Advances in Decellularization of Fish Wastes for Extracellular Matrix Extraction in Sustainable Tissue Engineering and Regenerative Medicine
Abstract
1. Introduction
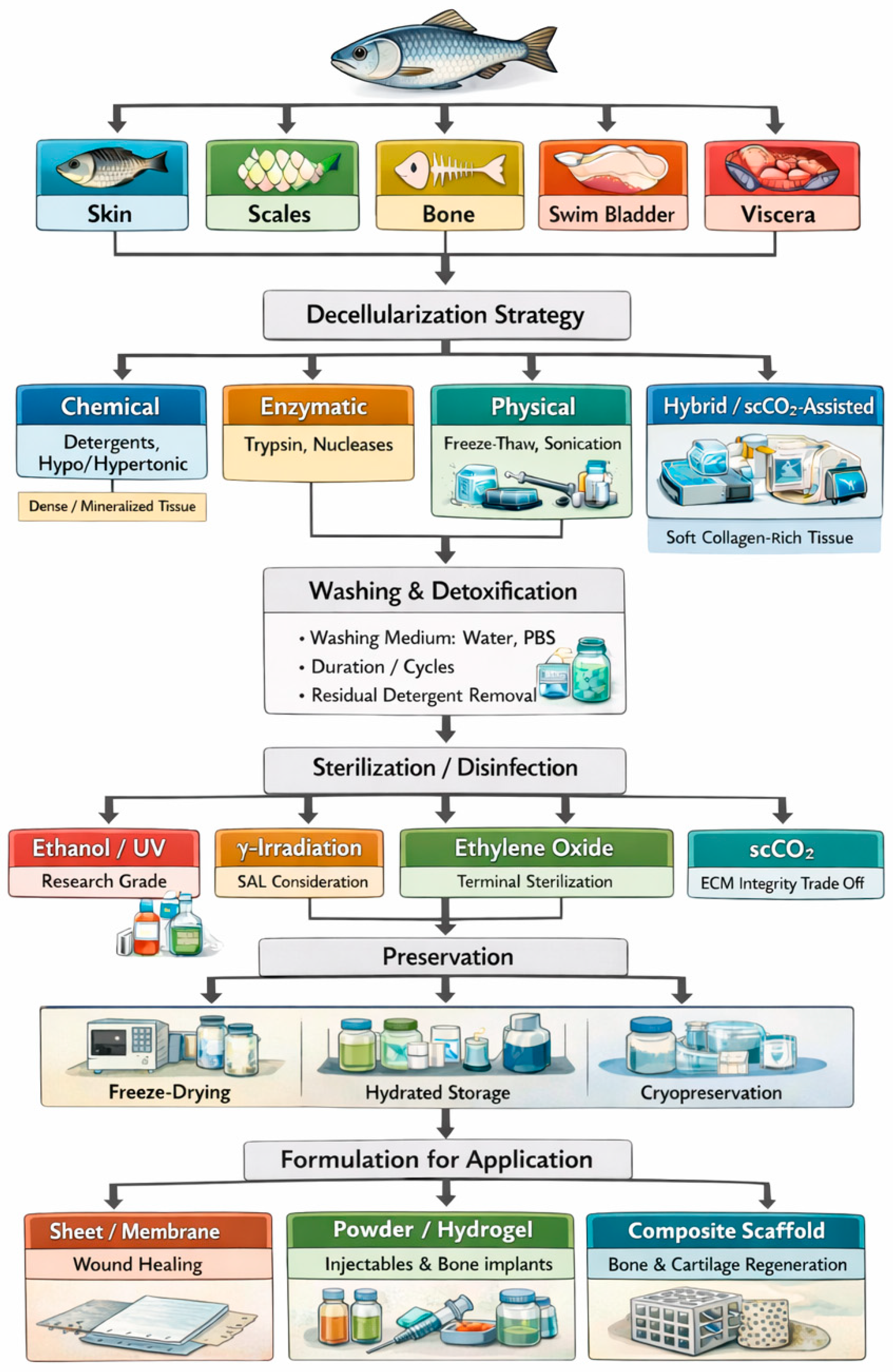
2. Fish Tissue Decellularization Techniques
2.1. Physical Treatment
2.1.1. Freeze–Thaw Process
2.1.2. Agitation
2.1.3. Sonication
2.2. Chemical Treatment
2.2.1. Acidic and Basic Solutions
2.2.2. Non-Ionic and Ionic Detergents
2.2.3. Hypotonic and Hypertonic Solutions
2.2.4. Chelating Agents
2.3. Biological Treatment
2.4. Strategic Selection and Optimization Framework
3. Sterilization and Preservation of Fish-Derived dECM
3.1. Sterilization Methods of Fish-Derived dECM
3.1.1. Gamma Irradiation
3.1.2. Ethylene Oxide and Chemical Disinfection
3.1.3. Supercritical CO2-Assissted Processing
3.2. Preservation Methods of Fish-Derived dECM
3.2.1. Freeze-Drying/Lyophilization
3.2.2. Hydration and Cryogenic Preservation
3.3. Clinical Translation Considerations for Fish-Derived dECM
4. Biomedical Applications of Fish-Derived dECM
4.1. Wound Healing and Skin Regeneration
4.2. Bone Regeneration
4.3. Cartilage Repair
4.4. Comparative Performance and Application-Driven Insights
5. Advances in Post-Processing of Fish-Derived dECM
5.1. Crosslinking for Surface Modification
5.2. Polymer–dECM Composites Fabrication
5.3. Three-Dimensional Printing
5.4. Engineering Trade-Offs and Design Considerations
6. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2024; FAO: Rome, Italy, 2024; ISBN 978-92-5-138763-4. [Google Scholar]
- Shahidi, F.; Varatharajan, V.; Peng, H.; Senadheera, R. Utilization of Marine By-Products for the Recovery of Value-Added Products. J. Food Bioact. 2019, 6, 10–61. [Google Scholar] [CrossRef]
- De Ungria, S.; Fernandez, L.T.; Sabado, S.E.; Santos, J.P.; Sararaña, A.R.; VinceCruz-Abeledo, C.C. Circular Economy in Fisheries: How Is Fish Market Waste Managed in the Philippines? Res. Sq. 2022. [Google Scholar] [CrossRef]
- Shavandi, A.; Hou, Y.; Carne, A.; McConnell, M.; Bekhit, A.E.A. Marine Waste Utilization as a Source of Functional and Health Compounds. In Advances in Food and Nutrition Research; Elsevier: Amsterdam, The Netherlands, 2019; Volume 87, pp. 187–254. ISBN 978-0-12-816049-7. [Google Scholar]
- Venugopal, V. Valorization of Seafood Processing Discards: Bioconversion and Bio-Refinery Approaches. Front. Sustain. Food Syst. 2021, 5, 611835. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Chowdhury, S.; Ikbal, A.; Murmu, P.; Nath, S. Fish Processing Wastes: Environmental Impacts and Mitigation Measures. Indian J. Anim. Health 2022, 61, 67–82. [Google Scholar] [CrossRef]
- Baclayon, L.; Bual, R.; Labares, M.; Valle, K.D.D.; Pague, J.; Alimasag, J.; Lumancas, G.; Arellano, F.; Nisperos, M.J.; Aron, J.; et al. Sonication-Assisted Decellularization of Waste Tilapia (Oreochromis niloticus) Heads for Extracellular Matrix Extraction. Biomass 2024, 4, 1078–1091. [Google Scholar] [CrossRef]
- Stevens, J.R.; Newton, R.W.; Tlusty, M.; Little, D.C. The Rise of Aquaculture By-Products: Increasing Food Production, Value, and Sustainability through Strategic Utilisation. Mar. Policy 2018, 90, 115–124. [Google Scholar] [CrossRef]
- Gadre, M.; Kasturi, M.; Agarwal, P.; Vasanthan, K.S. Decellularization and Their Significance for Tissue Regeneration in the Era of 3D Bioprinting. ACS Omega 2024, 9, 7375–7392. [Google Scholar] [CrossRef]
- Poole, J.J.A.; Mostaço-Guidolin, L.B. Optical Microscopy and the Extracellular Matrix Structure: A Review. Cells 2021, 10, 1760. [Google Scholar] [CrossRef] [PubMed]
- Dzobo, K.; Dandara, C. The Extracellular Matrix: Its Composition, Function, Remodeling, and Role in Tumorigenesis. Biomimetics 2023, 8, 146. [Google Scholar] [CrossRef]
- Dong, C.; Lv, Y. Application of Collagen Scaffold in Tissue Engineering: Recent Advances and New Perspectives. Polymers 2016, 8, 42. [Google Scholar] [CrossRef]
- Chen, W.; Chen, M.; Chen, S.; Wang, S.; Huang, Z.; Zhang, L.; Wu, J.; Peng, W.; Li, H.; Wen, F. Decellularization of Fish Tissues for Tissue Engineering and Regenerative Medicine Applications. Regen. Biomater. 2025, 12, rbae138. [Google Scholar] [CrossRef]
- Yi, S.; Ding, F.; Gong, L.; Gu, X. Extracellular Matrix Scaffolds for Tissue Engineering and Regenerative Medicine. Curr. Stem Cell Res. Ther. 2017, 12, 233–246. [Google Scholar] [CrossRef]
- Mundra, S.; Shetty, P.J.; Shetty, N.J. Tilapia Fish Waste: An Asset for Tissue Engineering—A Review. J. Indian Soc. Periodontol. 2023, 27, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, A.; Biazar, E.; Ebrahimi, M.; Heidari Keshel, S.; Kheilnezhad, B.; Saeedi Landi, F. Acellular Fish Skin for Wound Healing. Int. Wound J. 2023, 20, 2924–2941. [Google Scholar] [CrossRef] [PubMed]
- Aron, J.; Bual, R.; Alimasag, J.; Arellano, F.; Baclayon, L.; Bantilan, Z.C.; Lumancas, G.; Nisperos, M.J.; Labares, M.; Valle, K.D.D.; et al. Effects of Various Decellularization Methods for the Development of Decellularized Extracellular Matrix from Tilapia (Oreochromis niloticus) Viscera. Int. J. Biomater. 2024, 2024, 6148496. [Google Scholar] [CrossRef] [PubMed]
- Gershlak, J.R.; Hernandez, S.; Fontana, G.; Perreault, L.R.; Hansen, K.J.; Larson, S.A.; Binder, B.Y.K.; Dolivo, D.M.; Yang, T.; Dominko, T.; et al. Crossing Kingdoms: Using Decellularized Plants as Perfusable Tissue Engineering Scaffolds. Biomaterials 2017, 125, 13–22. [Google Scholar] [CrossRef]
- Alves, A.; Marques, A.; Martins, E.; Silva, T.; Reis, R. Cosmetic Potential of Marine Fish Skin Collagen. Cosmetics 2017, 4, 39. [Google Scholar] [CrossRef]
- Chen, L.; Cheng, G.; Meng, S.; Ding, Y. Collagen Membrane Derived from Fish Scales for Application in Bone Tissue Engineering. Polymers 2022, 14, 2532. [Google Scholar] [CrossRef]
- Schultz, H.S.; Nitze, L.M.; Zeuthen, L.H.; Keller, P.; Gruhler, A.; Pass, J.; Chen, J.; Guo, L.; Fleetwood, A.J.; Hamilton, J.A.; et al. Collagen Induces Maturation of Human Monocyte-Derived Dendritic Cells by Signaling through Osteoclast-Associated Receptor. J. Immunol. 2015, 194, 3169–3179. [Google Scholar] [CrossRef]
- Harris, A.F.; Lacombe, J.; Liyanage, S.; Han, M.Y.; Wallace, E.; Karsunky, S.; Abidi, N.; Zenhausern, F. Supercritical Carbon Dioxide Decellularization of Plant Material to Generate 3D Biocompatible Scaffolds. Sci. Rep. 2021, 11, 3643. [Google Scholar] [CrossRef]
- Leonard, A.R.; Cumming, M.H.; Ali, M.A.; Cabral, J.D. Fish Collagen Cross-Linking Strategies to Improve Mechanical and Bioactive Capabilities for Tissue Engineering and Regenerative Medicine. Adv. Funct. Mater. 2024, 34, 2405335. [Google Scholar] [CrossRef]
- Proksch, E.; Schunck, M.; Zague, V.; Segger, D.; Degwert, J.; Oesser, S. Oral Intake of Specific Bioactive Collagen Peptides Reduces Skin Wrinkles and Increases Dermal Matrix Synthesis. Skin Pharmacol. Physiol. 2014, 27, 113–119. [Google Scholar] [CrossRef]
- Li, M.; Chen, L.M.; Jiang, B.Q. Enzymatic Reaction Kinetics of Papain-Extracted Collagen from Bighead Carp Scales. Appl. Mech. Mater. 2015, 727–728, 56–60. [Google Scholar] [CrossRef]
- Ge, B.; Hou, C.; Bao, B.; Pan, Z.; De Val, J.E.M.S.; Elango, J.; Wu, W. Comparison of Physicochemical and Structural Properties of Acid-Soluble and Pepsin-Soluble Collagens from Blacktip Reef Shark Skin. Mar. Drugs 2022, 20, 376. [Google Scholar] [CrossRef] [PubMed]
- Klink, C.D.; Binnebösel, M.; Alizai, H.P.; Lambertz, A.; vonTrotha, K.T.; Junker, E.; Disselhorst-Klug, C.; Neumann, U.P.; Klinge, U. Tension of Knotted Surgical Sutures Shows Tissue Specific Rapid Loss in a Rodent Model. BMC Surg. 2011, 11, 36. [Google Scholar] [CrossRef]
