Muscle Fatigue in Dynamic Movement: Limitations and Challenges, Experimental Design, and New Research Horizons
Abstract
1. Introduction
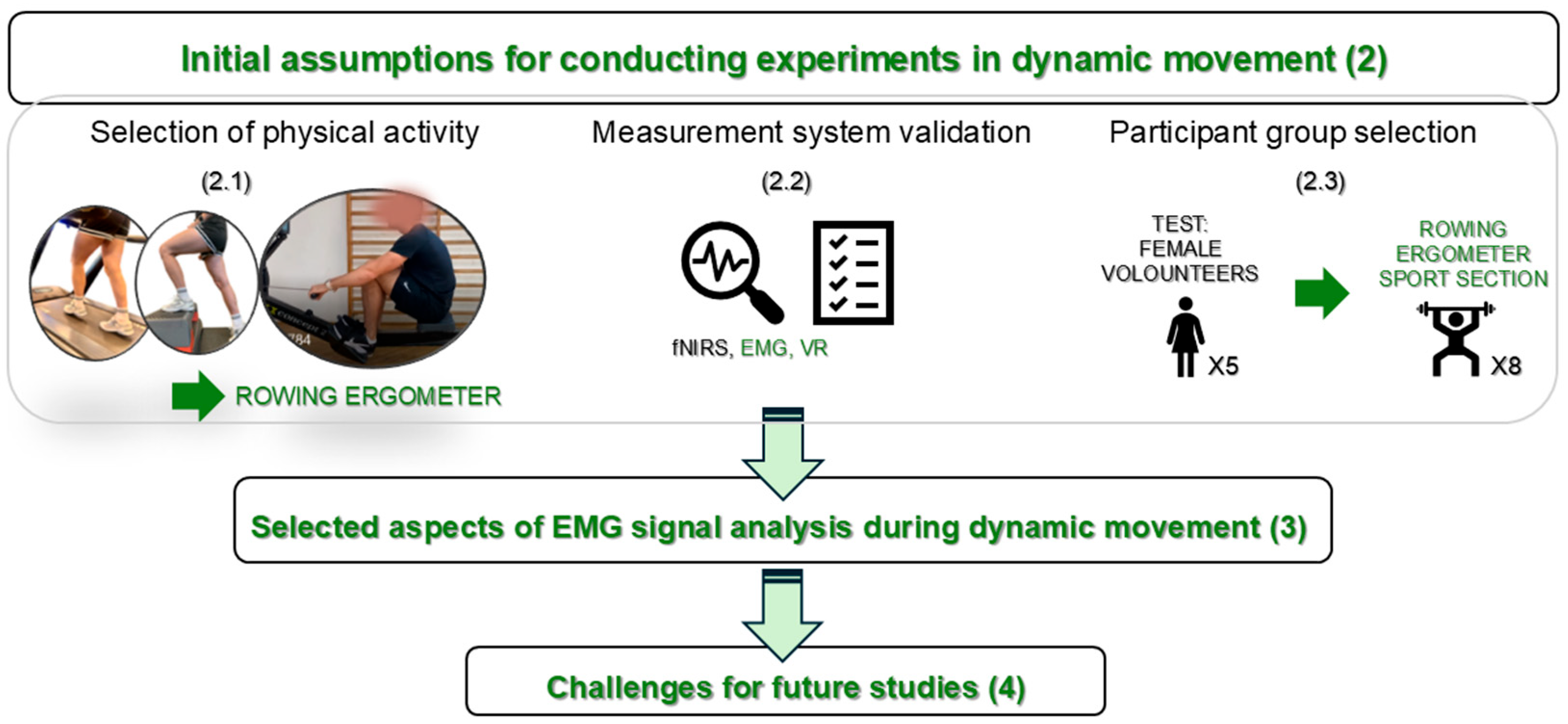
2. Initial Assumptions for Conducting Experiments in Dynamic Movement
2.1. Selection of Physical Activity
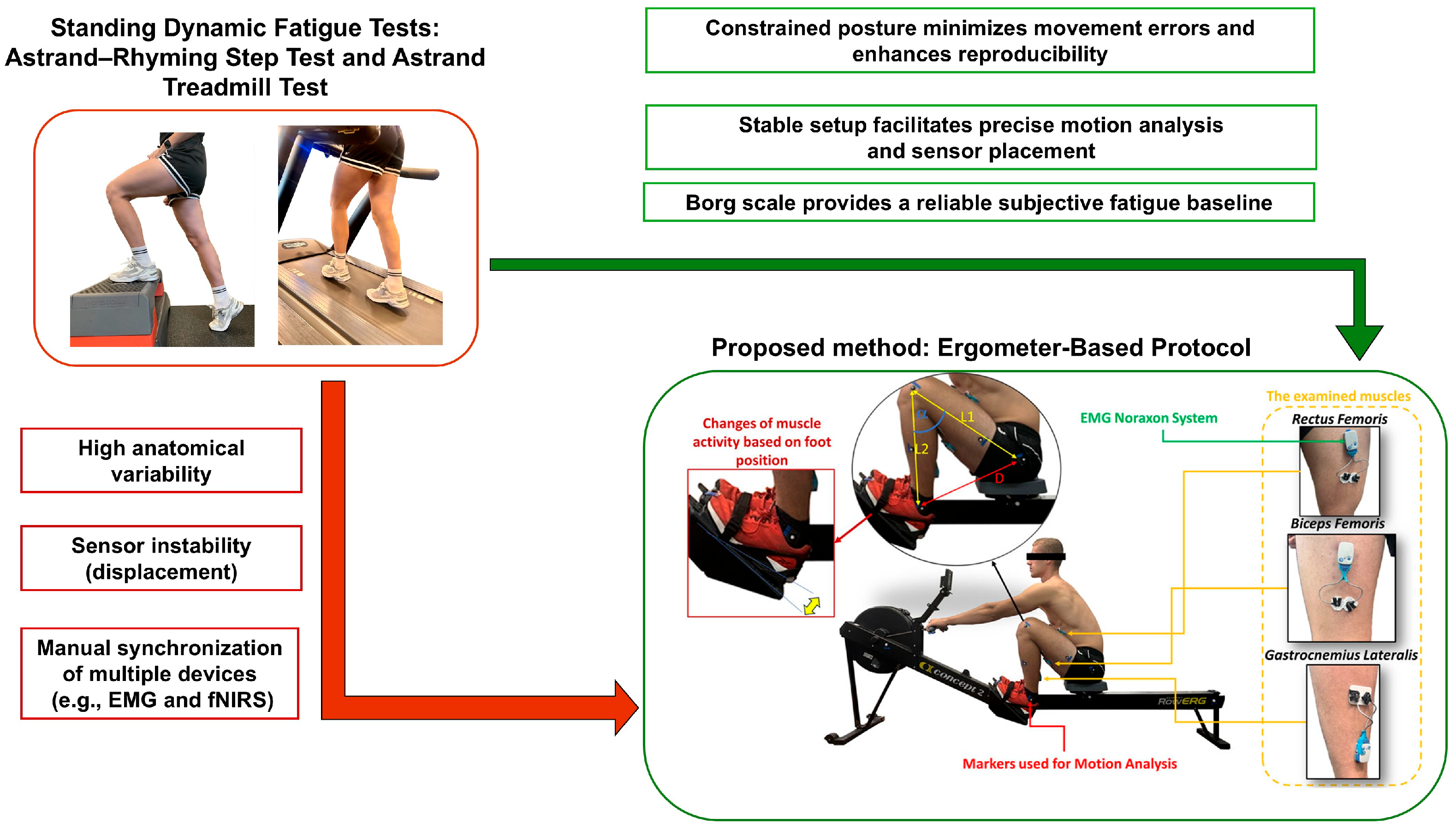
2.2. Measurement System Validation
- Due to differences in running technique and significant changes associated with leg movement, electrodes are often moved and detached.
- Despite adhering to the literature guidelines, the problem remains unresolved.
- Fatigue tests, although defined by detailed experimental conditions, introduce many artifacts in the readings of both sensors.
- The color of the skin, its thickness and the individual anatomical differences cause disturbances in the fNIRS sensors.
