Structural Features of Nerve Guidance Conduits and Scaffolds in Preventing Axonal Misdirection: A Systematic Review of Retrograde Tracing Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.1.1. Population Exclusions
2.1.2. Intervention Exclusions
2.1.3. Comparator Exclusions
2.1.4. Outcome Exclusions
2.2. Information Sources and Search Strategy
2.3. Selection Process
2.4. Data Collection
2.5. Data Items (Outcomes)
- LMN/LSN ratio—LMN counts relative to the number of retrogradely labeled sensory neurons (LSNs). The LMN/LSN ratio is defined as the proportion of labeled motoneurons to labeled sensory neurons, with lower ratios indicating higher misdirection of motor axons.
- TLN/TF ratio—total number of labeled motoneurons (TLN) relative to the total number of myelinated fibers (TF). To ensure comparability across studies, TF was standardized as a regenerated nerve (RN) measure, defined as values obtained at the mid-graft or distal graft; when both were reported, the mean value was used.
- Incorrect projections—The number of double or multiple labeled motoneurons following simultaneous retrograde tracing studies, expressed as a percentage of incorrectly projected neurons, indicating axonal dispersion, in the form of distal arborization and consequential polyinnervation and/or missinervation.
- Accurate projections—The number of double labeled motorneurons following sequential retrograde tracing studies, expressed as a percentage of correctly projecting neurons.
- AD/FD ratio—the relationship between axon diameter (AD) and fiber density (FD), where lower values reflect dense networks of fine sprouts indicative of axonal dispersion, and higher values reflect sparser networks of more mature fibers. Both AD and FD were standardized as RN measures, taken at the mid- or distal graft, or averaged when both sites were reported.
- TF ratios between nerve segments (e.g., MG.TF/PDS.TF, DDS.TF/MG.TF, DDS.TF/PDS.TF) indicating the extent of axonal continuity, dispersion, or loss.
2.6. Data Items (Other Variables)
- Study identifiers: Including the first author, year of publication, journal name, and funding.
- Population characteristics: Genus (strain), sex, weight, number of animals per group, injured nerve, length of nerve gap (mm).
- Intervention characteristics: Autologous/isogenous nerve graft orientation, artifitial nerve graft material and fabrication method, porosity, degradability, permeability, internal structure.
- Retrograde tracing methodology: The tracer compounds used, labeled nerves, and whether single, double, triple simultaneous or sequential labeling is performed.
2.7. Study Risk of Bias Assessment
2.8. Effect Measures
2.9. Synthesis Methods
- Unstructured ArtNGs (hollow NGCs without internal framework, further categorized by architecture to smooth and rough).
- Structured ArtNGs (NSCs or filled NGCs, with internal framework, stratified by architecture to fibrous, multichannel, microporous).
- Direct ETE repair versus ArtNG repair.
- ANG versus ArtNG repair assessed by single-dye tracing methods.
- ANG versus ArtNG repair assessed by multiple-dye tracing methods.
- Risk of bias: based on SYRCLE tool
- Heterogeneity: noted when SD of the study means was large (>50% of mean)
- Indirectness: variation in species (rat vs. dog) or gap length
- Imprecision: small n of study arms per group (<5)
- No publication bias assessment was possible.
2.10. Automatization Tools and Language Based Models
3. Results
3.1. Study Selection
3.2. Excluded Studies
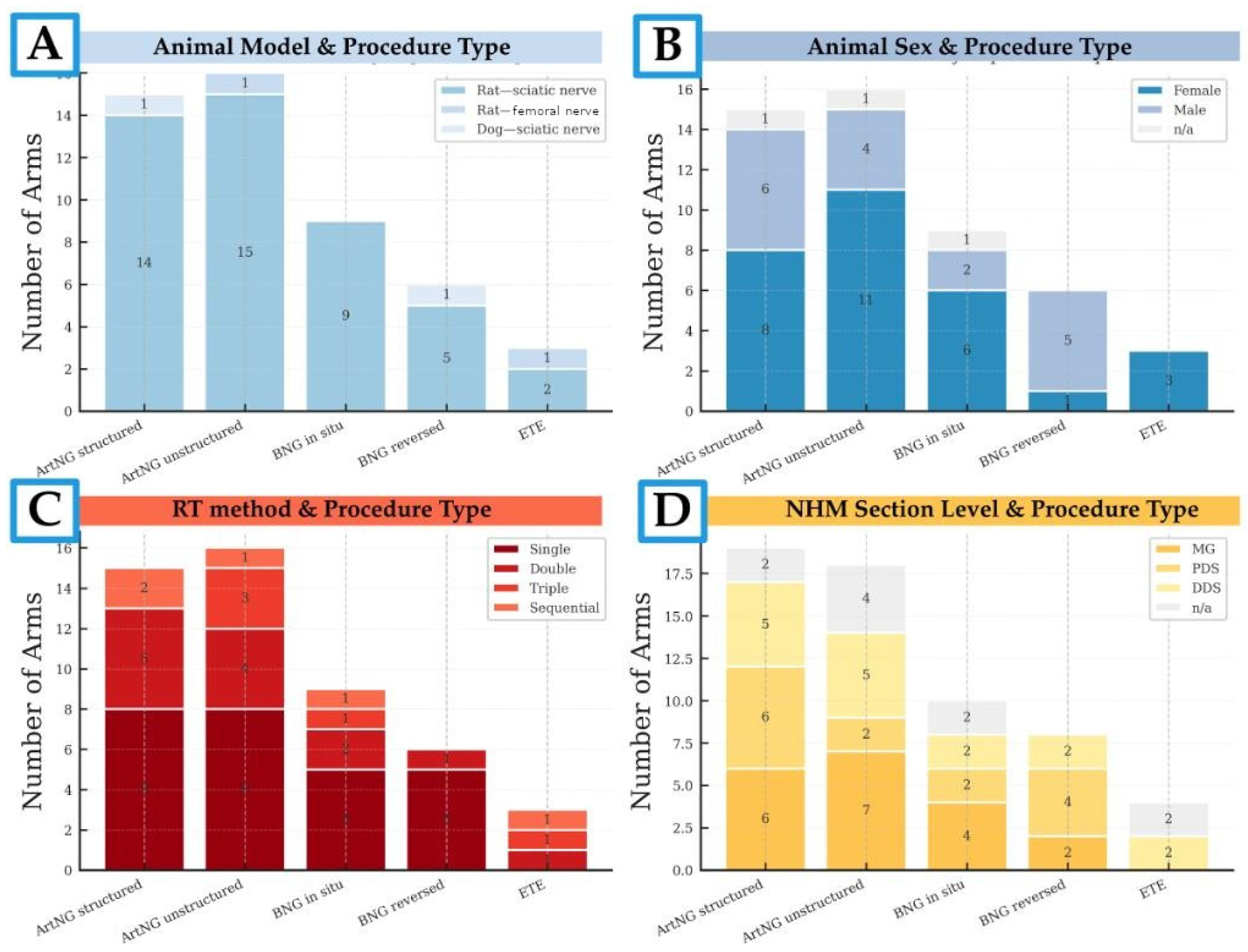
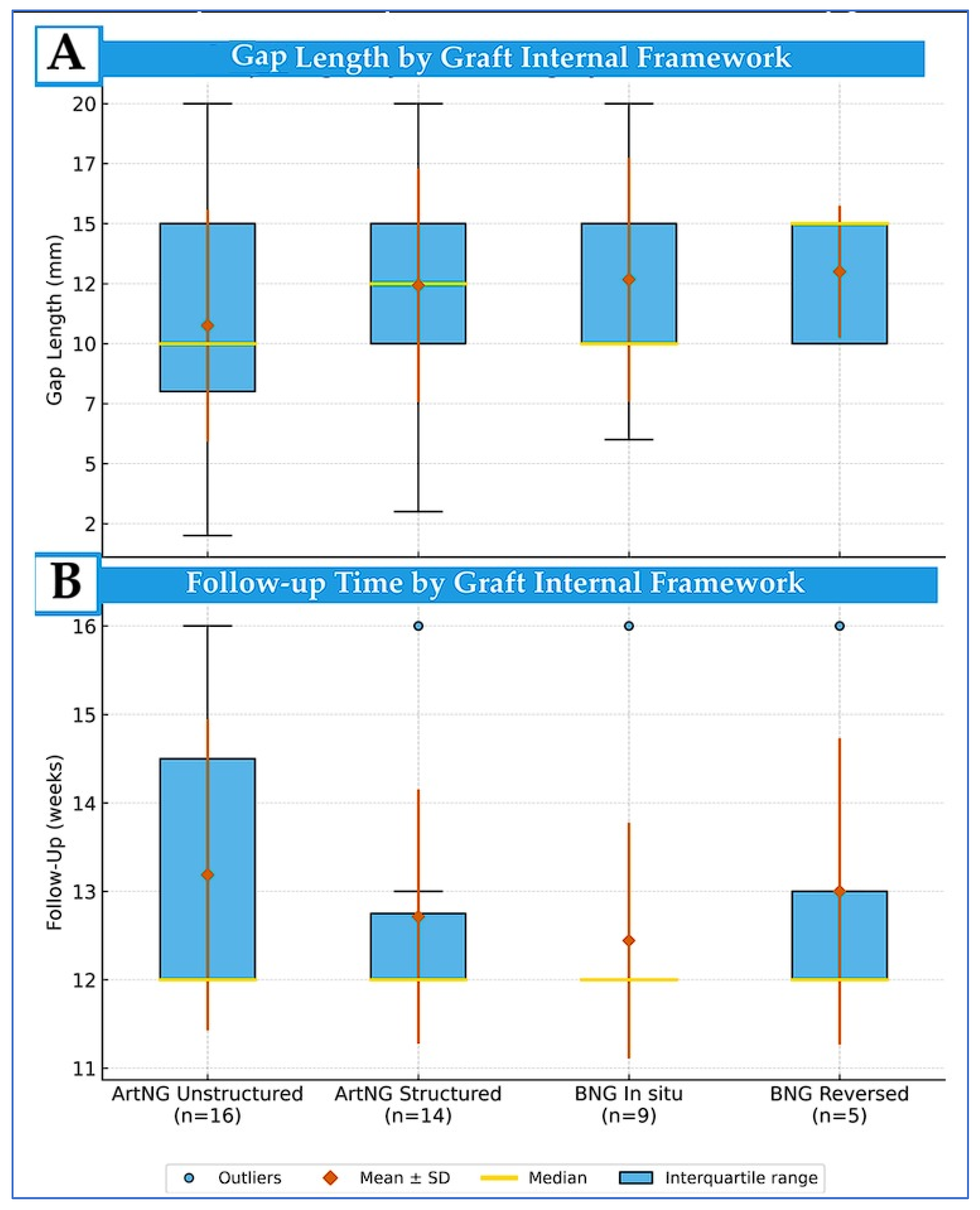
3.3. Study Characteristics
3.4. Risk of Bias in Studies
3.5. Results of Individual Studies
3.5.1. LMN Counts
ArtNG vs. Direct ETE Repair
ArtNG vs. ANG Repair
- Unstructured ArtNG arms, provided similar LMN counts in 2/11 and lower in 9/11 cases compared to ANGs.