- Batioglu-Karaaltin, A.; Ovali, E.; Karaaltin, M.V.; Yener, M.; Yılmaz, M.; Eyüpoğlu, F.; Yılmaz, Y.Z.; Bozkurt, E.R.; Demir, N.; Konuk, E.; et al. Decellularization of Trachea with Combined Techniques for Tissue-Engineered Trachea Transplantation. Clin. Exp. Otorhinolaryngol. 2019, 12, 86–94. [Google Scholar] [CrossRef]
- Zhou, P.; Huang, Y.; Guo, Y.; Wang, L.; Ling, C.; Guo, Q.; Wang, Y.; Zhu, S.; Fan, X.; Zhu, M.; et al. Decellularization and Recellularization of Rat Livers with Hepatocytes and Endothelial Progenitor Cells. Artif. Organs 2016, 40, E25–E38. [Google Scholar] [CrossRef] [PubMed]
- Koo, M.-A.; Jeong, H.; Hong, S.H.; Seon, G.M.; Lee, M.H.; Park, J.-C. Preconditioning Process for Dermal Tissue Decellularization Using Electroporation with Sonication. Regen. Biomater. 2022, 9, rbab071. [Google Scholar] [CrossRef]
- Liao, J.; Xu, B.; Zhang, R.; Fan, Y.; Xie, H.; Li, X. Applications of Decellularized Materials in Tissue Engineering: Advantages, Drawbacks and Current Improvements, and Future Perspectives. J. Mater. Chem. B 2020, 8, 10023–10049. [Google Scholar] [CrossRef]
- Tajima, K.; Kuroda, K.; Otaka, Y.; Kinoshita, R.; Kita, M.; Oyamada, T.; Kanai, K. Decellularization of Canine Kidney for Three-Dimensional Organ Regeneration. Vet. World 2020, 13, 452–457. [Google Scholar] [CrossRef]
- Chen, H.; Yin, B.; Hu, B.; Zhang, B.; Liu, J.; Jing, Y.; Fan, Z.; Tian, Y.; Wei, X.; Zhang, W. Acellular Fish Skin Enhances Wound Healing by Promoting Angiogenesis and Collagen Deposition. Biomed. Mater. 2021, 16, 045011. [Google Scholar] [CrossRef]
- Howaili, F.; Mashreghi, M.; Shahri, N.M.; Kompany, A.; Jalal, R. Development and Evaluation of a Novel Beneficent Antimicrobial Bioscaffold Based on Animal Waste-Fish Swim Bladder (FSB) Doped with Silver Nanoparticles. Environ. Res. 2020, 188, 109823. [Google Scholar] [CrossRef] [PubMed]
- Kamalvand, M.; Biazar, E.; Daliri-Joupari, M.; Montazer, F.; Rezaei-Tavirani, M.; Heidari-Keshel, S. Design of a Decellularized Fish Skin as a Biological Scaffold for Skin Tissue Regeneration. Tissue Cell 2021, 71, 101509. [Google Scholar] [CrossRef]
- Lin, X.; Kong, B.; Zhu, Y.; Zhao, Y. Bioactive Fish Scale Scaffolds with MSCs-Loading for Skin Flap Regeneration. Adv. Sci. 2022, 9, 2201226. [Google Scholar] [CrossRef]
- Chen, W.C.W.; Wang, Z.; Missinato, M.A.; Park, D.W.; Long, D.W.; Liu, H.-J.; Zeng, X.; Yates, N.A.; Kim, K.; Wang, Y. Decellularized Zebrafish Cardiac Extracellular Matrix Induces Mammalian Heart Regeneration. Sci. Adv. 2016, 2, e1600844. [Google Scholar] [CrossRef]
- Thomas-Porch, C.; Li, J.; Zanata, F.; Martin, E.C.; Pashos, N.; Genemaras, K.; Poche, J.N.; Totaro, N.P.; Bratton, M.R.; Gaupp, D.; et al. Comparative Proteomic Analyses of Human Adipose Extracellular Matrices Decellularized Using Alternative Procedures. J. Biomed. Mater. Res. 2018, 106, 2481–2493. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhang, F.; Wang, H.; Pan, T. Preparation and Characterization of a Novel Acellular Swim Bladder as Dura Mater Substitute. Neurol. Res. 2019, 41, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, W.; Dai, Y.; Huang, Y.; Chen, Z.; Xi, T.; Zhou, Z.; Liu, H. Decellularized Sturgeon Cartilage Extracellular Matrix Scaffold Inhibits Chondrocyte Hypertrophy In Vitro and In Vivo. J. Tissue Eng. Regen. Med. 2021, 15, 732–744. [Google Scholar] [CrossRef]
- Guo, F.; Jin, J.; Lin, W.; Gorostidi, M.; Yang, J.; Liu, L.; Chen, X. Preparation and Characterization of a Rat Uterine Decellularized Scaffold. Gland. Surg. 2024, 13, 2372–2382. [Google Scholar] [CrossRef]
- Suzuki, M.; Kimura, T.; Yoshida, Y.; Kobayashi, M.; Hashimoto, Y.; Takahashi, H.; Shimizu, T.; Anzai, S.; Nakamura, N.; Kishida, A. In Vitro Tissue Reconstruction Using Decellularized Pericardium Cultured with Cells for Ligament Regeneration. Polymers 2022, 14, 2351. [Google Scholar] [CrossRef]
- Lin, C.-H.; Hsia, K.; Su, C.-K.; Chen, C.-C.; Yeh, C.-C.; Ma, H.; Lu, J.-H. Sonication-Assisted Method for Decellularization of Human Umbilical Artery for Small-Caliber Vascular Tissue Engineering. Polymers 2021, 13, 1699. [Google Scholar] [CrossRef]
- Bera, A.K.; Sriya, Y.; Pati, F. Formulation of Dermal Tissue Matrix Bioink by a Facile Decellularization Method and Process Optimization for 3D Bioprinting toward Translation Research. Macromol. Biosci. 2022, 22, 2200109. [Google Scholar] [CrossRef]
- Li, D.; Sun, W.Q.; Wang, T.; Gao, Y.; Wu, J.; Xie, Z.; Zhao, J.; He, C.; Zhu, M.; Zhang, S.; et al. Evaluation of a Novel Tilapia-Skin Acellular Dermis Matrix Rationally Processed for Enhanced Wound Healing. Mater. Sci. Eng. C 2021, 127, 112202. [Google Scholar] [CrossRef]
- Fang, Z.; Wang, Y.; Feng, Q.; Kienzle, A.; Müller, W.E.G. Hierarchical Structure and Cytocompatibility of Fish Scales from Carassius Auratus. Mater. Sci. Eng. C 2014, 43, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Gullabzada, M. A Systematic Review of Tissue Engineering of a Total Artificial Heart: Focus on Perfusion Decellularization. J. Protoc. Biol. Methods 2022, 2, 1–19. [Google Scholar] [CrossRef]
- Chun, S.Y.; Lee, J.N.; Ha, Y.-S.; Yoon, B.H.; Lee, E.H.; Kim, B.M.; Gil, H.; Han, M.-H.; Oh, W.S.; Kwon, T.G.; et al. Optimization of Extracellular Matrix Extraction from Human Perirenal Adipose Tissue. J. Biomater. Appl. 2021, 35, 1180–1191. [Google Scholar] [CrossRef] [PubMed]
- Irastorza, A.; Vazquez Aristizabal, P.; Zumeta Olaskoaga, L.; Mateo Abad, M.; Guerrero, P.; de la Caba, K.; Izeta, A. Comparative Proteomic Analysis of the Composition of Decellularized Extracellular Matrix (dECM) and dECM-Based Inks as Compared to the Native Tissue. arXiv 2024, arXiv:2410.07064. [Google Scholar]
- Parekh, M.; Van den Bogerd, B.; Zakaria, N.; Ponzin, D.; Ferrari, S. Fish Scale-Derived Scaffolds for Culturing Human Corneal Endothelial Cells. Stem Cells Int. 2018, 2018, 8146834. [Google Scholar] [CrossRef]
- Song, M.; Liu, Y.; Hui, L. Preparation and Characterization of Acellular Adipose Tissue Matrix Using a Combination of Physical and Chemical Treatments. Mol. Med. Rep. 2017, 17, 138–146. [Google Scholar] [CrossRef]
- Wu, W.; Zhou, Z.; Sun, G.; Liu, Y.; Zhang, A.; Chen, X. Construction and Characterization of Degradable Fish Scales for Enhancing Cellular Adhesion and Potential Using as Tissue Engineering Scaffolds. Mater. Sci. Eng. C 2021, 122, 111919. [Google Scholar] [CrossRef]
- Bual, R.; Labares, M.; Valle, K.D.D.; Pague, J.; Bantilan, Z.C.; Ducao, P.G.; Alimasag, J.; Acibar, C. Characterization of Decellularized Extracellular Matrix from Milkfish (Chanos chanos) Skin. Biomimetics 2022, 7, 213. [Google Scholar] [CrossRef]
- Roth, S.P.; Glauche, S.M.; Plenge, A.; Erbe, I.; Heller, S.; Burk, J. Automated Freeze-Thaw Cycles for Decellularization of Tendon Tissue—A Pilot Study. BMC Biotechnol. 2017, 17, 13. [Google Scholar] [CrossRef]
- Su, S.; Wang, R.; Bai, J.; Chen, Z.; Zhou, F. Novel Decellularization Scheme for Preparing Acellular Fish Scale Scaffolds for Bone Tissue Engineering. ACS Omega 2025, 10, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Arellano, F.M.; Bual, R.; Bantilan, Z.C.; Alimasag, J.; Aron, J.; Baclayon, L.; Nisperos, M.J.; Valle, K.D.D.; Ducao, P.G.; Lumancas, G.; et al. Upcycling Waste Tilapia (Oreochromis niloticus) Scales through a Decellularization Process for Extracellular Matrix Extraction. Mater. Res. Express 2024, 11, 025101. [Google Scholar] [CrossRef]