- Synchronizing two independent sensors requires additional time, which affects the quality of research and the well-being of participants.
- The Borg scale provides a good starting point to determine the level of fatigue.
2.3. Participant Group Selection
2.4. Summary
3. Selected Aspects of EMG Signal Analysis During Dynamic Movement
3.1. Standard Spectral Analysis Under Static Conditions
3.2. Time–Frequency Analysis for Dynamic Movements
4. Challenges for Future Studies on Fatigue During Dynamic Movement
4.1. Integrated Research Framework and Preprocessing
4.2. Wavelet-Based Feature Extraction Strategies
4.3. Optimization of the WT-AI Synergy
4.4. Current State-of-the-Art: A Comparative Review
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| sEMG | Surface Electromyography |
| WT | Wavelet Transform |
| fNIRS | Functional Near-Infrared Spectroscopy |
| EEG | Electroencephalography |
| AI | Artificial Intelligence |
| EMG | Electromyography |
| MDF | Median Frequency |
| MNF | Mean Frequency |
| EMD | Empirical Mode Decomposition |
| HHT | Hilbert–Huang Transform |
| FFT | Fast Fourier Transform |
| VR | Virtual Reality |
| DWT | Discrete Wavelet Transform |
| CWT | Continuous Wavelet Transform |
| STFT | Short-Time Fourier Transform |
| db4 | Daubechies 4 |
| ML | Machine Learning |
| DL | Deep Learning |
| SVM | Support Vector Machine |
| CNN | Convolutional Neural Network |
| PCA | Principal Component Analysis |
| ANN | Artificial Neural Network |
| DNN | Deep Neural Network |
| MLP | Multilayer Perceptron |
| GRNN | General Regression Neural Network |
| GA-SVM | Genetic Algorithm-based SVM |
| FFBPNN | Feed-Forward Back-Propagation Neural Network |
| ANFIS | Adaptive Neuro-Fuzzy Inference System |
| KNN | K-Nearest Neighbors |
| XMANet | Cross-layer Mutual Attention Learning Network |
References
- Viitasalo, J.H.T.; Komi, P.V. Signal Characteristics of EMG during Fatigue. Eur. J. Appl. Physiol. Occup. Physiol. 1977, 37, 111–121. [Google Scholar] [CrossRef]
- Potvin, J.R.; Bent, L.R. A Validation of Techniques Using Surface EMG Signals from Dynamic Contractions to Quantify Muscle Fatigue during Repetitive Tasks. J. Electromyogr. Kinesiol. 1997, 7, 131–139. [Google Scholar] [CrossRef]
- Ball, N.; Scurr, J. Electromyography Normalization Methods for High-Velocity Muscle Actions: Review and Recommendations. J. Appl. Biomech. 2013, 29, 600–608. [Google Scholar] [CrossRef]
- Cifrek, M.; Medved, V.; Tonković, S.; Ostojić, S. Surface EMG Based Muscle Fatigue Evaluation in Biomechanics. Clin. Biomech. 2009, 24, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Tankisi, H.; Burke, D.; Cui, L.; de Carvalho, M.; Kuwabara, S.; Nandedkar, S.D.; Rutkove, S.; Stålberg, E.; van Putten, M.J.A.M.; Fuglsang-Frederiksen, A. Standards of Instrumentation of EMG. Clin. Neurophysiol. 2020, 131, 243–258. [Google Scholar] [CrossRef]
- Turker, K.S. Electromyography: Some Methodological Problems and Issues. Phys. Ther. 1993, 73, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Liu, G.; Sun, Y.; Lin, K.; Zhou, Z.; Cai, J. Application of Surface Electromyography in Exercise Fatigue: A Review. Front. Syst. Neurosci. 2022, 16, 893275. [Google Scholar] [CrossRef]
- Chuang, T.D.; Acker, S.M. Comparing Functional Dynamic Normalization Methods to Maximal Voluntary Isometric Contractions for Lower Limb EMG from Walking, Cycling and Running. J. Electromyogr. Kinesiol. 2019, 44, 86–93. [Google Scholar] [CrossRef]