- Structured ArtNG arms provided similar LMN counts in 8/15 and lower in 7/15 cases compared to ANGs. In 5/5 cases, longitudinal multichannel NSCs led to similar LMN counts, while randomized multichannel NSCs (2/2) and multichannel NGCs (3/3) led to lower LMN counts in all scenarios. Fibrous designs led to similar LMN counts in 3/5, and lower in 2/5 cases compared to ANGs.
3.5.2. TLN Counts and Associated Ratios
- Structured ArtNG arms matched ANGs in TLN counts in 4/5, LSN counts in 3/5, and LMN/LSN ratio in 4/5 cases. LSN counts exceeded ANGs in one case. Regarding TLM/TF ratio, structured ArtNGs were similar to ANGs in one and superior in other case, out of two.
- Unstructured ArtNG arms matched ANGs in TLN counts in 1/6, LSN counts in 1/6 and LMN/LSN ratio in 3/5 cases. Regarding TLM/TF ration, unstructured ArtNGs were superior to ANGs in 4/5 and inferior in 1/5 cases.
3.5.3. Motoneuron Projection Accuracy
ArtNG vs. Direct ETE Repair
ArtNG vs. ANG Repair
- Structured ArtNGs had lower percentage of incorrect projecting LMNs compared to ANGs in 4/7, similar in 2/7, and higher in 1/7 cases. Accurate projecting LMNs were reported only for the two arms and were similar to ANG in both cases.
- Unstructured ArtNGs provided similar percentage of incorrect projecting LMNs as ANGs in 3/5 and lower percentage in 2/5 cases. There were no data about accurate projecting LMNs across unstructured ArtNG arms.
3.5.4. Other Outcomes
- Structured ArtNGs had similar AD/FD ratios in 2/5, lower in 2/5 and higher in 1/5 arms, when compared to ANGs.
- Unstructured ArtNGs had higher AD/FD ratios in 2/2 arms when compared to ANGs.
3.6. Synthesis of Results
- LMN counts: In 26 arms with ANG comparators, structured ArtNGs preserved LMN numbers in approximately half of cases (8/15), with longitudinal microchannel NSCs consistently matching ANGs (5/5). In contrast, unstructured ArtNGs provided similar LMN counts in only 2/11 arms, with 9/11 showing reductions.
- TLN counts: Structured ArtNGs preserved TLN in 4/5 arms, whereas unstructured guides showed preservation in just 1/6, with the remainder reporting decreases.
- LSN counts: Structured conduits preserved LSN in 3/5 arms and exceeded autografts in one arm, while unstructured conduits preserved LSN in only 1/6, with reductions in the others.
- LMN/LSN ratio: Structured conduits preserved motor–sensory balance in 4/5 arms, whereas unstructured guides were preserved in 3/5 but decreased in 2/5 arms.
- Projection accuracy: For incorrect projecting LMNs, structured conduits outperformed autografts in 4/7 arms, were similar in 2/7, and worse in 1/7; accurate projections were reported in only two structured arms, both comparable to ANG. Unstructured conduits reduced misdirected projections in 2/5 arms, matched in 3/5, and did not report accurate projection data.
3.7. Risk of Bias Due to Missing Values
3.8. Certainty of Evidence
4. Discussion
4.1. Limitations of the Evidence
4.2. Limitations in the Review Process
4.3. Implications for Practice and Policy
5. Future Perspectives
- Adopt standardized and comprehensive outcome reporting. Future studies should consistently include retrograde tracing alongside histomorphometry to assess both axonal dispersion and reinnervation accuracy.
- Improve methodological rigor. Prospective protocol registration, randomization, blinding, and complete outcome reporting should become standard practice in preclinical nerve regeneration research.
- Use larger animal models and longer follow-up periods. These steps would enhance the clinical relevance of findings and better simulate human nerve injury and repair conditions.
- Isolate structural effects. Future experimental designs should separate the impact of the conduit structure from functional enhancements such as growth factors, stem cells, etc., to clarify the contribution of architecture alone.
- Explore personalized, AI-assisted NGC design. Emerging imaging and 3D printing technologies, combined with machine learning, offer promising avenues for the development of patient-specific NGCs that replicate native fascicular anatomy.
- Improve biomaterials, to provide microenvironment for axonal growth, by maintaining its physical structure, while being able to dissolve immediately when axons reach the distal stump.
6. Summary
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
Appendix A
| PICO | Key Words | Search Terms | |
|---|---|---|---|
| Population | Animals | Animals Mammals Models | (’’Animals’’[MH]) OR (‘’Models, Animal’’[MH]) OR (‘’Animal Experimentation’’[MH]) OR (rodent[Title/Abstract]) OR (rat[Title/Abstract]) OR (mouse[Title/Abstract]) OR (mice[Title/Abstract]) OR (hamster[Title/Abstract]) OR (monkey[Title/Abstract]) OR (’’guinea pig’’[Title/Abstract]) OR (pig[Title/Abstract]) OR (rabbit[Title/Abstract]) OR (cat[Title/Abstract]) OR (dog[Title/Abstract]) OR (sheep[Title/Abstract]) |
| Peripheral Nerve Injury | Nerve Injury Axotomy Nerve Gap Nerve Transection | (‘’Peripheral Nerve Injuries’’[MH]) OR (‘’Axotomy’’[MH]) OR (‘’Nerve Crush’’[MH]) OR (’’Nerve Regeneration’’[MH]) OR (Nerve Injury Model’’[Title/Abstract]) OR (’’Nerve Crush’’[Title/Abstract]) OR (’’Nerve Crush Model’’[Title/Abstract]) OR (’’Axonotmesis’’[Title/Abstract]) OR (’’Neurotmesis’’[Title/Abstract]) OR (’’Nerve Transection’’[Title/Abstract]) OR (’’Nerve Transection Model’’[Title/Abstract]) OR (’’Nerve Cut’’[Title/Abstract]) OR (’’Nerve Gap’’[Title/Abstract]) OR (’’Nerve Defect’’[Title/Abstract]) | |
| Intervention | Nerve Guidance Conduit | Nerve Guide Nerve Conduit Artificial Nerve Graft Nerve Scaffold | (‘’Tissue Scaffolds’’[MeSH terms]) OR (‘’Nerve Guidance Conduits’’[Title/Abstract]) OR (‘’Nerve Guidance Channels’’[Title/Abstract]) OR (NGCs[Title/Abstract]) OR (‘’Nerve Conduits’’[Title/Abstract]) OR (‘’Neural Conduits’’[Title/Abstract]) OR (‘’Neural Tubes’’[Title/Abstract]) OR (‘’Nerve Tubes’’[Title/Abstract]) OR (‘’Nerve Scaffold’’[Title/Abstract]) OR (‘’Neural Scaffold’’[Title/Abstract]) OR (‘’Artificial Nerve Graft’’[Title/Abstract]) OR (‘’Artificial Nerve Conduit’’[Title/Abstract]) OR (‘’Fabricated Nerve Graft’’[Title/Abstract]) OR (‘’Synthetic Nerve Graft’’[Title/Abstract]) OR (‘’Nerve Guide’’[Title/Abstract]) OR (‘’Nerve Connector’’[Title/Abstract]) |
| Structural design | Hollow Porous Grooved Nanofiber Nanosponge Multichannel | (‘’Biomimetics’’[MeSH terms]) OR (‘’Nanostructures’’[MeSH terms]) OR (’’Tissue’’Guided Tissue Regeneration’’[MeSH terms]) OR (Structure[Title/Abstract]) OR (Design[Title/Abstract]) OR (Property[Title/Abstract]) OR (‘’Structural Design Strategy’’[Title/Abstract]) OR (Architecture[Title/Abstract]) OR (Microarchitecture[Title/Abstract]) OR (Microstructure[Title/Abstract]) OR (Superstructure[Title/Abstract]) OR (Topography[Title/Abstract]) OR (‘’Lumen Topography’’[Title/Abstract]) OR (‘’Lumen Feature’’[Title/Abstract]) OR (‘’Lumen Surface Feature’’[Title/Abstract]) OR (‘’Structural Feature’’[Title/Abstract]) OR (’’Mechanical Cues’’[Title/Abstract]) OR (’’Topographic Cues’’[Title/Abstract]) OR (Topographic alignment[Title/Abstract]) OR (Porous[Title/Abstract]) OR (Nanopores[Title/Abstract]) (Grooved[Title/Abstract]) OR (Microgrooved[Title/Abstract]) OR (Filled[Title/Abstract]) OR (’’Luminal Fillers’’[Title/Abstract]) OR (Nanofiber[Title/Abstract]) OR (‘’Filament fiber’’[Title/Abstract]) OR (Hydrogel[Title/Abstract]) OR (Nanosponge[Title/Abstract]) OR (Micropore[Title/Abstract]) OR (Multichannel[Title/Abstract]) OR (Microchannel[Title/Abstract]) OR (Multilumen[Title/Abstract]) OR (‘’Single Channel’’[Title/Abstract]) OR (‘’Single Lumen’’[Title/Abstract]) OR (‘’Single Hollow’’[Title/Abstract]) OR (Multibranched[Title/Abstract]) OR (Branched[Title/Abstract]) OR (Micropatterned[Title/Abstract]) OR (Bibranched[Title/Abstract]) OR (Tubular[Title/Abstract]) OR (Spiral[Title/Abstract]) OR (’’Channel Orientation’’[Title/Abstract]) OR (’’Channel Number’’[Title/Abstract]) OR (’’Pores’’[Title/Abstract]) OR (’’Holes’’[Title/Abstract]) | |
| Comparison | Surgical nerve repair | Nerve repair End-to-end repair Autograft | (Neurotization[MeSH terms]) OR (‘’Suture Techniques’’[MeSH terms]) OR (’’Sutureless Surgical Procedures’’[MeSH terms]) OR (Autografts[MeSH terms]) OR (Neurotization[Title/Abstract]) OR (Autografts[Title/Abstract]) OR (’’Nerve Reconstruction’’[Title/Abstract]) OR (’’End-to-end suture’’[Title/Abstract]) OR (’’epineurial suture’’[Title/Abstract]) OR (’’Graft Repair’’[Title/Abstract]) OR (’’Nerve Repair’’[Title/Abstract]) OR (’’Direct Nerve Repair’’[Title/Abstract]) OR (’’Direct Nerve Suture’’[Title/Abstract]) OR (Microsuture[Title/Abstract]) OR (’’Nerve Surgery’’[Title/Abstract]) |