- Barlisan, C.L.; Bual, R.; Valle, K.D.D.; Bacosa, H. Assessment of the Decellularized Extracellular Matrix (dECM) from the Skin of Yellowfin Tuna (Thunnus albacares). Philipp. J. Sci. 2024, 153, 2165–2170. [Google Scholar] [CrossRef]
- Khajavi, M.; Hajimoradloo, A.; Zandi, M.; Pezeshki-Modaress, M.; Bonakdar, S.; Zamani, A. Fish Cartilage: A Promising Source of Biomaterial for Biological Scaffold Fabrication in Cartilage Tissue Engineering. J. Biomed. Mater. Res. 2021, 109, 1737–1750. [Google Scholar] [CrossRef]
- Wang, Y.; Song, W.; Yu, D.; Dai, Y.; Li, B. Preparation and Evaluation of Fish Skin Acellular Dermal Matrix for Oral Guided Tissue Regeneration. Chin. J. Tissue Eng. Res. 2019, 23, 1526–1532. [Google Scholar]
- Huh, M.-I.; Lee, K.-P.; Kim, J.; Yi, S.; Park, B.-U.; Kim, H.K. Generation of Femtosecond Laser-Cut Decellularized Corneal Lenticule Using Hypotonic Trypsin-EDTA Solution for Corneal Tissue Engineering. J. Ophthalmol. 2018, 2018, 2590536. [Google Scholar] [CrossRef] [PubMed]
- Cornelison, R.C.; Wellman, S.M.; Park, J.H.; Porvasnik, S.L.; Song, Y.H.; Wachs, R.A.; Schmidt, C.E. Development of an Apoptosis-Assisted Decellularization Method for Maximal Preservation of Nerve Tissue Structure. Acta Biomater. 2018, 77, 116–126. [Google Scholar] [CrossRef]
- Jiwangga, D.; Mahyudin, F.; Mastutik, G.; Juliana; Meitavany, E.N. Current Strategies for Tracheal Decellularization: A Systematic Review. Int. J. Biomater. 2024, 2024, 3355239. [Google Scholar] [CrossRef]
- Kara, A.; Tamburaci, S.; Tihminlioglu, F.; Havitcioglu, H. Bioactive Fish Scale Incorporated Chitosan Biocomposite Scaffolds for Bone Tissue Engineering. Int. J. Biol. Macromol. 2019, 130, 266–279. [Google Scholar] [CrossRef] [PubMed]
- Biazar, E.; Heidari Keshel, S.; Rezaei Tavirani, M.; Kamalvand, M. Healing Effect of Acellular Fish Skin with Plasma Rich in Growth Factor on Full-thickness Skin Defects. Int. Wound J. 2022, 19, 2154–2162. [Google Scholar] [CrossRef]
- Lv, K.; Wang, L.; He, X.; Li, W.; Han, L.; Qin, S. Application of Tilapia Skin Acellular Dermal Matrix to Induce Acute Skin Wound Repair in Rats. Front. Bioeng. Biotechnol. 2022, 9, 792344. [Google Scholar] [CrossRef]
- Lau, C.S.; Hassanbhai, A.; Wen, F.; Wang, D.; Chanchareonsook, N.; Goh, B.T.; Yu, N.; Teoh, S. Evaluation of Decellularized Tilapia Skin as a Tissue Engineering Scaffold. J. Tissue Eng. Regen. Med. 2019, 13, 1779–1791. [Google Scholar] [CrossRef]
- Williams, C.; Liao, J.; Joyce, E.M.; Wang, B.; Leach, J.B.; Sacks, M.S.; Wong, J.Y. Altered Structural and Mechanical Properties in Decellularized Rabbit Carotid Arteries. Acta Biomater. 2009, 5, 993–1005. [Google Scholar] [CrossRef]
- Vettese, J.; Manon, J.; Chretien, A.; Evrard, R.; Fievé, L.; Schubert, T.; Lengelé, B.G.; Behets, C.; Cornu, O. Collagen Molecular Organization Preservation in Human Fascia Lata and Periosteum after Tissue Engineering. Front. Bioeng. Biotechnol. 2024, 12, 1275709. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Z.; Yu, Q.; Xi, H.; Weng, J.; Du, X.; Chen, D.; Ma, J.; Mei, J.; Chen, C. Comparative Study of Two Perfusion Routes with Different Flow in Decellularization to Harvest an Optimal Pulmonary Scaffold for Recellularization. J. Biomed. Mater. Res. 2016, 104, 2567–2575. [Google Scholar] [CrossRef]
- Adhikari, S.P.; Paudel, A.; Sharma, A.; Thapa, B.; Khanal, N.; Shastri, N.; Rai, S.; Adhikari, R. Development of Decellularized Fish Skin Scaffold Decorated with Biosynthesized Silver Nanoparticles for Accelerated Burn Wound Healing. Int. J. Biomater. 2023, 2023, 8541621. [Google Scholar] [CrossRef]
- Ibrahim, A.; Fahmy, H.M.; Mahmoud, G.A.-E.; Soliman, M.; Elshahawy, A.M. New Strategies for Sterilization and Preservation of Fresh Fish Skin Grafts. Sci. Rep. 2024, 14, 1253. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Zhang, B.; Li, J.; Cen, L.; Zhao, L.; Xi, Z. Preparation of Extracellular Matrix of Fish Swim Bladders by Decellularization with Supercritical Carbon Dioxide. Bioresour. Bioprocess. 2023, 10, 14. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.-Y.; Kim, T.-H.; Kim, S.-C.; Oh, G.-W.; Heo, S.-J.; Jung, W.-K. Therapeutic Effect of Decellularized Extracellular Matrix from Fish Skin for Accelerating Skin Regeneration. Mar. Drugs 2024, 22, 437. [Google Scholar] [CrossRef]
- Lee, S.H.; Noh, S.; Pack, S.P.; Kim, G.; Lee, H. Low-Temperature 3D-Printed Aloe Vera/Fish-Derived Decellularized ECM Biocomposites for Wound Dressing Application: Fabrication, Characterization, and in Vitro Evaluation. Mater. Des. 2025, 258, 114556. [Google Scholar] [CrossRef]
- Jo, S.; Hwangbo, H.; Francis, N.; Lee, J.; Pei, M.; Kim, G. Fish-Derived Biomaterials for Tissue Engineering: Advances in Scaffold Fabrication and Applications in Regenerative Medicine and Cancer Therapy. Theranostics 2025, 15, 5666–5692. [Google Scholar] [CrossRef] [PubMed]
- Panggabean, J.A.; Adiguna, S.P.; Hardhiyuna, M.; Rahmawati, S.I.; Sadi, N.H.; Yoga, G.P.; Nafisyah, E.; Bayu, A.; Putra, M.Y. Cutting Edge Aquatic-Based Collagens in Tissue Engineering. Mar. Drugs 2023, 21, 87. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Chen, X.; Liu, X.; Wen, G.; Yu, Y. Recent Advances in Decellularized Biomaterials for Wound Healing. Mater. Today Bio 2023, 19, 100589. [Google Scholar] [CrossRef]
- Fiakos, G.; Kuang, Z.; Lo, E. Improved Skin Regeneration with Acellular Fish Skin Grafts. Eng. Regen. 2020, 1, 95–101. [Google Scholar] [CrossRef]
- Le, K.M.; Le, M.-A.T.; Quan, V.M.; Vu, T.L.; Tran, T.A.; Vo, P.P.; Lieu, A.H.; To, H.T.N.; Nguyen, T.-H.; Doan, H.N. Preparation and Characterization of Acellular Membrane from Yellowfin Tuna (Thunnus albacares) Skin for Skin Regeneration. Collagen Leather 2025, 7, 32. [Google Scholar] [CrossRef]
- Wang, M.-H.; Kuan, C.-H.; Ma, H.; Cheng, N.-C. Axolotl-Derived Decellularized Skin ECM as a pro-Regenerative Scaffold for Attenuating Fibrotic Wound Healing. Mater. Today Bio 2025, 35, 102443. [Google Scholar] [CrossRef]
- Esmaeili, A.; Rahimi, A.; Abbasi, A.; Hasannejad-Asl, B.; Bagheri-Mohammadi, S.; Farjami, M.; Keshel, S.H. Processing and Post-Processing of Fish Skin as a Novel Material in Tissue Engineering. Tissue Cell 2023, 85, 102238. [Google Scholar] [CrossRef]
- Liu, C.; Sun, J. Potential Application of Hydrolyzed Fish Collagen for Inducing the Multidirectional Differentiation of Rat Bone Marrow Mesenchymal Stem Cells. Biomacromolecules 2014, 15, 436–443. [Google Scholar] [CrossRef]
- Zheng, P.; Jia, Q.; Li, Z.; Jiang, H.B.; Zhou, L. Enhanced Osteogenic and Angiogenic Capabilities of Adipose-Derived Stem Cells in Fish Collagen Scaffolds for Treatment of Femoral Head Osteonecrosis. Sci. Rep. 2025, 15, 18300. [Google Scholar] [CrossRef]
- Rizzo, M.G.; Briglia, M.; Zammuto, V.; Morganti, D.; Faggio, C.; Impellitteri, F.; Multisanti, C.R.; Graziano, A.C.E. Innovation in Osteogenesis Activation: Role of Marine-Derived Materials in Bone Regeneration. Curr. Issues Mol. Biol. 2025, 47, 175. [Google Scholar] [CrossRef]
- Liu, Z.; Jiang, X.; Wang, K.; Zhou, Y.; Li, T.; Gao, J.; Wang, L. Preparation of Fish Decalcified Bone Matrix and Its Bone Repair Effect in Rats. Front. Bioeng. Biotechnol. 2023, 11, 1134992. [Google Scholar] [CrossRef]
- Dean, M.N.; Shahar, R. The Structure-Mechanics Relationship and the Response to Load of the Acellular Bone of Neoteleost Fish: A Review: Acellular Bone Structure-Mechanics. J. Appl. Ichthyol. 2012, 28, 320–329. [Google Scholar] [CrossRef]
- Jo, S.; Lee, H.; Jo, Y.; Jin, E.-J.; Kim, D.; Ryu, D.; Kim, G.H. Bioengineered Cell-Constructs Using Decellularized Fish Skin-Based Composite Bioink for Regenerating Muscle Tissue. Appl. Phys. Rev. 2024, 11, 021404. [Google Scholar] [CrossRef]