- Ball, N.; Scurr, J. An Assessment of the Reliability and Standardisation of Tests Used to Elicit Reference Muscular Actions for Electromyographical Normalisation. J. Electromyogr. Kinesiol. 2010, 20, 81–88. [Google Scholar] [CrossRef]
- Sae-lim, W.; Phukpattaranont, P.; Thongpull, K. Effect of Electrode Skin Impedance on Electromyography Signal Quality. In Proceedings of the 2018 15th International Conference on Electrical Engineering/Electronics, Computer, Telecommunications and Information Technology (ECTI-CON); IEEE: Chiang Rai, Thailand, 2018; pp. 748–751. [Google Scholar] [CrossRef]
- Hung, G.; Pallis, J. Biomedical Engineering Principles in Sports; Springer: Berlin/Heidelberg, Germany, 2004; Volume 1, ISBN 978-1-4613-4706-4. [Google Scholar]
- Williams, N. The Borg Rating of Perceived Exertion (RPE) Scale. Occup. Med. 2017, 67, 404–405. [Google Scholar] [CrossRef]
- Halaki, M.; Ginn, K. Normalization of EMG Signals: To Normalize or Not to Normalize and What to Normalize To? In Computational Intelligence in Electromyography Analysis; Naik, G.R., Ed.; IntechOpen: Rijeka, Croatia, 2012. [Google Scholar]
- Gates, D.H.; Dingwell, J.B. The Effects of Muscle Fatigue and Movement Height on Movement Stability and Variability. Exp. Brain Res. 2011, 209, 525–536. [Google Scholar] [CrossRef]
- Chowdhury, S.K.; Nimbarte, A.D. Comparison of Fourier and Wavelet Analysis for Fatigue Assessment during Repetitive Dynamic Exertion. J. Electromyogr. Kinesiol. 2015, 25, 205–213. [Google Scholar] [CrossRef]
- Jero, S.E.; Ramakrishnan, S. Analysis of Muscle Fatigue Conditions in Surface EMG Signal with A Novel Hilbert Marginal Spectrum Entropy Method. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC); IEEE: Piscataway, NJ, USA, 2019; Volume 2019, pp. 2675–2678. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, B.; Wang, H.; Gouda, M.A. Surface Electromyography Monitoring of Muscle Changes in Male Basketball Players During Isotonic Training. Sensors 2025, 25, 1355. [Google Scholar] [CrossRef]
- Fougner, A.; Scheme, E.; Chan, A.D.C.; Englehart, K.; Stavdahl, Ø. Resolving the Limb Position Effect in Myoelectric Pattern Recognition. IEEE Trans. Neural Syst. Rehabil. Eng. 2011, 19, 644–651. [Google Scholar] [CrossRef]
- Shakya, S.; Ranjitkar, B. Forearm Bio-Medical Signal Processing. Int. J. Eng. Technol. 2024, 2, 49–59. [Google Scholar] [CrossRef]
- Song, J.; Choi, Y.S.; Lee, S.; Park, D.; Park, J. Changes in Muscle Oxygenation and Activity during Cumulative Isometric Muscle Contraction: New Insight into Muscle Fatigue. Front. Physiol. 2025, 16, 1559893. [Google Scholar] [CrossRef]
- Jayarathne, M.; Wickramanayake, D.; Afsheenjinan, A.; Ranaweera, R.; Weerasingha, V. EMG Based Biofeedback System Using a Virtual Reality Method. In Proceedings of the 2015 IEEE 10th International Conference on Industrial and Information Systems (ICIIS); IEEE: Peradeniya, Sri Lanka, 2015; pp. 111–116. [Google Scholar] [CrossRef]
- Subasi, A.; Kiymik, M.K. Muscle Fatigue Detection in EMG Using Time-Frequency Methods, ICA and Neural Networks. J. Med. Syst. 2010, 34, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhang, C.; Liu, G.; Cui, W.; Sun, Y.; Zhang, C. Detecting Muscle Fatigue during Lower Limb Isometric Contractions Tasks: A Machine Learning Approach. Front. Physiol. 2025, 16, 1547257. [Google Scholar] [CrossRef] [PubMed]