| Outcome | Axonal misdirection | Axonal guidance Axonal pathfinding Axonal sprouting Axonal dispersion Axonal alignment Reinnervation mismatch | (Neurogenesis[MeSH terms]) OR (’’Neuronal Outgrowth’’[MeSH terms]) OR (’’Axon Pathfinding’’[MeSH terms]) OR (’’Axon Fasciculation’’[MeSH terms]) OR (’’Axon Guidance’’[MeSH terms]) OR (’’Axon* Bundling’’[Title/Abstract) OR (’’Neurite Fasciculation’’[Title/Abstract]) OR (’’Neural Pathfinding’’[MeSH terms]) OR (’’Neural Guidance’’[Title/Abstract]) OR (’’Axon* Pruning’’[Title/Abstract]) OR (’’Neurite Pruning’’[Title/Abstract]) OR (’’Neurite Arborization’’[Title/Abstract]) OR (’’Axon* Arborization’’[Title/Abstract]) OR (’’Synaptic Pruning’’[Title/Abstract]) OR (‘’Axon* Pathway’’[Title/Abstract]) OR (‘’Axon* Misdirection’’[Title/Abstract]) OR (‘’Axon* Direction’’[Title/Abstract]) OR (‘’Axon* Alignment’’[Title/Abstract]) OR (‘’Axon* Misalignment’’[Title/Abstract]) OR (‘’Axon* Orientation’’[Title/Abstract]) OR (‘’Axon* Navigation’’[Title/Abstract]) OR (‘’Axon* Misrouting’’[Title/Abstract]) OR (‘’Axon* Sprouting’’[Title/Abstract]) OR (‘’Axon Arbors’’[Title/Abstract]) OR (‘’Axon* Dispersion’’[Title/Abstract]) OR (‘’Axon* Deviation’’[Title/Abstract]) OR (‘’Reinnervation Mismatch’’[Title/Abstract]) OR (‘’Targeted Reinnervation’’[Title/Abstract]) OR (‘’Selective Regeneration’’[Title/Abstract]) OR (‘’’Selective Reinnervation’’[Title/Abstract]) OR (‘’Preferential Reinnervation’’[Title/Abstract]) OR (‘’Preferential Regeneration’’[Title/Abstract]) OR (‘’Random Reinnervation’’[Title/Abstract]) OR (‘’Failed Target Reinnervation’’[Title/Abstract]) OR (‘’Specific Reinnervation’’[Title/Abstract]) OR (‘’Aberrant Reinnervation’’[Title/Abstract]) |
| Labeling | Retrograde axonal tracing Anterograde axonal tracing | (‘’Radioactive Tracers’’[MeSH terms]) OR (‘’Neuroanatomical Tract- Tracing Techniques’’[MeSH terms]) OR (‘’Neuronal Tract- Tracers’’[MeSH terms]) OR (‘’Staining and Labeling’’[MeSH terms]) OR (‘’Fluorescent Antibody Technique’’[MeSH terms]) OR (‘’Neural Pathway Tracing’’[Title/Abstract]) OR (‘’Axon* Tracing’’[Title/Abstract]) OR (‘’Axon* Targeting’’[Title/Abstract]) OR (‘’Axon* Labeling’’[Title/Abstract]) OR (‘’Neural Tract-Tracers’’[Title/Abstract]) OR (‘’Retrograde Tracing’’[Title/Abstract]) OR (‘’Anterograde Tracing’’[Title/Abstract]) OR (‘’Retrograde Labeling’’[Title/Abstract]) OR (‘’Anterograde Labeling’’[Title/Abstract]) OR (‘’Fluoro-Gold’’[Title/Abstract]) OR (‘’DiI’’[Title/Abstract]) OR (‘’DiO’’[Title/Abstract]) OR (‘’Cholera Toxin B Subunit’’[Title/Abstract]) OR (’’Immunohistochemistry’’[MeSH Terms]) OR (‘’Neurofilament Proteins’’[MeSH Terms]) OR (‘’GAP-43’’[Title/Abstract]) OR (‘’βIII-Tubulin’’[Title/Abstract]) OR (‘’Fluoro-Gold’’[Title/Abstract]) OR (‘’Fast Blue’’[Title/Abstract]) OR (’’Nerve count’’[Title/Abstract]) OR (’’Axon count’’[Title/Abstract]) | |
| Functionality | Sciatic, Peroneal and Tibial function index | (’’Enhanced Recovery After Surgery’’[MeSH terms]) OR (‘’Functional Status’’[MeSH terms]) OR (‘’Functional Performance’’[MeSH terms]) OR (‘’Recovery of Function’’[MeSH terms]) OR (‘’Behavioral Tests’’[MeSH Terms]) OR (‘’Sciatic Functional Index’’[Title/Abstract]) OR (’’SFI’’[Title/Abstract]) OR (‘’Pinch Test’’[Title/Abstract]) OR (‘’Withdrawal Reflex’’[Title/Abstract]) | |
| Visualization | Microscopy Fluoromicroscopy | (‘’Electron Microscopy’’[MeSH Terms]) OR (‘’3D Imaging’’[Title/Abstract]) OR (‘’CLARITY’’[Title/Abstract]) OR (‘’iDISCO’’[Title/Abstract]) OR (‘’Light-Sheet Microscopy’’[Title/Abstract]) OR (‘’Confocal Microscopy’’[MeSH Terms]) OR (‘’Magnetic Resonance Imaging’’[MeSH Terms]) OR (‘’Diffusion Tensor Imaging’’[Title/Abstract]) OR (‘’DTI’’[Title/Abstract]) OR (‘’Optical Imaging’’[Title/Abstract]) OR (‘’Two-Photon Microscopy’’[Title/Abstract]) OR (’’Optogenetics’’[MeSH Terms]) OR (‘’Light-Activated Stimulation’’[Title/Abstract]) OR (‘’Axon* Functional Mapping’’[Title/Abstract]) | |
| Electrostudies | Electrostimulation Tetanic force | (‘’Electrophysiology’’[MeSH Terms]) OR (‘’Nerve Conduction Studies’’[MeSH Terms]) OR (‘’Nerve Conduction Velocity’’[Title/Abstract]) OR (‘’Compound Muscle Action Potential’’[Title/Abstract]) OR (’’Electrostimulation’’[Title/Abstract]) OR (’’Electroneurography’’[Title/Abstract]) OR (’’EMNG’’[Title/Abstract]) | |
| #1 | Animals AND (Nerve Injury AND Nerve Repair AND NGC) AND Outcome |
| #2 | (Nerve Injury AND Nerve Repair AND NGC) AND Outcome |
| #3 | (Nerve Injury AND (Nerve Repair OR NGC) AND Structural Design AND Outcome |
| #4 | Nerve Injury AND Axonal Guidance AND Outcome |
| #5 | Nerve Repair AND Axonal Guidance AND Assessment |
| #6 | NGC AND Structural Design AND Outcome |
| #7 | ((excludepreprints[Filter]) AND (fft[Filter]) AND (english[Filter])) NOT (’’review’’[Publication Type] OR ’’meta-analysis’’[Publication Type]) NOT (’’Spinal cord’’[Title]) NOT (‘’Brain’’[Title]) NOT (‘’Stroke’’[Title]) AND (medline[Filter]) AND (1000/1/1:2024/12/31[pdat]) |
| #8 | Final Search Code = (#1 OR #2 OR #3 OR #4 OR #5 OR #6) AND #7 = 2617 |
| Study ID | Sequence Generation | Baseline Characteristics | Allocation Concealment | Random Housing | Blinded Intervention | Random Outcome Assessment | Blinded Outcome Assessment | Incomplete Outcome Data | Selective Outcome Reporting | Other Sources of Bias |
|---|---|---|---|---|---|---|---|---|---|---|
| Bodine-Fowler et al., 1997 [77] | Unclear | Low risk | Unclear | Unclear | High risk | Unclear | Unclear | High risk | High risk | Unclear |
| Valero Cabre et al., 2001 [78] | Unclear | Low risk | Unclear | Unclear | High risk | Low risk | Low risk | Low risk | Low risk | Unclear |
| Valero Cabre et al., 2004 [79] | Low risk | Low risk | Low risk | Low risk | Unclear | Low risk | Low risk | Low risk | Unclear | Unclear |
| De Ruiter et al., 2008 [80] | High risk | High risk | High risk | High risk | High risk | Low risk | Low risk | Low risk | Unclear | Unclear |
| Hu et al., 2009 [81] | Unclear | Low risk | Unclear | Unclear | High risk | High risk | Low risk | Low risk | Low risk | High risk |
| Huang et al., 2010 [82] | Unclear | Low risk | Unclear | Unclear | High risk | Low risk | Low risk | Low risk | Unclear | Unclear |
| Yao et al., 2010 [55] | Unclear | Low risk | Unclear | Unclear | High risk | Low risk | Low risk | Low risk | Low risk | Unclear |
| Zhang et al., 2011 [83] | Unclear | Low risk | Unclear | Unclear | High risk | Low risk | Low risk | Low risk | Low risk | Unclear |
| Daly et al., 2012 [31] | Unclear | Low risk | High risk | High risk | High risk | Low risk | Low risk | Unclear | Low risk | Unclear |
| Jiang et al., 2012 [84] | High risk | Unclear | High risk | Unclear | High risk | Low risk | Low risk | High risk | Unclear | Unclear |
| Yu et al., 2015 [85] | Unclear | Low risk | Unclear | Unclear | High risk | Unclear | Low risk | Unclear | Unclear | Unclear |
| Bozkurt et al., 2016 [86] | High risk | Unclear | High risk | Unclear | High risk | Low risk | Low risk | High risk | Unclear | Unclear |
| Van Neerven et al., 2017 [87] | High risk | Low risk | High risk | Unclear | High risk | Low risk | Low risk | Low risk | Low risk | Unclear |
| Zhu et al., 2017 [88] | Unclear | Low risk | High risk | Unclear | High risk | Low risk | Low risk | Unclear | Unclear | Unclear |
| Xue et al., 2018 [90] | Unclear | Low risk | High risk | Unclear | High risk | Low risk | Low risk | Low risk | Unclear | High risk |
| Liu et al., 2018 [89] | Unclear | High risk | High risk | Unclear | High risk | Unclear | Low risk | Unclear | Low risk | Unclear |
| Zhu et al., 2020 [91] | Unclear | Low risk | High risk | Unclear | High risk | Unclear | Low risk | Low risk | Unclear | Unclear |
| Lu et al., 2021 [92] | Unclear | Low risk | Unclear | Unclear | High risk | Unclear | Low risk | Low risk | Unclear | Unclear |
| Yang et al., 2023 [93] | Low risk | Low risk | Unclear | Unclear | High risk | Low risk | Low risk | Low risk | Unclear | High risk |
References
- Akbari, H.; Saraee, A.; Mirzaei, L.; Abolfazli, M.; Bagheri, H.; Akbari, P. Comparison of Functionality Level, Disability and Quality of Life in Subjects with Peripheral Nerve Repair in the Upper Extremity. World J. Plast. Surg. 2024, 13, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.R.; Karpinski, M.; Davidge, K.M.; Klassen, A.; Dengler, J. Patient-reported Outcome Measures for Peripheral Nerve Injuries: A Systematic Review. Plast. Reconstr. Surg. Glob. Open 2024, 12, e6408. [Google Scholar] [CrossRef]
- Faroni, A.; Mobasseri, S.A.; Kingham, P.J.; Reid, A.J. Peripheral nerve regeneration: Experimental strategies and future perspectives. Adv. Drug Deliv. Rev. 2015, 82–83, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Grosu-Bularda, A.; Vancea, C.V.; Hodea, F.V.; Cretu, A.; Bordeanu-Diaconescu, E.M.; Dumitru, C.S.; Ratoiu, V.-A.; Teodoreanu, R.-N.; Lascar, I.; Hariga, C.-S. Optimizing Peripheral Nerve Regeneration: Surgical Techniques, Biomolecular and Regenerative Strategies-A Narrative Review. Int. J. Mol. Sci. 2025, 26, 3895. [Google Scholar] [CrossRef]