- Wu, T.; Han, L.; Zhu, Y.; Zeng, X.; Kang, Y.; Zheng, S.; Wang, Z.; Wang, J.; Gao, Y. Application of Decalcified Bone Matrix in Salmon Bone for Tibial Defect Repair in Rat Model. Int. J. Artif. Organs 2024, 47, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yu, M.-Z.; Peng, H.; Liu, R.-T.; Lim, T.; Zhang, C.-Q.; Zhu, Z.-Z.; Wei, X.-J. Decellularized Tilapia Fish Skin: A Novel Candidate for Tendon Tissue Engineering. Mater. Today Bio 2022, 17, 100488. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Zhang, X.; Jin, Y.; Chien, P.N.; Heo, C.Y. Acellular Extracellular Matrix Scaffolds in Regenerative Medicine: Advances in Decellularization and Clinical Applications. J. Funct. Biomater. 2025, 16, 383. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, X.; Gao, L.; Jing, L.; Zhou, Q.; Chang, J. The Role of the Micro-Pattern and Nano-Topography of Hydroxyapatite Bioceramics on Stimulating Osteogenic Differentiation of Mesenchymal Stem Cells. Acta Biomater. 2018, 73, 509–521. [Google Scholar] [CrossRef]
- Jin, S.; Sun, F.; Zou, Q.; Huang, J.; Zuo, Y.; Li, Y.; Wang, S.; Cheng, L.; Man, Y.; Yang, F.; et al. Fish Collagen and Hydroxyapatite Reinforced Poly(Lactide-Co-Glycolide) Fibrous Membrane for Guided Bone Regeneration. Biomacromolecules 2019, 20, 2058–2067. [Google Scholar] [CrossRef]
- Lin, W.; Wang, G.; Jin, M.; Li, Z.; Ao, R.; Wu, H.; Sun, C.; An, Y. Research on Cartilage Repair and Biomimetic Applications of dECM Derived from Various Tissues. Tissue Eng. Part B Rev. 2025, 2025, 19373368251391032. [Google Scholar] [CrossRef]
- Chen, Y.-W.; Lin, Y.-H.; Lin, T.-L.; Lee, K.-X.A.; Yu, M.-H.; Shie, M.-Y. 3D-Biofabricated Chondrocyte-Laden Decellularized Extracellular Matrix-Contained Gelatin Methacrylate Auxetic Scaffolds under Cyclic Tensile Stimulation for Cartilage Regeneration. Biofabrication 2023, 15, 045007. [Google Scholar] [CrossRef]
- Tavares, D.F.; Mano, J.F.; Oliveira, M.B. Advances in Abiotic Tissue-Based Biomaterials: A Focus on Decellularization and Devitalization Techniques. Mater. Today Bio 2025, 32, 101735. [Google Scholar] [CrossRef]
- Karageorgou, D.; Rova, U.; Christakopoulos, P.; Katapodis, P.; Matsakas, L.; Patel, A. Benefits of Supplementation with Microbial Omega-3 Fatty Acids on Human Health and the Current Market Scenario for Fish-Free Omega-3 Fatty Acid. Trends Food Sci. Technol. 2023, 136, 169–180. [Google Scholar] [CrossRef]
- Rahim, M.A.; Imran, M.; Khan, M.K.; Haseeb Ahmad, M.; Nadeem, M.; Khalid, W.; Lorenzo, J.M.; Afifi, M.M.; AL-Farga, A.; Aljobair, M.O. Omega-3 Fatty Acid Retention and Oxidative Stability of Spray-Dried Chia–Fish-Oil-Prepared Microcapsules. Processes 2022, 10, 2184. [Google Scholar] [CrossRef]
- Djuricic, I.; Calder, P.C. N-3 Fatty Acids (EPA and DHA) and Cardiovascular Health—Updated Review of Mechanisms and Clinical Outcomes. Curr. Atheroscler. Rep. 2025, 27, 116. [Google Scholar] [CrossRef]
- Gzik-Zroska, B.; Joszko, K.; Wolański, W.; Suchoń, S.; Burkacki, M.; Ples, M.; Malachowski, J.; Tomaszewski, M.; Szarek, A.; Stradomski, G.; et al. Assessment of the Impact of Decellularization Methods on Mechanical Properties of Biocomposites Used as Skin Substitute. Materials 2021, 14, 4785. [Google Scholar] [CrossRef] [PubMed]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound Repair and Regeneration: Mechanisms, Signaling, and Translation. Sci. Transl. Med. 2014, 6, 265sr1. [Google Scholar] [CrossRef]
- Boleti, A.P.D.A.; Jacobowski, A.C.; Frihling, B.E.F.; Cruz, M.V.; Santos, K.F.D.P.; Migliolo, L.; De Andrade, L.R.M.; Macedo, M.L.R. Wound Healing: Molecular Mechanisms, Antimicrobial Peptides, and Emerging Technologies in Regenerative Medicine. Pharmaceuticals 2025, 18, 1525. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lin, Z.; Liu, R.; Liu, Z.; Peng, H.; Hu, Z.; Fu, W.; Jin, L.; Zhang, C.; Tang, Q.; et al. Fish Acellular Dermal Matrix Promotes Repair of Full-Thickness Skin Defects in Mice and Bama Pigs. Wound Repair Regen. 2025, 33, e70091. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Mao, Z.; Zhao, C.; Fan, Z.; Yang, H.; Xia, A.; Zhang, X. Fish Skin Dressing for Wound Regeneration: A Bioactive Component Review of Omega-3 PUFAs, Collagen and ECM. Int. J. Biol. Macromol. 2024, 283, 137831. [Google Scholar] [CrossRef]
- Luze, H.; Nischwitz, S.P.; Smolle, C.; Zrim, R.; Kamolz, L.-P. The Use of Acellular Fish Skin Grafts in Burn Wound Management—A Systematic Review. Medicina 2022, 58, 912. [Google Scholar] [CrossRef]
- Lin, X.; Zhang, H.; Zhang, H.; Zhang, Z.; Chen, G.; Zhao, Y. Bio-Printed Hydrogel Textiles Based on Fish Skin Decellularized Extracellular Matrix for Wound Healing. Engineering 2023, 25, 120–127. [Google Scholar] [CrossRef]
- Stone, R.; Saathoff, E.C.; Larson, D.A.; Wall, J.T.; Wienandt, N.A.; Magnusson, S.; Kjartansson, H.; Natesan, S.; Christy, R.J. Accelerated Wound Closure of Deep Partial Thickness Burns with Acellular Fish Skin Graft. Int. J. Mol. Sci. 2021, 22, 1590. [Google Scholar] [CrossRef]
- Liang, R.; Pan, R.; He, L.; Dai, Y.; Jiang, Y.; He, S.; Li, B.; Li, Y. Decellularized Extracellular Matrices for Skin Wound Treatment. Materials 2025, 18, 2752. [Google Scholar] [CrossRef]
- Chen, M.; Jiang, Z.; Zou, X.; You, X.; Cai, Z.; Huang, J. Advancements in Tissue Engineering for Articular Cartilage Regeneration. Heliyon 2024, 10, e25400. [Google Scholar] [CrossRef]
- Dechwongya, P.; Sathirakul, K.; Kristjánsson, B.; Chirachanchai, S.; Honsawek, S. Preparation and Characterization of Demineralized Bone Matrix/Chitosan Composite Scaffolds for Bone Tissue Engineering. Chulalongkorn Med. J. 2019, 63, 119–126. [Google Scholar] [CrossRef]
- Lingam, P.C.; Senthil, R.; Kumar, P.; Kumar, J.K. Preparation and Characterization of Demineralised Bone Matrix from Fish Waste. J. Pioneer. Med. Sci. 2023, 12, 17–20. [Google Scholar] [CrossRef]
- Elango, J.; Zhang, J.; Bao, B.; Palaniyandi, K.; Wang, S.; Wenhui, W.; Robinson, J.S. Rheological, Biocompatibility and Osteogenesis Assessment of Fish Collagen Scaffold for Bone Tissue Engineering. Int. J. Biol. Macromol. 2016, 91, 51–59. [Google Scholar] [CrossRef]
- Liu, C.; Sun, J. Hydrolyzed Tilapia Fish Collagen Induces Osteogenic Differentiation of Human Periodontal Ligament Cells. Biomed. Mater. 2015, 10, 065020. [Google Scholar] [CrossRef]
- Wang, Y.; Kong, B.; Chen, X.; Liu, R.; Zhao, Y.; Gu, Z.; Jiang, Q. BMSC Exosome-Enriched Acellular Fish Scale Scaffolds Promote Bone Regeneration. J. Nanobiotechnol 2022, 20, 444. [Google Scholar] [CrossRef]
- Xia, C.; Mei, S.; Gu, C.; Zheng, L.; Fang, C.; Shi, Y.; Wu, K.; Lu, T.; Jin, Y.; Lin, X.; et al. Decellularized Cartilage as a Prospective Scaffold for Cartilage Repair. Mater. Sci. Eng. C 2019, 101, 588–595. [Google Scholar] [CrossRef]
- Pfeiffer, E.; Vickers, S.M.; Frank, E.; Grodzinsky, A.J.; Spector, M. The Effects of Glycosaminoglycan Content on the Compressive Modulus of Cartilage Engineered in Type II Collagen Scaffolds. Osteoarthr. Cartil. 2008, 16, 1237–1244. [Google Scholar] [CrossRef]
- Golebiowska, A.A.; Intravaia, J.T.; Sathe, V.M.; Kumbar, S.G.; Nukavarapu, S.P. Decellularized Extracellular Matrix Biomaterials for Regenerative Therapies: Advances, Challenges and Clinical Prospects. Bioact. Mater. 2024, 32, 98–123. [Google Scholar] [CrossRef]
- Abbasnezhad, S.; Biazar, E.; Aavani, F.; Kamalvand, M.; Heidari Keshel, S.; Pourjabbar, B. Chemical Modification of Acellular Fish Skin as a Promising Biological Scaffold by Carbodiimide Cross-linker for Wound Healing. Int. Wound J. 2023, 20, 1566–1577. [Google Scholar] [CrossRef]