- Reaz, M.B.I.; Hussain, M.S.; Mohd-Yasin, F. Techniques of EMG Signal Analysis: Detection, Processing, Classification and Applications. Biol. Proced. Online 2006, 8, 11–35. [Google Scholar] [CrossRef]
- Daniel, N.; Sybilski, K.; Kaczmarek, W.; Siemiaszko, D.; Małachowski, J. Relationship between EMG and FNIRS during Dynamic Movements. Sensors 2023, 23, 5004. [Google Scholar] [CrossRef]
- Daniel, N.; Małachowski, J. Wavelet Analysis of the EMG Signal to Assess Muscle Fatigue in the Lower Extremities during Symmetric Movement on a Rowing Ergometer. Acta Bioeng. Biomech. 2023, 25, 15–27. [Google Scholar] [CrossRef]
- Daniel, N.; Małachowski, J.; Sybilski, K.; Siemiaszko, D. Quantitative Assessment of Muscle Fatigue during Rowing Ergometer Exercise Using Wavelet Analysis of Surface Electromyography (SEMG). Front. Bioeng. Biotechnol. 2024, 12, 1344239. [Google Scholar] [CrossRef]
- Daniel, N.; Małachowski, J.; Sybilski, K.; Siemiaszko, D.; Klicki, K. Research on the Influence of Virtual Reality on Muscle Fatigue during Rowing Ergometer Exercise—Pilot Study. PLoS ONE 2026, 21, e0342166. [Google Scholar] [CrossRef] [PubMed]
- Di Giminiani, R.; Cardinale, M.; Ferrari, M.; Quaresima, V. Validation of Fabric-Based Thigh-Wearable EMG Sensors and Oximetry for Monitoring Quadricep Activity during Strength and Endurance Exercises. Sensors 2020, 20, 4664. [Google Scholar] [CrossRef]
- González-Izal, M.; Malanda, A.; Gorostiaga, E.; Izquierdo, M. Electromyographic Models to Assess Muscle Fatigue. J. Electromyogr. Kinesiol. 2012, 22, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Phinyomark, A.; Thongpanja, S.; Hu, H.; Phukpattaranont, P.; Limsakul, C. The Usefulness of Mean and Median Frequencies in Electromyography Analysis. In Computational Intelligence in Electromyography Analysis—A Perspective on Current Applications and Future Challenges; IntechOpen: London, UK, 2012. [Google Scholar] [CrossRef]
- Kim, C.-B.; Park, C.-H.; Kim, C.-H.; Lee, H.-S.; Kim, M.-O. Changes in Surface Electromyography Signal According to Severity in Patients with Carpal Tunnel Syndrome. J. Electrodiagn. Neuromuscul. Dis. 2020, 22, 15–22. [Google Scholar] [CrossRef]
- Chowdhury, R.; Reaz, M.B.I.I.; Islam, M.T. Wavelet Transform to Recognize Muscle Fatigue. In Proceedings of the 2012 Third Asian Himalayas International Conference on Internet; IEEE: Kathmundu, Nepal, 2012; pp. 1–5. [Google Scholar] [CrossRef]
- Dantas, J.L.; Camata, T.V.; Brunetto, M.A.O.C.; Moraes, A.C.; Abrão, T.; Altimari, L.R. Fourier and Wavelet Spectral Analysis of EMG Signals in Isometric and Dynamic Maximal Effort Exercise. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, EMBC’10; IEEE: Buenos Aires, Argentina, 2010; pp. 5979–5982. [Google Scholar] [CrossRef]
- Yousif, H.A.; Zakaria, A.; Rahim, N.A.; Salleh, A.F.B.; Mahmood, M.; Alfarhan, K.A.; Kamarudin, L.M.; Mamduh, S.M.; Hasan, A.M.; Hussain, M.K. Assessment of Muscles Fatigue Based on Surface EMG Signals Using Machine Learning and Statistical Approaches: A Review. In IOP Conference Series: Materials Science and Engineering; IOP Publishing Ltd.: Bristol, UK, 2019; Volume 705. [Google Scholar]
- Chang, K.M.; Liu, S.H.; Wu, X.H. A Wireless SEMG Recording System and Its Application to Muscle Fatigue Detection. Sensors 2012, 12, 489–499. [Google Scholar] [CrossRef]
- Kuthe, C.D.; Uddanwadiker, R.V.; Ramteke, A.A. Surface Electromyography Based Method for Computing Muscle Strength and Fatigue of Biceps Brachii Muscle and Its Clinical Implementation. Inform. Med. Unlocked 2018, 12, 34–43. [Google Scholar] [CrossRef]