- Costa, N.N.; Santos, J.F.D.; Aranha, M.; Coelho, E.W.F.; Paes, V.L.S.; Oliveira, R.C.S. Repair methods in peripheral nerves after traumatic injuries: A systematic literature review. Acta Cir. Bras. 2025, 40, e401225. [Google Scholar] [CrossRef]
- Bertelli, J.A.; Tuffaha, S.; Sporer, M.; Seltser, A.; Cavalli, E.; Soldado, F.; Hill, E. Distal nerve transfers for peripheral nerve injuries: Indications and outcomes. J. Hand Surg. Eur. Vol. 2024, 49, 721–733. [Google Scholar] [CrossRef]
- Rasulic, L.; Savic, A.; Grujic, J.; Micic, A.; Radojevic, S.; Ivic, J.; Lepic, M. Distal nerve transfer (PT-AIN, SUP-PIN) for regaining hand function in C8, T1 root injury following extirpation of the right C8, T1 schwannoma. Neurosurg. Focus Video 2023, 8, V10. [Google Scholar] [CrossRef] [PubMed]
- DeLeonibus, A.; Rezaei, M.; Fahradyan, V.; Silver, J.; Rampazzo, A.; Bassiri Gharb, B. A meta-analysis of functional outcomes in rat sciatic nerve injury models. Microsurgery 2021, 41, 286–295. [Google Scholar] [CrossRef]
- IJpma, F.F.; Nicolai, J.P.A.; Meek, M.F. Sural nerve donor-site morbidity: Thirty-four years of follow-up. Ann. Plast. Surg. 2006, 57, 391–395. [Google Scholar] [CrossRef]
- Muheremu, A.; Ao, Q. Past, Present, and Future of Nerve Conduits in the Treatment of Peripheral Nerve Injury. BioMed Res. Int. 2015, 2015, 237507. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S. Nerve guide conduits for peripheral nerve injury repair: A review on design, materials and fabrication methods. Acta Biomater. 2020, 106, 54–69. [Google Scholar] [CrossRef]
- Stocco, E.; Barbon, S.; Emmi, A.; Tiengo, C.; Macchi, V.; De Caro, R.; Porzionato, A. Bridging Gaps in Peripheral Nerves: From Current Strategies to Future Perspectives in Conduit Design. Int. J. Mol. Sci. 2023, 24, 9170. [Google Scholar] [CrossRef]
- Sarker, M.; Naghieh, S.; McInnes, A.D.; Schreyer, D.J.; Chen, X. Strategic Design and Fabrication of Nerve Guidance Conduits for Peripheral Nerve Regeneration. Biotechnol. J. 2018, 13, e1700635. [Google Scholar] [CrossRef]
- Zhou, W.; Rahman, M.S.U.; Sun, C.; Li, S.; Zhang, N.; Chen, H.; Han, C.C.; Xu, S.; Liu, Y. Perspectives on the Novel Multifunctional Nerve Guidance Conduits: From Specific Regenerative Procedures to Motor Function Rebuilding. Adv. Mater. 2024, 36, e2307805. [Google Scholar] [CrossRef]
- Dogny, C.; Andre-Levigne, D.; Kalbermatten, D.F.; Madduri, S. Therapeutic Potential and Challenges of Mesenchymal Stem Cell-Derived Exosomes for Peripheral Nerve Regeneration: A Systematic Review. Int. J. Mol. Sci. 2024, 25, 6489. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Bai, J.; Zhou, X.; Chen, N.; Jiang, Q.; Ren, Z.; Li, X.; Su, T.; Liang, L.; Jiang, W.; et al. Electrical stimulation with polypyrrole-coated polycaprolactone/silk fibroin scaffold promotes sacral nerve regeneration by modulating macrophage polarisation. Biomater Transl. 2024, 5, 157–174. [Google Scholar]
- Amalakanti, S.; Mulpuri, R.P.; Avula, V.C.R. Recent advances in biomaterial design for nerve guidance conduits: A narrative review. Adv. Technol. Neurosci. 2024, 1, 32–42. [Google Scholar] [CrossRef]
- Cao, S.; Deng, Y.; Zhang, L.; Aleahmad, M. Chitosan nanoparticles, as biological macromolecule-based drug delivery systems to improve the healing potential of artificial neural guidance channels: A review. Int. J. Biol. Macromol. 2022, 201, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Yihao, W.; Qi, T.; Xiong, A.; Liu, P.; Chen, Y.; Zeng, H.; Yu, F.; Weng, J. Combination of stem cells and nerve guide conduit for the treatment of peripheral nerve injury: A meta-analysis. Muscle Nerve 2024, 69, 227–238. [Google Scholar] [CrossRef]
- Ronchi, G.; Fregnan, F.; Muratori, L.; Gambarotta, G.; Raimondo, S. Morphological Methods to Evaluate Peripheral Nerve Fiber Regeneration: A Comprehensive Review. Int. J. Mol. Sci. 2023, 24, 1818. [Google Scholar] [CrossRef]
- Orfahli, L.M.; Rezaei, M.; Figueroa, B.A.; Crawford, A.V.; Annunziata, M.J.; Rao, M.; Fahradyan, V.; Ordenana, C.X.; Silver, J.; Rampazzo, A.; et al. Histomorphometry in Peripheral Nerve Regeneration: Comparison of Different Axon Counting Methods. J. Surg. Res. 2021, 268, 354–362. [Google Scholar] [CrossRef]
- Kemp, S.W.; Cederna, P.S.; Midha, R. Comparative outcome measures in peripheral regeneration studies. Exp. Neurol. 2017, 287, 348–357. [Google Scholar] [CrossRef]
- Wood, M.D.; Kemp, S.W.; Weber, C.; Borschel, G.H.; Gordon, T. Outcome measures of peripheral nerve regeneration. Ann. Anat. 2011, 193, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Vleggeert-Lankamp, C.L. The role of evaluation methods in the assessment of peripheral nerve regeneration through synthetic conduits: A systematic review. J. Neurosurg. 2007, 107, 1168–1189. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.A.; Moradzadeh, A.; Whitlock, E.L.; Brenner, M.J.; Myckatyn, T.M.; Wei, C.H.; Tung, T.H.; Mackinnon, S.E. Binary imaging analysis for comprehensive quantitative histomorphometry of peripheral nerve. J. Neurosci. Methods 2007, 166, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.I.; Gurjar, A.A.; Talukder, M.A.H.; Rodenhouse, A.; Manto, K.; O’Brien, M.; Karuman, Z.; Govindappa, P.K.; Elfar, J.C. Purposeful Misalignment of Severed Nerve Stumps in a Standardized Transection Model Reveals Persistent Functional Deficit With Aberrant Neurofilament Distribution. Mil. Med. 2021, 186, 696–703. [Google Scholar] [CrossRef]
- Alant, J.D.; Senjaya, F.; Ivanovic, A.; Forden, J.; Shakhbazau, A.; Midha, R. The impact of motor axon misdirection and attrition on behavioral deficit following experimental nerve injuries. PLoS ONE 2013, 8, e82546. [Google Scholar] [CrossRef]
- De Ruiter, G.C.; Malessy, M.J.; Alaid, A.O.; Spinner, R.J.; Engelstad, J.K.; Sorenson, E.J.; Kaufman, K.; Dyck, P.J.; Windebank, A.J. Misdirection of regenerating motor axons after nerve injury and repair in the rat sciatic nerve model. Exp. Neurol. 2008, 211, 339–350. [Google Scholar] [CrossRef]
- Wang, K.; Qin, B. [Research progress of peripheral nerve mismatch regeneration]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2021, 35, 387–391. [Google Scholar]
- Gordon, T. Peripheral Nerve Regeneration and Muscle Reinnervation. Int. J. Mol. Sci. 2020, 21, 8652. [Google Scholar] [CrossRef]
- Daly, W.T.; Yao, L.; Abu-rub, M.T.; O’Connell, C.; Zeugolis, D.I.; Windebank, A.J.; Pandit, A.S. The effect of intraluminal contact mediated guidance signals on axonal mismatch during peripheral nerve repair. Biomaterials 2012, 33, 6660–6671. [Google Scholar] [CrossRef]
- Uschold, T.; Robinson, G.A.; Madison, R.D. Motor neuron regeneration accuracy: Balancing trophic influences between pathways and end-organs. Exp. Neurol. 2007, 205, 250–256. [Google Scholar] [CrossRef]
- Brushart, T.M.; Gerber, J.; Kessens, P.; Chen, Y.G.; Royall, R.M. Contributions of pathway and neuron to preferential motor reinnervation. J. Neurosci. 1998, 18, 8674–8681. [Google Scholar] [CrossRef]
- Abdullah, M.; O’Daly, A.; Vyas, A.; Rohde, C.; Brushart, T.M. Adult motor axons preferentially reinnervate predegenerated muscle nerve. Exp. Neurol. 2013, 249, 1–7. [Google Scholar] [CrossRef]
- Brushart, T.; Kebaish, F.; Wolinsky, R.; Skolasky, R.; Li, Z.; Barker, N. Sensory axons inhibit motor axon regeneration in vitro. Exp. Neurol. 2020, 323, 113073. [Google Scholar] [CrossRef] [PubMed]
- Maki, Y.; Yoshizu, T.; Tajima, T.; Narisawa, H. The selectivity of regenerating motor and sensory axons. J. Reconstr. Microsurg. 1996, 12, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Maki, Y.; Yoshizu, T.; Tsubokawa, N. Selective regeneration of motor and sensory axons in an experimental peripheral nerve model without endorgans. Scand. J. Plast Reconstr. Surg. Hand Surg. 2005, 39, 257–260. [Google Scholar] [CrossRef]
- Rotterman, T.M.; Garcia, V.V.; Housley, S.N.; Nardelli, P.; Sierra, R.; Fix, C.E.; Cope, T.C. Structural Preservation Does Not Ensure Function at Sensory Ia-Motoneuron Synapses following Peripheral Nerve Injury and Repair. J. Neurosci. 2023, 43, 4390–4404. [Google Scholar] [CrossRef] [PubMed]
- Koerber, H.R.; Seymour, A.W.; Mendell, L.M. Mismatches between peripheral receptor type and central projections after peripheral nerve regeneration. Neurosci. Lett. 1989, 99, 67–72. [Google Scholar] [CrossRef]
- Valero-Cabre, A.; Navarro, X. Functional impact of axonal misdirection after peripheral nerve injuries followed by graft or tube repair. J. Neurotrauma 2002, 19, 1475–1485. [Google Scholar] [CrossRef]