- Qiao, S.; Peijie, T.; Nan, J. Crosslinking Strategies of Decellularized Extracellular Matrix in Tissue Regeneration. J. Biomed. Mater. Res. 2024, 112, 640–671. [Google Scholar] [CrossRef]
- Kasravi, M.; Ahmadi, A.; Babajani, A.; Mazloomnejad, R.; Hatamnejad, M.R.; Shariatzadeh, S.; Bahrami, S.; Niknejad, H. Immunogenicity of Decellularized Extracellular Matrix Scaffolds: A Bottleneck in Tissue Engineering and Regenerative Medicine. Biomater. Res. 2023, 27, 10. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.; Wang, Y.; Li, B.; Hou, H. Cross-Linking Effects of Carbodiimide, Oxidized Chitosan Oligosaccharide and Glutaraldehyde on Acellular Dermal Matrix of Basa Fish (Pangasius bocourti). Int. J. Biol. Macromol. 2020, 164, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Akhshabi, S.; Biazar, E.; Singh, V.; Heidari Keshel, S.; Geetha, N. The Effect of the Carbodiimide Cross-Linker on the Structural and Biocompatibility Properties of Collagen-Chondroitin Sulfate Electrospun Mat. Int. J. Nanomed. 2018, 13, 4405–4416. [Google Scholar] [CrossRef]
- Lan, X.; Luo, M.; Li, M.; Mu, L.; Li, G.; Chen, G.; He, Z.; Xiao, J. Swim Bladder-Derived Biomaterials: Structures, Compositions, Properties, Modifications, and Biomedical Applications. J. Nanobiotechnol. 2024, 22, 186. [Google Scholar] [CrossRef]
- Sun, L.; Li, B.; Yao, D.; Song, W.; Hou, H. Effects of Cross-Linking on Mechanical, Biological Properties and Biodegradation Behavior of Nile Tilapia Skin Collagen Sponge as a Biomedical Material. J. Mech. Behav. Biomed. Mater. 2018, 80, 51–58. [Google Scholar] [CrossRef]
- Rowland, C.R.; Lennon, D.P.; Caplan, A.I.; Guilak, F. The Effects of Crosslinking of Scaffolds Engineered from Cartilage ECM on the Chondrogenic Differentiation of MSCs. Biomaterials 2013, 34, 5802–5812. [Google Scholar] [CrossRef]
- Bui, H.-T.D.; Cho, W.; Park, J.K.; Lee, M.S.; Kim, H.K.; Yoo, H.S. Korean Amberjack Skin-Inspired Hyaluronic Acid Bioink for Reconstruction of Human Skin. ACS Omega 2023, 8, 22752–22761. [Google Scholar] [CrossRef]
- Ma, C.; Choi, J.-B.; Jang, Y.-S.; Kim, S.-Y.; Bae, T.-S.; Kim, Y.-K.; Park, J.-M.; Lee, M.-H. Mammalian and Fish Gelatin Methacryloyl–Alginate Interpenetrating Polymer Network Hydrogels for Tissue Engineering. ACS Omega 2021, 6, 17433–17441. [Google Scholar] [CrossRef]
- Yoon, H.J.; Shin, S.R.; Cha, J.M.; Lee, S.-H.; Kim, J.-H.; Do, J.T.; Song, H.; Bae, H. Cold Water Fish Gelatin Methacryloyl Hydrogel for Tissue Engineering Application. PLoS ONE 2016, 11, e0163902. [Google Scholar] [CrossRef]
- Masoumi Shahrbabak, S.; Jalali, S.M.; Fathabadi, M.F.; Tayebi-Khorrami, V.; Amirinejad, M.; Forootan, S.; Saberifar, M.; Fadaei, M.R.; Najafi, Z.; Askari, V.R. Modified Alginates for Precision Drug Delivery: Advances in Controlled-Release and Targeting Systems. Int. J. Pharm. X 2025, 10, 100381. [Google Scholar] [CrossRef]
- Vasile, C.; Pamfil, D.; Stoleru, E.; Baican, M. New Developments in Medical Applications of Hybrid Hydrogels Containing Natural Polymers. Molecules 2020, 25, 1539. [Google Scholar] [CrossRef]
- Pasanaphong, K.; Pukasamsombut, D.; Boonyagul, S.; Pengpanich, S.; Tawonsawatruk, T.; Wilairatanarporn, D.; Jantanasakulwong, K.; Rachtanapun, P.; Hemstapat, R.; Wangtueai, S.; et al. Fabrication of Fish Scale-Based Gelatin Methacryloyl for 3D Bioprinting Application. Polymers 2024, 16, 418. [Google Scholar] [CrossRef]
- Jia, Z.; Wang, Z. Photo-Crosslinking Hydrogel Based on Porcine Small Intestinal Submucosa Decellularized Matrix/Fish Collagen/GelMA for Culturing Small Intestinal Organoids and Repairing Intestinal Defects. Int. J. Mol. Sci. 2025, 26, 663. [Google Scholar] [CrossRef]
- Ren, H.; Huang, D.; Qiu, M.; Xue, L.; Zhu, S.; Gan, J.; Chen, C.; Chen, D.; Wang, J. Microfluidic 3D Printing Hydrogels Based on Fish Liver Decellularized Extracellular Matrix for Liver Regeneration. Smart Med. 2024, 3, e20240056. [Google Scholar] [CrossRef]
- Zhou, T.; Sui, B.; Mo, X.; Sun, J. Multifunctional and Biomimetic Fish Collagen/Bioactive Glass Nanofibers: Fabrication, Antibacterial Activity and Inducing Skin Regeneration In Vitro and In Vivo. Int. J. Nanomed. 2017, 12, 3495–3507. [Google Scholar] [CrossRef]
- Yano, S.; Yamaguchi, K.; Shibata, M.; Ifuku, S.; Teramoto, N. Photocrosslinked Fish Collagen Peptide/Chitin Nanofiber Composite Hydrogels from Marine Resources: Preparation, Mechanical Properties, and an In Vitro Study. Polymers 2023, 15, 682. [Google Scholar] [CrossRef]
- Chung, J.J.; Im, H.; Kim, S.H.; Park, J.W.; Jung, Y. Toward Biomimetic Scaffolds for Tissue Engineering: 3D Printing Techniques in Regenerative Medicine. Front. Bioeng. Biotechnol. 2020, 8, 586406. [Google Scholar] [CrossRef]
- Ibrahim, M.; Ayyoubi, H.S.; Alkhairi, L.A.; Tabbaa, H.; Elkins, I.; Narvel, R. Fish Skin Grafts Versus Alternative Wound Dressings in Wound Care: A Systematic Review of the Literature. Cureus 2023, 15, e36348. [Google Scholar] [CrossRef]
- Figueiró, S.D.; Macêdo, A.A.M.; Melo, M.R.S.; Freitas, A.L.P.; Moreira, R.A.; De Oliveira, R.S.; Góes, J.C.; Sombra, A.S.B. On the Dielectric Behaviour of Collagen–Algal Sulfated Polysaccharide Blends: Effect of Glutaraldehyde Crosslinking. Biophys. Chem. 2006, 120, 154–159. [Google Scholar] [CrossRef]
- Liu, S.; Lau, C.-S.; Liang, K.; Wen, F.; Teoh, S.H. Marine Collagen Scaffolds in Tissue Engineering. Curr. Opin. Biotechnol. 2022, 74, 92–103. [Google Scholar] [CrossRef]
- Lima-Verde, M.E.Q.; Parthiban, S.P.; Junior, A.E.C.F.; de Barros Silva, P.G.; Junior, E.M.L.; de Moraes, M.O.; de Paulo Aragão Sabóia, V.; Bertassoni, L.E.; Alves, A.P.N.N. Nile Tilapia Fish Skin, Scales, and Spine as Naturally Derived Biomaterials for Tissue Regeneration. Curr. Oral Health Rep. 2020, 7, 335–343. [Google Scholar] [CrossRef]
- Zimba, B.L.; Rwiza, M.J.; Sauli, E. Utilizing Tilapia Fish Skin Biomaterial for Burn Wound Dressing: A Systematic Review. Sci. Afr. 2024, 24, e02245. [Google Scholar] [CrossRef]
- Sultana, N.; Cole, A.; Strachan, F. Biocomposite Scaffolds for Tissue Engineering: Materials, Fabrication Techniques and Future Directions. Materials 2024, 17, 5577. [Google Scholar] [CrossRef]
- Valério, N.; Soares, M.; Vilarinho, C.; Correia, M.; Carvalho, J. Diving into Fish Valorisation: Review Opportunities and Analyzing Azorean Fish Data. Processes 2023, 11, 1998. [Google Scholar] [CrossRef]

| Chemical Treatment | Fish Species | Fish Tissue/Organ | Decellularization Protocol | Results | References |
|---|---|---|---|---|---|
| Ionic detergent | Oreochromis niloticus (Tilapia) | Scales | Immersed in varying SDS concentrations (0.1%, 0.5%, and 1%) at 4 °C for 72 h, then washed. | Higher SDS concentrations resulted in more protein loss due to its denaturing effect. | [56] |
| Non-ionic detergent | Oreochromis niloticus (Tilapia) | Scales | Immersed in varying TX-100 concentrations (0.1%, 0.5%, and 1%) at 4 °C for 72 h, then washed. | TX-100 was better at preserving the structure and protein content of the dECM than SDS. | [56] |
| Ionic detergent + Hypotonic solution + Chelating agent | Sparus aurata (Gilt-head bream) | Scales | Treated with 0.1% SDS in Tris-HCl buffer and 0.1% EDTA at 4 °C for 3 days. | Fully removed cells, preserved collagen I and porosity (70–78%), as confirmed by SEM and FTIR. Mechanical stability remained high (compression modulus: 13.5–14.3 kPa). No cytotoxic effects were observed, with increasing cell viability and enhanced osteogenic differentiation. | [63] |