- Siecinski, S.; Kostka, P.S.; Tkacz, E.J. Assessment of Muscle Fatigue During The Walk on a Treadmill in Healthy Subjects Based on Median Frequency of Surface Electromyogram. In Proceedings of the 2022 IEEE 21st Mediterranean Electrotechnical Conference (MELECON); IEEE: Palermo, Italy, 2022; pp. 436–441. [Google Scholar] [CrossRef]
- Macisaac, D.; Parker, P.A.; Scott, R.N. The Short-Time Fourier Transform and Muscle Fatigue Assessment in Dynamic Contractions. J. Electromyogr. Kinesiol. 2001, 11, 439–449. [Google Scholar] [CrossRef]
- Sparto, P.J.; Parnianpour, M.; Barria, E.A.; Jagadeesh, J.M. Wavelet and Short-Time Fourier Transform Analysis of Electromyography for Detection of Back Muscle Fatigue. IEEE Trans. Rehabil. Eng. 2000, 8, 433–436. [Google Scholar] [CrossRef]
- Boyer, M.; Bouyer, L.; Roy, J.S.; Campeau-Lecours, A. A Real-Time Algorithm to Estimate Shoulder Muscle Fatigue Based on Surface EMG Signal for Static and Dynamic Upper Limb Tasks. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, EMBS; IEEE: Mexico City, Mexico, 2021; pp. 100–106. [Google Scholar] [CrossRef]
- Thongpanja, S.; Phinyomark, A.; Phukpattaranont, P.; Limsakul, C. Mean and Median Frequency of EMG Signal to Determine Muscle Force Based on Time Dependent Power Spectrum. Elektron. Elektrotechnika 2013, 19, 51–56. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Y.; Han, R.P.S. Wavelet Transform Theory and Its Application in EMG Signal Processing. In Proceedings of the 2010 Seventh International Conference on Fuzzy Systems and Knowledge Discovery; IEEE: Yantai, China, 2010; Volume 5, pp. 2234–2238. [Google Scholar] [CrossRef]
- Chowdhury, S.K.; Nimbarte, A.D.; Jaridi, M.; Creese, R.C. Discrete Wavelet Transform Analysis of Surface Electromyography for the Fatigue Assessment of Neck and Shoulder Muscles. J. Electromyogr. Kinesiol. 2013, 23, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Vannozzi, G.; Conforto, S.; D’Alessio, T. Automatic Detection of Surface EMG Activation Timing Using a Wavelet Transform Based Method. J. Electromyogr. Kinesiol. 2010, 20, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, S.; Gerdle, B. Mean Frequency and Signal Amplitude of the Surface EMG of the Quadriceps Muscles Increase with Increasing Torque—A Study Using the Continuous Wavelet Transform. J. Electromyogr. Kinesiol. 2001, 11, 131–140. [Google Scholar] [CrossRef]
- Canal, M.R. Comparison of Wavelet and Short Time Fourier Transform Methods in the Analysis of EMG Signals. J. Med. Syst. 2010, 34, 91–94. [Google Scholar] [CrossRef]
- Reaz, M.B.I.; Hussain, M.; Mohd-Yasin, F. EMG Analysis Using Wavelet Functions to Determine Muscle Contraction. In Proceedings of the 8th International Conference on e-Health Networking, Applications and Services; IEEE: New Delhi, India, 2006; pp. 132–134. [Google Scholar] [CrossRef]
- Wang, J.; Sun, S.; Sun, Y. A Muscle Fatigue Classification Model Based on LSTM and Improved Wavelet Packet Threshold. Sensors 2021, 21, 6369. [Google Scholar] [CrossRef]
- Li, Z.; Wang, X.; Li, Q.; Wang, F.; Tao, X. Muscle Fatigue Identification and Prediction in Motion Using Wearable Device with Power and Torque-Based Features. Wearable Electron. 2025, 2, 62–68. [Google Scholar] [CrossRef]