- Dun, X.P.; Parkinson, D.B. Visualizing peripheral nerve regeneration by whole mount staining. PLoS ONE 2015, 10, e0119168. [Google Scholar] [CrossRef]
- Brushart, T.M. Motor axons preferentially reinnervate motor pathways. J. Neurosci. 1993, 13, 2730–2738. [Google Scholar] [CrossRef] [PubMed]
- Madison, R.D.; Robinson, G.A.; Chadaram, S.R. The specificity of motor neurone regeneration (preferential reinnervation). Acta Physiol. 2007, 189, 201–206. [Google Scholar] [CrossRef]
- Hizay, A.; Ozsoy, U.; Demirel, B.M.; Ozsoy, O.; Angelova, S.K.; Ankerne, J.; Sarikcioglu, S.B.; Dunlop, S.A.; Angelov, D.N.; Sarikcioglu, L. Use of a Y-tube conduit after facial nerve injury reduces collateral axonal branching at the lesion site but neither reduces polyinnervation of motor endplates nor improves functional recovery. Neurosurgery 2012, 70, 1544–1556; discussion 56. [Google Scholar] [CrossRef]
- Guntinas-Lichius, O.; Hundeshagen, G.; Paling, T.; Angelov, D.N. Impact of different types of facial nerve reconstruction on the recovery of motor function: An experimental study in adult rats. Neurosurgery 2007, 61, 1276–1283; discussion 83-5. [Google Scholar] [CrossRef]
- Puigdellivol-Sanchez, A.; Prats-Galino, A.; Molander, C. Estimations of topographically correct regeneration to nerve branches and skin after peripheral nerve injury and repair. Brain Res. 2006, 1098, 49–60. [Google Scholar] [CrossRef]
- De Ruiter, G.C.; Spinner, R.J.; Verhaagen, J.; Malessy, M.J. Misdirection and guidance of regenerating axons after experimental nerve injury and repair. J. Neurosurg. 2014, 120, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Witzel, C.; Rohde, C.; Brushart, T.M. Pathway sampling by regenerating peripheral axons. J. Comp. Neurol. 2005, 485, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Naumann, T.; Hartig, W.; Frotscher, M. Retrograde tracing with Fluoro-Gold: Different methods of tracer detection at the ultrastructural level and neurodegenerative changes of back-filled neurons in long-term studies. J. Neurosci. Methods 2000, 103, 11–21. [Google Scholar] [CrossRef]
- Hayashi, A.; Moradzadeh, A.; Hunter, D.A.; Kawamura, D.H.; Puppala, V.K.; Tung, T.H.; Mackinnon, S.E.; Myckatyn, T.M. Retrograde labeling in peripheral nerve research: It is not all black and white. J. Reconstr. Microsurg. 2007, 23, 381–389. [Google Scholar] [CrossRef]
- Zygelyte, E.; Bernard, M.E.; Tomlinson, J.E.; Martin, M.J.; Terhorst, A.; Bradford, H.E.; Lundquist, S.A.; Sledziona, M.; Cheetham, J. RetroDISCO: Clearing technique to improve quantification of retrograde labeled motor neurons of intact mouse spinal cords. J. Neurosci. Methods 2016, 271, 34–42. [Google Scholar] [CrossRef]
- Negredo, P.; Castro, J.; Lago, N.; Navarro, X.; Avendano, C. Differential growth of axons from sensory and motor neurons through a regenerative electrode: A stereological, retrograde tracer, and functional study in the rat. Neuroscience 2004, 128, 605–615. [Google Scholar] [CrossRef]
- Yu, Y.L.; Li, H.Y.; Zhang, P.X.; Yin, X.F.; Han, N.; Kou, Y.H.; Jiang, B.G. Comparison of commonly used retrograde tracers in rat spinal motor neurons. Neural Regen Res. 2015, 10, 1700–1705. [Google Scholar]
- Sarker, M.D.; Naghieh, S.; McInnes, A.D.; Schreyer, D.J.; Chen, X. Regeneration of peripheral nerves by nerve guidance conduits: Influence of design, biopolymers, cells, growth factors, and physical stimuli. Prog. Neurobiol. 2018, 171, 125–150. [Google Scholar] [CrossRef]
- Yao, L.; de Ruiter, G.C.; Wang, H.; Knight, A.M.; Spinner, R.J.; Yaszemski, M.J.; Windebank, A.J.; Pandit, A. Controlling dispersion of axonal regeneration using a multichannel collagen nerve conduit. Biomaterials 2010, 31, 5789–5797. [Google Scholar] [CrossRef]
- Grinsell, D.; Keating, C.P. Peripheral nerve reconstruction after injury: A review of clinical and experimental therapies. BioMed Res. Int. 2014, 2014, 698256. [Google Scholar] [CrossRef]
- Scheib, J.; Hoke, A. Advances in peripheral nerve regeneration. Nat. Rev. Neurol. 2013, 9, 668–676. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, A.P.; Lokai, T.; Albin, B.; Yang, I.H. A Review on the Technological Advances and Future Perspectives of Axon Guidance and Regeneration in Peripheral Nerve Repair. Bioengineering 2022, 9, 562. [Google Scholar] [CrossRef]
- Zeng, Z.; Yang, Y.; Deng, J.; Saif Ur Rahman, M.; Sun, C.; Xu, S. Physical Stimulation Combined with Biomaterials Promotes Peripheral Nerve Injury Repair. Bioengineering 2022, 9, 292. [Google Scholar] [CrossRef] [PubMed]
- Castro, V.O.; Merlini, C. Aligned electrospun nerve conduits with electrical activity as a strategy for peripheral nerve regeneration. Artif. Organs 2021, 45, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Mankavi, F.; Ibrahim, R.; Wang, H. Advances in Biomimetic Nerve Guidance Conduits for Peripheral Nerve Regeneration. Nanomaterials 2023, 13, 2528. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Brushart, T.M. Outcomes Of Experimental Nerve Repair and Grafting. In Nerve Repair; Oxford University Press: Oxford, UK, 2011; pp. 159–195. [Google Scholar]
- Navarro, X. Functional evaluation of peripheral nerve regeneration and target reinnervation in animal models: A critical overview. Eur. J. Neurosci. 2016, 43, 271–286. [Google Scholar] [CrossRef]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef]
- Campbell, M.; McKenzie, J.E.; Sowden, A.; Katikireddi, S.V.; Brennan, S.E.; Ellis, S.; Hartmann-Boyce, J.; Ryan, R.; Shepperd, S.; Thomas, J.; et al. Synthesis without meta-analysis (SWiM) in systematic reviews: Reporting guideline. BMJ 2020, 368. [Google Scholar] [CrossRef]
- Haddaway, N.R.; Page, M.J.; Pritchard, C.C.; McGuinness, L.A. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Syst. Rev. 2022, 18, e1230. [Google Scholar] [CrossRef]
- Madison, R.D.; Da Silva, C.F.; Dikkes, P. Entubulation repair with protein additives increases the maximum nerve gap distance successfully bridged with tubular prostheses. Brain Res. 1988, 447, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ding, F.; Wu, J.; Hu, W.; Liu, W.; Liu, J.; Gu, X. Development and evaluation of silk fibroin-based nerve grafts used for peripheral nerve regeneration. Biomaterials 2007, 28, 5526–5535. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.D.; MacEwan, M.R.; French, A.R.; Moore, A.M.; Hunter, D.A.; Mackinnon, S.E.; Moran, D.W.; Borschel, G.H.; Sakiyama-Elbert, S.E. Fibrin matrices with affinity-based delivery systems and neurotrophic factors promote functional nerve regeneration. Biotechnol. Bioeng. 2010, 106, 970–979. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.; Deng, M.; Yang, M.; Liu, L.; Zhang, Y.; Wen, X. Axonal regeneration and remyelination evaluation of chitosan/gelatin-based nerve guide combined with transforming growth factor-beta1 and Schwann cells. Cell Biochem. Biophys. 2014, 68, 163–172. [Google Scholar] [CrossRef]
- Herda, T.J.; Zuniga, J.M.; Ryan, E.D.; Camic, C.L.; Bergstrom, H.C.; Smith, D.B.; Weir, J.P.; Cramer, J.T.; Housh, T.J. Quantifying the effects of electrode distance from the innervation zone on the electromyographic amplitude versus torque relationships. Physiol. Meas. 2013, 34, 315–324. [Google Scholar] [CrossRef]
- Shen, C.C.; Yang, Y.C.; Huang, T.B.; Chan, S.C.; Liu, B.S. Neural regeneration in a novel nerve conduit across a large gap of the transected sciatic nerve in rats with low-level laser phototherapy. J. Biomed. Mater. Res. A 2013, 101, 2763–2777. [Google Scholar] [CrossRef]
- Wang, X.; Pan, M.; Wen, J.; Tang, Y.; Hamilton, A.D.; Li, Y.; Qian, C.; Liu, Z.; Wu, W. A novel artificial nerve graft for repairing long-distance sciatic nerve defects: A self-assembling peptide nanofiber scaffold-containing poly(lactic-co-glycolic acid) conduit. Neural Regen. Res. 2014, 9, 2132–2141. [Google Scholar]
- Gan, L.; Zhao, L.; Zhao, Y.; Li, K.; Tong, Z.; Yi, L.; Wang, X.; Li, Y.; Tian, W.; He, X.; et al. Cellulose/soy protein composite-based nerve guidance conduits with designed microstructure for peripheral nerve regeneration. J. Neural Eng. 2016, 13, 056019. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Wang, X.; Li, X.; Shi, W.; Wang, X.; Zou, Y.; Yang, G.; Tang, X.; Zhang, Z.; Xiang, W.; et al. EMSCs-Seeded Micro-Stripe Patterned Polycaprolactone Promoting Sciatic Nerve Regeneration. Adv. Mater. Interfaces 2022, 10, 2201929. [Google Scholar] [CrossRef]
- Bodine-Fowler, S.C.; Meyer, R.S.; Moskovitz, A.; Abrams, R.; Botte, M.J. Inaccurate projection of rat soleus motoneurons: A comparison of nerve repair techniques. Muscle Nerve 1997, 20, 29–37. [Google Scholar] [CrossRef]