| Ctenopharyngodon idella (Grass carps) | Scales | The sample was incubated in 10 mM Tris-HCl and 0.1% EDTA at 4 °C for 24 h, followed by 0.1% SDS treatment at 4 °C for three days. It was then rinsed five times with distilled water to remove residual chemicals. | SDS treatment led to the complete removal of cells with DNA content less than 50 ng/mg. Hydroxyapatite in the scaffold decreased its content by 55.61%, while there was 55.49% collagen lost. The surface microstructure was destroyed, as shown in SEM. The material exhibited a degradation rate of 49.89 ± 6.56% over eight weeks. The Young’s Modulus decreased significantly to 3.63 ± 2.34 GPa. | [55] | |
| Non-ionic detergent + Hypertonic and hypotonic solutions + Chelating agent | Ctenopharyngodon idella (Grass carps) | Skin | Hypertonic treatment (0.5 M NaCl, 25–50 mM Tris, 10 mM EDTA) to break cell membranes, followed by 0.5% TX-100 treatment (24 h) for further cell removal. Finally, the sample was washed with PBS and cold distilled water to eliminate any remaining detergents. | The scaffold had a porous structure (20–100 μm), enhancing cell adhesion and proliferation. SEM confirmed strong epithelial cell attachment, supporting skin regeneration. | [64] |
| Basic solution + Hypertonic solution | Oreochromis niloticus (Tilapia) | Skin | Soaked in 0.1 M NaOH for 8 h, followed by soaking in 1 M NaCl solution for 12 h, then soaked in 0.1 M NaOH again for 6 h. | The scaffold was porous and spongy, with complete cell removal and preserved collagen integrity. Hydroxyproline analysis showed minimal collagen loss, and SEM revealed a two-layered, highly porous structure supporting fibroblast adhesion. FTIR confirmed collagen presence, Calcein AM-PI staining confirmed high cell viability, while mechanical testing revealed a tensile strength of 21.76 MPa. | [65] |
| Acid solution + Chelating agent | Carassius auratus (Goldfish) | Scales | Treated with 5% nitric acid (10 h), and decalcified in 10 wt.% EDTA + 2% nitric acid (3 days, 4 °C, with daily renewal). | Final material was mainly organic (3.5 wt.% inorganic content) with intact fibrillary collagen layers. Cytocompatibility tests showed high cell adhesion and proliferation, with cells aligning along natural ridge channels. | [57] |
| Physical-Chemical Treatment | Fish Species | Fish Tissue/Organ | Decellularization Protocol | Results | References |
|---|---|---|---|---|---|
| Ionic detergent + Agitation | Thunnus albacares (Yellowfin Tuna) | Skin | Treated with 0.1% SDS for 2 days at 300 rpm and 4 °C, with solution changes every 24 h, then washed. | H&E staining confirmed significant nuclear loss, while ATR-FTIR showed retention of the collagen triple-helix structure. SEM imaging revealed a smooth inner surface and well-organized collagen fibers, maintaining ECM integrity. DSC analysis indicated thermal stability (71.93 °C). | [57] |
| Chanos chanos (Milkfish) | Skin | Treated with 0.1% and 1% SDS at 4 °C for 24 h with constant 300 rpm agitation, then washed. | 1% SDS achieved the highest DNA removal (3.9 ± 0.65 ng/mg) but caused ECM disruption, with fragmented collagen fibers seen in SEM. Tensile strength dropped (67.22 ± 12.47 MPa), while hydrophilicity increased, aiding cell adhesion. | [53] | |
| Oreochromis niloticus (Tilapia) | Heads | Treated with 0.1% and 1% SDS at 4 °C for 24 h with constant 300 rpm agitation, then washed. | Achieved moderate DNA removal, but higher residual DNA remained compared to sonication-assisted protocols. Collagen structure showed some disruption, and residual SDS levels were high, potentially affecting biocompatibility. | [7] | |
| Oreochromis niloticus (Tilapia) | Viscera | Treated with 1% SDS with agitation for 5 and 10 min. Then washed thrice with distilled water. | 0.3% SDS achieved higher DNA removal (94.6% efficiency). However, SDS-treated scaffolds exhibited some ECM disruption, as evidenced by SEM imaging, which showed a denser and more disorganized fiber structure. The mechanical properties declined, exhibiting lower tensile strength and Young’s modulus compared to the raw tissue. | [17] | |
| Non-ionic detergent + Agitation | Chanos chanos (Milkfish) | Skin | Treated with 0.1% and 1% TX-100 at 4 °C for 24 h with constant 300 rpm agitation, then washed. | 1% SDS preserved the ECM structure, with higher collagen retention and organized fibers. It maintained high tensile strength (118.14 ± 10.27 MPa), had lower residual detergent, and ensured better biocompatibility, though DNA removal was slightly lower than SDS. | [53] |
| Oreochromis niloticus (Tilapia) | Heads | Treated with 1% TX-100 with agitation for 5 and 10 min. Then washed thrice with distilled water. | 1% TX-100 with agitation preserved ECM integrity and porosity better than SDS, though DNA removal was slightly lower. It also had lower residual detergent, improving biocompatibility. | [7] | |
| Oreochromis niloticus (Tilapia) | Viscera | Treated with 0.1% and 0.3% TX-100 with agitation via orbital shaker and magnetic stirrer | TX-100 with agitation preserved ECM integrity better than SDS. At 0.3% concentration, TX-100 maintained a more porous structure with less ECM damage. While DNA removal was slightly lower than SDS, TX-100-treated scaffolds demonstrated better biocompatibility and higher cell adhesion. | [17] | |
| Non-ionic detergent + Hypertonic solution + Basic solution + Agitation | Oreochromis niloticus (Tilapia) | Skin | Samples were pretreated with 3% NaCl at 8 °C for 6 h, then placed on filter paper pre-soaked in 0.3% acetic acid. They were subsequently incubated in 0.1% TX-100 at 8 °C for 16 h, followed by 0.1 M NaOH treatment at 8 °C for 6 h with continuous shaking at 150 rpm. | Residual DNA content was 1.4 ± 0.7 ng/mg, well below the 50 ng/mg medical industry limit. | [45] |
| Freeze–thaw cycle + Acid-base solutions + Ionic detergent | Oreochromis niloticus (Tilapia) | Scales | Fish scales were freeze-thawed (−80 °C, 4 cycles), then soaked in 0.1% NaOH and 3% H2O2. Then, finally, immersed in 2% SDS. | H&E and Masson’s staining confirmed complete decellularization, and SEM showed an intact collagen network. The scaffold retained high tensile strength (5.89 ± 0.74 MPa) and exhibited slow degradation, supporting MSC viability and proliferation, making it biocompatible for tissue regeneration. | [36] |
| Freeze–thaw cycle + Hypertonic and hypertonic solutions | Ctenopharyngodon idella (Grass carps) | Skin | Samples were frozen (−20 °C, 3 cycles, 30 min each) and thawed at room temperature, then treated with a hypertonic solution for 24 h. Finally, incubated in a hypotonic solution for 24 h. | Has high residual DNA content (>100 ng/mg), indicating incomplete decellularization. SEM showed dense collagen fibers, potentially limiting cell infiltration. While mechanical strength remained high (23.15 MPa, 205.8 MPa Young’s modulus), biocompatibility was lower due to remaining cellular debris. | [35] |
| Freeze–thaw cycle + Hypertonic and hypertonic solutions + Non-ionic detergent | Ctenopharyngodon idella (Grass carps) | Skin | Samples were frozen (−20 °C, 3 cycles, 30 min each) and thawed at room temperature, followed by a hypertonic solution for 24 h. Then, treated with a variation in the concentration of TX-100. Finally, incubated in a hypotonic solution for 24 h. | Improved DNA removal (<50 ng/mg in Protocol 4) and collagen preservation, enhancing porosity and cell attachment. Protocol 4 (0.5% TX-100) achieved the best ECM integrity, tensile strength (21.25 MPa), and Young’s modulus (192.15 MPa). High cell adhesion confirmed superior biocompatibility. | [53] |
