: Mexico City, Mexico, 2008; pp. 1–41. [Google Scholar]
- Chisti, Y. Large-Scale Production of Algal Biomass: Raceway Ponds. In Algae Biotechnology; Products and Processes; Bux, F., Chisti, Y., Eds.; Springer: Cham, Switzerland, 2016; pp. 21–35. [Google Scholar] [CrossRef]
- Amini, H.; Hashemisohi, A.; Wang, L.; Shahbazi, A.; Bikdash, M.; Yuan, W. Numerical and experimental investigation of hydrodynamics and light transfer in open raceway ponds at various algal cell concentrations and medium depths. Chem. Eng. Sci. 2016, 156, 11–23. [Google Scholar] [CrossRef]
- Kusmayadi, A.; Philippidis, G.P.; Yen, H.-W. Application of computational fluid dynamics to raceways combining paddlewheel and CO2 spargers to enhance microalgae growth. J. Biosci. Bioeng. 2020, 129, 93–98. [Google Scholar] [CrossRef] [PubMed]









| Zone | 20 RPM Velocity (m/s) | 25 RPM Velocity (m/s) | 30 RPM Velocity (m/s) |
|---|---|---|---|
| A | 0.3195 | 0.3537 | 0.4053 |
| B | 0.3403 | 0.5274 | 0.5246 |
| C | 0.2867 | 0.3548 | 0.3867 |
| D | 0.0243 | 0.2508 | 0.3929 |
| E | 0.0326 | 0.2884 | 0.2662 |
| F | 0.0294 | 0.0985 | 0.1309 |
| G | 0.0172 | 0.0100 | 0.1039 |
| Paddlewheel Velocity (RPM) | % Relative Error |
|---|---|
| 20 | 7.6 |
| 25 | 3.9 |
| 30 | 12.1 |
| Average % error | 7.9 |
| Zona | 20 RPM | 25 RPM | 30 RPM |
|---|---|---|---|
| A | 39,323 | 43,532 | 49,883 |
| B | 41,883 | 64,910 | 64,566 |
| C | 35,286 | 43,667 | 47,593 |
| D | 2990 | 30,867 | 48,356 |
| E | 4012 | 35,495 | 32,763 |
| F | 3618 | 12,123 | 16,110 |
| G | 2116 | 1230 | 12,787 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zamora-Campos, L.A.; Rivera-Arreola, D.E.; Rojas-Hernández, R.; Trujillo-Mora, V.; Márquez-Vera, M.A.; Salgado-Ramírez, J.C.; Cadena-Ramírez, A. CFD Modelling Validated by PIV of Hydrodynamics in a Raceway Bioreactor: Dead Zone Detection and Flow Field Analysis. Bioengineering 2026, 13, 285. https://doi.org/10.3390/bioengineering13030285
Zamora-Campos LA, Rivera-Arreola DE, Rojas-Hernández R, Trujillo-Mora V, Márquez-Vera MA, Salgado-Ramírez JC, Cadena-Ramírez A. CFD Modelling Validated by PIV of Hydrodynamics in a Raceway Bioreactor: Dead Zone Detection and Flow Field Analysis. Bioengineering. 2026; 13(3):285. https://doi.org/10.3390/bioengineering13030285
Chicago/Turabian StyleZamora-Campos, Luis Alberto, Daniel Eduardo Rivera-Arreola, Rafael Rojas-Hernández, Valentín Trujillo-Mora, Marco Antonio Márquez-Vera, Julio César Salgado-Ramírez, and Arturo Cadena-Ramírez. 2026. "CFD Modelling Validated by PIV of Hydrodynamics in a Raceway Bioreactor: Dead Zone Detection and Flow Field Analysis" Bioengineering 13, no. 3: 285. https://doi.org/10.3390/bioengineering13030285
APA StyleZamora-Campos, L. A., Rivera-Arreola, D. E., Rojas-Hernández, R., Trujillo-Mora, V., Márquez-Vera, M. A., Salgado-Ramírez, J. C., & Cadena-Ramírez, A. (2026). CFD Modelling Validated by PIV of Hydrodynamics in a Raceway Bioreactor: Dead Zone Detection and Flow Field Analysis. Bioengineering, 13(3), 285. https://doi.org/10.3390/bioengineering13030285