- Valero-Cabre, A.; Tsironis, K.; Skouras, E.; Perego, G.; Navarro, X.; Neiss, W.F. Superior muscle reinnervation after autologous nerve graft or poly-L-lactide-epsilon-caprolactone (PLC) tube implantation in comparison to silicone tube repair. J. Neurosci. Res. 2001, 63, 214–223. [Google Scholar] [CrossRef]
- Valero-Cabre, A.; Tsironis, K.; Skouras, E.; Navarro, X.; Neiss, W.F. Peripheral and spinal motor reorganization after nerve injury and repair. J. Neurotrauma 2004, 21, 95–108. [Google Scholar] [CrossRef] [PubMed]
- De Ruiter, G.C.; Spinner, R.J.; Malessy, M.J.; Moore, M.J.; Sorenson, E.J.; Currier, B.L.; Yaszemski, M.J.; Windebank, A.J. Accuracy of motor axon regeneration across autograft, single-lumen, and multichannel poly(lactic-co-glycolic acid) nerve tubes. Neurosurgery 2008, 63, 144–153; discussion 53-5. [Google Scholar] [CrossRef]
- Hu, X.; Huang, J.; Ye, Z.; Xia, L.; Li, M.; Lv, B.; Shen, X.; Luo, Z. A novel scaffold with longitudinally oriented microchannels promotes peripheral nerve regeneration. Tissue Eng. Part A 2009, 15, 3297–3308. [Google Scholar] [CrossRef]
- Huang, J.; Lu, L.; Hu, X.; Ye, Z.; Peng, Y.; Yan, X.; Geng, D.; Luo, Z. Electrical stimulation accelerates motor functional recovery in the rat model of 15-mm sciatic nerve gap bridged by scaffolds with longitudinally oriented microchannels. Neurorehabil. Neural Repair. 2010, 24, 736–745. [Google Scholar] [CrossRef]
- Zhang, Y.G.; Huang, J.H.; Hu, X.Y.; Sheng, Q.S.; Zhao, W.; Luo, Z.J. Omentum-wrapped scaffold with longitudinally oriented micro-channels promotes axonal regeneration and motor functional recovery in rats. PLoS ONE 2011, 6, e29184. [Google Scholar] [CrossRef]
- Jiang, X.; Mi, R.; Hoke, A.; Chew, S.Y. Nanofibrous nerve conduit-enhanced peripheral nerve regeneration. J. Tissue Eng. Regen. Med. 2014, 8, 377–385. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, P.; Yin, X.; Han, N.; Kou, Y.; Jiang, B. Specificity of motor axon regeneration: A comparison of recovery following biodegradable conduit small gap tubulization and epineurial neurorrhaphy. Am. J. Transl. Res. 2015, 7, 53–65. [Google Scholar]
- Bozkurt, A.; Boecker, A.; Tank, J.; Altinova, H.; Deumens, R.; Dabhi, C.; Tolba, R.; Weis, J.; Brook, G.; Pallua, N.; et al. Efficient bridging of 20 mm rat sciatic nerve lesions with a longitudinally micro-structured collagen scaffold. Biomaterials 2016, 75, 112–122. [Google Scholar] [CrossRef]
- Van Neerven, S.G.A.; Haastert-Talini, K.; Boecker, A.; Schriever, T.; Dabhi, C.; Claeys, K.; Deumens, R.; Brook, G.; Weis, J.; Pallua, N.; et al. Two-component collagen nerve guides support axonal regeneration in the rat peripheral nerve injury model. J. Tissue Eng. Regen. Med. 2017, 11, 3349–3361. [Google Scholar] [CrossRef]
- Zhu, L.; Wang, K.; Ma, T.; Huang, L.; Xia, B.; Zhu, S.; Yang, Y.; Liu, Z.; Quan, X.; Luo, K.; et al. Noncovalent Bonding of RGD and YIGSR to an Electrospun Poly(epsilon-Caprolactone) Conduit through Peptide Self-Assembly to Synergistically Promote Sciatic Nerve Regeneration in Rats. Adv. Healthc. Mater. 2017, 6, 1600860. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Mi, D.; Zhang, T.; Zhang, Y.; Yan, J.; Wang, Y.; Tan, X.; Yuan, Y.; Yang, Y.; Gu, X.; et al. Tubulation repair mitigates misdirection of regenerating motor axons across a sciatic nerve gap in rats. Sci. Rep. 2018, 8, 3443. [Google Scholar] [CrossRef] [PubMed]
- Xue, C.; Zhu, H.; Tan, D.; Ren, H.; Gu, X.; Zhao, Y.; Zhang, P.; Sun, Z.; Yang, Y.; Gu, J.; et al. Electrospun silk fibroin-based neural scaffold for bridging a long sciatic nerve gap in dogs. J. Tissue Eng. Regen. Med. 2018, 12, e1143–e1153. [Google Scholar] [CrossRef]
- Zhu, L.; Jia, S.; Liu, T.; Yan, L.; Huang, D.; Wang, Z.; Chen, S.; Zhang, Z.; Zeng, W.; Zhang, Y.; et al. Aligned PCL Fiber Conduits Immobilized with Nerve Growth Factor Gradients Enhance and Direct Sciatic Nerve Regeneration. Adv. Funct. Mater. 2020, 30, 2002610. [Google Scholar] [CrossRef]
- Lu, C.F.; Wang, B.; Zhang, P.X.; Han, S.; Pi, W.; Kou, Y.H.; Jiang, B. Combining chitin biological conduits with small autogenous nerves and platelet-rich plasma for the repair of sciatic nerve defects in rats. CNS Neurosci. Ther. 2021, 27, 805–819. [Google Scholar] [CrossRef]
- Yang, J.; Hsu, C.C.; Cao, T.T.; Ye, H.; Chen, J.; Li, Y.Q. A hyaluronic acid granular hydrogel nerve guidance conduit promotes regeneration and functional recovery of injured sciatic nerves in rats. Neural Regen. Res. 2023, 18, 657–663. [Google Scholar]
- Tomita, K.; Kubo, T.; Matsuda, K.; Hattori, R.; Fujiwara, T.; Yano, K.; Hosokawa, K. Effect of conduit repair on aberrant motor axon growth within the nerve graft in rats. Microsurgery 2007, 27, 500–509. [Google Scholar] [CrossRef]
- Papaloizos, M.Y.; Holmquist, B.; Lundborg, G. An experimental study of nerve grafting combined with silicone tubes in the rat model: Functional outcome and specificity of muscle reinnervation. Restor. Neurol. Neurosci. 1997, 11, 161–168. [Google Scholar] [CrossRef]
- Johnson, B.N.; Lancaster, K.Z.; Zhen, G.; He, J.; Gupta, M.K.; Kong, Y.L.; Engel, E.A.; Krick, K.D.; Ju, A.; Meng, F.; et al. 3D Printed Anatomical Nerve Regeneration Pathways. Adv. Funct Mater. 2015, 25, 6205–6217. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Tringale, K.R.; Woller, S.A.; You, S.; Johnson, S.; Shen, H.; Schimelman, J.; Whitney, M.; Steinauer, J.; Xu, W.; et al. Rapid continuous 3D printing of customizable peripheral nerve guidance conduits. Mater Today 2018, 21, 951–959. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Yan, L.W.; Wang, T.; Qiu, S.; Lin, T.; He, F.L.; Yuan, R.H.; Liu, X.L.; Qi, J.; Zhu, Q.T. A rapid micro-magnetic resonance imaging scanning for three-dimensional reconstruction of peripheral nerve fascicles. Neural Regen Res. 2018, 13, 1953–1960. [Google Scholar]
- Yao, Z.; Yan, L.W.; Qiu, S.; He, F.L.; Gu, F.B.; Liu, X.L.; Qi, J.; Zhu, Q.-T. Customized Scaffold Design Based on Natural Peripheral Nerve Fascicle Characteristics for Biofabrication in Tissue Regeneration. BioMed Res. Int. 2019, 2019, 3845780. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.S.; Lee, T.Y.; Kneiber, D.; Dy, C.J.; Ward, P.M.; Kazarian, G.; Apostolakos, J.; Brogan, D.M. Design and In Vivo Testing of an Anatomic 3D-Printed Peripheral Nerve Conduit in a Rat Sciatic Nerve Model. HSS J. 2024, 21, 414–423. [Google Scholar] [CrossRef]
- Stewart, C.E.; Kan, C.F.K.; Stewart, B.R.; Sanicola, H.W., 3rd; Jung, J.P.; Sulaiman, O.A.R.; Wang, D. Machine intelligence for nerve conduit design and production. J. Biol. Eng. 2020, 14, 25. [Google Scholar] [CrossRef]
- Venkata Krishna, D.; Ravi Sankar, M. Engineered approach coupled with machine learning in biofabrication of patient-specific nerve guide conduits—Review. Bioprinting 2023, 30, e00264. [Google Scholar] [CrossRef]
- Chaker, S.C.; Reddy, A.P.; King, D.; Manzanera Esteve, I.V.; Thayer, W.P. Diffusion Tensor Imaging: Techniques and Applications for Peripheral Nerve Injury. Ann. Plast Surg. 2024, 93, S113–S115. [Google Scholar] [CrossRef] [PubMed]




| Inclusion Criteria | Exclusion Criteria | |
|---|---|---|
| Population |
|
|
| Intervention |
|
|
| Comparison |
|
|
| Outcome |
|
|
| Author, Year | Animal Species | Gap Length (mm) | Follow-Up (Weeks) | Repair Technique | Graft Type | Material | Internal Framework | Internal Achitecture | RT Technique | RT Cohort (n) | NHM Level | NHM Cohort (n) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bodine-Fowler et al., 1997 [77] | SD female rat SN | NA | 14 | ETE | NA | NA | NA | NA | Sequential | 9 | NA | NA |
| 5 | 14 | ArtNG | NGC | SIL | Unstructured | Smooth | Sequential | 8 | NA | NA | ||
| Valero Cabre et al., 2001 [78] | W female rat SN | 8 | 12 | BNG | ANG | - | In situ | - | Triple | 6 | NA | NA |
| 8 | 12 | ArtNG | NGC | SIL | Unstructured | Smooth | Triple | 6 | NA | NA | ||
| 8 | 12 | ArtNG | NGC | PLC | Unstructured | Smooth | Triple | 8 | NA | NA | ||
| Valero Cabre et al., 2004 [79] | W female rat SN | NA | 12 | ETE | NA | NA | NA | NA | Triple | 9 | NA | NA |
| 4 | 12 | ArtNG | NGC | SIL | Unstructured | Smooth | Triple | 13 | NA | NA | ||
| De Ruiter et al., 2008 [80] | SD rat SN | 10 | 12 | BNG | ANG | NA | In situ | NA | Double | 7 | MG & DDS | 6 |
| 10 | 12 | ArtNG | NGC | PLGA | Unstructured | Rough | Double | 4 | MG & DDS | 4 | ||
| 10 | 12 | ArtNG | NGC | PLGA | Structured | Multichannel (7) * | Double | 6 | MG & DDS | 2 | ||
| Hu et al., 2009 [81] | SD male rat SN | 15 | 12 | ArtNG | NGC | SIL | Unstructured | Smooth | Single | 3 | DDS | 3 |
| 15 | 12 | ArtNG | NSC | COL/CS | Structured | Multichannel (R) * | Single | 3 | DDS | 5 | ||
| 15 | 12 | ArtNG | NSC | COL/CS ** | Structured | Multichannel (L) * | Single | 3 | DDS | 9 | ||