- Murillo-Escobar, J.; Jaramillo-Munera, Y.E.; Orrego-Metaute, D.A.; Delgado-Trejos, E.; Cuesta-Frau, D. Muscle Fatigue Analysis during Dynamic Contractions Based on Biomechanical Features and Permutation Entropy. Math. Biosci. Eng. 2020, 17, 2592–2615. [Google Scholar] [CrossRef]
- Al-Mulla, M.R.; Sepulveda, F.; Colley, M. A Review of Non-Invasive Techniques to Detect and Predict Localised Muscle Fatigue. Sensors 2011, 11, 3545–3594. [Google Scholar] [CrossRef]
- Ghofrani Jahromi, M.; Parsaei, H.; Zamani, A.; Dehbozorgi, M. Comparative Analysis of Wavelet-Based Feature Extraction for Intramuscular EMG Signal Decomposition. J. Biomed. Phys. Eng. 2017, 7, 365–378. [Google Scholar] [PubMed]
- Achmamad, A.; Jbari, A. A Comparative Study of Wavelet Families for Electromyography Signal Classification Based on Discrete Wavelet Transform. Bull. Electr. Eng. Inform. 2020, 9, 1420–1429. [Google Scholar] [CrossRef]
- Berger, P.D.A.; Nascimento, F.A.D.O.; Do Carmo, J.C.; Da Rocha, A.F. Compression of EMG Signals with Wavelet Transform and Artificial Neural Networks. Physiol. Meas. 2006, 27, 457–465. [Google Scholar] [CrossRef]
- Ozsert, M.; Yavuz, O.; Durak-Ata, L. Analysis and Classification of Compressed EMG Signals by Wavelet Transform via Alternative Neural Networks Algorithms. Comput. Methods Biomech. Biomed. Eng. 2011, 14, 521–525. [Google Scholar] [CrossRef]
- Shilaskar, S.; Bhatlawande, S.; Chavare, R.; Ingale, A.; Joshi, R.; Vaishale, A. Human Hand Movement Classification Based on EMG Signal Using Different Feature Extractor. Biomed. Pharmacol. J. 2024, 17, 71–82. [Google Scholar] [CrossRef]
- Rong, Y.; Hao, D.; Han, X.; Zhang, Y.; Zhang, J.; Zeng, Y. Classification of Surface EMGs Using Wavelet Packet Energy Analysis and a Genetic Algorithm-Based Support Vector Machine. Neurophysiology 2013, 45, 39–48. [Google Scholar] [CrossRef]
- Aljebory, K.M.; Jwmah, Y.M.; Mohammed, T.S. Classification of EMG Signals: Using DWT Features and ANN Classifier. IAENG Int. J. Comput. Sci. 2024, 51, 23–31. [Google Scholar]
- Côté-Allard, U.; Fall, C.L.; Drouin, A.; Campeau-Lecours, A.; Gosselin, C.; Glette, K.; Laviolette, F.; Gosselin, B. Deep Learning for Electromyographic Hand Gesture Signal Classification Using Transfer Learning. IEEE Trans. Neural Syst. Rehabil. Eng. 2019, 27, 760–771. [Google Scholar] [CrossRef] [PubMed]
- Arozi, M.; Putri, F.T.; Ariyanto, M.; Caesarendra, W.; Widyotriatmo, A.; Setiawan, J.D. Electromyography (EMG) Signal Recognition Using Combined Discrete Wavelet Transform Based Adaptive Neuro-Fuzzy Inference Systems (ANFIS). In AIP Conference Proceedings; AIP Publishing LLC: Melville, NY, USA, 2017; Volume 1788. [Google Scholar] [CrossRef]
- Tuncer, T.; Dogan, S.; Subasi, A. Surface EMG Signal Classification Using Ternary Pattern and Discrete Wavelet Transform Based Feature Extraction for Hand Movement Recognition. Biomed. Signal Process. Control 2020, 58, 101872. [Google Scholar] [CrossRef]
- Aarotale, P.N.; Rattani, A. Time Frequency Analysis of EMG Signal for Gesture Recognition Using Fine Grained Features. arXiv 2025, arXiv:2504.14708. [Google Scholar] [CrossRef]

| Source | Technique | AI Methods | Application | Results |
|---|---|---|---|---|
| [55] | DWT | Intelligent dynamic bit allocation scheme implemented using a Kohonen layer (neural network) | Data compression, noise reduction | It has been demonstrated that the compression performance of the EMG signal is superior compared to standard wavelet algorithms, minimizing distortions at a given compression ratio. |