| Freeze–thaw cycle + Ionic detergent + Agitation | Rutilus frisii | Swim bladder | The fish swim bladder (FSB) was snap freeze-dried by soaking in liquid nitrogen (3 min) followed by rapid thawing in distilled water (5 min). It was then washed in PBS (37 °C) and treated with 0.5% and 1% SDS for 24 h in a shaker incubator. | Histological staining showed the absence of nuclei. SEM imaging revealed a porous structure with collagen fibers intact. The scaffold exhibited high collagen content and a pore size of ~0.5–2 µm, suitable for cell migration. Cytotoxicity tests showed a cell viability of 84–142% after 24 h. | [34] |
| Sonication + Ionic detergent | Oreochromis niloticus (Tilapia) | Heads | Sonication-assisted (40 kHz, 5–10 min) with 1% SDS. Then washed thrice with distilled water. | 1% SDS treatment was the most effective in DNA removal (93.7%), with the lowest residual DNA (7.67 ng/mg). However, SEM analysis showed greater ECM disruption, and SDS retention remained high, which may impact cytocompatibility. | [7] |
| Oreochromis niloticus (Tilapia) | Viscera | Sonication-assisted (40 kHz, 10 h) with 0.1% and 0.3% SDS at room temperature. Then washed 3 times. | Sonication enhanced DNA removal, with 0.3% SDS achieving 96.5% efficiency. It accelerated decellularization, but SEM revealed greater ECM disruption, showing fragmented collagen fibers and rougher surfaces. | [17] | |
| Sonication + Non-ionic detergent | Oreochromis niloticus (Tilapia) | Heads | Sonication-assisted (40 kHz, 5–10 min) with 1% TX-100. Then washed thrice with distilled water. | 1% TX-100 also achieved high DNA removal, but better collagen preservation and porosity retention than SDS-treated samples. Additionally, residual TX-100 was significantly reduced (99% lower for 5 min treatment), enhancing biocompatibility. | [7] |
| Oreochromis niloticus (Tilapia) | Viscera | Sonication-assisted (40 kHz, 10 h) with 0.1% and 0.3% TX-100 at room temperature. Then washed 3 times. | Sonication with 0.3% TX-100-preserved ECM integrity while ensuring high DNA removal. Collagen remained more intact than in SDS-treated samples, with better cell viability and attachment, offering a balance between decellularization and ECM preservation. | [17] |
| Physical-Chemical Treatment | Fish Species | Fish Tissue/Organ | Decellularization Protocol | Results | References |
|---|---|---|---|---|---|
| Ionic detergent + Enzyme + Chelating agent | Sturgeon fish | Cartilage | Cartilage samples were treated with 1% SDS in PBS (4 °C, 24 h) with solution changes every 8 h, followed by 0.1% EDTA in PBS (24 h). They were then digested in 1 U/mL DNase I (24 h) and washed in PBS. | The H&E staining showed no visible nuclear remnants, indicating effective decellularization. | [58] |
| Non-Ionic detergent + Enzyme + Hypotonic solution + Chelating agent | Ctenopharyngodon idella (Grass carps) | Scales | The samples were incubated in 10 mM Tris-HCl and 0.1% EDTA at 4 °C for 24 h, followed by 0.1% TX-100 treatment at 4 °C. They were then digested with a nuclease solution containing 500 U/mL DNase I and 1 mg/mL RNase A at 37 °C for 24 h. | TX-100 treatment for 3 days could not completely remove the cellular components of the scaffolds, while extending the treatment time to 4 and 5 days completely removed them. Treatment for 3 days and 4 days has better effects in retaining ECM components while preserving the intact surface microstructure. The degradation rate of 3- and 4-day treatments was also lower and had no significant difference between them. Their Young’s modulus decreased slightly to 6.26 ± 1.49 and 5.80 ± 1.17 GPa. In general, the 4-day treatment proved to be the best protocol. | [55] |
| Ionic detergent + Non-Ionic detergent + Enzyme + Hypotonic solution | Oreochromis niloticus (Tilapia) | Skin | Samples were incubated in PBS with 0.02% sodium azide and 0.5% TX-100 (RT, 2 h), washed with HBSS (RT, 10 min), treated with 0.5% SDS (RT, 1 h), and digested in trypsin (0.05 g/mL) with 1 M Tris-HCl. | H&E staining confirmed that the decellularization process effectively removed cells, increased scaffold porosity, and preserved the ECM’s original structure. Mechanical testing showed that the AFS scaffold exhibited high tensile strength and flexibility. Degradation studies revealed that approximately 70% of the matrix degraded within 28 days, exhibiting behavior similar to collagen-based scaffolds. | [70] |
| Physical-Chemical Treatment | Fish Species | Fish Tissue/Organ | Decellularization Protocol | Results | References |
|---|---|---|---|---|---|
| Freeze–thaw cycle + Non-ionic detergent + Hypertonic and hypotonic solutions + Chelating agent + Enzyme | Ctenopharyngodon idella (Grass carps) | Skin | Samples were frozen (−20 °C, 3 cycles, 30 min each) and thawed at room temperature, then treated with a hypertonic solution for 24 h. Followed by a variation in concentration of Trypsin-EDTA for 90 min. Then, TX-100 treatment was performed at varying concentrations. Finally, incubated in a hypotonic solution for 24 h. | Trypsin-EDTA (0.25%) enhanced decellularization (DNA < 20 ng/mg in Protocol 6) but caused greater ECM disruption, reducing collagen content, mechanical strength, and cell adhesion. Weaker scaffolds degraded faster in PBS, compromising stability despite effective cell removal. | [35] |
| Freeze–thaw cycle + Non-ionic detergent + Enzymes in Hypotonic solution + Agitation | Mylopharyngodon piceus (Black carps) | Skin | Samples were frozen at −40 °C, thawed in tap water, and stirred in 1% TX-100 (12 h) and 0.5 μg/mL trypsin (18 h, pH 8.0 Tris–HCl buffer) for effective decellularization while preserving the extracellular matrix. | H&E and SEM confirmed a cell-free, porous AFS with preserved 3D structure and well-arranged collagen fibers. Cytotoxicity tests showed no toxicity to L929 cells. AFS exhibited super hydrophilicity, a swelling ratio that stabilized by day 5, and slow degradation over 13 weeks with high wet-condition resistance (0.07 ± 0.04%). | [33] |
| Freeze–thaw cycle + Ionic detergents + Enzyme + Agitation | Hypopththalmichthys molitrix (Chubs) | Swim bladder | The SBs underwent three freeze–thaw cycles (−80 °C, 1 h; 36 °C thaw), followed by detergent treatment (0.5% sodium deoxycholate, 0.05% sodium orthovanadate in PBS, RT, 12 h, 110 r/min) and DNase-I treatment (20 U/mL, 37 °C, 2 h, shaking). | The freeze–thaw and DNase-I treatment (Group E) was the most effective for decellularization, yielding low residual DNA. The decellularized ASBs had a loosened fiber layer and 3D porous structure, promoting cell adhesion and migration. Additionally, the hemolysis rate (2.8 ± 0.15%) was well below the 5% threshold, confirming good biocompatibility for medical applications. | [39] |
| Freeze–thaw cycles + Ionic detergent + Enzyme | Astroconger Myriaster | Skin | The samples underwent three freeze–thaw cycles (−80 °C), followed by 2.0% deoxycholic acid treatment (12 h) for decellularization. They were then rinsed and treated with 0.5% SDS (1 h) and subjected to enzymatic digestion with 0.2 μg/L trypsin (4 h). | Cellular components were successfully removed as confirmed by H&E, Masson, and PAS staining, leaving a collagen-rich matrix with no PAS-positive substances. DNA content was reduced to 18.64 ± 2.51 ng/mg, significantly lower than natural fish skin (234.17 ± 13.24 ng/mg) and commercial oral membranes (32.41 ± 4.87 ng/mg), indicating effective decellularization. | [59] |