| 15 | 12 | BNG | ANG | - | Reversed | - | Single | 3 | DDS | 9 | ||
| Huang, 2010 [82] | SD male rat SN | 15 | 12 | ArtNG | NSC | CS | Structured | Multichannel (R) * | Single | 5 | PDS | 10 |
| 15 | 12 | ArtNG | NSC | CS | Structured | Multichannel (L) * | Single | 5 | PDS | 10 | ||
| 15 | 12 | BNG | ANG | NA | Reversed | NA | Single | 5 | PDS | 10 | ||
| Yao et al., 2010 [55] | L female rat SN | 10 | 16 | BNG | ANG | NA | In situ | NA | Double | 6 | MG | 8 |
| 10 | 16 | ArtNG | NGC | COL | Unstructured | Rough | Double | 6 | MG | 8 | ||
| 10 | 16 | ArtNG | NGC | COL | Structured | Multichannel (2) * | Double | 6 | MG | 8 | ||
| 10 | 16 | ArtNG | NGC | COL | Structured | Multichannel (4) * | Double | 6 | MG | 8 | ||
| Zhang, 2011 [83] | SD male rat SN | 15 | 12 | ArtNG | NSC | COL | Structured | Multichannel (L) * | Single | 6 | MG & PDS | 6 |
| 15 | 12 | BNG | ANG | NA | Reversed | NA | Single | 6 | MG & PDS | 6 | ||
| Daly, 2012 [31] | L female rat SN L female rat SN L female rat SN L female rat SN | 10 | 13 | BNG | ANG | NA | Reversed | NA | Double | 6 | MG | 7 |
| 10 | 13 | ArtNG | NGC | COL | Structured | Fibrous (S) * | Double | 6 | MG | 8 | ||
| 10 | 13 | ArtNG | NGC | COL | Structured | Fibrous (U) * | Double | 6 | MG | 8 | ||
| 10 | 13 | ArtNG | NGC | COL | Unstructured | Rough | Double | 6 | MG | 8 | ||
| Jiang, 2012 [84] | SD female rat SN | 15 | 16 | ArtNG | NGC | PCL | Unstructured | Rough (M) * | Single | 8 | MG | 8 |
| SD female rat SN | 15 | 16 | ArtNG | NGC | PCL | Unstructured | Rough (N) * | Single | 9 | MG | 9 | |
| Yu, 2015 [85] | SD female rat SN | - | 12 | ETE | NA | NA | NA | NA | Double | 10 | DDS | 6 |
| SD female rat SN | 2 | 12 | ArtNG | NGC | CHIT | Unstructured | Smooth | Double | 10 | DDS | 6 | |
| Bozkurt, 2016 [86] | L female rat SN | 20 | 12 | ArtNG | NSC | COL | Structured | Multichannel (L) * | Single | 4 | PDS & DDS | 7 |
| L female rat SN | 20 | 12 | BNG | ANG | - | In situ | NA | Single | 5 | PDS & DDS | 6 | |
| Van Neerven et al., 2017 [87] | L female rat SN | 20 | 12 | ArtNG | NGC | COL | Structured | Multichannel (L) * | Single | 4 | PDS & DDS | 6 |
| L female rat SN | 20 | 12 | ArtNG | NGC | COL | Unstructured | Rough | Single | 4 | PDS & DDS | 8 | |
| L female rat SN | 20 | 12 | BNG | ANG | NA | In situ | NA | Single | 5 | PDS & DDS | 5 | |
| Zhu, 2017 [88] | SD male rat SN | 15 | 12 | ArtNG | NGC | PCL | Unstructured | Rough | Single | 9 | MG | 3 |
| SD male rat SN | 15 | 12 | BNG | ANG | NA | In situ | NA | Single | 9 | MG | 3 | |
| Liu, 2018 [89] | SD female rat SN | 6 | 12 | BNG | ANG | NA | In situ | NA | Sequential | 6 | NA | NA |
| SD female rat SN | 3 | 12 | ArtNG | NGC | PLGA-CS *** | Structured | Fibrous | Sequential | 6 | NA | NA | |
| SD female rat SN | 6 | 12 | ArtNG | NGC | PLGA-CS *** | Structured | Fibrous | Sequential | 6 | NA | NA | |
| Xue, 2018 [90] | B male dog SN | 30 | 52 | ArtNG | NGC | SF | Structured | Fibrous | Single | 3 | PDS | 6 |
| 30 | 52 | BNG | ANG | NA | Reversed | NA | Single | 3 | PDS | 6 | ||
| Zhu, 2020 [91] | SD male rat SN | 15 | 12 | ArtNG | NGC | PCL | Unstructured | Rough | Single | 6 | MG | 6 |
| 15 | 12 | BNG | ANG | NA | In situ | NA | Single | 6 | MG | 6 | ||
| Lu, 2021 [92] | SD female rat SN | 10 | 12 | ArtNG | NGC | CHIT | Unstructured | Smooth | Single | 4 | DDS | 5 |
| 10 | 12 | BNG | ANG | NA | In situ | NA | Single | 4 | DDS | 5 | ||
| Yang, 2023 [93] | SD male rat SN | 10 | 16 | ArtNG | NGC | CS | Unstructured | Smooth | Single | 3 | PDS | 3 |
| 10 | 16 | BNG | ANG | NA | Reversed | NA | Single | 3 | PDS | 3 |
| Study ID | Animal Model | Repair Technique Characteristics | LMN | Incorrect Projecting LMNs (%) | Accurate Projecting LMN (%) | |||
|---|---|---|---|---|---|---|---|---|
| Bodine-Fowler et al., 1997 [77] | Rat (SD), SN, | ETE | 112 ± 38 | REF | NA | NA | 19 ± 9 | REF |
| Rat (SD), SN, 5 mm | ArtNG, NGC, SIL, Unstructured, Smooth | 133 ± 41 | = | NA | NA | 14 ± 5 | = | |
| Key finding: Despite similar LMN survival, there were no significant differences between the amount of accurate projecting LMNs between the hollow SIL NGC and direct ETE repair (14 ± 5 vs. 19 ± 9). | ||||||||
| Valero Cabre et al., 2004 [79] | Rat (W), SN, | ETE | 1175 ± 106 | REF | 2.2 | REF | NA | NA |
| Rat (W), SN, 4 mm | ArtNG, NGC, SIL, Unstructured, Smooth | 1250 ± 182 | = | 6.3 | ↑ (p < 0.05) | NA | NA | |
| Key finding: Despite similar LMN survival, the percentage of incorrect projections was significantly higher in NGC (6%) compared to ETE repair (2.2%), with p < 0.05. | ||||||||
| Yu et al., 2015 [85] | Rat (SD), FN, | ETE | 334 ± 38 | REF | 112 ± 13 | REF | 55.89 ± 1.63 | REF |
| Rat (SD), FN, 2 mm | ArtNG, NGC, CHIT, Unstructured, Smooth | 326 ± 42 | = | 74 ± 11 | ↓ (p < 0.05) | 68.18 ± 2.04 | ↑ (p < 0.05) | |
| Key finding: Despite similar LMN survival, the percentage of incorrect projections (74 ± 11%) was significantly lower in NGC compared to ETE repair (112 ± 13) (p < 0.05). This observation is reflected in a significant difference in the specificity of motor axon regeneration between the two groups (small gap tubulization group: 68.18 ± 2.04% vs. epineurial neurorrhaphy group: 55.89 ± 1.63%, p < 0.05) | ||||||||
| Study ID | Animal Model | Repair Technique Characteristics | LMN | LSN | TLN | LMN/LSN | TLN/RN.TF | TLN/DDS | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hu et al. [81] 2009 | Rat (SD), SN, 15 mm | ArtNG, NGC, SIL, Unstructured, Smooth | 230 ± 30 | ↓ (p < 0.05) | 260 ± 50 | ↓ (p < 0.05) | 490 ± 58.3 | ↓ (Δ ≥ 10%) | 0.88 ± 0.21 | = (overlap) | NR | NA | 0.09 | ↑ |
| ArtNG, NSC, COL-CS*, Structured, Multichannel (R) | 550 ± 85 | ↓ (p < 0.05) | 480 ± 60 | ↓ (p < 0.05) | 1030 ± 104.0 | ↓ (Δ ≥ 10%) | 1.15 ± 0.23 | = (overlap) | NR | NA | 0.10 | ↑ | ||
| ArtNG, NSC, COL-CS*, Structured, Multichannel (L) | 700 ± 90 | = (p > 0.05) | 720 ± 70 | = (p > 0.05) | 1420 ± 114.0 | = (overlap) | 0.97 ± 0.16 | = (overlap) | NR | NA | 0.06 | ↓ | ||
| BNG, ANG, Reversed | 800 ± 80 | REF | 820 ± 80 | REF | 1620 ± 113.1 | REF | 0.98 ± 0.14 | REF | NR | NA | 0.07 | REF | ||
| Key findings: There was no significant difference in LMN and LSN counts between longitudinal multichannel NSCs and reversed ANGs (p > 0.05), while significantly lower in two other ArtNg Groups (p < 0.05). | ||||||||||||||
| Huang et al., 2010 [82] | Rat (SD), SN, 15 mm | ArtNG, NSC, CS, Structured, Multichannel (R) | 390 ± 12 | ↓ (p < 0.05) | NR | NA | NR | NA | NR | NA | NR | NA | NR | NA |
| ArtNG, NSC, CS, Structured, Multichannel (L) | 520 ± 12 | = (p > 0.05) | NR | NA | NR | NA | NR | NA | NR | NA | NR | NA | ||
| BNG, ANG, Reversed | 530 ± 12 | REF | NR | NA | NR | NA | NR | NA | NR | NA | NR | NA | ||
| Key findings: There was a significant difference in LMN counts between longitudinal and randomized multichannel NSCs (p < 0.05). | ||||||||||||||
| Zhang et al., 2011 [83] | Rat (SD), SN, 15 mm | ArtNG, NSC, COL, Structured, Multichannel (L) | 520 ± 20 | = (p > 0.05) | 580 ± 20 | = (p > 0.05) | 1100 ± 28.3 | = (Δ < 10%) | 0.90 ± 0.05 | = (overlap) | 0.06 | = | NR | NA |
| BNG, ANG, Reversed | 550 ± 20 | REF | 650 ± 20 | REF | 1200 ± 28.3 | REF | 0.85 ± 0.04 | REF | 0.06 | REF | NR | NA | ||
| Key findings: There was no significant difference in LMN and LSN counts between longitudinal multichannel NSCs and reversed ANG (p > 0.05). | ||||||||||||||
| Jiang et al., 2012 [84] | Rat (SD), SN, 15 mm | ArtNG, NGC, PCL, Unstructured, Rough Microfibrous | 270 ± 141 | NA | 980 ± 849 | NA | 1250 ± 860 | NA | 0.28 ± 0.28 | NA | 0.39 | NA | NR | NA |
| ArtNG, NGC, PCL, Unstructured, Rough Nanofibrous | 360 ± 180 | NA | 1930 ± 2130 | NA | 2290 ± 2138 | NA | 0.19 ± 0.23 | NA | 0.34 | NA | NR | NA | ||
| Key findings: There was no significant difference in LMN counts, while there were significantly higher LSN and TF counts in nanofibrous compared to microfibrous NGCs (p < 0.05). | ||||||||||||||
| Bozkurt et al., 2016 [86] | Rat (L), SN, 20 mm | ArtNG, NSC, COL, Structured, Multichannel (L) | 454 ± 358 | = (overlap) | 760 ± 174 | = (overlap) | 1214 ± 398 | = (overlap) | 0.60 ± 0.49 | = (overlap) | NR | NA | NR | NA |
| BNG, ANG, In situ | 650 ± 168 | REF | 900 ± 179 | REF | 1550 ± 245 | REF | 0.72 ± 0.24 | REF | NR | NA | NR | NA | ||
| Key findings: No reported significant difference in outcomes between in situ ANG and longitudinal multichannel NSCs. | ||||||||||||||
| Van Neerven et al., 2017 [87] | Rat (L), SN, 20 mm | ArtNG, NGC, COL, Structured, Multichannel (L) | 290 ± 184 | = (overlap) | 1424 ± 338 | ↑ (Δ ≥ 10%) | 1714 ± 385 | = (overlap) | 0.20 ± 0.14 | ↓ (Δ ≥ 10%) | 0.44 | ↑ | 0.8 | = |