| [56] | WT | ANN—MLP and GRNN (input comprises the coefficients of the auto-regressive signal model after WT) | Muscle type classification, feature extraction | The high effectiveness in classifying muscle types was confirmed, and it was demonstrated that preliminary processing of EMG signals using the wavelet transform significantly improves the results. |
| [57] | DWT | Random Forest, KNN, Decision Tree | Hand movement classification, feature extraction | The effectiveness of different feature extractors and classifiers was compared, identifying combinations that produced good results; DWT features showed competitive effectiveness. |
| [58] | WPT (wavelet packet transform) | BPNN SVM GA-SVM | Muscle fatigue, muscle activity classification | Identification of muscle fatigue using the GA-SVM classifier, which was more accurate than other approaches. |
| [59] | DWT | Feed Forward Back Propagation Neural Network (FFBPNN) (ANN) | Hand movement classification, feature extraction | High accuracy was achieved in the classification of hand movements using DWT features and a neural network, indicating the effectiveness of the selected decomposition level. |
| [60] | CWT | Deep neural networks (ConvNets) | Gesture recognition, Feature extraction (automatic) | Transfer learning was shown to systematically and significantly improve the performance of deep neural networks in EMG gesture recognition, particularly for CWT models. |
| [61] | DWT | Adaptive Neuro-Fuzzy Inference System (ANFIS). | EMG signal recognition (for prosthetics) | High accuracy in the recognition of EMG signals was achieved, indicating the potential to combine DWT and ANFIS as a control signal for prosthetics. |
| [62] | DWT | KNN | Recognition of hand movements. | A practical and computationally lightweight multilevel feature extraction method (TP-DWT) was proposed for sEMG signals, which allowed for achieving high accuracy to be achieved in hand movement recognition. |
| [63] | CWT (cumulative scalograms) | XMANet (Cross-layer Mutual Attention Learning Network) with different CNNs | Gesture recognition, advanced feature extraction | A novel network architecture (XMANet) with attention mechanisms was proposed, which consistently improves the performance of gesture recognition based on CWT scalograms. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Daniel, N.; Małachowski, J.; Sybilski, K.; Błażkiewicz, M. Muscle Fatigue in Dynamic Movement: Limitations and Challenges, Experimental Design, and New Research Horizons. Bioengineering 2026, 13, 248. https://doi.org/10.3390/bioengineering13020248
Daniel N, Małachowski J, Sybilski K, Błażkiewicz M. Muscle Fatigue in Dynamic Movement: Limitations and Challenges, Experimental Design, and New Research Horizons. Bioengineering. 2026; 13(2):248. https://doi.org/10.3390/bioengineering13020248
Chicago/Turabian StyleDaniel, Natalia, Jerzy Małachowski, Kamil Sybilski, and Michalina Błażkiewicz. 2026. "Muscle Fatigue in Dynamic Movement: Limitations and Challenges, Experimental Design, and New Research Horizons" Bioengineering 13, no. 2: 248. https://doi.org/10.3390/bioengineering13020248
APA StyleDaniel, N., Małachowski, J., Sybilski, K., & Błażkiewicz, M. (2026). Muscle Fatigue in Dynamic Movement: Limitations and Challenges, Experimental Design, and New Research Horizons. Bioengineering, 13(2), 248. https://doi.org/10.3390/bioengineering13020248