| Non-ionic detergent + Hypotonic solution + Enzyme + Agitation | Crisp flesh grass carp | Scales | Fish scales were stirred in Tris-buffer with PMSF (4 °C, 36 h), treated with 1% TX-100 (4 °C, 36 h), then digested with DNase/RNase (37 °C, 2 h) and extracted in TX-100 (4 °C, 24 h). | Cells were effectively removed (DNA: 23.5 ± 1.8 ng/mg) while preserving ECM integrity. H&E and DAPI staining confirmed complete cell removal, and SEM showed a porous collagen network. The scaffold retained high tensile strength (92.7 ± 6.3 MPa) and Young’s modulus (2.5 ± 0.4 GPa). Degradation studies showed 54.2% weight retention (28 days, PBS), and cell viability (>90%) confirmed high biocompatibility. | [52] |
| Ionic detergent + Enzymes + Agitation | Oreochromis niloticus (Tilapia) | Skin | The skin samples were shaken in 2.5 U/mL dispase (3 h), followed by 1% SDS treatment (6 h) and gentle scraping. They were then shaken in 25 U/mL nuclease (3 h), treated with 1% SDS (1 h), and finally freeze-dried. | Decellularization removed pigments, preserved 69.3% collagen, and loosened fibers while maintaining structure. SEM showed increased porosity, strong cell attachment, and minimal surface changes. Tensile strength decreased, but thermal stability remained high (>60 °C). Degradation: 81.0% retained in PBS (8 weeks), 13.7% in collagenase (72 h). | [66] |
| Fish-Tissue Type | Best-Performing Protocol Families | Key Parameters | Typical Pitfalls | Recommended Characterization Endpoints |
|---|---|---|---|---|
| Skin | Basic-Hypertonic Sequence or Mild Chemical | 0.1 M NaOH (8 h)/1 M NaCl (12 h); or 0.5% TX-100 | High lipid content in skin can hinder the penetration of aqueous reagents; SDS risks fiber orientation disruption. | H&E for nuclear loss, ATR-FTIR for triple-helix integrity |
| Scales | Hybrid: Non-ionic detergent + Chelating agent + Nuclease | 0.1% TX-100 + 10 mM EDTA; 4-day incubation at 4 °C | SDS treatment (0.1%) causes ~55% collagen loss and destroys surface microstructure. | DNA quantification (<50 ng/mg), SEM for porosity (70–78%) |
| Viscera/Soft organs | Physical-assisted Chemical: Sonication + Detergent | 40 kHz sonication (10 min); 0.3% SDS | Higher protein loss is observed in sonicated samples; requires strict control of burst times to prevent thermal denaturation. | DNA removal efficiency (>95%), DSC for thermal stability (>60 °C) |
| Bones | Acid-Chelating: Acidic demineralization + EDTA | 5% Nitric acid (10 h); 10% EDTA (3 days, 4 °C) | Acidic hydrolysis can lead to mechanical weakening if exposure is too long. | Residual inorganic content wt.%, SEM for cell-alignment channels |
| Swim Bladder | Mild Chemical + Crosslinking | Low-concentration TX-100; Post-process with GA or EDC/NHS | High mechanical fragility; susceptible to tearing during agitation or rinsing. | Degradation rate and porosity analysis |
| Biomedical Application | Biological Property Assessed | Observations Reported | Fish Source and Tissue | Observed Biological Advantages | References |
|---|---|---|---|---|---|
| Wound healing/Skin regeneration | Cell adhesion and proliferation | Enhanced fibroblast and keratinocyte adhesion and proliferation reported when collagen fibrillar structure and GAG content are preserved | Tilapia, cod, salmon skin | Strongly dependent on detergent type, exposure time, and tissue thickness | [13,16,73,76,77] |
| Angiogenic response | Increased neovascularization observed in select in vivo wound models; magnitude varied across studies | Fish skin ECM | Sensitive to GAG retention and lipid preservation during decellularization | [13,16,33,75,78,79] | |
| Inflammatory response | Reduced inflammatory cell infiltration reported under minimally processed conditions; variable outcomes reported following aggressive chemical treatments | Fish skin grafts | Influenced by residual detergent content and matrix disruption | [13,16,75,80,81] | |
| Bone regeneration | Osteogenic differentiation | Upregulation of osteogenic markers and mineral deposition observed in vitro | Fish scale- and bone-derived dECM | Sensitive to mineral preservation and collagen integrity | [75,82,83,84] |
| Structural integrity under load | Limited mechanical stability reported without secondary reinforcement | Fish-derived demineralized bone matrix (DBM) | Strongly affected by decalcification and processing severity | [13,52,85,86,87,88,89,90,91,92] | |
| Cartilage repair | Chondrogenic response | Chondrocyte attachment and differentiation supported; suppression of hypertrophic markers reported in select studies | Sturgeon cartilage dECM | Sensitive to GAG depletion during decellularization | [40,75,93,94,95] |
| Compressive performance | Reduced compressive properties reported following loss of sulfated GAGs | Fish cartilage dECM | Strong dependence on decellularization chemistry | [58,93] |
| Strategy | Specific Method/Material | Targeted Limitation | Reported Qualitative Effect | Trade-Offs/Limitations | References |
|---|---|---|---|---|---|
| Crosslinking | EDC/NHS | Mechanical weakness | ↑ tensile strength; ↓ degradation rate | Reduced porosity at high density | [89,117,118,119,120,121,122] |
| DHT | Structural instability | ↑ thermal stability | Limited tunability | [89,120,123,124] | |
| Polymer composite | dECM–Alg/MA | Poor printability | ↑ shape fidelity; ↑ porosity | Polymer dilution of ECM cues | [125,126,127,128,129] |
| dECM–GelMA | Weak mechanics | ↑ modulus; ↑ cell viability | UV exposure concerns | [126,130,131,132] | |
| Nanocomposite | Collagen nanofibers | Low strength | ↑ mechanical reinforcement | Scale-up challenges | [133,134] |
| 3D Printing (dECM bio-inks) | Low viscosity; Poor print fidelity | ↑ shape fidelity; ↑ precise microarchitecture control; ↑ preserved bioactivity | Complexity of ink formulation and dependency on crosslinking/additives | [32,105,125,132,135,136] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Amarillas, J.L.; Dingcong, R., Jr.; Sabugaa, L.G.; Domingo, M.I.K.; Samulde, C.A.; Pingoy, G.I.; Ananoria, A.; Malaluan, R.; Bual, R.; Dumancas, G.; et al. Advances in Decellularization of Fish Wastes for Extracellular Matrix Extraction in Sustainable Tissue Engineering and Regenerative Medicine. Bioengineering 2026, 13, 255. https://doi.org/10.3390/bioengineering13020255
Amarillas JL, Dingcong R Jr., Sabugaa LG, Domingo MIK, Samulde CA, Pingoy GI, Ananoria A, Malaluan R, Bual R, Dumancas G, et al. Advances in Decellularization of Fish Wastes for Extracellular Matrix Extraction in Sustainable Tissue Engineering and Regenerative Medicine. Bioengineering. 2026; 13(2):255. https://doi.org/10.3390/bioengineering13020255
Chicago/Turabian StyleAmarillas, Jady Lee, Roger Dingcong, Jr., Lornie Grace Sabugaa, Maree Ivonne Kyla Domingo, Carl Angelo Samulde, Gerard Ian Pingoy, Abhel Ananoria, Roberto Malaluan, Ronald Bual, Gerard Dumancas, and et al. 2026. "Advances in Decellularization of Fish Wastes for Extracellular Matrix Extraction in Sustainable Tissue Engineering and Regenerative Medicine" Bioengineering 13, no. 2: 255. https://doi.org/10.3390/bioengineering13020255
APA StyleAmarillas, J. L., Dingcong, R., Jr., Sabugaa, L. G., Domingo, M. I. K., Samulde, C. A., Pingoy, G. I., Ananoria, A., Malaluan, R., Bual, R., Dumancas, G., & Lubguban, A. (2026). Advances in Decellularization of Fish Wastes for Extracellular Matrix Extraction in Sustainable Tissue Engineering and Regenerative Medicine. Bioengineering, 13(2), 255. https://doi.org/10.3390/bioengineering13020255