| ArtNG, NGC, COL, Unstructured, Rough | 206 ± 180 | = (overlap) | 1126 ± 522 | = (overlap) | 1332 ± 552 | = (overlap) | 0.18 ± 0.18 | ↓ (Δ ≥ 10%) | 0.58 | ↑ | 0.8 | = | ||
| BNG, ANG, In situ | 456 ± 94 | REF | 785 ± 192 | REF | 1241 ± 214 | REF | 0.58 ± 0.19 | REF | 0.08 | REF | 0.1 | REF | ||
| Key findings: Structured ArtNG resulted in significantly higher PDS.FD compared to unstructured ArtNG (p < 0.05), and higher DDS.AD and DDS.FD compared to BNGs (p < 0.01). | ||||||||||||||
| Zhu et al., 2017 [88] | Rat (SD), SN, 15 mm | ArtNG, NGC, PCL, Unstructured, Rough | 490 ± 40 | ↓ (Δ ≥ 10%) | 570 ± 50 | ↓ (Δ ≥ 10%) | 1060 ± 64.0 | ↓ (Δ ≥ 10%) | 0.86 ± 0.10 | = (overlap) | 0.44 | ↑ | NR | NA |
| BNG, ANG, In situ | 580 ± 35 | REF | 680 ± 40 | REF | 1260 ± 53.2 | REF | 0.85 ± 0.07 | REF | 0.12 | REF | NR | NA | ||
| Key findings: No reported significant difference in outcomes between in situ ANG and unstructured rough NGC. | ||||||||||||||
| Xue et al., 2018 [90] | Dog (B), SN, 30 mm | ArtNG, NGC, SF, Structured, Fibrous | 1700 ± 300 | = (overlap) | NR | NA | NR | NA | NR | NA | NR | NA | NR | NA |
| BNG, ANG, Reversed | 1800 ± 200 | REF | NR | NA | NR | NA | NR | NA | NR | NA | NR | NA | ||
| Key findings: No reported significant difference in outcomes between reversed ANG and structured fibrous NSC. | ||||||||||||||
| Zhu et al., 2020 [91] | Rat (SD), SN, 15 mm | ArtNG, NGC, PCL, Unstructured, Rough | 500 ± 30 | ↓ (p < 0.01) | 580 ± 30 | ↓ (Δ ≥ 10%) | 1080 ± 42.4 | ↓ (Δ ≥ 10%) | 0.86 ± 0.07 | = (Overlap) | 0.09 | ↑ | NR | NA |
| BNG, ANG, In situ | 635 ± 69 | REF | 705 ± 56 | REF | 1340 ± 88.9 | REF | 0.90 ± 0.12 | REF | 0.07 | REF | NR | NA | ||
| Key findings: ANGs provided significantly higher LMN (p < 0.01) and TF (p < 0.05) counts compared to unstructured ArtNGs. | ||||||||||||||
| Lu et al., 2021 [92] | Rat (SD), SN, 10 mm | ArtNG, NGC, CHIT, Unstructured, Smooth | 195 ± 10 | ↓ (p < 0.01) | 300 ± 10 | ↓ (p < 0.01) | 495 ± 14.1 | ↓ (Δ ≥ 10%) | 0.65 ± 0.04 | ↓ (Δ ≥ 10%) | NR | NA | NR | NA |
| BNG, ANG, In situ | 420 ± 20 | REF | 570 ± 10 | REF | 990 ± 22.4 | REF | 0.74 ± 0.04 | REF | NR | NA | NR | NA | ||
| Key findings: ANGs provided significantly higher LMN, LSN, and DDS.FD (p < 0.01) counts compared to unstructured ArtNGs. | ||||||||||||||
| Yang et al., 2023 [93] | Rat (SD), SN, 10 mm | ArtNG, NGC, CS, Unstructured, Smooth | 95 ± 10 | ↓ (p < 0.01) | 310 ± 20 | ↓ (Δ ≥ 10%) | 405 ± 22.4 | ↓ (Δ ≥ 10%) | 0.31 ± 0.04 | ↓ (Δ ≥ 10%) | 0.04 | ↓ | NR | NA |
| BNG, ANG, Reversed | 140 ± 5 | REF | 375 ± 5 | REF | 515 ± 7.1 | REF | 0.37 ± 0.01 | REF | 0.11 | REF | NR | NA | ||
| Key findings: ANGs provided significantly higher LMN (p < 0.01) counts compared to unstructured ArtNGs. | ||||||||||||||
| Study ID | Animal Model | Repair Technique Characteristics | LMN | Incorrect Projecting LMNs (%) | Accurate Projecting LMNs (%) | |||
|---|---|---|---|---|---|---|---|---|
| Valero Cabre et al., 2001 [78] | Rat (W), SN, 8 mm | BNG, ANG, In situ | 1186 ± 56 | REF | 5.8 ± 0.6 | REF | NA | NA |
| ArtNG, NGC, SIL, Unstructured, Smooth | 935 ± 213 | = (Overlap) | 10 ± 0.1 | ↓ (p < 0.05) | NA | NA | ||
| ArtNG, NGC, PLC, Unstructured, Smooth | 802 ± 101 | ↓ (p < 0.05) | 6.0 ± 1.6 | = (Δ < 10%) | NA | NA | ||
| Key finding: The percentage of double projecting neurons was significantly higher with hollow SIL NGC (10.1%) compared to autograft (5.8%) and PLC NGC (6.0%), with (p < 0.05). | ||||||||
| De Ruiter et al., 2008 [80] | Rat (SD), SN, 10 mm | BNG, ANG, In situ | 1140 ± 179 | REF | 5.9 ± 2.9 | REF | NA | NA |
| ArtNG, NGC, PLGA, Unstructured, Rough | 406 ± 156 | ↓ (Δ ≥ 10%) | 21.4 ± 4.9 | ↓ (p < 0.01) | NA | NA | ||
| ArtNG, NGC, PLGA, Structured, Multichannel (7) | 448 ± 108 | ↓ (Δ ≥ 10%) | 16.9 ± 6.0 | ↓ (Δ ≥ 10%) | NA | NA | ||
| Key finding: The percentage of double projecting neurons was 21.4% compared to autograft repair (5.9%) being significantly lower with a p value less than 0.01 | ||||||||
| Yao et al., 2010 [55] | Rat (L), SN, 10 mm | BNG, ANG, In situ | 1000 ± 167 | REF | 4.5 ± 2.4 | REF | NA | NA |
| ArtNG, NGC, COL, Unstructured, Rough | 270 ± 64 | ↓ (p < 0.01) | 7.1 ± 2.7 | = (Overlap) | NA | NA | ||
| ArtNG, NGC, COL, Structured, Multichannel (2) | 245 ± 50 | ↓ (p < 0.01) | 2.7 ± 2.9 | = (Overlap) | NA | NA | ||
| ArtNG, NGC, COL, Structured, Multichannel (4) | 325 ± 75 | ↓ (p < 0.01) | 2.4 ± 1.5 | = (Overlap) | NA | NA | ||
| Key finding; Percentage of LMNs with double projections in hollow COL NGC (7.1% ± 2.7%), was significantly higher when compared with treated 2-channel and 4-channel conduits (p < 0.05). | ||||||||
| Daly et al., 2012 [31] | Rat (L), SN, 10 mm | BNG, ANG, Reversed | 980 ± 269 | REF | 17.83 ± 13.57 | REF | NA | NA |
| ArtNG, NGC, COL, Structured, Fibrous (S) | 880 ± 184 | = (Overlap) | 0.84 ± 1.19 | ↑ (p > 0.05) | NA | NA | ||
| ArtNG, NGC, COL, Structured, Fibrous (U) | 730 ± 171 | = (Overlap) | 2.42 ± 2.33 | ↑ (p > 0.05) | NA | NA | ||
| ArtNG, NGC, COL, Unstructured, Rough | 260 ± 147 | ↓ (Δ ≥ 10%) | 6.8 ± 1.1 | ↑ (Δ ≥ 10%) | NA | NA | ||
| Key finding: Structured fibers and unstructured fibers showed significantly lower misdirection rates (Structured: 0.84% ± 1.19%, Unstructured: 2.42% ± 2.33%) compared to autograft (17.83% ± 13.57%) (p < 0.05). | ||||||||
| Liu et al., 2018 [89] | Rat (SD), SN, 6 mm | BNG, ANG, In situ | 2144 ± 205 | REF | 30.50 ± 3.25 | REF | 83.98 ± 5.41 | REF |
| ArtNG, NGC, PLGA/CS, Structured, Fibrous | 1886 ± 292 | ↓ (p < 0.01) | 20.33 ± 8.10 | ↑ (p < 0.05) | 83.43 ± 9.48 | = (Overlap) | ||
| ArtNG, NGC, PLGA/CS, Structured, Fibrous | 1544 ± 228 | ↓ (p < 0.01) | 19.45 ± 9.21 | ↑ (p < 0.05) | 83.98 ± 6.76 | = (Overlap) | ||
| Key finding: The tubulation repair with a 3 mm chitosan/PGLA conduit significantly reduced motor axon misdirection compared to autograft repair, despite slightly lower total motor neuron regeneration. | ||||||||
| Outcome | Structured ArtNGs | Unstructured ArtNGs | No. of Arms (Studies) | Certainty of Evidence | Plain Language Summary |
|---|---|---|---|---|---|
| LMN counts | 8/15 similar, 7/15 lower; microchannel 5/5 preserved; microporous & multichannel 100% lower | 2/11 similar, 9/11 lower | 26 arms (19 studies) | ⬤◯◯◯ Very low | Structured ArtNGs often preserved LMNs; unstructured conduits mostly reduced LMNs vs. ANG |
| TLN counts | 4/5 similar | 1/6 similar, 5/6 lower | 11 arms (13 studies) | ⬤◯◯◯ Very low | Structured ArtNGs generally preserved TLN; unstructured conduits mostly reduced TLN |
| LSN counts | 3/5 similar, 1/5 higher | 1/6 similar, 5/6 lower | 11 arms (13 studies) | ⬤◯◯◯ Very low | Structured ArtNG occasionally increased LSN; unstructured conduits often reduced LSN |
| LMN/LSN ratio | 4/5 similar | 3/5 similar, 2/5 lower | 10 arms (13 studies) | ⬤◯◯◯ Very low | Structured conduits more often preserved motor–sensory specificity, while unstructured more often worsened it |
| Innervation accuracy—Incorrect projecting LMNs | 4/7 lower, 2/7 similar, 1/7 higher | 2/5 lower, 3/5 similar | 12 arms (8 studies) | ⬤◯◯◯ Very low | Structured conduits more often reduced misdirection; unstructured presented with mixed results |
| Innervation accuracy—Accurate projecting LMNs | 2/7 similar | 0/5 reported | 2 arms (2 studies) | ⬤◯◯◯ Very low | Sparse reporting was present—structured conduits matched ANG in two arms |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mićić, A.; Aksić, M.; Savić, A.; Poleksić, J.; Grujić, J.; Lepić, M.; Aleksić, D.; Vujić, L.; Rasulić, L. Structural Features of Nerve Guidance Conduits and Scaffolds in Preventing Axonal Misdirection: A Systematic Review of Retrograde Tracing Studies. Bioengineering 2026, 13, 220. https://doi.org/10.3390/bioengineering13020220
Mićić A, Aksić M, Savić A, Poleksić J, Grujić J, Lepić M, Aleksić D, Vujić L, Rasulić L. Structural Features of Nerve Guidance Conduits and Scaffolds in Preventing Axonal Misdirection: A Systematic Review of Retrograde Tracing Studies. Bioengineering. 2026; 13(2):220. https://doi.org/10.3390/bioengineering13020220
Chicago/Turabian StyleMićić, Aleksa, Milan Aksić, Andrija Savić, Joko Poleksić, Jovan Grujić, Milan Lepić, Dubravka Aleksić, Lazar Vujić, and Lukas Rasulić. 2026. "Structural Features of Nerve Guidance Conduits and Scaffolds in Preventing Axonal Misdirection: A Systematic Review of Retrograde Tracing Studies" Bioengineering 13, no. 2: 220. https://doi.org/10.3390/bioengineering13020220
APA StyleMićić, A., Aksić, M., Savić, A., Poleksić, J., Grujić, J., Lepić, M., Aleksić, D., Vujić, L., & Rasulić, L. (2026). Structural Features of Nerve Guidance Conduits and Scaffolds in Preventing Axonal Misdirection: A Systematic Review of Retrograde Tracing Studies. Bioengineering, 13(2), 220. https://doi.org/10.3390/bioengineering13020220









